Kim Y.J. (Ed.) Advanced Environmental Monitoring
Подождите немного. Документ загружается.

404 S. Mülhopt et al.
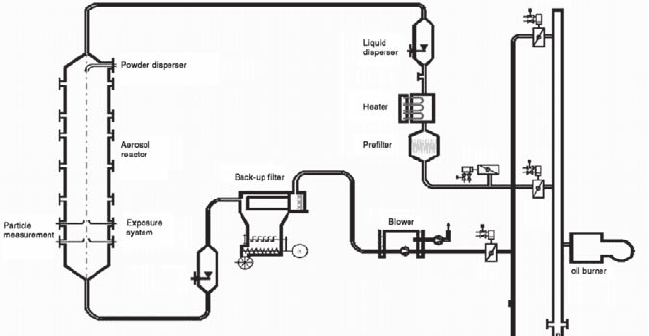
etc.) may be added to the gas. At flow rates of up to 800 Nm
3
/h and temperatures
from 20 to 200°C, gas and particle concentrations can be varied over wide ranges.
The carrier gas first flows through a HEPA filter. This filter ensures that the gas is
particle-free and later measurements are not falsified by impurities. Further down-
stream is an air heater, which allows heating the gas to the desired temperature.
Fluids like fluorescein-sodium solution or organics can be sprayed into the heated
gas stream via a two-phase nozzle (Schlick, Typ S4) driven with 2 bar and 1.2 l/h
fluid. Dry powders are suspended by an aerosol generator using a rotating brush.
For the exposure experiments AEOLA is operated at a volume flow of 500 Nm
3
/h
and a temperature of 80°C. Stability of total number and mass concentration of the
generated aerosol was controlled using an optical particle counter (scattered light
analyser PCS 2000, PALAS, Karlsruhe).
The aerosol samples are taken at the lower part of an 8 m high flow reactor
which produces constant and homogeneous aerosol flow. Afterwards the off gas
passes a bag filter and the blower before leaving to a stack.
For the bioassay the fine fraction of fly ash from a municipal waste incinerator
is dispersed in the upper part of the reactor as described above. The fly ash was
collected in a municipal waste incinerator and the fraction (MAF02) below 20 µm
was prepared by air separation (Fig. 31.2).
The chemical composition of this fly ash is determined by Total Reflection
X-Ray Fluorescence Analysis, ion chromatography and elemental analysis. Main
components are nearly 70% salts, metals and heavy metals and approximately 1%
carbon (Table 31.1). Birnbaum et al. (1996) reported a density similar to quartz of
about 2.2 g/cm
3
for complete fly ash. For the fine fraction below 20 µm we deter-
mined the density to 2.7 g/cm
3
by a helium pyknometer (Micromeritics, average of
10 measurements). By the same method the density of solid fluorescein sodium is
determined to 1.5 g/cm
3
.
Fig. 31.1 Schematic representation of the AEOLA aerosol laboratory

31 In Vitro Testing of Inhalable Fly Ash at the Air Liquid Interface 405
Fig. 31.2 Left: Scanning electron microscopy of a filter sample of fly ash. Right: Transmission
electron microscopy of the insoluble fraction of fly ash
Table 31.1 Main components of the fine fraction of fly ash in percent by weight
Al Mg Na Si K Ca Fe Cl S
1.2 0.5 7.3 3.6 12.0 6.4 0.9 6.5 6.0
31.2.2 Aerosol Measurement
The resuspended aerosol is described by its number size distribution (Fig. 31.3) that
is determined using a scanning mobility analyser SMPS 3071 (TSI). This instrument
is also used to determine the grade efficiency by measuring the size distribution
upstream and downstream of the exposure chamber (Fig. 31.4). The size distribu-
tions and the calculated grade efficiencies correspond to the gas composition of
mobility analyser and are not corrected for particle growth in humid air. At relative
humidity of 85% as used in this study this factor was determined to 1.3 in previous
experiments. Similar data are reported for sodium sulphate (Mätzing et al., 1996)
and for other combustion aerosols (Weingartner et al., 1995). For the scanning
electron microscopy Nuclepore filters are sampled according to VDI 2066 by
drawing the aerosol for a defined time at a defined volume flow through the filter
and correcting the volume to normal conditions.
