Ahmed S.N. Physics and Engineering of Radiation Detection
Подождите немного. Документ загружается.

16 Chapter 1. Properties and Sources of Radiation
versus time in such a case will deviate from a straight line of single isotopes. The
best way to understand this is by assuming that the composite material has one
effective decay constant. But this decay constant will have time dependence since as
time passes the sample runs out of the short lived isotope. Hence equations 1.3.20
and 1.3.21 will not be linear any more.
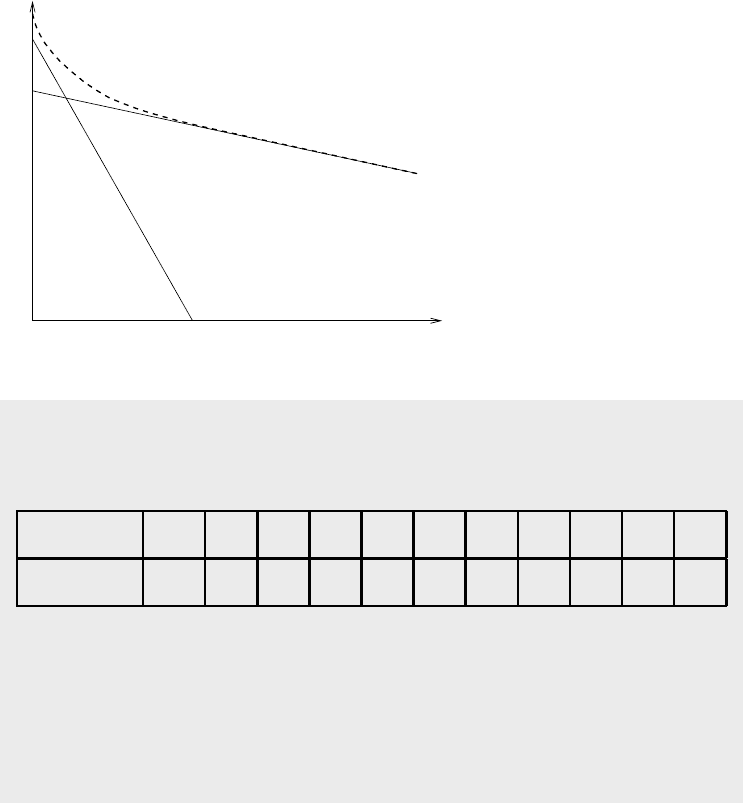
Figure 1.3.3 shows the activity plot of a composite radioactive material. Since
we know that each individual isotope should in fact yield a straight line therefore
we can extrapolate the linear portion of the graph backwards to get the straight
line for the isotope with longer half life. We can do this because the linear portion
shows that the shorter lived isotope has fully decayed and the sample now essentially
contains only one radioactive isotope. Then the straight line for the other isotope
can be determined by subtracting the total activity from the activity of the long
lived component.
ln(A)
Tim
e
Slow decaying component
Fast decaying component
Measured activity
Figure 1.3.3: Experimental de-
termination of decay constants
of two nuclides in a composite
decaying material.
Example:
The following table gives the measured activity in counts of a composite ra-
dioactive sample with respect to time. Assuming that the sample contains two
radioactive isotopes, compute their decay constants and half lives.
