Benton M.J. Introduction to paleobiology and the fossil record
Подождите немного. Документ загружается.


88 INTRODUCTION TO PALEOBIOLOGY AND THE FOSSIL RECORD
Trace fossil associations show that burrows
may be organized in an infaunal, tiered hier-
archy (see Chapter 19). Ausich and Bottjer
(1982) defi ned three levels with increasing
depth from the sediment–water interface: 0 to
−60 mm, −60 to −120 mm and −120 mm to
−1 m. During the earliest Paleozoic, only the
fi rst tier was consistently occupied, the second
tier was occupied from the Late Silurian and,
fi nally, the third tier was populated in the
Carboniferous. Tiering was also selectively
affected by extinction events, and tiers deeper
than 500 mm are rare after the Late Creta-
ceous because of predation by bony fi shes.
Trophic structures: bottom or
top of the food chain?
Food pyramids form the basis of most eco-
logical systems, defi ning the energy fl ow
through a chain of different organisms from
extremely abundant primary producers to
relatively few predators. A number of basic
trophic or feeding strategies are known (Fig.
4.10). Several marine food chains (basically,
who eats what) have been documented includ-
ing those dominated by suspension feeders
such as brachiopods, bryozoans and sponges.
These fed mainly on phytoplankton and other
organic detritus. Suspension feeding was par-
ticularly common in Paleozoic benthos; the
Mesozoic and Cenozoic faunas were more
dominated by detritus feeders, such as echi-
noids, and food chains were generally longer
and more complex (Fig. 4.11).
It might seem rather easy at fi rst sight to
reconstruct a food chain for a fossil assem-
blage, providing you can work out who ate
what. But that is easier said than done. One
of the most spectacular fossil lake deposits,
dominated by amphibians, has been docu-
mented from the Upper Carboniferous of
Seaweeds (brown, green algae)
Sea grasses, mangroves (angiosperms)
Epibenthic, sessile (bryozoans,
forams, spirorbids)
Crinozoans, octocorals
High-level suspension feeders
Intermediate-level suspension feeders
Low-level suspension feeders
25 cm
5 cm
Giant bivalves, corals, sponges,
giant brachiopods
Most brachiopods,
bivalves, bryozoans
Figure 4.9 Epifaunal tiering of marine benthic communities; infaunal tiering recorded in trace fossil
assemblages is discussed on p. 205. (From Copper 1988.)

1
Sediment surface
Sea surface
Primary
production
Phytoplankton
2
Herbivores
Primary
consumers
Plankton
and
nekton
Nektobenthos
Shallow
Deep
3
Deposit feeders
Primary/secondary
consumers
4
Suspension feeders
Primary/secondary
consumers
5
Carnivores
Epifauna
Infauna
Active
or passive
Life site and activity
Secondary/tertiary
consumers
(a)
(a)
(a)
(a)
(b)
(b)
(b)
(b)
(c)
(c)
(d)
(d)
(e)
bivalves
carnivores
bryozoans
brachiopods
rain of live plankton / organic detritus
forams
corals sponges
carnivoressuspension feederszoo / phytoplankton
(a)
(b)
placoderms
cephalopods
ostracodes
dead organic plankton detritus
trilobites
gastropods
asterozoans
organic detritus
labial palp
deposit feeders
carnivoresdeposit feeders
phyllocarids
polychaetes
Figure 4.10 Trophic groups, activity of members and their life sites. 1, Primary producers:
phytoplankton in surface waters with (a) cyanobacteria and (b) benthic algae. 2, Herbivores: browsing
and grazing gastropods. 3, Deposit feeders: (a) deposit-feeding gastropod and (b) shallow infaunal
bivalve. 4, Suspension feeders: (a) semi-infaunal, byssally-attached bivalve, (b) shallow infaunal bivalve,
(c) crinoid, (d) epifaunal bivalve, and (e) deep infaunal bivalve. 5, Carnivores: (a) nektonic fi shes, (b)
nekton-benthic fi shes, (c) epifaunal gastropod, and (d) infaunal gastropod. (From Brenchley & Harper
1998.)