31.2.3 Sampling System and Exposure
For the in vitro exposure of cultured pulmonary cell lines to ultra fine particles a
sampling system which simulates the human respiratory tract is required. The
exposure of the cell cultures takes place in the exposure unit CULTEX which is
integrated in the developed exposure system (Mülhopt et al., 2004a + b). The flow
chart is shown in Fig. 31.4.
406 S. Mülhopt et al.
Fig. 31.4 Flow chart of the exposure system installed in the AEOLA aerosol laboratory
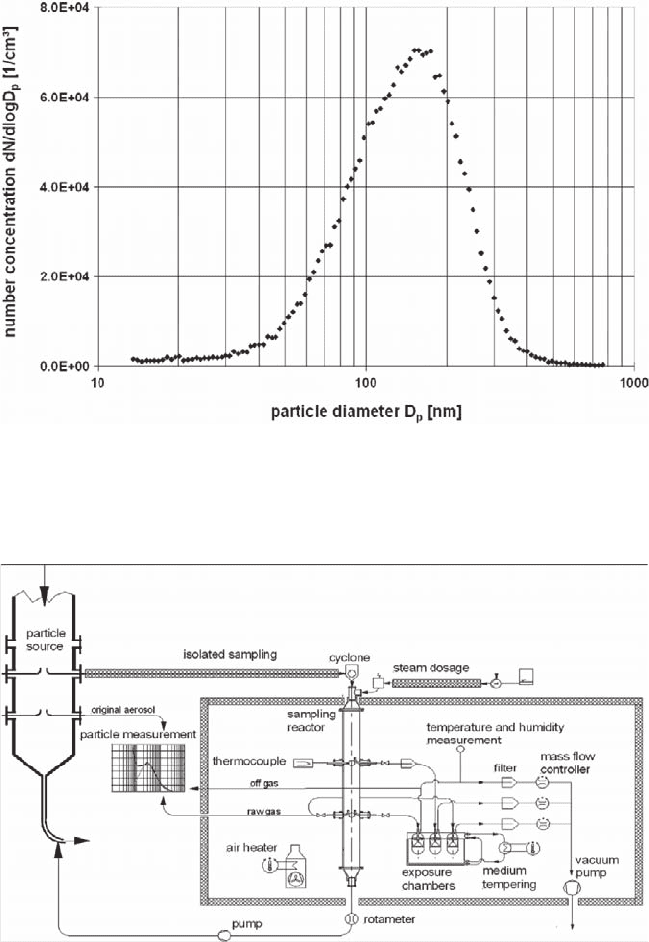
Fig. 31.3 Size distribution of the fine fraction of fly ash (MAF02) suspended in air, sampled from
AEOLA, and determined by the Scanning Mobility Particle Sizer SMPS (TSI), (X
modal
= 165 nm;
σ
g
= 1.65)

31 In Vitro Testing of Inhalable Fly Ash at the Air Liquid Interface 407
The main components in the sampling and exposure system are:
1. Sampling of the aerosol from the AEOLA reactor at a flow rate of 2 m
3
/h.
2. Removal of coarse particles by a cyclone (Stage 1 of the Sierra Stack 5 Cascade
Cyclone) with a cut off diameter of ∼1 µm. Thus, individual large particles
are removed that would contribute considerably to the deposited mass and there-
fore to the dosimetry. Additionally, large particles would probably disturb
the bioassay.
3. Humidification of the aerosol: To avoid drying of the cell cultures the aerosol is
humidified to conditions corresponding to the lower region of human lungs.
Therefore, water vapor is injected at a rate of 80 g/h into the humidification
reactor downstream of the cyclone to achieve a relative humidity of >85% at
37 ± 2°C.
4. Exposure of the lung cells: The exposure units CULTEX (Vitrocell, Gutach)
(Aufderheide et al., 2000) consist of an inlet system into which the aerosol flows,
and the lower unit that contains the cell cultures. The aerosol is directed via con-
centrical steel funnels into the center above the cell layer and directed to the
outside (Fig. 31.5). It leaves the exposure chamber and enters the off gas line. For
each cell culture an extra aerosol sample is drawn from the reactor. The advantage
of this separation of aerosol flows is to avoid fluctuations by linking the chambers
and particle losses due to dividing the flows. Each aerosol flow is regulated by its
own mass flow controller downstream of the exposure chambers. The separation
is also necessary to have the possibility of filtering one or two aerosol flows before
entering the exposure chamber so there are simultaneous exposures of cell cultures
to particles and to particle free tracer gas. The lower unit of the exposure system
accommodates the Transwell inserts (Corning, Wiesbaden) with the cell culture.