t (min) 0 30 60 90 120 150 180 210 240 270 300
A (cts/min) 2163 902 455 298 225 183 162 145 133 120 110
Solution:
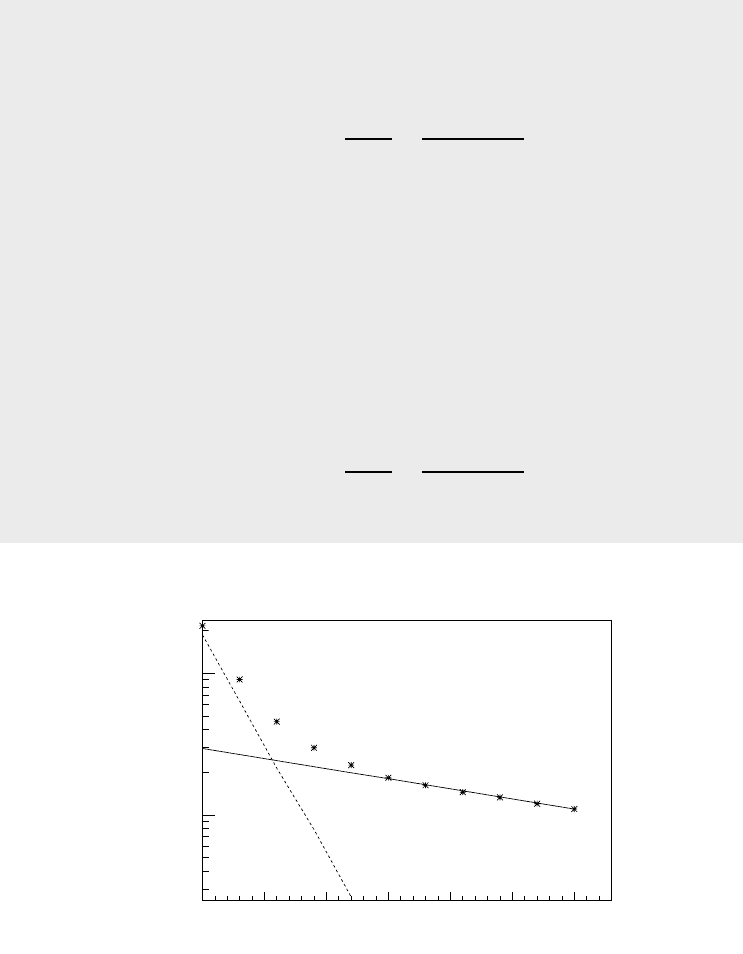
Following the procedure outlined in this section, we plot the activity as a
function of time on a semilogarithmic graph (see Fig.1.3.4). It is apparent from
theplotthataftert = 120 minutes ln(A) varies linearly with time. Using least
square fitting algorithm we fit a straight line through points between t = 150
and t = 300 minutes. The equation is found to be
ln(A)=−3.28 × 10
−3
t +5.68.
1.3. Radioactivity and Radioactive Decay 17
This straight line represents the activity of the long lived component in the
sample. Its slope gives the decay constant of the long lived component, which
can then be used to obtain the half life. Hence we get
λ
1
=3.28 × 10
−3
min
−1
⇒ T
1/2,1
=
0.693
λ
1
=
0.693
3.28 × 10
−3
= 211.3days.
To obtain the decay constant of the short lived component, we extrapolate
the straight line obtained for the long lived component up to t =0andthen
subtract it from the observed data (see Fig.1.3.4). The straight line thus
obtained is given by
ln(A)=−3.55 × 10
−2
t +7.53.
The slope of this line gives the decay constant of the second isotope, which
can then be used to determine its half life. Hence we have
λ
2
=3.55 × 10
−2
min
−1
⇒ T
1/2,2
=
0.693
λ
1
=
0.693
3.55 × 10
−2
=19.5days.
Time (min)
0 50 100 150 200 250 300
Counts
2
10
3
10
Figure 1.3.4: Determination of decay parameters of two
nuclides from observed effective activity. The actual data
are represented by (*). The solid and dashed lines represent
the long lived and short lived components respectively.

18 Chapter 1. Properties and Sources of Radiation
1.3.D Radioactive Chain
We saw earlier that when a radionuclide decays, it may change into another element
or another isotope. This new daughter radionuclide may as well be unstable and
radioactive. The decay mode and half life of the daughter may also be different
from the parent. Let us see how our radioactive decay equations can be modified for
such a situation.
I will start with a sample composed of a parent and a daughter radionuclide.
There will be two processes happening at the same time: production of daughter
(or decay of parent) and decay of daughter. The net rate of decay of the daughter
will then be the difference of these two rates, that is
dN
D
dt
= λ
dP
N
P
− λ
dD
N
D
, (1.3.24)
where subscripts P and D represent parent and daughter respectively.
