Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.


and
κ
ea
,
κ
cc
,
κ
ec
,
κ
sb
are the transmission factors for the different reactions; Γ
s
is
the surface density of H adsorption sites (number per unit area); z
1
[in k
cc
, Eqs.
(27) and (28)] is the number of adsorption sites adjacent to a given site (lateral or
surface coordination), so that the surface density of dual sites is equal to s/2z
1
Γ
s
[85]. The dimension of the rates is given by the product kT / h Γ
s
(cm
–2
s
–1
). Also,
γ
≠
ea
, γ
≠
cc
, γ
≠
ec
, are the activity coefficients of the respective activated complexes,
dependent on c
H
+
and/or p
H2
; ΔG
≠
ea
, ΔG
≠
cc
, ΔG
≠
ec
, ΔG
≠
sb
are the intrinsic activation
free energies (standard activation free energies of the respective reactions for
ΔG = 0) [86,87]; β, γ, δ,
ν
are energy-barrier symmetry factors or Brönsted
coefficients (0/ < β, γ, δ, v < 1). The
κ
'S, ΔG
≠
's and the symmetry factors are
constant for a given reaction and independent of ΔG (i.e., of the potential and of
the nature of the substrate) in a range of moderate variation of ΔG [87]. So k
ea
, k
cc
,
k
ec
, k
sb
(intrinsic rate constants) are constant for a given reaction, provided that z
1
and Γ
s
, i.e., the surface crystallography, are unchanged.
Steady-State Equations for the H Cathodic Reactions
The relation between the H atomic fraction in the bulk sites of the first planes
beneath the surface and the surface variables depends on the mechanism of the
global cathodic reaction, which is determined by the steady-state conditions. The
following analysis is restricted to the current case of overpotential adsorption, with
the simplifications stated earlier. In the steady state, the OP H
ads
surface coverage
is constant with time so the net rate of the electroadsorption step is equal to the sum
of the net rates of the consecutive parallels steps consuming adsorbed H:
76 Protopopoff and Marcus
The overall steady cathodic current density, i
cat
, is given by
According to the conditions at the surface of the sample, two main cases are
distinguished in electrochemical experiments: charging or permeation experiments.
1. In charging experiments, the H concentration under the surface is imposed by
galvanostatic or potentiostatic conditions. The steady state is reached when this
concentration is reached in the whole sample; i.e., the rate of diffusion in the bulk
is zero. This condition applied to Eqs. (33) and (34) leads to i
c
= Fv
HER
and to
the isotherm expressing the equilibrium of the surface-bulk transfer [Eq. (12)].
2. In permeation experiments, a concentration gradient in the bulk is imposed
by different conditions at the two faces of a membrane, so H permeates
through the sample and a steady diffusion (permeation) rate is established
through the membrane (see Fig. 9). The H concentration under the entry
surface is imposed by galvanostatic or potentiostatic conditions, and the
Copyright © 2002 Marcel Dekker, Inc.
