Heidersbach R. Metallurgy and Corrosion Control in Oil and Gas Production
Подождите немного. Документ загружается.

CHEMISTRY OF CORROSION 9
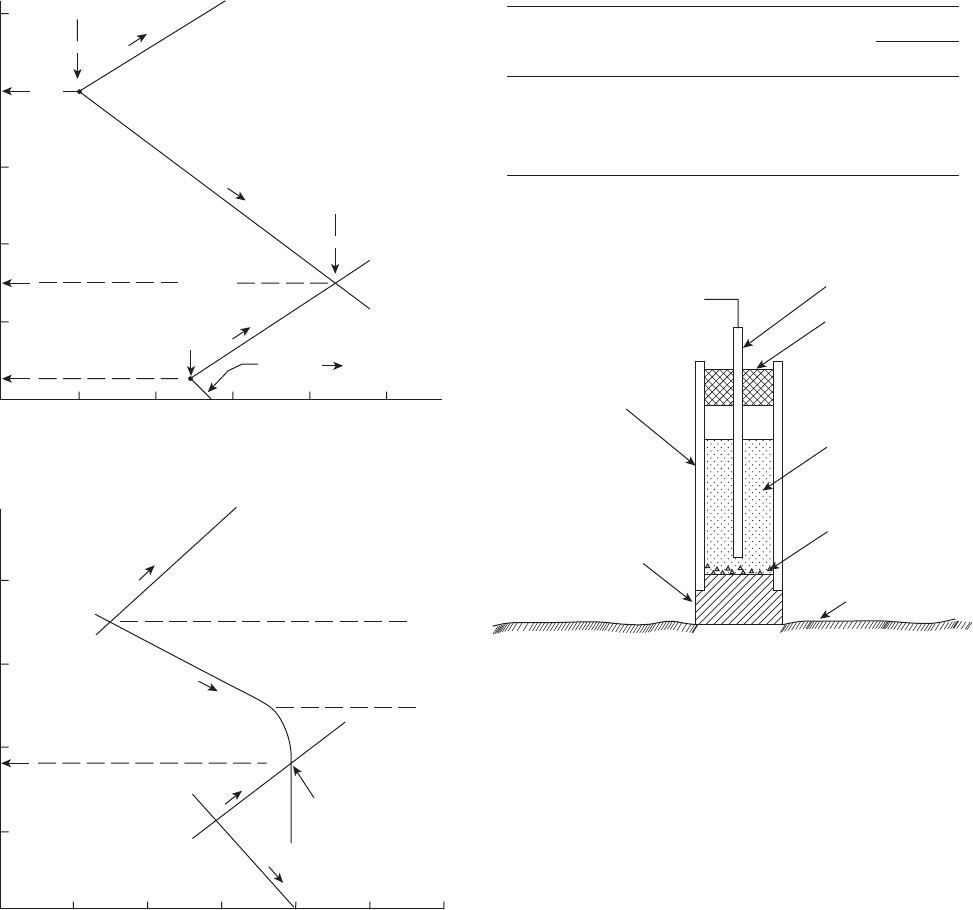
Figure 2.6 (a) Corrosion current and potential of iron deter-
mined by the polarization of iron and the hydrogen reduction
reaction.
9
(b) Corrosion current and potential of iron deter-
mined by the concentration polarization of oxygen.
9
E
corr
E
0
(M
2+
/M)
M
2+
+2e
–
M
E
0
(H
+
/H
2
)
i
0
(H
+
/H
2
)
i
0
(M
2+
/M)
i
corr
X
Log Current Density
a
Potential +–
2H
+
+2e
–
H
2
2H
+
+2e
–
H
2
M
2+
+2e
–
M
E
corr
M
2+
+2e
–
M
i
corr
Activation
Polarization
Concentration
Polarization
Log Current Density
b
Potential +–
4e
–
+2H
2
O+O
2
4OH
–
O
2
+2H
2
O+4e
–
4OH
–
M
2+
+2e
–
M
•
Electrical current associated with corrosion
•
Time to failure
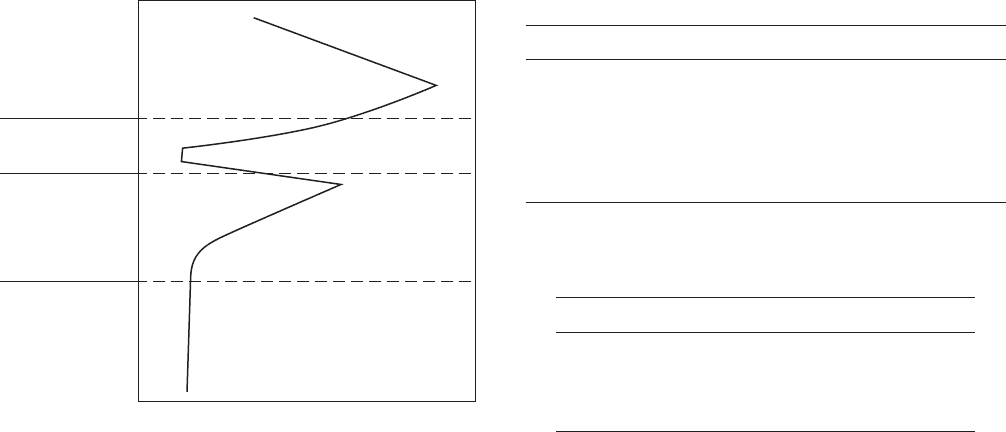
The simplest of these concepts to understand is depth
of penetration. It can be expressed in mm/yr (millime-
TABLE 2.2 Potential Values for Common Reference
Electrodes
Name
Potential
V vs. SHE
Copper - copper sulfate
+ 0.318
Saturated calomel
+ 0.241
Silver - silver chloride
+ 0.222
Standard hydrogen
+ 0.000
Adapted from Reference 3 .