Using N
P
= N
0P
e
−λ
dP
t
, this equation can be written as
dN
D
dt
+ λ
dD
N
D
− λ
dP
N
0P
e
−λ
dP
t
=0. (1.3.25)
Solution of this first order linear differential equation is
N
D
=
λ
dP
λ
dD
− λ
dP
N
0P
e
−λ
dP
t
− e
−λ
dD
t
+ N
0D
e
−λ
dD
t
. (1.3.26)
Here N
0P
and N
0D
are the initial number of parent and daughter nuclides respec-
tively. In terms of activity A(= λN), the above solution can be written as
A
D
=
λ
dD
λ
dD
− λ
dP
A
0P
e
−λ
dP
t
− e
−λ
dD
t
+ A
0D
e
−λ
dD
t
. (1.3.27)
Equations 1.3.26 and 1.3.27 have decay as well as growth components, as one would
expect. It is apparent from this equation that the way a particular material decays
depends on the half lives (or decay constants) of both the parent and the daughter
nuclides. Let us now use equation 1.3.27 to see how the activity of a freshly pre-
pared radioactive sample would change with time. In such a material, the initial
concentration and activity of daughter nuclide will be zero N
0D
=0,A
0D
=0. This
condition reduces equation 1.3.26 to
A
D
=
λ
dD
λ
dD
− λ
dP
A
0P
e
−λ
dP
t
− e
−λ
dD
t
. (1.3.28)
The first term in parenthesis on the right side of this equation signifies the buildup
of daughter due to decay of parent while the second term represents the decay of
daughter. This implies that the activity of the daughter increases with time and,
after reaching a maximum, ultimately decreases (see figure 1.3.5). This point of
maximum daughter activity t
max
D
can be easily determined by requiring
dA
D
dt
=0.
Applying this condition to equation 1.3.28 gives
t
max
D
=
ln(λ
dD
/λ
dP
)
λ
dD
− λ
dP
. (1.3.29)

1.3. Radioactivity and Radioactive Decay 19
Activity
max
Tim
e
A
P
t
A
D
D
Figure 1.3.5: Typical parent
and daughter nuclide activities.
Example:
Derive the relation for the time behavior of buildup of a stable nuclide from
a radioactive element.
Solution:
Assuming the initial concentration of daughter to be zero (N
0D
= 0), equation
1.3.26 can be used to determine the concentration of daughter nuclide at time
t.
N
D
=
λ
dP
λ
dD
− λ
dP
N
0P
e
−λ
dP
t
− e
−λ
dD
t
Since the daughter nuclide is stable therefore we can substitute λ
dD
=0in
the above equation to get the required relation.
N
D
= N
0P
1 − e
−λ
dP
t
In most cases the radioactive decay process does not stop at the decay of the
daughter nuclide as depicted by equation 1.3.26. Instead the nuclides continue to
decay into other unstable nuclides until a stable state is reached. Assuming that the
initial concentrations of all the nuclides except for the parent is zero, equation 1.3.28
can be generalized for a material that undergoes several decays. The generalization
was first done by Bateman in 1910 (8). The Bateman equation for the concentration
of i
th
radionuclide is
N
i
(t)=λ
d1
λ
d2
.....λ
d(i−1)
N
01
i
j=1
e
−λ
dj
t
k=1,k=j
(λ
dk
− λ
dj
)
, (1.3.30)
provided N
0i
=0fori>1. In terms of activity the Bateman equation can be
written as
A
i
(t)=λ
d2
.....λ
di
A
01
i
j=1
e
−λ
dj
t
k=1,k=j
(λ
dk
− λ
dj
)
. (1.3.31)

20 Chapter 1. Properties and Sources of Radiation
Example:
A50µCi radioactive sample of pure
222
88
Rn goes through the following series
of decays.
222
86
Rn (T
1/2
=3.82 days ) →
218
84
Po (T
1/2
=3.05 min ) →
214
82
Pb (T
1/2
=26.8
min ) →
214
83
Bi (T
1/2
=19.7min)
Compute the activity of its decay products after 3 hours.
Solution:
Activity of
222
86
Rn
The decay constant of
222
86
Rn can be calculated from its half life as follows.
λ
d1
=
ln(2)
T
1/2
=
0.693
3.82 × 24 × 60
=1.26 × 10
−4
min
−1
Since we have a pure sample of radon-222 therefore its activity after 3 hours
can be calculated from equation 1.3.16.
A
1
= A
01
e
−λ
d1
t
=50
e
−1.26×10
−4
×3×60
=48.88 µCi
Activity of
218
84
Po
The decay constant of
218
84
Po is
λ
d2
=
ln(2)
T
1/2
=
0.693
3.05
=0.227 min
−1
.