The cell cultures adhere to the membrane of Transwell-Clear inserts with a diam-
eter of 24 mm and 0.4 µm pores (catalogue number 3450). The cell layer is wetted
by the nutrient medium (RPMI 1640, Invitrogen, Karlsruhe) supplemented with
10 mM HEPES (4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid) from below.
The cell cultures used are kept at 37±2 °C by circulating warmed water in the
housing of the exposure unit.
Fig. 31.5 Left: Exposure chamber with three cell culture inserts. Right: 3D image of the aerosol
inlet above a Transwell membrane insert with cell culture
408 S. Mülhopt et al.
31.2.4 Determination of the Deposited Dose
To determine the deposited mass on the surface of a cell culture a dosimetric
procedure is developed. An aerosol of fluorescein-sodium is generated in the
AEOLA flow reactor as described above. The size fraction below 1 µm is separated
by the cyclone and the membrane surface without cells is exposed to the aerosol for
1 to 2 hours. The polyethylene membranes are cut out of the Transwell-clear inserts
by a scalpel and extracted with 10 ml of distilled water in an ultrasonic bath for
10 min at room temperature. Depending on the concentration, the solution is diluted
such that spectroscopy could be performed in the range of the calibration curve
with known concentrations of fluorescein sodium. The deposited mass is calculated
from the fluorescence intensity of the solution measured with a fluorescence
spectrometer (Aminco Bowman Series 2 Luminescence Spectrometer) at the wave-
length of 509 nm. The fluorescence intensity of fluorescein-sodium standards
exhibits a linear behaviour in the concentration range of 0.01 – 0.1 µg/ml.
31.2.5 Biotest
For the biotest, a co-culture system of BEAS-2B, immortalized cells from normal
human bronchial epithelium (ATCC, Rockville, MD) and differentiated THP-1
macrophages, was seeded onto porous Transwell-clear membranes. At the time of
the experiments approximately 1 × 10
6
cells covered the membrane surface area of
4.5 cm
2
. The confluent monolayer is exposed to filtered air or to aerosol at the air-
liquid interface having access to medium through the pores (0.4 µm in diameter).
These samples were compared to controls kept in the laboratory.
After a post-incubation period of 20 h, the medium is analyzed for lactate
dehydrogenase (LDH) (Roche Mannheim) and interleukin-8 (IL-8) release (ELISA kit
from BD Pharmingen, Heidelberg). The cells are tested for viability with alamarBlue
(Serotec, Düsseldorf), a reagent to determine the relative metabolic activity of the
cells (Diabaté et al., 2006). The values are reported as arithmetic mean values with
the error bars representing the standard error of the mean (s.e.m.) of independent
experiments with several samples each as indicated in the figure captions.
31.3 Results and Discussion
31.3.1 Characterization of the Exposure System
To get reproducible data of biological responses all details of the exposure system
were characterized. For this purpose we used the dosimetry method described
above to optimize and characterize the system for parameters like deposition
31 In Vitro Testing of Inhalable Fly Ash at the Air Liquid Interface 409
efficiency, reproducibility, deposited mass per membrane and grade efficiency for
the model aerosol of fluorescein-sodium particles.
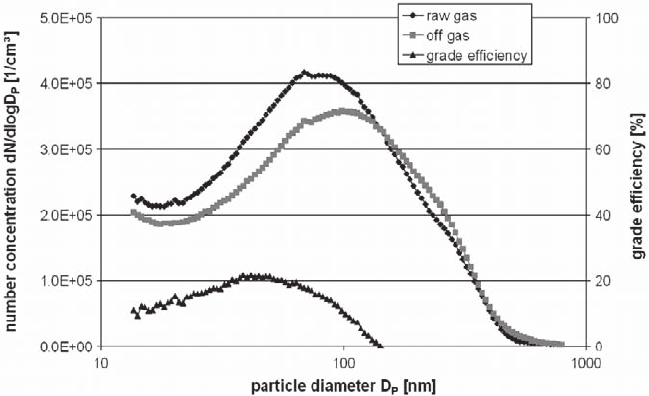
To describe the deposition behaviour of the whole exposure system the grade
efficiency is determined by measuring the size distribution upstream and down-
stream of the exposure chambers containing cell cultures with SMPS (Fig. 31.6).