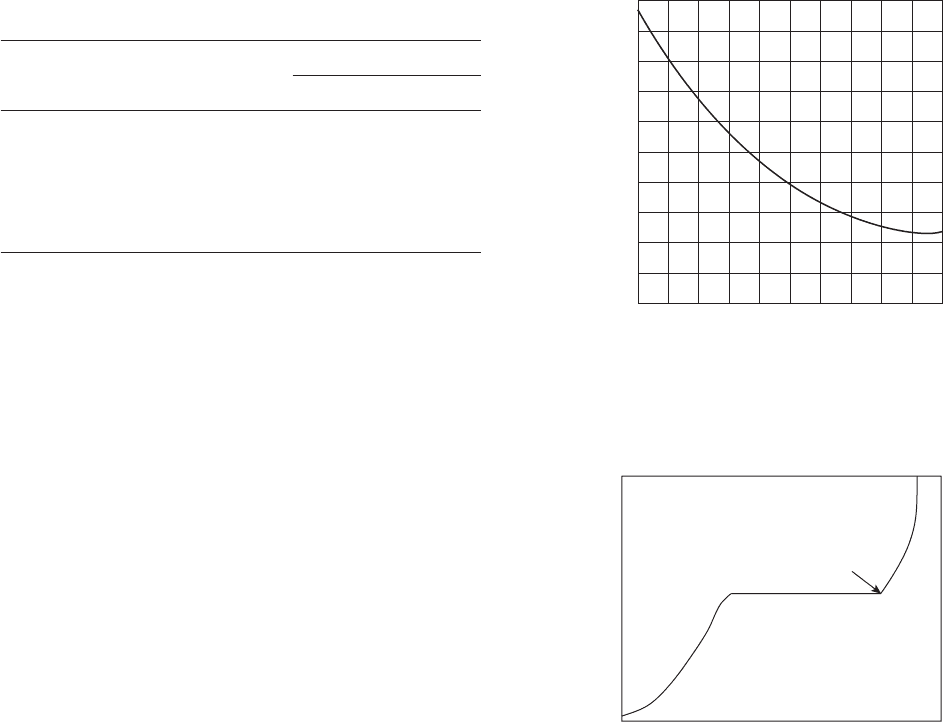
Figure 2.7 Saturated copper - copper sulfate reference
electrode.
9
Porous Plug (Usually
Wood or Ceramic)
Container of Insulating
Material. Often Transparent
or Translucent
Pure Copper Rod
(Electrolytic Copper)
Insulating Seal
Saturated Solution
of Copper Sulfate
Surplus Crystals of
Copper Sulfate
Ground Surface
ters per year) or mpy (mils or thousandths of an inch
per year). The loss of wall thickness is often used to
determine remaining equipment life or safe operating
pressures for piping systems, storage tanks, and so on.
Table 2.3 shows a commonly used classifi cation of
relative corrosion rates. The U.S. Standard units, mpy,
produce small numbers that are easy to understand,
and corrosion rates in mpy are commonly used world-
wide, although other expressions are also common .
1
Weight loss measurements are commonly used on
exposure samples used to monitor corrosion rates in oil
and gas production. It is a simple matter to convert
these weight loss measurements into average depths of
penetration, although this can be very misleading,
because most corrosion is localized in nature and the
average penetration rate seldom gives an indication of
the true condition of oilfi eld equipment.
The electrical current associated with anodic dissolu-
tion of a metal can be used to determine the corrosion
rate using Faraday ’ s law. This calculation of mass loss
c02.indd 9c02.indd 9 10/29/2010 10:35:04 AM10/29/2010 10:35:04 AM
10 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
PASSIVITY
Passivity is a phenomenon that is frequently misunder-
stood. Most metals form oxide fi lms in most corrosive
environments. These passive fi lms can be protective and
retard or even effectively stop corrosion, but they can
also lead to fairly deep localized corrosion in situations
where the protective fi lms are removed or defective.
Except in rare circumstances, the oxide fi lms formed on
carbon steel are not adequately protective, and other
means of corrosion control are necessary. This is in
contrast to stainless steels, titanium, and aluminum —
oilfi eld metals that form protective passive fi lms which
are commonly the primary means of corrosion control
for these alloys. On many corrosion - resistant alloys
can be converted into remaining thickness. Once again,
the reader is cautioned that most corrosion is localized
in nature and calculations assuming uniform loss of
cross section are frequently misleading.
The time to failure, however defi ned, is the most
common concern of managers and operators of equip-
ment. For some forms of corrosion testing, for example,
stress corrosion cracking, the time to failure is used to
screen alloys, environments, or other variables.
p H
The pH of an environment is one of the major factors
determining if corrosion will occur. It also infl uences the
type of corrosion that is experienced.
pH is defi ned as:
pH [H ]=−
+
log
(Eq. 2.13)
where the [H
+
] expression shows the hydrogen ion activ-
ity of the environment.
The [H
+
] depends on the ionization of water and
varies with temperature. The pH of neutral water at
standard temperature (25 ° ) is 7.00, but neutrality varies
with temperature as shown in Figure 2.8 . Downhole
oilfi eld temperatures are usually elevated, and it is
common to calculate the in situ pH of any fl uids that
might affect corrosion or scale deposition. There are
many software packages available for this purpose.
Figure 2.9 shows the effects of pH on the corrosion rates
of iron in water. At low pHs, bare metal is exposed to
the environment, and acid reduction on the surface con-
trols corrosion rates. For intermediate pHs, a partially
protective fi lm of iron oxide reduces the corrosion rate
and the diffusion of oxygen to cathodic locations on the
metal surface controls. As the pH increases to even
higher values, the surface becomes covered with mineral
scales and corrosion is reduced.