Figure 4.11 Reconstructions of two different food chain communities. (a) A community with a
suspension-feeding food chain, displaying a variety of suspension feeders, collecting food in different
ways (bivalves with a mucous trap or setae, bryozoans and brachiopods with lophophores,
foraminiferans with cilia, corals with tentacles, and sponges with fl agellae). (b) A community with a
detritus-feeding food chain dominated by various types of bottom-dwelling deposit feeders and nektonic
carnivores represented by a cephalopod and placoderm. (From Copper 1988.)
90 INTRODUCTION TO PALEOBIOLOGY AND THE FOSSIL RECORD
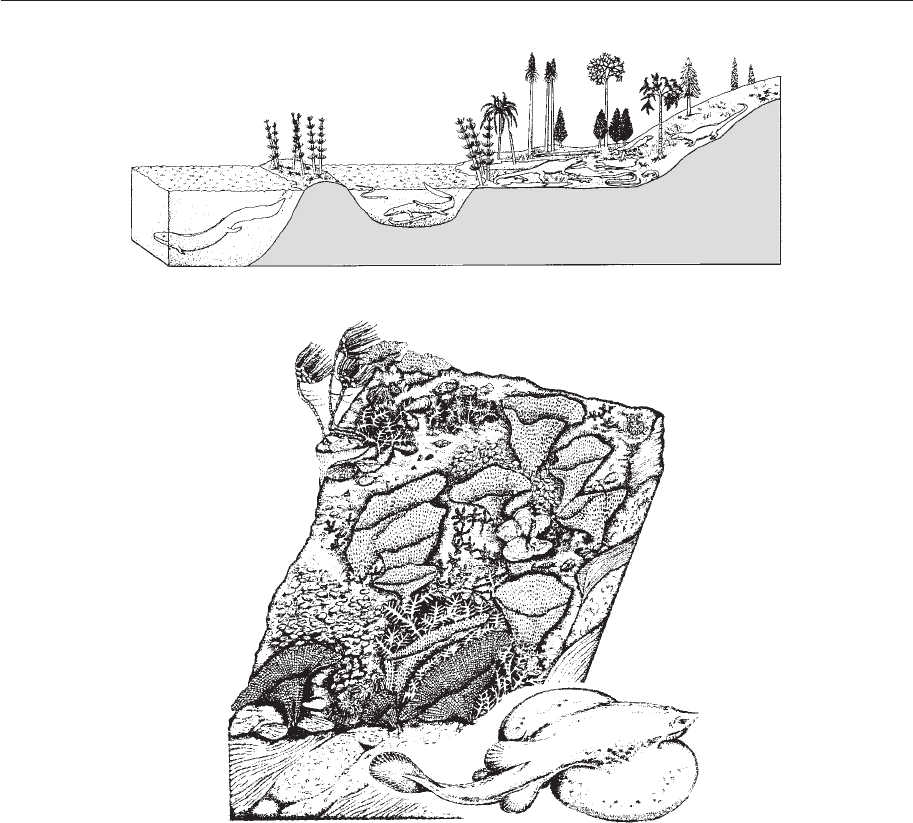
Czechoslovakia (Fig. 4.12a). The lake ecosys-
tem recreated for the inhabitants of the Ny´rˇany
Lake complex has three main ecological com-
munities: an open water and lake association,
dominated by fi shes together with various
larger amphibians; a shallow water and
swamp/lake association with amphibians,
small fi shes, land plants and other plant
debris; and fi nally a terrestrial–marginal asso-
ciation with microsaur (small, primitive)
amphibians and primitive reptiles. Food
chains have been worked out for each of these
associations by careful study of the teeth (was
it a herbivore with grinding teeth or a carni-
vore with slashing teeth?) of each beast, and
comparisons with modern relatives. For
example, in the open-water environments
fi shes, such as the spiny acanthodians, fed on
plankton but were themselves attacked by the
amphibians, presumably at the top of the
food chain. In the associated terrestrial envi-
ronments, plant material was consumed by a
variety of invertebrates, including insects,
millipedes, spiders, snails and worms; these
provided food and nutrients for a range of
small amphibians, themselves prey for larger
amphibians and reptiles.