Measurement of raw gas takes place in the reactor after cyclone and humidification,
the measurement of the clean gas was full flow after the CULTEX chamber. The
total number concentration amounts 4.1E + 05 ± 15% in the raw gas and 3.8E +
05 ± 30%in the off gas over the measurement range of 13 – 800 nm. That means
10% of the particles remain in the exposure system.
For determining the grade efficiency a flow rate of 300 ml/min is used because
of the defined aerosol inlet flow of the SMPS. We assume there is nearly no differ-
ence to the flow rate of 100 ml/min used for the exposure of cell cultures because
in both cases the gas velocities above the membrane are below 0.012 m/s. Out of
this and the low particle diameter the main deposition mechanism to expect above
the cell culture is diffusion. In the exhaust part of the exposure chamber small
diameters and sharp changes in flow direction may create other deposition mecha-
nism like impaction.
The deposition efficiency on single membranes is determined from the deposited
mass by fluorescence spectrometry as described above. For a 1 h exposure at
300 ml/min, the deposited mass was 3.9 µg per 24 mm membrane with a standard
deviation of 11%. This corresponds to 2.3% of the total mass that passed the
membrane.
Fig. 31.6 Mean size distribution and grade efficiencies for an exposure of cell cultures (A549
cells, r.h. 85%) to fluorescein-sodium particles, mean values, n(raw gas) = 5, standard deviation
20%, n(offgas) = 20, standard deviation 38%
410 S. Mülhopt et al.
In the results of the grade efficiency the influence of the geometry of the system
is shown. Losses in the off gas part of the exposure system and in the tubes of the
sampling is part of the grade efficiency. Due to this the deposition efficiency meas-
ured by the fluorescein-sodium dosimetry is much lower than the efficiency
calculated from the grade efficiency.
31.3.2 Exposure of Human Lung Cells Towards Fly Ash
For the exposure experiments with fly ash during the exposure of the cell cultures
a similar behaviour as determined for fluorescein sodium particles is observed. This
indicates that the dose on a mass basis was extremely low. While exposure
experiments the total number concentration amounts to 4 E + 04 ± 25% 1/cm
3
.
Based on the deposition rate of 2.3% of mass determined for fluorescein-sodium
the deposited mass of fly ash per membrane can be estimated. The basis for this
estimation is the similarity of the aerosols in structure (both are salts), size
distribution, number concentration and deposition behaviour. So the deposited mass
is approximately 0.09 µg per membrane and hour. Similar results have been
reported for the exposure of A549 cells to carbonaceous particles by Bitterle et al.
(2006). The 1-hour dose per unit of area of cell culture is in the range of 20 ng/
cm
2*
h. This concentration of 20 ng/cm
2
corresponds to about the 20 fold of a
24 hour-dose of PM
10
inhaled by an adult at a polluted urban site (100 µg/m
3
).
A co-culture of BEAS-2B cells with characteristics of the human bronchial
epithelium and THP-1 macrophages was used as target cells for the exposure
experiments. Prior studies revealed that its response to fly ash was most sensitive
compared to other cell lines. However, to detect an effect due to deposited fly ash
it is necessary to demonstrate that the cells did not suffer from the exposure
conditions themselves. Comparably to in vivo conditions, where bronchial epithe-
lial cells are directly exposed to inhaled air, it is of essential importance for the
viability of the cells not to dry out. Therefore, the evaporation of water by the gas
flow must be replaced by moistening through the membranes. The influence of
the volume flow is shown by passing filtered humidified air at different flow rates
over the cells.
The viability of the cells, which was indicated by reduction of the alamarBlue
reagent, is analysed 20 h after exposure. No difference is observed between the
submerged controls which are kept unchanged in the incubator and the control
cells which are kept at air-liquid conditions without flow in normal air at 37°C
(Fig. 31.7).