TABLE 2.3 Relative Corrosion Resistance versus Annual
Penetration Rates
Relative Corrosion Resistance
Corrosion Rate
mpy mm/yr
Outstanding
< 1 < 0.02
Excellent 1 – 5 0.02 – 0.1
Good 5 – 20 0.1 – 0.5
Fair 20 – 50 0.5 – 1
Poor 50 – 200 1 – 5
Unacceptable
200 + 5 +
Source : Adapted from M. Fontana, Corrosion Engineering (McGraw -
Hill, 1986).
1
Figure 2.8 pH values of pure water at various
temperatures.
10
Temperature (°C)
0
7. 5
7. 0
6.5
6.0
5.5
5.0
50
pH
100 150 200 250
Figure 2.9 The effect of pH on the corrosion rate of iron
in water at room temperature. Adapted from W. R. Revie
and H. H. Uhlig, Corrosion and Corrosion Control , Wiley -
Interscience, 2008.
pH
H
2
evolution
begins
10 4
Corrosion rate
c02.indd 10c02.indd 10 10/29/2010 10:35:04 AM10/29/2010 10:35:04 AM
CHEMISTRY OF CORROSION 11
available for most structural metals for which thermo-
dynamic data are available (Figure 2.11 ).
2,11 – 14
These diagrams make a number of important points
useful for oilfi eld corrosion control:
•
Water is only stable over a potential range of
slightly more than one volt. This is very important
in cathodic protection.
•
Iron (carbon steel) is covered with iron oxides
(passive fi lms) in most aqueous environments.
Unfortunately, these passive fi lms are usually not
suffi ciently protective and other means of corro-
sion control are necessary.
•
The potentials at which iron (carbon steel) is pro-
tected from corrosion do not coincide with the
immunity regions on the Pourbaix diagram. This
point is discussed in greater detail in Chapter 6 ,
Corrosion Control.
The diagrams for zinc, aluminum, and cadmium, com-
monly referred to as the amphoteric coating metals,
have passive regions in neutral environments. These
metals also have low corrosion rates in neutral environ-
ments and higher corrosion rates in both acids and
bases.
Pourbaix diagrams have limitations in addition to the
inability of thermodynamics to predict the protective-
ness of passive fi lms. These include the idea that they
can only be calculated for alloys, although experimental
Pourbaix diagrams have been reported.
12,13
Revie and
Uhlig list other limitations.
2
SUMMARY
The following ideas have been discussed in detail in this
chapter:
such as stainless steels, the passive fi lms may be only
dozens of atoms thick. This means that they are very
weak and are subject to mechanical damage, and this
can lead to localized corrosion at the damaged
locations.
Potential - pH (Pourbaix) Diagrams
Marcel Pourbaix developed a means of explaining the
thermodynamics of corrosion systems by plotting
regions of thermodynamic stability of metals and their
reaction plots on potential versus pH plots.
10 – 12
The
regions of a Pourbaix diagram can be described as:
•
Immunity The metal cannot oxidize or corrode
(although it may still be subject to hydrogen
embrittlement).
•
Corrosion Ions of the metal are thermodynami-
cally stable and the metal will corrode.
•
Passivity Compounds of the metal and chemicals
from the environment are thermodynamically
stable, and the metal may be protected from
corrosion if the passive fi lm is adherent and
protective.
Many users of Pourbaix diagrams miss the fi nal point
above. Thermodynamics alone cannot predict if passive
fi lms will be protective or not.
2,11 – 14
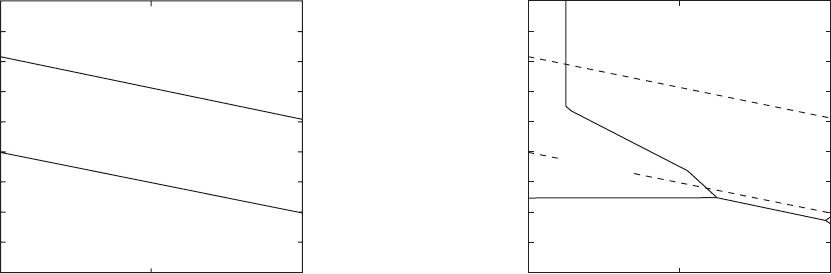
Figure 2.10 shows the Pourbaix diagram for water.
Water is thermodynamically stable over a potential
region of 1.23 V, and the potentials at which oxidation
and evolution (bubbling off) of oxygen from water at
the top of the diagram or the evolution of hydrogen at
the bottom of the diagram depend on the pH of the
environment.
The Pourbaix diagram of iron is superimposed on the
diagram for water in Figure 2.11 . Similar diagrams are
Figure 2.10 Potential - pH (Pourbaix) diagram for water.
14
pH
O
2
stable
H
2
O stable
H
2
stable
Equilibrium potential
0
2.0
1. 6
1. 2
0.8
0.4
0
–0.4
–0.8
–1.2
–1.6
714
Figure 2.11 Potential - pH (Pourbaix) diagram for iron.
14
pH
Passive
Fe stable
Corrosion
Equilibrium potential
0
2.0
1. 6
1. 2
0.8
0.4
0
–0.4
–0.8
–1.2
–1.6
714
c02.indd 11c02.indd 11 10/29/2010 10:35:04 AM10/29/2010 10:35:04 AM
12 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
2 W. R. Revie and H. H. Uhlig . 2008 . Corrosion and
Corrosion Control . New York : Wiley - Interscience .
3 D. A. Jones . 1996 . Principles and Prevention of Corrosion ,
2nd ed. Upper Saddle River, NJ : Prentice - Hall .
4 P. R. Roberge . 2006 . Basics — An Introduction , 2nd ed.
Houston, TX : NACE International .