A good example of a marine food web
comes from the Zechstein Reef facies of north-
ern Europe, dating from the Late Permian
(a)
upland
lake margin
shallow lake
open water
(b)
Figure 4.12 (a) Trophic structures in and around a Late Carboniferous lake complex, Ny´rˇany,
Czechoslavakia. (b) Trophic structures in a Late Permian reef complex, northeast England. (a, based on
Benton 1990; b, from Hollingworth & Pettigrew 1998.)
PALEOECOLOGY AND PALEOCLIMATES 91
(Fig. 4.12b). The Zechstein benthos was dom-
inated by diverse associations of brachiopods,
overshadowed in the higher tiers by fan- and
vase-shaped bryozoans (Hollingworth & Pet-
tigrew 1988). Both groups were sessile fi lter
feeders. Stalked echinoderms were rarer and
occupied the highest tiers. Mollusks such as
bivalves and gastropods were important
deposit feeders and grazers. One of the largest
predators was Janassa, a benthic ray, equipped
with a formidable battery of teeth capable of
crushing the shells of the sedentary benthos.
Megaguilds
Assignment of organisms to megaguilds pro-
vides another way to classify and understand
the components of a fossil community. Guilds
are groups of functionally similar organisms
occurring together in a community. Megagu-
ilds are simply a range of adaptive strategies
based on a combination of life position (e.g.
shallow, active, infaunal burrower) and
feeding type (e.g. suspension feeder). Some
paleontologists have used the term “guild”
for these categories; however, these were
probably fi ner ecological divisions within the
so-called Bambachian megaguilds, named
after the American paleontologist Richard
Bambach, who fi rst used the concept (Bambach
1983). Megaguilds have also become an effec-
tive tool in assessing long-term ecological
change (see p. 105).
Controlling factors
The ecological niche of an organism is deter-
mined by a huge range of limiting factors,
many of which are not recorded in the rock
record (Fig. 4.13). Key limiting factors for
marine organisms are light, oxygen levels,
temperature, salinity, depth and substrate
(Pickerill & Brenchley 1991).
Light is the main energy source for primary
producers, thus diatoms, dinofl agellates, coc-
coliths and cyanobacteria are dependent on
light and usually occupy the photic zone.
Most biological productivity occurs in the top
10–20 m of the water column. Virtually all
eukaryotic organisms require oxygen for their
metabolic processes, absorbing oxygen by dif-
fusion, in the case of small-bodied organisms,
or through gills or lungs in the case of the
larger metazoans. There is a well-developed
oxygen–depth profi le in the world’s seas and
oceans. Oxygen levels generally decrease
down to 100–500 m, where the amount of
oxygen absorbed by organic matter exceeds
primary oxygen production. Here in the
oxygen minimum zone (OMZ), the lowest
oxygen values are reached. The numbers of
many organisms, such as corals, echinoderms,
mollusks, polychaetes and sponges drop off
dramatically in the OMZ.
Levels of oxygen in marine environments
are important in determining who lives where.
Aerobic (normoxic) environments have
>1.0 ml L
−1
concentrations of oxygen, dys-
aerobic (hypoxic) environments have 0.1–
1.0 ml L
−1
and anaerobic (hypoxic-anoxic)
have <0.1 ml L
−1
. Although there is marked
decrease of biodiversity in oxygen-poor envi-
ronments, these environments encourage more
unusual adaptations such as the fl at shells of
the “paper pectens” (e.g. the genus Dun-
barella) and the compressed bodies of the fl at
worms; the increased surface areas of both
presumably helped the diffusion of oxygen.
Temperature is one of the most important
limiting factors. Most marine animals are poi-
kilotherms, having the same body tempera-
ture as their surroundings, and they live within
a temperature range of about −1.5 to 30°C.