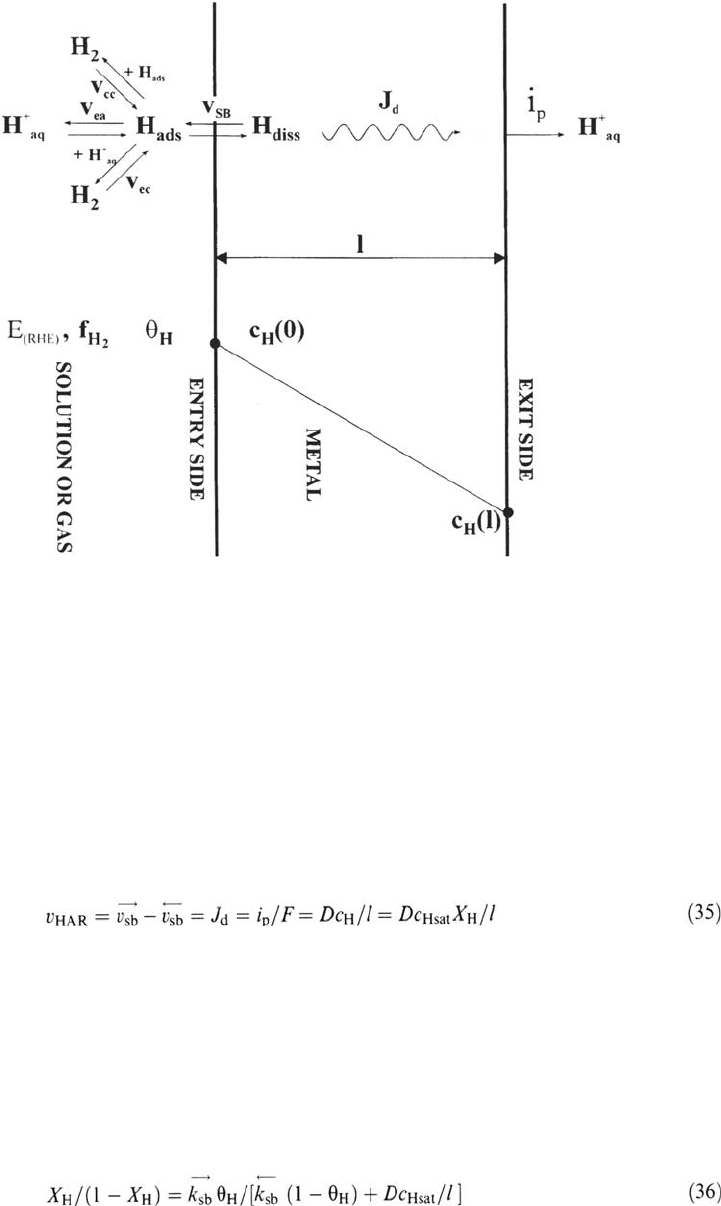
concentration under the exit surface is usually fixed to zero by potentiostatic
anodic conditions [28,89], such that H diffusing out of the exit face of the
membrane is electrochemically oxidized. In this case, the anodic current
density i
p
provides a direct measure of the H permeation rate J
d
. When the
steady state is reached, the stationary H permeation rate, proportional to the
concentration gradient through the membrane according to the first Fick
law, is equal to the net absorption rate at the entry side. If the concentration
under the exit face is zero, we obtain:
Surface Effects on Hydrogen Entry into Metals 77
Figure 9 Balance of reactions during permeation of H through a metal membrane, in gas
or aqueous solution, and concentration profile in the bulk.
where D is the H diffusion coefficient in the bulk, L the thickness of the membrane,
c
H
the volumic concentration of H under the entry face, and c
Hsat
is the same
quantity at saturation.
From the steady-state Equations (33) and (35), the variations of the surface
and bulk atomic fractions θ
H
and X
H
are interrelated and depend on the mechanism
of the whole cathodic reaction, i.e., both the HER and HAR.
Equality (35), expressing
v
sb
and
v
sb
from Eqs. (31) and (32), yields the
following expression for X
H
versus θ
H
:
Copyright © 2002 Marcel Dekker, Inc.