5 Z. Ahmad . 2006 . Principles of Corrosion Engineering and
Corrosion Control . Oxford : Butterworth - Heinemann .
6 S. A. Bradford . 2001 . Corrosion Control , 2nd ed.
Edmonton, Alberta, Canada : CASTI .
7 K. R. Trethewey and J. Chamberlin . 1995 . Corrosion for
Science and Engineering , 2nd ed. London : Longman
Scientifi c and Technical .
8 E. McCafferty . 2009 . Introduction to Corrosion Science .
Berlin : Springer - Verlag .
9 J. Beavers . 2000 . Fundamentals of corrosion . In Peabody ’ s
Control of Pipeline Corrosion , 2nd ed. , ed. R. Bianchetti ,
297 – 317 . Houston, TX : NACE International .
10 R. Baboian , ed. 2002 . Corrosion Engineer ’ s Reference
Book , 3rd ed. Houston, TX : NACE International , 78 .
11 M. Pourbaix . 1974 . Atlas of Electrochemical Equilibria
in Aqueous Solutions , 2nd English ed. Houston, TX :
National Association of Corrosion Engineers, and
Brussels: Centre Belge d ’ Etude de la Corrosion
(CELBECOR).
12 E. D. Verink Jr . 2000 . Simplifi ed procedure for construct-
ing Pourbaix diagrams . In Uhlig ’ s Corrosion Handbook ,
2nd ed. , ed. R. W. Revie , 111 – 124 . New York : Wiley -
Interscience .
13 W. T. Thompson , M. H. Kaye , C. W. Bale , and A. D.
Pelton . 2000 . Pourbaix diagrams for multielement
systems . In Uhlig ’ s Corrosion Handbook , 2nd ed. , ed. R.
W. Revie , 125 – 136 . New York : Wiley - Interscience .
14 B. Cottis . 2002 . Introduction to corrosion, UMIST
Corrosion and Protection Centre . http://corrosiontest.
its.manchester.ac.uk/lecturenotes/civils/Civils%20
Corrosion%20Notes.pdf (accessed May 28, 2009).
•
Corrosion is electrochemical in nature.
•
Most metal surfaces have both oxidation and
reduction occurring simultaneously.
•
If the predominant reaction is oxidation, the metal
will corrode.
•
The most important reduction reaction is oxygen
reduction for many oilfi eld systems. If no oxygen
is available, the corrosion rate will often be very
low.
Electrode potentials are determined by:
•
Metal chemistry
•
Chemicals in the environment
•
Temperature
These potentials are usually measured against either
copper - copper sulfate or silver - silver chloride elec-
trodes, depending on the environment.
Corrosion rates are often expressed by average depth
of penetration, and this can be misleading because most
oilfi eld corrosion is localized in nature.
The pH of the environment has a major effect on
corrosivity.
Passive fi lms may limit corrosion in many environ-
ments, but carbon steel, the most common oilfi eld
metal, seldom forms adequately protective passive
fi lms, and other means of corrosion are often
necessary.
REFERENCES
1 M. Fontana . 1986 . Corrosion Engineering . New York :
McGraw - Hill .
c02.indd 12c02.indd 12 10/29/2010 10:35:04 AM10/29/2010 10:35:04 AM

3
CORROSIVE ENVIRONMENTS
A very limited amount of oilfi eld corrosion is associated
with very high temperature atmospheric exposures,
common in fl ares, and with liquid metals, usually
mercury found in natural gas and some crude oils. The
great majority of oilfi eld corrosion requires liquid water.
For this reason, as an oil fi eld ages and the water cut
increases, corrosion also increases. This is shown in
Figure 3.1 , which shows the effect of water cut on cor-
rosion rates.
1
Many operators use rules of thumb such
as the idea that corrosion is not a problem until the
water cut reaches 40% or 50%. For some oil fi elds, this
may take several years before corrosion becomes a
problem. Unfortunately, this means that corrosion and
other maintenance problems become more important
at a time when maintenance funds, often related to
production rates, decrease.
Water has very limited solubility in hydrocarbons,
and the presence of a separated water phase is neces-
sary for corrosion. The low - corrosion region in Figure
3.1 is where most of the metal surface is in contact with
a water - in - oil emulsion. The small water droplets are
not continuous, and most of the metal surface is in
contact with nonconductive hydrocarbons. As the water
cut increases, the amount of the metal surface in contact
with water gradually increases until the emulsion
reverses, and the liquid becomes continuous water
with entrained hydrocarbon droplets. Production and
fl uid fl ow rates, along with temperature and pressure
considerations, determine when this will happen.
Figure 3.2 shows how water separates out on production
tubing .
In contrast to oil wells, natural gas wells are corrosive
from the beginning. This is due to the fact that all
natural gas reservoirs will produce some water, and
minor components of the natural gas, which condense
from the gas stream as temperatures and pressures are
reduced, dissolve in this water and make it corrosive.
Most downhole hydrocarbon reservoirs have virtually
no dissolved oxygen in the fl uids, and this is fortunate,
because the presence of oxygen at the parts - per - billion
(ppb) level has been shown to promote corrosion. This is
in contrast to carbon dioxide (CO
2
) and hydrogen sulfi de
(H
2
S), which may be present in varying quantities in both
oil and gas fi elds. The relative effects of these three gases
are shown in Figure 3.3 . Oxygen is approximately 50
times more corrosive than CO
2
and more than a hundred
times more corrosive than H
2
S .
3
Downhole corrosion, in the absence of oxygen, is
largely determined by the concentrations of CO
2
or H
2
S
in the produced fl uids. The terms “ sweet corrosion ” to
describe corrosion caused by CO
2
, and “ sour corrosion ”
to describe problems with H
2
S, have been used for
many years to differentiate which of these two gases is
likely to predominate in a given fi eld.