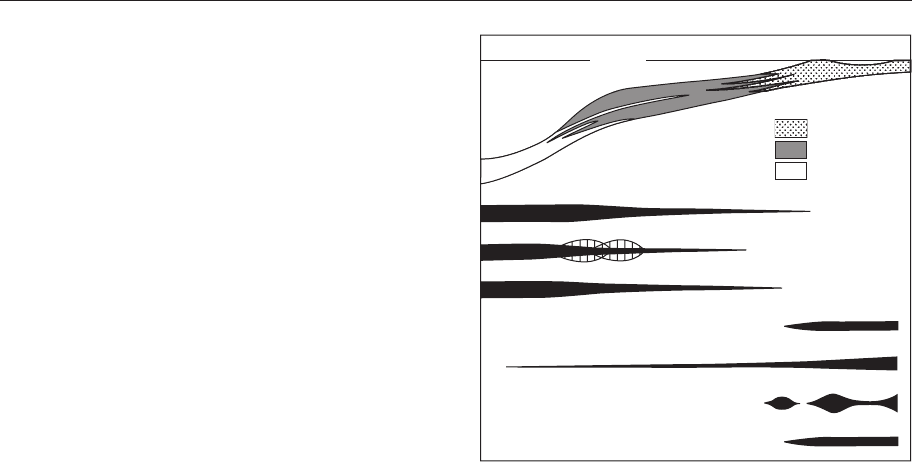
Sea level
sand
oxic mud
anoxic mud
Factors affecting benthos
light
oxygen
food
salinity
substrate
Substrate mobility
tidal shoals
turbidity
Figure 4.13 Shoreline to basin transect showing
the relative importance of different factors on
the distribution of organisms. (From Brenchley
& Harper 1998.)
92 INTRODUCTION TO PALEOBIOLOGY AND THE FOSSIL RECORD
Water temperature in the oceans decreases
steadily to the base of the thermocline, the
layer within a body of water where the tem-
perature changes rapidly, at around 1000 m
depth, where it reaches about 6°C. Tempera-
tures on the ocean fl oor rarely exceed more
than 2°C. Temperature also changes with lati-
tude and obviously affects the broad geo-
graphic distribution of organisms; those from
the poles are generally quite different from
those from the tropics.
Salinity, too, controls the distribution of
organisms. Most marine animals are isotonic
(“same salinity”) with seawater and live
within narrow (stenohaline) rather than wide
(euryhaline) ranges of salinity, commonly
with 30–40‰ dissolved salts in seawater. In
broad terms normal marine water is charac-
terized by stenohaline groups such as the
ammonites, belemnites, brachiopods, corals,
echinoderms and large benthic forams. Brack-
ish waters have mainly low-diversity assem-
blages with bivalves, crustaceans, ostracodes
and small benthic forams, whereas hypersa-
line assemblages are of very low diversity
with just a few bivalves, gastropods and
ostracodes.
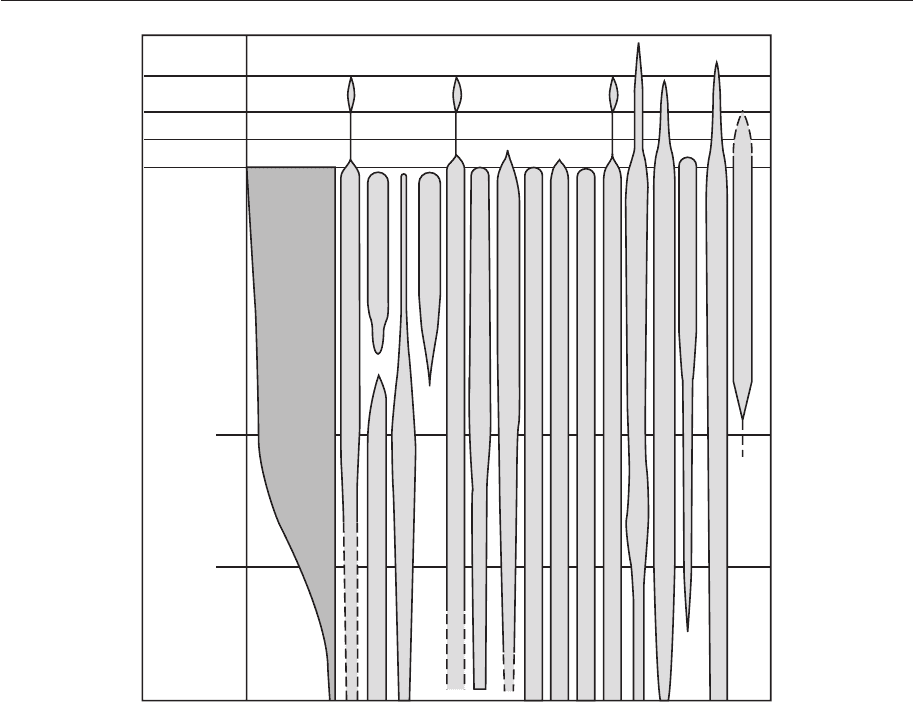
Depth is one of the most often quoted con-
trols on the distribution of marine organisms
(Fig. 4.14). Although the direct affects of
depth are related to hydrostatic pressure,
many other factors, both chemical and physi-
cal, are related to depth; for example, in
general terms, the grain size of sediment and
water temperature decreases with depth.