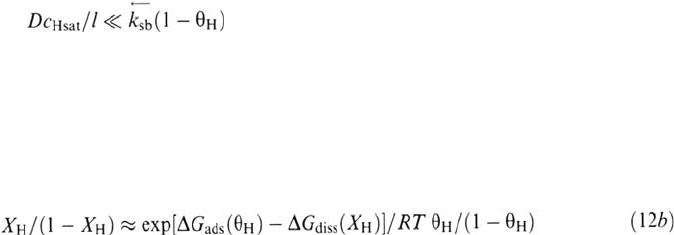
In the limiting case where
78 Protopopoff and Marcus
(this corresponds to conditions where D is low, the membrane is thick, θ
H
is not
close to unity, and
k
sb
is high), the permeation rate is limited by the bulk diffusion
rate in the membrane, so the surface-bulk transfer step is quasi-equilibrium
(
υ
sb
~
υ
sb
). It gives, as an approximation, the same expression for X
H
versus θ
H
as
in the charging conditions [Eq. (12)] except that this equation applies only to the
region of the bulk close to the entry surface:
As the permeation rate is negligible with respect to the H
2
evolution rate (or the
overall rate), as for charging, (
υ
sb
–
υ
sb
) may be neglected in Eq. (33) and then the
variation of θ
H
with the potential depends only on the HER mechanism and so does
the variation of X
H
. This is a good approximation for most experiments on
permeation through iron, nickel, and their alloys, with relatively thick membranes,
where i
p
/ i
cat
< 2/100 (low permeation rates). In this case, X
H
is thickness independent
so, from the last equality of Eq. (35), i
p
is proportional to 1/l [28]. It must be noted
that this limiting case no longer applies when θ
H
approaches unity. As θ
H
increases,
the complete Eq. (36) must be considered.
Dependence of the HER and HAR Rates on the Potential and the
Adsorption Gibbs Free Energy
Only the current conditions, charging conditions, or permeation limiting case
described before are considered here, so Eq. (12b) applies. Application of the
steadystate Equations (33) and (12b), using the expressions (23)–(32), allows one
to derive analytical expressions for θ
H
and X
H
versus the potential and ΔG
ads
for
each HER mechanism [61].
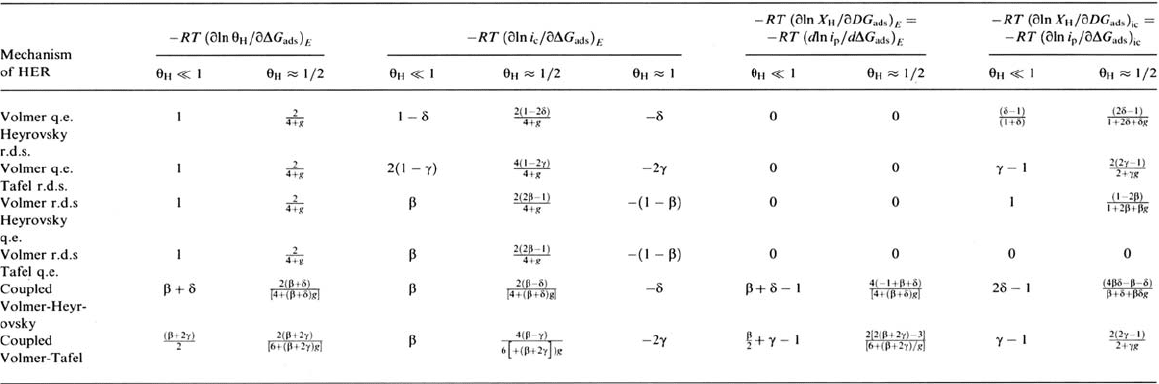
Analysis of the Dependence on the Potential
Differentiation of the preceding equations yields the (partial) derivatives of the
logarithms of θ
H
, X
H
(proportional to the stationary permeation current i
p
), and the
HER current with respect to the potential for a given metal and surface composition,
usually called diagnostic parameters, which may be compared with the experimental
variations for the particular cases of low H coverage θ
H
<< 1, midcoverage θ
H
≈ ½,
or saturation coverage θ
H
≈ 1. The parameters calculated for six different HER
mechanisms are reported in Table 1 and may be compared with previous calculations
[28,56,90,91] made for particular values of the symmetry factors (β = δ = ½),
assuming Langmuir-type adsorption for low coverage and Temkin-type adsorption
for medium coverage, with γ = ½ (activated molecular adsorption) or 1 (nonactivated),
and X
H
<< 1. The simplified form of the Temkin adsorption model was used where
the variations of θ
H
, 1 – θ
H
, θ
H
/ (1 – θ
H
) at θ
H
≈ ½ are neglected, whereas the
Fowler-Frumkin adsorption model used here is more general. In Table 1 it can be
checked that at θ≈½, only for large positive values of g (g >> 4), corresponding to
strong repulsive interactions between adsorbed H atoms or high surface heterogeneity,
the Tafel slopes are independent of the coverage and hence of the potential and the
transfer coefficient α takes the values calculated previously [28,56,90,91]
[α= –RT / F(∂ ln i
c
/ ∂E) is related to the Tafel slope b for the HER by the relation
Copyright © 2002 Marcel Dekker, Inc.