Since polonium-218 is the first daughter down the radioactive chain of radon-
222, we use i = 2 in Bateman equation 1.3.31 to get
A
2
= λ
d2
A
01
e
−λ
d1
t
(λ
d2
− λ
d1
)
+
e
−λ
d2
t
(λ
d1
− λ
d2
)
=0.227 × 50
e
−1.26×10
−4
×3×60
(0.227 − 1.26 ×10
−4
)
+
e
−0.227×3×60
(1.26 ×10
−4
− 0.227)
=48.9 µCi
A point worth noting here is that the second term in the parenthesis on the
right side of the above equation is negligible as compared to the first term
and could have safely been omitted from the calculations.
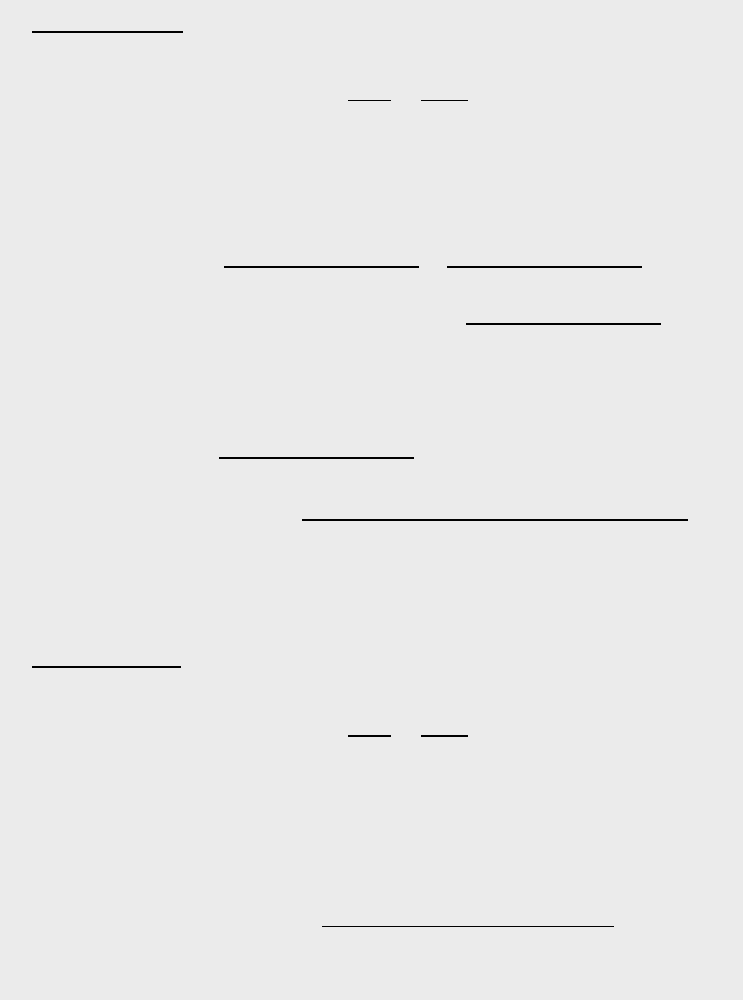
1.3. Radioactivity and Radioactive Decay 21
Activity of
214
82
Pb
The decay constant of
214
82
Pb is
λ
d3
=
ln(2)
T
1/2
=
0.693
26.8
=0.0258 min
−1
.
To calculate the activity of this isotope of lead after 3 hours we use i =3in
Bateman equation 1.3.31.
A
3
= λ
d2
λ
d3
A
01
e
−λ
d1
t
(λ
d2
− λ
d1
)(λ
d3
− λ
d1
)
+
e
−λ
d2
t
(λ
d1
− λ
d2
)(λ
d3
− λ
d2
)
+
λ
d2
λ
d3
A
01
e
−λ
d3
t
(λ
d1
− λ
d3
)(λ
d2
− λ
d3
)
Due to high decay constants of
218
84
Po and
214
82
Pb the second and third terms
on right hand side can be neglected. Hence
A
3
≈ λ
d2
λ
d3
A
01
e
−λ
d1
t
(λ
d2
− λ
d1
)(λ
d3
− λ
d1
)
=0.227 × 0.0258 ×50
e
−1.26×10
−4
×3×60
(0.227 −1.26 ×10
−4
)(0.0258 − 1.26 × 10
−4
)
=49.14 µCi.