Although hydrostatic pressure does not
usually distort the shells and soft tissues of
organisms it can dramatically affect organ-
isms with pockets of gas in their bodies, such
as fi shes and nautiloids. Apart from the effects
of hydrostatic pressure, depth can also control
the solubility of calcium carbonate; cold water
2000 m
continental slope shelfabyssal
200 m
intertidal zone
supratidal zone
freshwater
dry land
demosponges
hexactinellids
calcisponges
ahermatypic corals
hermatypic corals
annelids with calcareous tubes
bryozoans
brachiopods
echinoderms
chitons and scaphopods
cephalopods
bivalves
gastropods
ostracodes
cirripedes
other crustacea
limulids
Figure 4.14 Distribution of living organisms across a depth gradient. (From Brenchley & Harper
1998.)

PALEOECOLOGY AND PALEOCLIMATES 93
contains more dissolved carbon dioxide
(CO
2
) providing a means to corrode carbon-
ate. At given depths in the world’s oceans,
carbonate material begins to dissolve at
so-called compensations depths. Below the
carbonate compensation depth (CCD) the
dissolution of calcium carbonate exceeds
supply and at about 4–5 km calcite is not
preserved. The depth is shallower for arago-
nite, with the aragonite compensation depth
(ACD) placed at 1–2 km. Both the CCD and
ACD vary with latitude, being shallower at
higher latitudes, and both parameters have
varied throughout geological time. Neverthe-
less, depth alone probably has little effect on
biotic distribution, rather the many depth-
related factors can be used to reconstruct
the water depths of ancient marine
communities.
Finally, the state of the substrate, rates of
sedimentation and turbidity dramatically
affect the distributions of benthic organisms
(Brenchley & Pickerill 1993). Organisms have
complex ecological requirements, some pre-
ferring a particular grain size, a certain type
of organic material or they even respond to
chemical signals (chemotaxic). There are also
complex taphonomic feedback processes,
where biogenic substrates such as shell
pavements can form attachment sites for new
communities. In general terms, within near-
shore environments, there is a broad correla-
tion between community distribution and
grain size. Diversity tends to be highest in
muddy sands, moderate in sandy muds,
low in pure sands and virtually zero in soft
muds. Moreover whether the sediments
form soupy muds, loose sands, fi rmgrounds
or hardgrounds will infl uence faunal
distributions.
Paleocommunities
Paleocommunities are recurrent groups of
organisms related to some specifi c set of envi-
ronmental conditions or limiting factors.
Many of the concepts and techniques applied
to marine fossil communities are based on the
work of biologists such as the Danish scientist
Carl Petersen, researching in the late 1800s
and early 1900s. Petersen recognized a series
of level-bottom benthic communities around
the Scandinavian coasts; the major control on
community distribution was water depth,
although other factors such as the substrate
were also infl uential.