b = RT ln 10 / αF]. The term (1 – X
H
) takes into account the fraction of available sites
on the surface and in the bulk under the surface. It cannot be neglected a priori when
θ
H
(and X
H
) approaches unity. The coefficient (
∂
ln X
H
) / (
∂
ln i
c
), also reported in
Table 1, gives the power of i
c
to which X
H
or the permeation current is proportional.
Analysis of the Dependence on the Gibbs Free Energy of Adsorption
Similarly, derivatives expressing the dependence of θ
H
, i
c
, and X
H
on the Gibbs free
energy of adsorption at fixed potential (potentiostatic control) are given in Table 2.
Table 2 shows that the variation of the logarithm of the rate of the HER i
c
, at
a given potential, with ΔG
ads
gives a volcano curve for each mechanism, increasing
for values of ΔG
ads
such that θ
H
<< 1, decreasing at values of ΔG
ads
such that θ
H
≈ 1.
In each case, the curve has a maximum for a value of θ
H
depending on the values of
β, δ, γ. This result is similar to a classic result obtained for the exchange current
densities of the three HER steps [92–95]. For very high values of g (so-called
Temkin case), the current is nearly independent of ΔG
ads
in the midcoverage range
[93,94]. This extreme situation does not apply to the adsorption of hydrogen on
smooth metal substrates.
The steady-state equations for each HER mechanism where one step is
quasi-equilibrium lead to the adsorption isotherms given in Eqs. (10b) and (14) to
(17), hence to a quasi-equilibrium between H species in solution and H dissolved in
the bulk [Eqs. (18) and (19)]. In this case, there is no dependence of the H bulk
concentration on the Gibbs energy of adsorption at fixed potential, as seen in Table 2.
The steady-state conditions for the HER mechanisms where the
electroadsorption step is coupled to one of the subsequent steps lead to
pseudoequilibrium adsorption isotherms [26,28,61]. It is seen in Table 2 that for a
coupled Volmer-Heyrovsky mechanism, the dependence of X
H
on ΔG
ads
is slight if
β + δ≈1. Only for the coupled Volmer-Tafel mechanism may X
H
depend
significantly on ΔG
ads
, unless γ + β/2 ≈ 1 for θ
H
<< 1 or β + 2γ << 3/2 for θ
H
≈ ½.