4
Other consider-
ations which affect corrosion rates include temperature
and pressure, which determine the nature of the fl uid
(gas, liquid, etc.) on the metal surface, and minor con-
stituents in the liquid water phase. Figure 3.4 shows how
complex the determination of corrosivity can be.
EXTERNAL ENVIRONMENTS
The external environments discussed in this section are
not unique to oil and gas production, but much of
the information comes from oilfi eld experience with
13
Metallurgy and Corrosion Control in Oil and Gas Production,
by Robert Heidersbach
Copyright © 2011 John Wiley & Sons, Inc.
c03.indd 13c03.indd 13 10/29/2010 11:52:44 AM10/29/2010 11:52:44 AM
14 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
production platforms, buried or subsea pipelines, and
similar equipment.
Atmospheric Corrosion
Like all other types of corrosion discussed in this book,
this form of corrosion requires the presence of con-
densed water on the metal surface in order for corrosion
to occur. The only exception to this general rule is at
very elevated temperatures, for example, those associ-
ated with fl ares and other combustion processes, where
corrosion can occur without liquid water. Even here
most corrosion occurs below the dew point, because
these high - temperature applications require special
alloys to withstand corrosion at high temperatures, and
this equipment often suffers the worst corrosion during
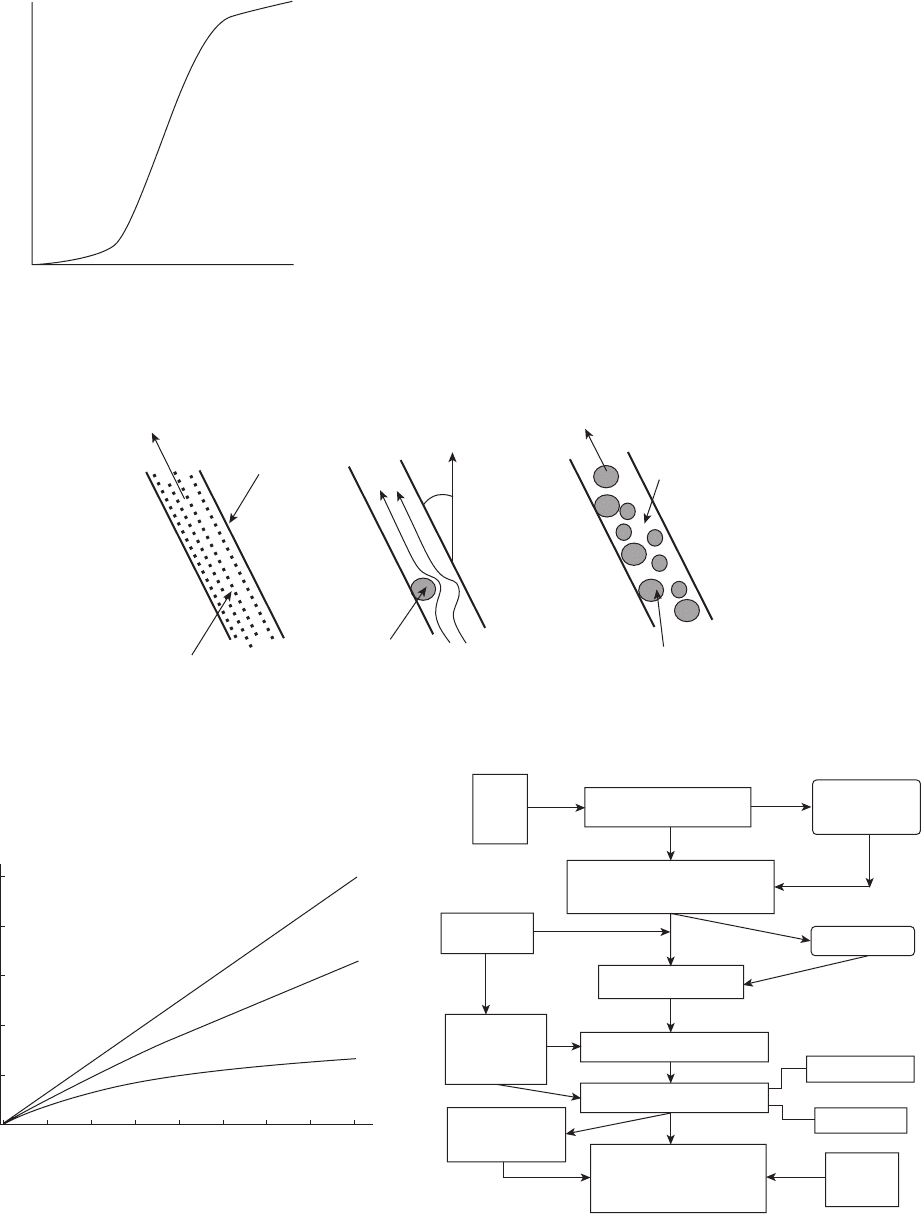
Figure 3.1 The effects of water cut on the corrosion rate of
oil well tubing.
1
Water Cut Percent
Corrosion Rate →
0100
Figure 3.2 Water wetting producing corrosion on deviated oil wells.
2
mode I
mode II
tubing
oil
α
mode III
oil/emulsion
moving water
droplets
stationary water
droplet
water in oil
emulsion
Figure 3.3 The effect of dissolved gases on the corrosion of
carbon steel.
3
Dissolved gas concentration in water phase (ppm)
Overall corrosion rate of carbon steel
(mpy)
25
20
15
10
5
O
2
O
2
12345678
100 200 300 400 500 600 700 800
50 100 150 200 250 300 350 400
CO
2
CO
2
H
2
S
H
2
S
Figure 3.4 A fl ow chart indicating the factors which deter-
mine the corrosion severity to be expected in an oil or gas
fi eld.