Paleontologists were slow to adopt these
insights from modern marine biology. There
were a few pioneer studies on Carboniferous
assemblages in the 1930s, but it was the classic
work by Alfred Ziegler in the 1960s that
really brought these methods to the attention
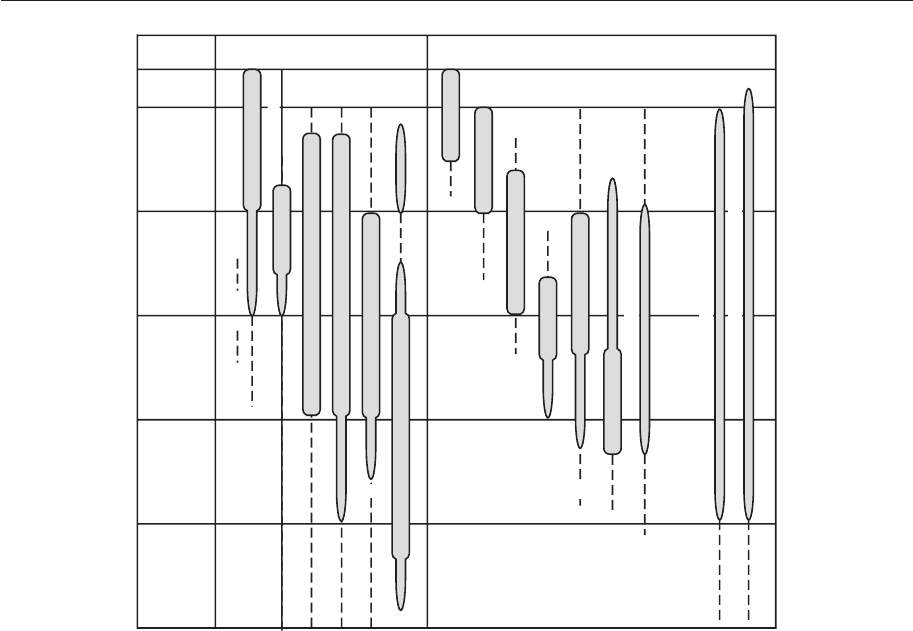
of paleontologists. He identifi ed fi ve depth-
related, brachiopod-dominated communities
in the Lower Silurian rocks of Wales and the
Welsh borderlands (see Chapter 12). These
communities stretched from the intertidal
zone in the east to the deep shelf and conti-
nental slope towards the west, matching per-
fectly the ancient paleogeography. This whole
system has been revamped and is now more
widely known as the benthic assemblage zones
(Fig. 4.15). These zones are defi ned on a wide
range of faunal and sedimentological criteria
and may be subdivided, internally, on the
basis of, for example, substrate type and the
degree of turbulence (Brett et al. 1993).
Describing fossil communities
Sometimes the simplest jobs are the hardest
to do properly. For over a hundred years,
paleontologists have provided lists of species
from particular localities, but these are not
helpful for ecological work unless the relative
abundances of the different species are docu-
mented as well. We need to know which
species dominate (sometimes one species
makes up more than 50% of the sample) and
which are rare (i.e. less than 5% of the col-
lection). Now it is more common to docu-
ment the absolute and relative abundance of
each organism, illustrated graphically with
frequency histograms, and based on data
derived from line transects, quadrats or more
commonly now from bed-by-bed collecting or
bulk samples.
Counting conventions remain a problem.
With many organisms it is relatively simple to
calculate how many individuals were actually
represented in a given assemblage: univalved
species (e.g. gastropods) count as one, whereas
twin-valved species (e.g. bivalves and brachio-
pods) may be assessed by adding the most
common valve (right or left, dorsal or ventral)
to the number of articulated or conjoined
shells. Animals that molt, such as ostracods
and trilobites, colonial organisms and those
94 INTRODUCTION TO PALEOBIOLOGY AND THE FOSSIL RECORD
that easily fragment (e.g. bryozoans) and
those with multi-element skeletons (e.g. cri-
noids and vertebrates) require more special-
ized counting techniques. These basic data are
then transformed into a more realistic picture
of ancient communities populating past land-
scapes and seascapes, through histograms and
pie charts. Raw numerical data are extremely
useful, but these can also be converted to
diversity, dominance and evenness parame-
ters, and parameters for taxonomic distinc-
tiveness (Box 4.2). These together can give
us a rich overview of the composition and
structure of the paleocommunity and allow
numerical comparisons with other similar
assemblages. Such approaches have become
routine in studies of invertebrate paleoecology
but it is much more diffi cult to apply these
methods to vertebrate assemblages where
sample sizes are generally much smaller.
Detailed analysis of paleocommunity struc-
tures has permitted recognition of a number
of specifi c community types. Pioneer commu-
nities are those that have just entered new
ecospace, and they may be dominated by one
or two very abundant opportunistic species,
in contrast to long-established and rather
stable equilibrium communities where rela-
tively high diversities of more or less equally
abundant animals are present. The ecological
relationships between organisms is also an
important aspect of community development
(Box 4.3).
Paleocommunity development through time
Communities undoubtedly change with time.