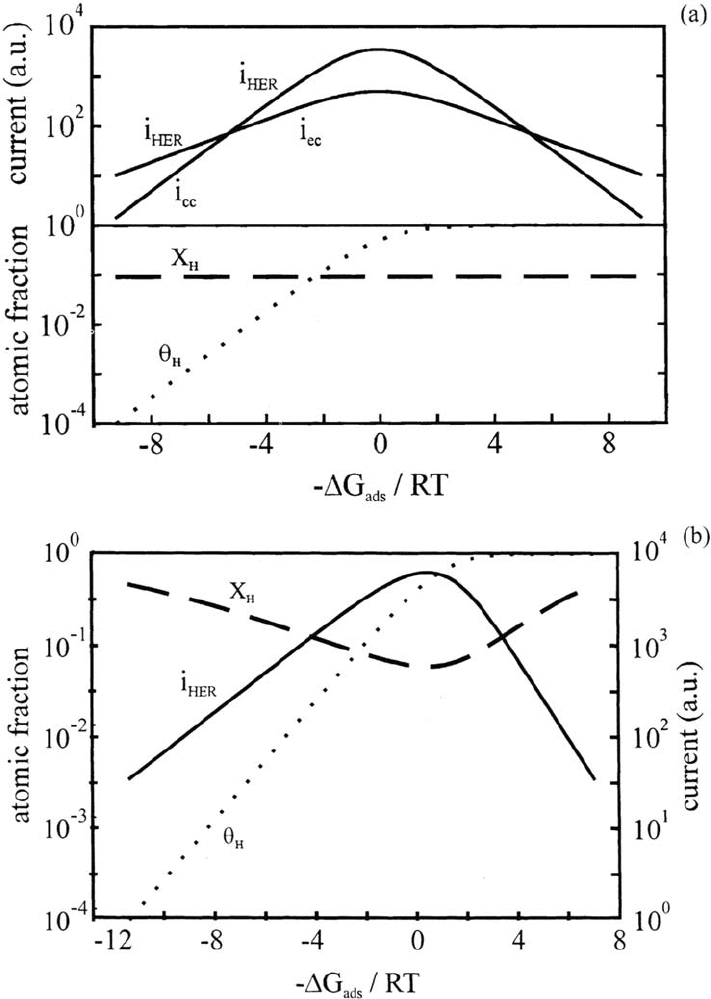
Figure 10a and b show the theoretical variations of log i
c
, θ
H
, and X
H
versus
–ΔG
ads
/ RT for an HER mechanism with the Volmer step in quasi-equilibrium and
for the coupled Volmer-Tafel mechanism. From Table 2, the maximum of the HER
current occurs at θ
H
= ½ if the symmetry factors are equal to ½ or for the two
coupled mechanisms if β = δ or β = γ. For the coupled Volmer-Tafel mechanism,
Figure 10b shows a case where X
H
is minimum for θ
H
≈ ½ and maximum for extreme
values of coverage (θ
H
<< 1 or θ
H
≈ 1), i.e., of the adsorption free energy.
In order to analyze experiments in which instead of the potential, the cathodic cur-
rent is the controlled variable (galvanostatic control), the partial derivatives of ln X
H
with respect to ΔG
ads
at fixed cathodic current density are also given in Table 2. At fixed
current density i
c
, X
H
depends on ΔG
ads
for most mechanisms, which may explain some
surface effects reported for H permeation in galvanostatic conditions (see later).
INFLUENCE OF THE SURFACE MODIFIERS ON H ENTRY
Effects of Metal Oxide Films
Perfect oxide surfaces are essentially inert to H
2
dissociative adsorption, although
atomic H may be adsorbed on and diffuse relatively fast in stoichiometric oxides
[96]. Oxide layers on metal surfaces impede or prevent the H
2
adsorption and H
Surface Effects on Hydrogen Entry into Metals 79
Copyright © 2002 Marcel Dekker, Inc.
80
Table 2 Dependence of θ
H
, i
c
, X
H
on the Adsorption Free Energy for Various HER Mechanisms, at fixed Potential or Fixed Current
Copyright © 2002 Marcel Dekker, Inc.
Surface Effects on Hydrogen Entry into Metals 81
Figure 10 Theoretical variations at a fixed potential of the HER current, the surface
coverage θ
H
, and the bulk fractional concentration X
H
with the H adsorption free energy
ΔG
ads
for two HER mechanisms: (a) electroadsorption step in quasi-equilibrium (a case
where the rate-determining step changes from electrocombination to chemical combination);
(b) electroadsorption coupled with chemical combination. The symmetry factors are all
taken equal to 1/2. The represented case is for Langmuir type H adsorption.