5
Operating
Temperature
Corrosion
Nomogram
CO
2
/H
2
S Ratio
Horizontal Flow
Vertical Flow
Chlorides
Aeration
Sulfur
Relative Domination
Determine Eff.
CO
2
Pressure
Film Effects
Gas/Oil Ratio
Water Content
Oil Type
Dew Point
Inhibition:
Delivery Method
Effectiveness
H
2
S
CO
2
HCO
3
Temp.
H
2
S Effect
Oil/Gas/Water Effects
Determine CO
2
-based
Corrosion Rate
Velocity Effects
Corrosion
Severity Index
Determine pH
c03.indd 14c03.indd 14 10/29/2010 11:52:44 AM10/29/2010 11:52:44 AM
CORROSIVE ENVIRONMENTS 15
Carbon steel, the most common metal used in oilfi eld
systems, corrodes at unacceptable rates in many aqueous
environments, and pH adjustment is a common means
of controlling corrosion.
Most readers are familiar with the idea that saltwater
is more corrosive than freshwater. The combined effects
of dissolved oxygen and salt concentration on the cor-
rosivity of water are shown in Figure 3.6 . As increasing
amounts of salt are added to water, the electrical con-
ductivity of the electrolyte increases and so does the
corrosion rate. At the same time, the oxygen solubility
decreases continuously with additional concentrations
of salt, and this limits the corrosion rate because oxygen
reduction is the rate - controlling chemical (reduction)
reaction.
5
The same phenomenon happens with all
other salts. The maximum corrosion rate is at approxi-
mately 3% salt — the exact concentration depends on
temperature and the salt involved.
8
This explains why
highly concentrated brines, such as those used in packer
fl uids, are noncorrosive, provided they are properly pH -
adjusted and have little or no dissolved oxygen.
Figure 3.6 explains why freshwater, low in salt, is less
corrosive than saltwater, but the most important point
to be learned from this picture is that, even at its most
corrosive, only about one - third of the corrosion in salt-
water is due to salt — most of the corrosion would occur
anyway due to the presence of oxygen.
Many reports on corrosion ascribe corrosion damage
to the presence of chlorides, the most common anions
found in seawater and often found in freshwater as well.
This dates back to analytical chemistry practices in the
early twentieth century, when qualitative analysis tech-
niques (methods of determining the presence of various
chemicals in the environment) were relatively new. The
fi eld methods for identifying chloride were relatively
easy, and many authors started blaming chlorides for
damage caused by salts. It was unnecessary to identify
the other components of salt, as there will always be
cations (positively charged ions) present to balance the
charge of the negatively charged anions. This practice
Figure 3.5 Simplifi ed diagram showing the effect of relative
humidity and pollution on the corrosion of carbon steel.
6
Corrosion loss
Relative humidity (%)
50 60 70 80 90 100
Pure air
Air
polluted
with SO
2
Air polluted
with SO
2
and
solid particles
Figure 3.6 The corrosion rate of iron in air - exposed fresh-
water at varying salt (sodium chloride) concentrations.
6
Conc. NaCl (wt %)
Relative Corrosion Rate
2
1
0
035 10 20 302515
shutdown, when acidic moisture can condense on rough
surfaces and cause corrosion similar to that on automo-
tive muffl ers during times when the system is cold
enough for condensation.
It would seem logical that atmospheric corrosion
would not occur until the relative humidity is 100%, but
this is not the case. Research dating back to the 1920s
has shown that corrosion can occur once the humidity
reaches a “ critical humidity ” of approximately 60 –
70%.
6 – 9
Many structures, especially on the away - from -
the - sun side (the north side in northern latitudes), stay
above this critical humidity virtually all the time, at least
whenever the temperature is above freezing.
8
It is
important to realize that heat sinks; for example, large
structural members on offshore structures can remain
below the critical humidity long after the sun comes up
and corrosion has diminished elsewhere on the same
structure. The presence of deliquescent salts means that
many surfaces remain wetted even in sunlight. This is a
very important consideration when painting structures,
because “ fl ash rusting ” due to surface moisture can
quickly form and severely degrade the adherence of
primary coatings to painted structures (Figure 3.5 ).
Most metal exposed to atmospheric corrosion is
carbon steel , and the most common method of corro-
sion control is by the use of protective coatings (paint-
ing). Some process equipment, storage tanks, and
electronic control systems are protected by the use of
inerting gases, heaters, deliquescing agents, or vapor
phase inhibitors. Control lines, conduit, and similar
tubing are often stainless steel on offshore structures.
Water as a Corrosive Environment
The effect of pH on corrosion of carbon steel was dis-
cussed in Chapter 2 , Chemistry of Corrosion, fi gure 2.9.
c03.indd 15c03.indd 15 10/29/2010 11:52:44 AM10/29/2010 11:52:44 AM
16 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
of the National Bureau of Standards (now the National
Institute of Standards and Technology) in 1957 and,
when the government report went out of print, it was
republished by the National Association of Corrosion
Engineers (NACE).
12
Many advances have been made
in the understanding of corrosion and cathodic protec-
tion since the original publication, but the data repre-
sent one of the most extensive sources of corrosion in
soil data that is available.
Water and gas occupy much of the space between the
solid particles of soil, and these are very important in
determining the corrosivity of soils. The air – water inter-
face, wherever located, is the most corrosive location
for buried structures, and this location often varies with
seasonal rainfall patterns. The minerals in soil dissolve
in water and affect the soil resistivity. This directly
affects corrosivity, as shown in Table 3.1 .