Factors such as environmental fl uctuation,
immigration and emigration of animals and
plants, evolution and extinction of species
and coevolutionary changes will alter the
composition and structure of a community.
But are the components of communities tightly
linked and thus evolve together or is it a rather
haphazard random process? Living communi-
ties, when fi rst established, show initial high
rates of replacement and instability, whereas
later stages are more stable with little change,
Benthic
assemblage
Wave
base
Storm evidence Photic zone evidence
BA–1
BA–2
BA–3
BA–4
BA–5
BA–6
wave ripples
amalgamated beds
gutter casts
winnowed coquinites
hummocky cross stratification
?
mud tempestites
stromatolites
thrombolites
tabulate-stromatolite reefs
small bioherms
receptaculitids
green algal borings
blue-green algal borings
trilobites with well developed eyes
cyclocrinitids
(large)
(small)
Figure 4.15 Silurian marine benthic assemblage zones and identifying criteria. (From Brenchley &
Harper 1998.)

PALEOECOLOGY AND PALEOCLIMATES 95
Box 4.2 Ecological statistics and sampling suffi ciency: are you getting
enough?
It is often diffi cult to assess the adequacy of a paleoecological sample. Some authorities have sug-
gested that samples of about 300 give a fairly accurate census of a fossil assemblage. Commonly,
investigators plot rarefaction curves (Fig. 4.16). These are produced simply by collecting samples of
10 and identifying the number of species in each. For each sample of 10 plotted along the x-axis,
the cumulative number of species is plotted along the y-axis. The curve may level off at the point
where no additional species are identifi ed with additional collecting and this fi xes the sample size
that is adequate to count the majority of species present (Fig. 4.16).
A range of statistics has been used to describe aspects of fossil communities. Although the number
of species collected from an assemblage provides a rough guide to the diversity of the association,
obviously in most cases the larger the sample, the higher the diversity. Diversity measures are usually
standardized against the sample size. Dominance measures have high values for communities with
a few abundant elements and low values where species are more or less evenly represented; measures
of evenness are usually the inverse of dominance.
Margalef diversity =−SN1/log
Dominance = ( / )nN
i
2
∑
Evenness = 1/ ( )p
i
2
∑
where S is the number of species, N is the number of specimens, n
i
is the number of the ith species,
and p
i
is the relative frequency of ith species.
Many numerical techniques have been used to analyze paleocommunities and their distributions.
Phenetic methods (see Chapter 2) are based on the investigation of a similarity or distance matrix
0 10 100 1000
No. of specimes collected
in batches of ten
Cumulative number of taxa
25
(a) (b) (c)
(d)
(e) (f)
Figure 4.16 Construction of a rarefaction curve based on data collected from a mid-Devonian
brachiopod-dominated fauna, northern France. The main types of brachiopod are illustrated: (a)
Schizophoria, (b) Douvillina, (c) Productella, (d) Cyrtospirifer, (e) Rhipidiorhynchus, and (f)
Athyris. The curve levels off at about 300 specimens, suggesting this sample size is a suffi cient
census of the fauna. Magnifi cation approximately ×0.5 for all.
Continued

96 INTRODUCTION TO PALEOBIOLOGY AND THE FOSSIL RECORD
derived from a raw data matrix of the presence or absence or numerical abundance of fossils at each
site. Cluster analysis is most commonly used in ecological studies and there is a wide range of both
distance and similarity measures, together with clustering techniques, to choose from. R-mode analy-
sis clusters the variables, in most paleoecological studies the taxa, whereas Q-mode analysis clusters
the cases, usually the localities or assemblages (Fig. 4.17).
For example, Late Ordovician brachiopod-dominated assemblages from South China have been
investigated by cluster analyses (Hammer & Harper 2005) and fall into a number of ecogroups.
These data are available at http://www.blackwellpublishing.com/paleobiology/.