Copyright © 2002 Marcel Dekker, Inc.
absorption reactions at temperatures below 400°C (97). Monolayer amounts of
oxygen on transition metals reduce the H
2
adsorption and desorption rate by orders
of magnitude [96,97]. The effect of oxygen precoverage on transition metals
increases with the degree of oxide formation. Thin suboxide layers (in which cations
are not in their highest valence state) formed during the initial stages of oxidation or
by partial oxide reduction in H
2
impede the H absorption much less than layers of the
maximal valence oxides. Transition metal overlayers on the oxide layer restore the
original uptake rate of the substrate, which confirms that the effect of oxygen is
essentially to impede the H
2
dissociation step [96,97]. Similarly, the effect of passive
films formed anodically in solution is to impede H absorption [98,121].
Effects of Electronegative Species
As the H cathodic reactions are controlled by surface adsorption processes, they
are drastically affected by electronegative species known to poison hydrogenation
reactions on transition metals [99]. Electronegative elements are strongly
chemisorbed (far more than H) on transition metal surfaces and, even at low
concentrations, inhibit coadsorption of reactive species in the gaseous phase [100]
as in aqueous medium [53,59,101,117,118]; they hinder H
2
dissociation in the gas
phase [100,102] and the HER in electrolytes, increasing the cathodic overpotential
[53,75,99,101,117,118], even if opposite effects have also been observed,
particularly on Fe [103]. Compounds of elements of columns 15 and 16 of the
periodic table, P, As, Sb, S, Se, and Te, added to the electrolyte, while being strong
inhibitors of H underpotential adsorption, are known to promote H penetration
into the bulk of transition metals in aqueous medium [99]: in iron, nickel, and
steels [31,33,34,104–112]; palladium [73]; and platinum [113]. Additions of CN
–
,
I
–
, and naphthalene to the electrolyte also increase the permeation rate into iron,
although less efficiently, and additions of organic nitriles decrease it; however, all
these additives increase the HER overpotential at fixed current density [31]. The
surface step is clearly the determining step in these processes. The mechanisms of
action of promoters are investigated in detail now.
MECHANISMS OF ACTION OF H ABSORPTION PROMOTERS
Conditions for H Absorption Promotion
It has been noted [104] that all the promoters of H entry into iron (and steel) formed
from elements of columns 15 and 16 form gaseous hydrides of the type AsH
3
, H
2
Se,
H
2
Te, H
2
S, PH
3
, and SbH
3
and that the promotion is effective only in the cathodic
overpotential region where the hydrides form [104,105,109], and in the range of pH
in which they are stable [108]. For example, after addition of a compound of arsenic
in the +III oxidation state, H entry inhibition is observed at low overpotentials,
attributed to deposition by reduction of elemental arsenic, whereas at higher
overpotentials, H uptake increases drastically [34,106,109] and AsH
3
gas is detected
[106]. An additional proof is that a significant acceleration of H permeation results if
PH
3
, H
2
S, or AsH
3
produced outside is bubbled into the electrolyte [110].
The paramount role of hydride in promoting H entry into iron and steel has been
further confirmed by investigations conducted with a rotating-disk electrode, which
82 Protopopoff and Marcus
Copyright © 2002 Marcel Dekker, Inc.
also permitted a comparison of the effects of different compounds over a wide range
of pH and electrode potential [56,110]. For a given compound, the critical cathodic
overpotential at which the H entry efficiency begins to increase was related to the
equilibrium potential of hydride formation from elements (for H
2
S the latter is
positive vs. NHE, which explains why the permeation efficiency increases from the
free corrosion potential of steel while for AsH
3
, where it is more negative, the
permeation efficiency increases at higher overpotentials). Moreover, for S, Se, and Te
compounds in moderately acid electrolyte, the permeation rate increases with the
cathodic overpotential, reaches a maximum, and then decreases before attainment
of the limiting cathodic current density for proton discharge; this was explained by
the fact that at high current densities, due to limited proton transport, the activity of
protons in the vicinity of the cathode falls to a value giving a local pH equal to the
critical value from which the molecular hydrides begin to dissociate into anionic
forms. For As and Sb, because the molecular hydrides AsH
3
and SbH
3
are stable in
the whole pH range, the permeation rate may increase until the limiting cathodic
current density for which the local pH is equal to 7 is reached, after which it
decreases because the HER from water molecules is slower [56,110]. These results
proved that only the molecular forms of the hydrides are active in promoting H entry
[56,108,110].