Sandy soils drain well and tend to have the highest
resistivities and lowest corrosion rates. Clays that can
swell when wetted sometimes produce situations where
drainage is prevented and buried structures remain wet
and corrode.
Soil pH can also affect corrosion. Table 3.2 shows the
effect of soil pH on corrosivity. Acidic soils are encoun-
tered in swampy locations, volcanic regions, and areas
with silicate rocks and high moisture.
Some dry soil, especially clay - rich soil, contracts
during dry seasons as shown in Figure 3.8 . This can lead
to air ingress down to the buried structure, usually a
pipeline, and lead to corrosion when rainy weather
returns. Soil expansion and contraction can also cause
movement of the buried structure. This produces
stresses that can lead to stress corrosion cracking (SCC).
continues. Any highly ionic salt would cause similar
damage, but chloride salts are the most common in most
natural environments.
Figure 3.7 shows the corrosion rates of piling in sea-
water at various elevations. The highest corrosion rates
are in the splash zone, where the metal is frequently
covered with air - saturated water. The relatively low
corrosion rates in the tidal region are due to the oxygen
concentration cells between the highly aerated tidal
zone and the fully submerged zone just below. The tidal
zone, having high oxygen concentrations, is cathodic to
the fully submerged zone just below, which is anodic.
As the water deepens, the oxygen concentrations dimin-
ish and corrosion decreases.
Produced water can vary from very salty, which is
common in oil wells, to almost pure, the condensate
associated with some gas wells. This “ pure ” water can
become very corrosive because the dissolved gases, CO
2
and H
2
S, plus acidic hydrocarbons can drastically lower
the pH, especially at downhole temperatures and
pressures.
Most oilfi eld metal exposed to corrosive waters
is carbon steel, and the most common method of
corrosion control is by the use of protective coatings
(painting) which is often supplemented by cathodic pro-
tection. Corrosion inhibitors and corrosion - resistant
alloys (CRAs) are also used, especially in downhole
environments.
Soils as Corrosive Environments
Much has been written on corrosion in soils.
12
The defi n-
itive work on this subject was published by M. Romanoff
TABLE 3.1 Corrosivity Ratings Based on Soil Resistivity
13
Soil Resistivity (ohm - cm) Corrosivity Rating
> 20,000
Essentially noncorrosive
10,000 – 20,000 Mildly corrosive
5000 – 10,000 Moderately corrosive
3000 – 5000 Corrosive
1000 – 3000 Highly corrosive
< 1000
Extremely corrosive
TABLE 3.2 Soil pH versus Corrosivity
14
pH Degree of Corrosivity
< 5.5
Severe
5.5 – 6.5 Moderate
6.5 – 7.5 Neutral
> 7.5
None (alkaline)
Figure 3.7 Zones of corrosion for steel piling in sea water.
10,11
Mean high tide
Mean low tide
Mud line
Relative loss in metal thickness
Zone 5:
Subsoil
Zone 3:
Tidal
Zone 4:
Continuously
submerged
Zone 1:
Atmospheric
corrosion
Zone 2:
Splash zone
above high tide
c03.indd 16c03.indd 16 10/29/2010 11:52:44 AM10/29/2010 11:52:44 AM

CORROSIVE ENVIRONMENTS 17
Figure 3.8 Cracked soil due to drying after the rainy season.
More common is coating damage due to motion of the
coated pipeline against rocks and other hard features in
the trench.
Peabody, in his classic book on pipeline corrosion,
cautioned against galvanic cells between new pipe and
old pipe.
15
There are a number of possibilities for gal-
vanic cells to form when a new structure is placed in the
soil adjacent to already - buried structures. The most
obvious reason for this corrosion is that the lack of soil
compaction over the recently disturbed soil is more
likely to leave void spaces and locations for enhanced
air and moisture ingress. The new structure usually acts
as an anode, indicating that increased moisture perme-
ability is corrosion rate controlling.
Buried pipelines are in disturbed soil near soil that
has been in place for many years. The differences in
aeration and moisture are evident in the vegetation pat-
terns over many pipelines. This is shown in Figure 3.9 ,
which shows two obvious right - of - way locations, each
of which has several parallel buried pipelines.
The most corrosive location in any buried structure is
usually where the structure crosses the air - to - soil inter-
face. This is shown in Figure 3.10 . It is important to
concentrate inspections in these locations because
cathodic protection, which protects buried structures,
cannot be effective in the loosely compacted soil at these
locations. Abrasion, motion due to solar - induced expan-
sion and contraction, and a variety of other factors, are
likely to cause coating damage at these locations.
Virtually all oilfi eld equipment buried in soil is pro-
tected by a combination of protective coatings supple-
mented by cathodic protection. The exceptions are in
those rare locations where the resistivity of the soil is so
high that corrosion is unlikely and cathodic protection
would be diffi cult to achieve. Even in these locations, it
is common to use protective coatings. Cathodic protec-
Figure 3.9 Differences in vegetation over two parallel pipe-
line rights of way.
Figure 3.10 Corroded pipeline at the air - to - soil interface.
tion also cannot work at elevated temperatures where
any moisture in the soil will quickly evaporate and no
electrolyte is available. Many locations where this
occurs, for example, in acidic volcanic soils, require spe-
cialized protective coatings, and the lack of widely
accepted coatings for these applications is a continuing
problem for many operators.
Corrosion under Insulation
Corrosion under insulation (CUI) is an increasingly
important problem. Most air - exposed insulated piping
and vessels are covered with porous insulation that
is protected from moisture by sheet metal covering.
Figure 3.11 shows the corrosion locations identifi ed by
a major operator. They are virtually identical to the
c03.indd 17c03.indd 17 10/29/2010 11:52:45 AM10/29/2010 11:52:45 AM
18 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
tain noncorrosive temperatures on the inside of pipe-
lines and gathering lines that would otherwise quickly
cool to the temperatures of the ambient seawater
environments.