Leptostro
Streptola
Clorinda
Glassia
Aegiria
Pentameru
Eoplectod
Atrypa
Stricklan
Tentaculi
Eocoelia
Bivalve
Stegerhyn
Lingula
Clorinda
Pentamerus
Stricklandia
Eocoelia
Lingula
1
0
1
1
0
1
1
1
1
0
1
0
0
0
0
1
0
0
0
0
1
0
0
0
0
0
1
0
1
0
1
1
0
1
0
1
1
1
0
0
0
1
1
0
0
1
1
1
1
1
1
1
1
1
1
0
1
1
1
1
0
1
1
1
1
0
0
0
1
1
Figure 4.17 In a two-way cluster analysis, an R-mode clusters the genera (bottom) and a Q-mode
clusters the community type (right). The original data matrix is in the center of the diagram. The
data indicate the reality of a shallow-water biofacies (Lingula and Eocoelia communities), and
mid to deep shelf (Pentamerus and Stricklandia communities) and outer shelf to slope (Clorinda
community) assemblages.
building up to a climax community in equi-
librium with its environment. There is still
some discussion among ecologists about
whether communities conform to Eltonian
models of change (predictable over long
periods of time), Gleasonian models (short-
term, rapid change and instability) or perhaps
even both. Evidence from Quaternary, mainly
Holocene, communities suggests them to be
rather ephemeral (Davis et al. 2005). Species
may evolve, become extinct or migrate out of
the immediate area during intervals of climate
change thus destroying the community struc-
ture. They may, however, return and recom-
bine to form the original communities during
intervals of more favorable climate (Bennett
1997). Nevertheless, paleocommunities domi-
nated by incumbent taxa such as the dino-
saurs during the Jurassic and Cretaceous or
pentameride brachiopods during the Silurian

PALEOECOLOGY AND PALEOCLIMATES 97
Box 4.3 Ecological interactions
Animals and plants have participated in a wide range of relationships throughout geological time.
Ecologists have classifi ed these arrangements in terms of gain (+), loss (−) and neutrality (0). Antago-
nistic arrangements include antibiosis (−,0), exploitation (0,+) and competition (0,0) whereas sym-
biosis involves both commensalism (+,0) and mutualism (+,+).
Antibiosis is diffi cult to demonstrate although mass mortalities of fi shes have been
ascribed to dinofl agellate blooms. Some paleontologists believe that the twisted skeleton of a
Late Cretaceous Struthiomimus from Alberta may show the animal died from strychnine
poisoning.
Exploitation includes predation and parasitism. There are many records of bite marks, particularly
by marine reptiles on mollusk shells, while the stomach contents of Jurassic ichthyosaurs have
revealed a diet of belemnites. Moreover a wide variety of nibble marks have been reported from
fossil leaves. The relationship between the Devonian tabulate coral Pleurodictyum and the worm
Hicetes fooled many paleontologists. Was this a bizarre compound organism? In fact the worm was
probably a parasite; the association is common throughout Europe and virtually every specimen of
Pleurodictyum has a parasitic worm at its core.
Competition is often diffi cult to observe directly in the fossil record. Encrusting bryozoans,
however, commonly compete for space and food resources on the seabed. Competition between the
cyclostome and cheilostome clades (see Chapter 12) may have infl uenced the post-Paleozoic history
of the phylum in favor of the latter. Encrusting bryozoans can also faithfully replicate their substrate,
recording the imprint of a soft-bodied animal or aragonitic mollusk. This process of bioimmuration
(“biological burial”) is a useful means of preserving an organism that otherwise may have escaped
detection.
Commensalism is one of the most common relationships apparent in the fossil record, where small
epifauna or epibionts use larger organisms for attachment and support. Small and immature pro-
ductoid brachiopods are often attached by clasping spines to crinoid stems while microconchids,
previously thought to be Spirobis worms (see Chapter 12), are commonly attached near the exhalent
currents of Carboniferous non-marine bivalves. Some of the most spectacular examples have been
reported from the shells of Devonian spiriferide brachiopods. Derek Ager (University of Wales,
Swansea) reported a succession of epifauna, commencing with Spirobis (microconchids) followed by
Hederella and Paleschara and fi nally the tabulate coral Aulopora, clustered near the inhalent current
of the brachiopod (Fig. 4.18).
Spirorbis sp.
Hederella filiformis
Paleschara incrustans
Aulopora elleri
(b)(a)
Figure 4.18 Commensalism between (a) the gastropod Platyceras and a Devonian crinoid and (b)
Spinocyrtia iowensis with an epifauna primarily located on the fold of the brachial valve adjacent
to inhalant or exhalent currents. (Based on Ager 1963.)