Activity of
214
83
Bi
The decay constant of
214
83
Bi is
λ
d4
=
ln(2)
T
1/2
=
0.693
19.7
=0.0352 min
−1
.
The Bateman’s equation for i = 4 will also contain negligible exponential
terms as we saw in the previous case. Hence we can approximate the activity
of
214
83
Bi after 3 hours by
A
4
≈ λ
d2
λ
d3
λ
d4
A
01
e
−λ
d1
t
(λ
d2
− λ
d1
)(λ
d3
− λ
d1
)(λ
d4
−λ
d1
)
=49.32 µCi.

22 Chapter 1. Properties and Sources of Radiation
1.3.E Decay Equilibrium
Depending on the difference between the decay constants of parent and daughter
nuclides it is possible that after some time their activities reach a state of equilib-
rium. Essentially there are three different scenarios leading to different long term
states of the radioactive material. These are termed as secular equilibrium, tran-
sient equilibrium,andno equilibrium states. For the discussion in this section we
will assume a radioactive material that has a parent and a daughter only. However
the assertions will also be valid for materials that go through a number of decays.
E.1 Secular Equilibrium
If the activity of parent becomes equal to that of the daughter, the two nuclides are
said to be in secular equilibrium. This happens if the half life of parent is much
greater than that of the daughter, i.e.
T
P
1/2
T
D
1/2
or λ
dP
λ
dD
Let us see if we can derive the condition of equal activity from equation 1.3.28. It
is apparent that if λ
dP
>λ
dD
then as t →∞
e
−λ
dD
t
e
−λ
dP
t
,
and hence we can neglect the second term on right side of equation 1.3.28. The
daughter activity in this case is given approximately by
A
D
λ
dD
λ
dD
− λ
dP
A
0P
e
−λ
dP
t
(1.3.32)
=
λ
dD
λ
dD
− λ
dP
A
P
Or
A
P
A
D
1 −
λ
dP
λ
dD
. (1.3.33)
As λ
dD
λ
dP
we can neglect second term on right hand side of this equation.
Hence
A
P
A
D
This shows that if the half life of parent is far greater than that of the daughter then
the material eventually reaches a state of secular equilibrium in which the activities
of parent and daughter are almost equal. The behavior of such a material with
respect to time is depicted in Fig.1.3.6.
An example of a material that reaches secular equilibrium is
237
93
Np. Neptunium-
237 decays into protactinium-233 through α-decay with a half life of about 2.14×10
6
years. Protactinium-233 undergoes β-decay with a half life of about 27 days.
Example:
How long would it take for protactanium-233 to reach secular equilibrium
with its parent neptunium-237?

1.3. Radioactivity and Radioactive Decay 23
Activity
A
P
A
D
T
P
1/2
>>
T
D
1/2
Tim
e
Figure 1.3.6: Activities of par-
ent and daughter nuclides as a
function of time for a material
that eventually reaches the state
of secular equilibrium. The half
life of parent in such a material
is so large that it can be consid-
ered stable.
Solution:
Since the half life of the parent is much larger than that of the daughter,
therefore we can safely assume that at the state of secular equilibrium the
activity of the daughter will be nearly equal to the initial activity of the
parent, that is.
A
D
A
0P
.
Substitution of this equality in equation 1.3.32 gives
λ
dD
λ
dD
− λ
dP
e
−λ
dP
t
1
⇒ t
1
λ
dP
ln
λ
dD
λ
dD
− λ
dP
.
The decay constants of the two materials are
λ
dP
=
ln(2)
T
P
1/2
=
ln(2)
2.14 × 10
6
× 365
=8.87 × 10
−10
day
−1
λ
dD
=
ln(2)
T
D
1/2
=
ln(2)
27
=2.57 × 10
−2
day
−1
Hence the required time is given by
t
1
8.87 × 10
−10
ln
2.57 × 10
−2
2.57 × 10
−2
− 8.87 × 10
−10
.
=38.9days

24 Chapter 1. Properties and Sources of Radiation
Interestingly enough, this is exactly the mean life of protactanium-233 and
it shows that after about one mean life of the daughter its activity becomes
approximately equal to that of the parent.
E.2 Transient Equilibrium
The parent and daughter nuclides can also exist in a transient state of equilibrium in
which their activities are not equal but differ by a constant fraction. This happens
when the half life of the parent is only slightly higher than that of the daughter, i.e.