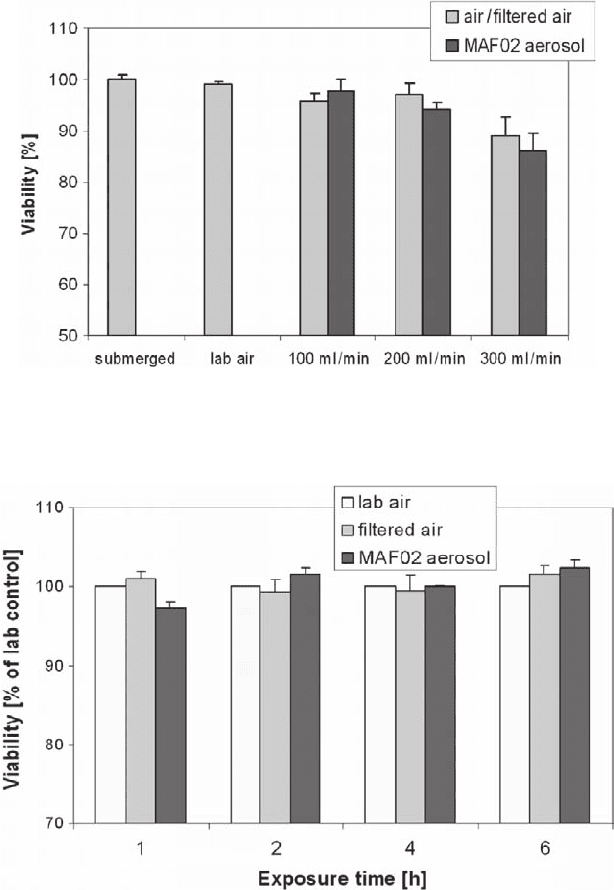
The viability of the cells which are transported to the AEOLA and exposed to
filtered air decreased with increasing flow rate. For exposure with aerosol we
decided to employ a flow rate of 100 ml/min per membrane. At 100 l/min, the
viability of the cells is not affected up to an exposure time of 6 h to filtered air or
MAF02 aerosol (Fig. 31.8). According to this for the toxicity tests the flow rate of
100 ml/min is used. As a marker for the inflammatory response the release of the
31 In Vitro Testing of Inhalable Fly Ash at the Air Liquid Interface 411
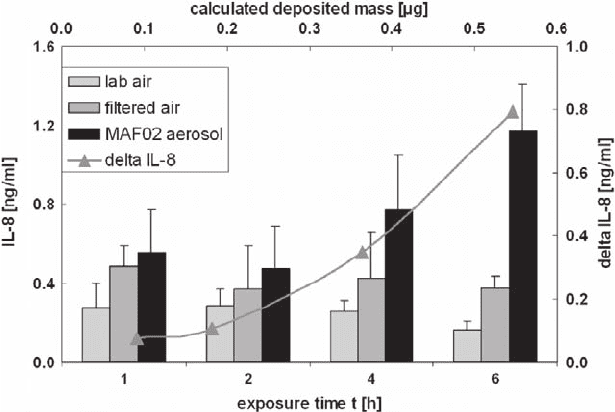
protein interleukin-8 is determined. The release of interleukin-8 increases in
dependence of the exposure time (Fig. 31.9). As the deposited mass of fly ash is
constant during the experiment this correlates with the dose.
Fig. 31.7 Viability of co-cultures of BEAS-2B and THP-1 cells exposed at the air-liquid interface
as a function of the flow rate (r.h. = 85%, t = 1 h, MAF02 = submicron fraction of fly ash)
Fig. 31.8 Viability of co-cultures of BEAS-2B and THP-1 cells as a function of the exposure time
(flow rate 100 ml/min per membrane, r.h. = 85%, MAF02 = submicron fraction of fly ash)
412 S. Mülhopt et al.
Fig. 31.9 Effects of exposure to filtered air and the submicron fraction of fly ash aerosol on
the IL-8 release of human lung cells (number of experiments n = 3 for t = 2, 4, 6 h and n = 2 for
t = 1 h, number of samples in each experiment m = 1 for lab air, m = 2 for filtered air, m = 3
for MAF02 aerosol) (co-culture of BEAS-2B with THP-1 cells, r.h. = 85%, particle number
concentration 3.58 E + 04 1/cm
3
) columns to left y-axis, curve to upper x-axis and right y-axis
31.4 Summary
An exposure system and a protocol was developed and characterized to measure the
dose response relationship between submicron particles and the inflammatory
response of human lung cells. The exposure system can be applied to measure-
ments up to six hours at industrial particle sources to assess the risk of submicron
particles.