The elements involved in the promoter species have been classified by various
investigators according to their effectiveness in promoting H entry; it seems to
depend upon the pH, the conditions of charging, and the method used for measuring
the H concentration, which explains why the order varied according to the authors
[28,56,107,108]. It has been suggested that the promoter efficiency is related to the
strength of the M–H bond in the hydride [104].
Usually the H concentrations for a given polarization increases with the con-
centration of promoter up to a maximum, for relatively low concentrations, beyond
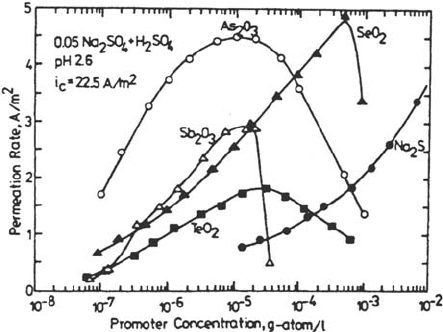
which it eventually decreases [56,108,110]. Figure 11 shows the H permeation rate at a
given cathodic current density as a function of the concentration of various compounds
of the elements discussed here. All the curves show maxima, except the one for Na
2
S,
Surface Effects on Hydrogen Entry into Metals 83
Figure 11 Effect of promoter concentration on hydrogen permeation rate through steel
membranes. (From Fig. 11–11 in Ref. 56.)
Copyright © 2002 Marcel Dekker, Inc.
where a continuous increase is observed. It is believed that a decrease for
concentrations higher than the optimal value is due to the formation of deposits of
elemental As, Sb, Se, or Te that block the metal surface for H entry and that there is
no such effect with Na
2
S solutions, probably because the thermodynamic conditions
for S formation are restrictive ad even deposited S does not form a protective film
[56,110].
The embrittlement of steels (stress corrosion cracking) in H
2
S-containing
media is of great industrial importance, so the effect of dissolved H
2
S deserves a
special analysis here. According to most authors [56,111,112], H
2
S enhances the
HER on iron. This is to be compared with similar effects observed on Fe single
crystals precovered by a fraction of a monolayer of adsorbed sulfur [53,103].
Classic explanations are that in acid medium H
2
S molecules adsorbed on the cathode
catalyze the proton discharge step by being proton transfer centers [56,112,114].
The HAR seems to be promoted only in the low pH region where H
2
S is dissolved
molecularly; the rate of H permeation through iron or steel increases with H
2
S
concentration, beginning from very low values [56].
The effects of surface oxides and dissolved H
2
S on the HER and the HAR
on/in steel have been investigated [111]. The effect of H
2
S on the HER and hydro-
gen permeation on/in passivated and anodically depassivated surfaces of steel was
analyzed, assuming that the HER occurs by the Volmer-Tafel path only. It was
concluded that H
2
S plays a multiple role: it increases the rates of iron dissolution
and proton discharge, probably by roughening the metal and removing the passivation
layer. It then poisons the chemical combination reaction on the depassivated surface,
thereby permitting a large fraction of the H atoms to enter the metal [111].
Proposed Mechanisms of Action of Promoters
From the many suggestions made in the past to explain the mechanism of action
of H entry promoters into ferrous metals, the more realistic ones are summarized
here. It was often considered that the promoter is adsorbed on the surface, although
for elements of columns 15 and 16 it was not clearly stated whether it is in elemental
or hydride from [108,115].
1. The promoter increases the strength of the M–H
ads
bond, thus decreasing the
rate of H–H combination and increasing the surface H coverage, which
thereby increases the permeation rate [116].
2. The promoter lowers the M–H
ads
bond energy (weakens the M–H
ads
bond).
This decreases the height of the energy barrier for H surface-bulk transfer
[31,54,108].