T
P
1/2
>T
D
1/2
or λ
dP
<λ
dD
.
The approximate activity 1.3.33 derived earlier is valid in this situation as well.
A
P
A
D
1 −
λ
dP
λ
dD
However now we can not neglect the second term on the right side as we did in the
case of secular equilibrium. In this case the equation depicts that the ratio of parent
to daughter activity is a constant determined by the ratio of parent to daughter
decay constant. Figure 1.3.5 shows the typical behavior of such a material. A
common example of transient equilibrium decay is the decay of Pb
212
00
into Bi
212
00
.
E.3 No Equilibrium
If the half life of parent is less than the half life of daughter, i.e.
T
P
1/2
<T
D
1/2
or λ
dP
>λ
dD
,
then the activity due to parent nuclide will diminish quickly as it decays into the
daughter. Consequently the net activity will be solely determined by the activity of
the daughter. Figure 1.3.7 depicts this behavior graphically.
1.3.F Branching Ratio
In the preceding sections we did not make any assumption with regard to whether
there was a single mode or multiple modes of decay of the nuclides. In fact the
majority of the nuclides actually decay through a number of modes simultaneously
with different decay constants. Branching ratio is a term that is used to characterize
the probability of decay through a mode with respect to all other modes. For example
if a nuclide decays through α and γ modes with branching ratios of 0.8 and 0.2, it
would imply that α-particle is emitted in 80% of decays while photons are emitted
in 20% of decays. The total decay constant λ
d,t
of such a nuclide having N decay
modes is obtained by simply adding the individual decay constants.
λ
d,t
=
n
i=1
λ
d,i
(1.3.34)
Here λ
d,i
represents the decay constant of the ith mode for a material that decays
through a total of n modes. The total decay constant can be used to determine the
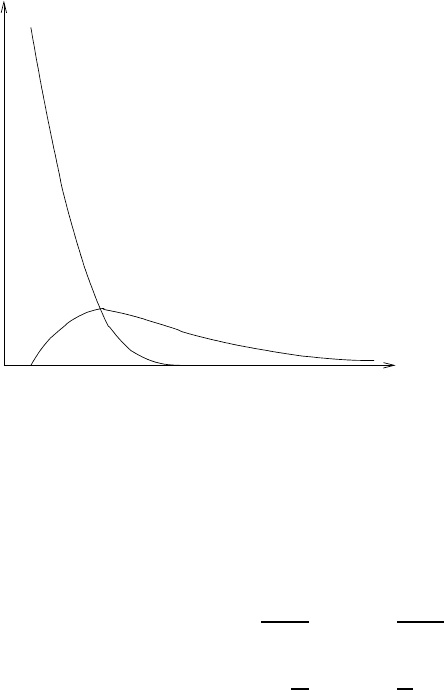
1.3. Radioactivity and Radioactive Decay 25
T
P
1/2
<
T
1/2
D
Activity
A
D
A
P
Tim
e
Figure 1.3.7: Activities of par-
ent and daughter nuclides as a
function of time for a material
that never reaches the state of
equilibrium. The parent in such
a material is shorter lived than
the daughter.
effective activity and other related quantities. The expressions for the effective half
and mean lives can be obtained by substituting T
1/2,i
=0.693/λ
d,i
and τ
i
=1/λ
d,i
for the ith decay mode in the above equation. This gives
1
T
1/2.e
=
n
i=1
1
T
1/2,i
(1.3.35)
and
1
τ
e
=
n
i=1
1
τ
i
, (1.3.36)
where T
1/2,e
and τ
e
represent the effective half and mean lives respectively.
1.3.G Units of Radioactivity
Since the most natural way to measure activity of a material is to see how many
disintegrations per unit time it is going through, therefore the units of activity are
defined in terms of disintegrations per second. For example 1 Becquerel corresponds
to 1 disintegration per second and 1 Curie is equivalent to 3.7 ×10
10
disintegrations
per second. Curie is a much bigger unit than Becquerel and is therefore more
commonly used. However for most practical sources used in laboratories, Curie is
too big. Therefore its subunits of milli-Curie and micro-Curie are more commonly
found in literature. The subunits of Curie and interconversion factors of Curie and
Becquerel are given below.