INTERNAL ENVIRONMENTS
The environments discussed in this section relate to
internal corrosion in oilfi eld production and piping
systems. These internal environments will vary depend-
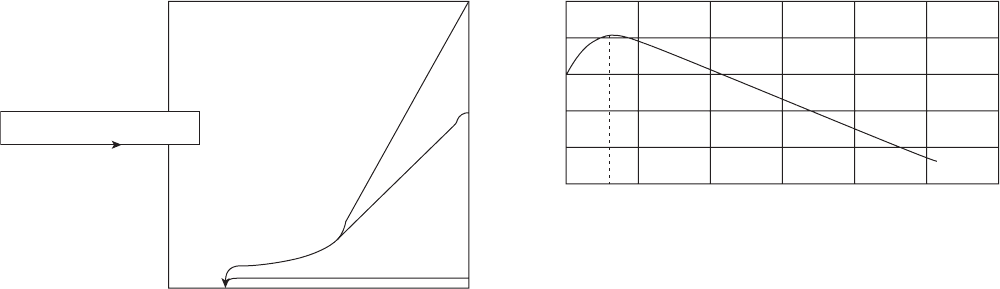
Figure 3.11 Problem locations for insulated above - ground pipelines.
Worst locations are
insulation joints at
saddles
Mid-span insulation
joints trap water
Vertical risers
shed water and
water traps at
lower insulation joints
Saddles trap water
• Prevent water egress
• Heat sink attracts vapor
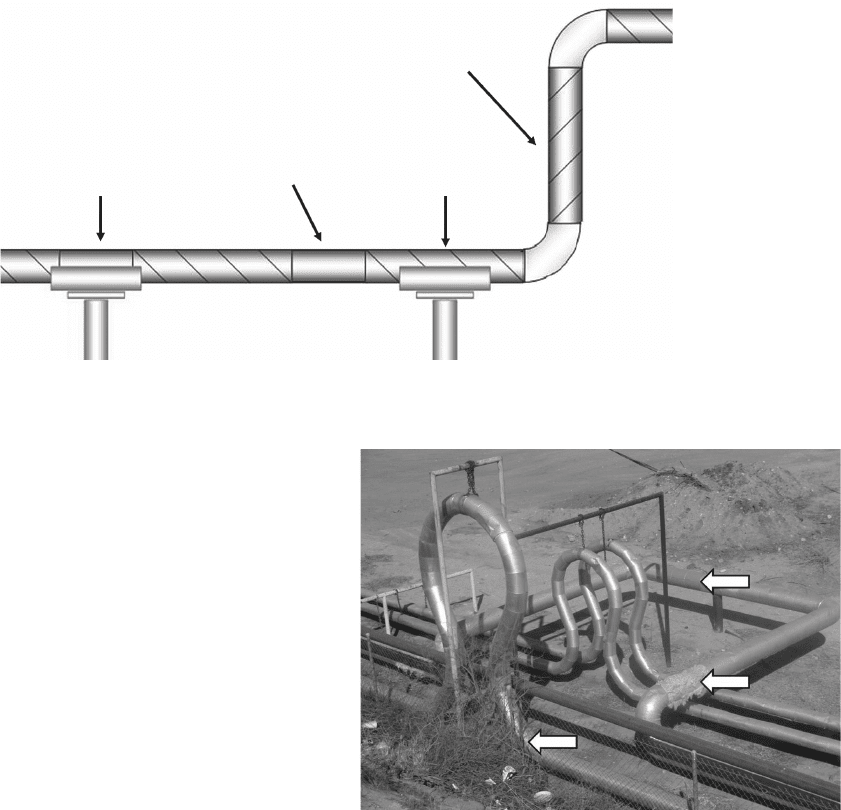
Figure 3.12 Vertical expansion loops in steam piping for
secondary recovery. Top arrow indicates where repair has
been covered with painted sheet metal, middle arrow indi-
cates location where sheet metal outer covering is missing, and
bottom arrow indicates the bottom of a vertical expansion
loop that may cause internal slugging problems, especially
with low - quality steam.
locations discussed in NACE RP0198, The Control of
Corrosion Under Thermal Insulation and Fireproofi ng
Materials — A Systems Approach, which contains
detailed discussions on how this corrosion occurs and
approaches to minimize it.
16
Breaks in the weather barrier usually occur at joints
and allow water into the insulation. This water can leach
contaminants from the insulation which are then con-
centrated and deposited on hot surfaces. This can even-
tually lead to SCC, especially with insulations containing
soluble chloride salts.
Corrosion under insulation is hard to inspect, and it
is important to conduct these inspections on a frequent
basis. It is also important to replace the weather barriers
after the inspection. A missing weather barrier is shown
in Figure 3.12 .
Slugging may occur at the bottom of upstream loca-
tions on vertical expansion joints like those shown in
Figure 3.12 . This creates fl uid fl ow problems, but it also
induces stresses into the system and may lead to corro-
sion fatigue failures. All expansion joints, whether hori-
zontal or vertical, are locations where corrosion and
crack detection inspections should be concentrated.
Buried or submerged thermally insulated piping
relies on protective coatings for corrosion control. The
application of cathodic protection to thermally insu-
lated structures has not worked in most instances.
17
In addition to the proper design of moisture shields
and draining for insulated piping, many operators have
adopted the practice of always coating the carrier piping
(even if it is made from a corrosion - resistant alloy) with
an immersion - grade coating. It is also important to
provide inspection capabilities for insulated piping. This
is becoming an increasingly important consideration as
more and more subsea pipelines are insulated to main-
c03.indd 18c03.indd 18 10/29/2010 11:52:45 AM10/29/2010 11:52:45 AM