3. The promoter inhibits the recombination step(s), so that it increases the H
entry rate [33,73,107,111].
4. The promoter is adsorbed in the sites of H
ads
, lowering the probability of
finding a pair of H adsorbed at contiguous sites, necessary for H
2
formation;
it thus poisons the H chemical combination step, so a high concentration of
atomic H is built up on the surface, which increases the rate of H entry into
the metal [112,115].
Explanation 1 is valid only for a Vomer-Tafel path with θ
H
close to unity (see
Fig. 10b). Furthermore, the promoters are known to be poisons for adsorption of H
in the gas phase [100], adsorption of UPD H on Pt [41,42,101,117,118], and also
84 Protopopoff and Marcus
Copyright © 2002 Marcel Dekker, Inc.
adsorption of the HER intermediate (OPD H) [75,117,118]. They lower the H
adsorption capacity and are likely to lower the M–H
ads
bond strength [41,42,101].
Explanation 2 was proposed in Ref. 31 for a coupled Volmer-Tafel path and is
valid at constant current density, assumed to be due mainly to the combination rate
(see Table 2). However, a lowering of the rate constant for surface-bulk transfer
cannot by itself explain why H permeation is increased under potentiostatic
conditions [113]. At fixed potential, the weakening of the M–H
ads
bond strength,
associated with a strong reduction of the number of surface adsorption sites, should
lead to a reduction of the H coverage, which would probably be the determining factor.
Explanation 3, rather phenomenological, is often given for an increase
of permeation current under gavanostatic conditions, for an HER mechanism
assumed to occur only by the Volmer-Tafel path. In this case, the constant cathodic
current is related only to the electroadsorption rate, which is equal to the
sum of the combination rate plus the permeation rate [see Eq. 34]. If the
promoter inhibits the chemical combination step, the permeation rate is increased
[33,111]. This trivial explanation fails to explain promoter effects occurring
under potentiostatic conditions.
Explanation 4 is valid if the reaction of consumption of H
ads
is solely the
combination step: in addition to assuming the Volmer-Tafel path for the HER,
the permeation rate must be negligible to allow an increase of the H coverage
[see Eq. 33]. This classic explanation is apparently not self-consistent because it
proposes that a high H coverage may appear whereas the H sites are occupied by
poison, unless the sites for electroadsorption are supposed to be different from sites
for combination, which would conflict with the present view [27,53,58,59].
However, a modeling of promoter effects shows that the relative coverage in the
sites left active may well increase while, due to the blocking effect, the overall
coverage decreases [53,61].
There are two recent significant contributions to the understanding of the
mechanism of action of promoters on H permeation into iron:
1. Some authors, using the rather rough assumption that the mechanism of HER
on iron in acid is only coupled Volmer-Tafel but taking the permeation
current into account in the establishment of the steady-state equations
(so-called IPZ model), analyzed their data on iron in acid solutions
containing H
2
S and concluded that H
2
S increases the proton discharge rate
constant and lowers the combination rate constant, leading to a coupled
Volmer-Tafel mechanism at low overpotentials, a decreased overvoltage, an
increased H coverage, and an increased permeation rate [112]. Their
conclusions are essentially similar to explanation 4.
2. This explanation is contested in the latest permeation study [34], in which the
promoting effects of dissolved species of As on iron are analyzed with accurate
fitting of the experimental current-potential curves with the complete steady-state
equations and taking into account the possibility of reduction of adsorbed As at
cathodic potentials into the hydride AsH
3
; yet a simplifying choice of parameters
could not be avoided. It is concluded that the mechanism of HER cannot be or
become the coupled Volmer-Tafel one. It is once again observed that the H
entry is inhibited a low overpotentials, which is attributed to deposition of bulk
As, and that promoting effects of As occur only at relatively high overpotentials,
where As is likely to desorb into AsH
3
. From the fit, the authors deduce that the
Surface Effects on Hydrogen Entry into Metals 85
Copyright © 2002 Marcel Dekker, Inc.