The exposure system was applied for in vitro testing of submicron fly ash from
a municipal waste incinerator. The IL-8 release of lung cells exposed to this fly
ash increased by a factor of 4 compared to the controls. This demonstrates the
potential of submicron fly ash to trigger the inflammation of lung cells. IL-8 is a
key biomarker of lung cells exposed to airborne pollutants and an indicator of a
pro- inflammatory response which may contribute to lung inflammation (Steerenberg
et al., 1998; Monn et al., 1999; Diabaté et al., 2002). A similar exposure system
has also been employed by other research groups e.g. to study the effects of diesel
exhaust particles (Cheng et al., 2003) or metallic nanoparticles (Cheng 2004) on
the IL-8 release from A549 cells which have been exposed at the air-liquid
interface.
The advantage of aerosol exposure at the air-liquid layer compared to submerged
exposure is the direct contact of the aerosol with the target cells without further
31 In Vitro Testing of Inhalable Fly Ash at the Air Liquid Interface 413
treatment of the particles such as deposition on filters and resuspension. The results
of this study show that an in vitro exposure of pulmonary cells at the air-liquid
interface can be conducted by the way described under methods without loss of cell
viability. That means transport and exposure to low flow rates has no effect on the
viability of the cells.
This exposure technique can therefore be used for screening the toxicological
potential of unknown aerosols in order to identify potential emitters of health
relevant particles. Furthermore, new developments in the technology of the
combustion process and flue gas cleaning can directly be evaluated.
Acknowledgements We thank H. Fischer for taking the SEM images, F. Seidenstricker and M.
Hauser for useful help in the laboratory. The financial support of BWPLUS (Baden-Württemberg
Research Program Securing a Sustainable Living Environment) is appreciated.
References
Aufderheide M., and Moor U. (2000), CULTEX—An alternative technique for cultivation and
exposure of cells of the respiratory tract to airborne pollutants at the air/liquid interface. Exp.
Toxicol. Pathol., 52, 265–270
Aufderheide M. (2005), Direct exposure methods for testing native atmospheres. Exp. Toxicol.
Pathol., 57, Supplement 1, 213–226
Birnbaum L., Richers U., and Koeppel W. (1996), Untersuchung der physikalisch/chemischen
Eigenschaften von Filterstäuben aus Müllverbrennungsanlagen (MVA). Wissenschaftliche
Berichte/Forschungszentrum Karlsruhe, FZKA-5693
Bitterle E., Karg E., Schroeppel A., Kreyling W. G., Tippe A., Ferron G. A., Schmid O., Heyder
J., Maier K. L., and Hofer T. (2006), Dose-controlled exposure of A549 epithelial cells at the
air-liquid interface to airborne ultrafine carbonaceous particles. Chemosphere, 65, (10),
1784–1790.
Cheng M. D. (2004), Effects of nanophase materials (≤20 nm) on biological responses. J. Env. Sci.
Health - Part A Toxic/Hazard. Subst. Env. Eng., 39, (10), 2691–2705
Cheng M. D., Malone B., and Storey J. M. E. (2003), Monitoring cellular responses of engine-
emitted particles by using a direct air-cell interface deposition technique. Chemosphere,
53, 237–243.
Diabaté S., Mülhopt S., Paur H.-R., and Krug H. F. (2002), Pro-inflammatory effects in lung cells
after exposure to fly ash aerosol via the atmosphere or the liquid phase. Ann. Occup. Hyg.,
46, 382–385.
Diabaté S., Mülhopt S., Paur H.-R., and Krug H. F. (2007). Responses of human lung cells after
exposure to ultrafine particles of incinerator fly ash at the air-liquid interface, submitted
Mätzing H., Baumann W., and Paur H.-R. (1996), Bimodal aerosol coagulation with simultaneous
condensation/evaporation, J. Aerosol Sci., 27, Supplement 1, S363–S364
Monn C., and Becker S. (1999), Cytotoxicity and induction of proinflammatory cytokines from
human monocytes exposed to fine (PM2.5) and coarse particles (PM10–2.5) in outdoor and
indoor air. Toxicol. App. Pharmacol., 155, 245–252.
Mülhopt S., Seifert H., and Paur H.-R. (2004a, June), Exposure technique for a lung specific
bioassay for the assessment of industrial ultra fine particle-emissions. (Paper presented at the
7
th
International Conference on Nanostructured Materials, Wiesbaden; Germany)
Mülhopt S., Paur H-R., and Seifert H. (2004b), Expositionsverfahren für einen lungen- spezifischen
Bioassay zur Bewertung industrieller Feinstpartikel-Emissionen. BWPLUS Report 2004,
Retrieved from http://www.bwplus.fzk.de/berichte/SBer/BWB21018SBer.pdf
