Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.


2.2 Interaction of radiation with matter 33
effect is not observed. The threshold frequency, ω
th
, which is equal to
ω
th
=
A
wf
h
-
, (2.58)
is called the cut-off frequency of the photoelectric effect.
For practical applications of the photoelectric effect an important quantitative
characteristic called the external quantum efficiency (or quantum efficiency), η,
is used. It defines the ratio of the number of emitted electrons to the number
of incident photons. Near the cut-off frequency of the photoelectric effect for
most metals the quantum efficiency is about η ∼ 10
−4
(electrons per photon).
The small efficiency in this frequency range is due to the fact that only those
electrons which are close to the surface of the metal possess sufficient energy
to leave the metal. At the same time the predominant portion of the radiation
incident on the metal is reflected. With increasing photon energy, i.e., increasing
frequency of the radiation, the quantum efficiency increases and η is about 10
−2
for the photons with energy close to 1 eV. For X-rays with the energy of photons
ε
ph
∼ 10
3
eV the quantum efficiency is about 10
−1
electrons per photon.
Example 2.3. Find the work function of an electron for an unknown metal, if after
exposure of its surface to light with wavelength λ = 0.35 µm the photocurrent
that occurs in the circuit of a photoelement disappears if the negative voltage
U
d
=−1.2 V is applied.
Reasoning. If we apply a negative (decelerating) voltage, U
d
, to the circuit with
the photoelement,
eU
d
=
m
e
v
2
max
2
, (2.59)
the photocurrent disappears. In this case Einstein’s equation for the photoelectric
effect can be rewritten in the form
h
-
ω = A
wf
+ eU
d
. (2.60)
Taking into account that ω = 2πc/λ, we obtain for the work function of the
unknown metal
A
wf
= h
-
2πc
λ
− eU
d
≈ 3.84 × 10
−19
J = 2.4eV. (2.61)
This unknown metal may be lithium, whose work function is equal to A
wf
=
2.39 eV.
2.2.2 X-ray bremsstrahlung
If the energy of a photon incident on a metal significantly exceeds the work
function of a metal, i.e., h
-
ω A
wf
, then A
wf
can be neglected in Einstein’s
equation (2.57) for the photoelectric effect. Thus, the equation can be simplified
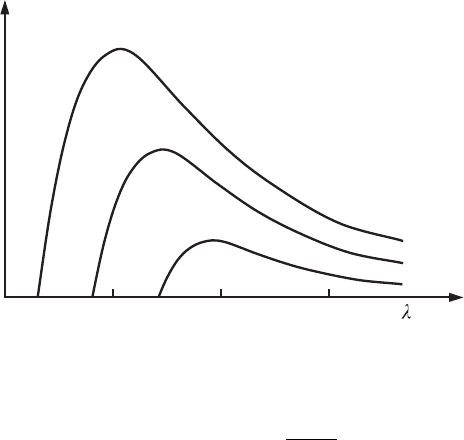
34 Wave–particle duality and its manifestation
2468
50 kV
35 kV
25 kV
I
(a.u.)
(nm)
Figure 2.9 The
dependences of the
distribution of the
intensity, I ,ofX-ray
bremsstrahlung on the
wavelength λ at
accelerating potentials U
equal to 50, 35, and
25 kV.
to
h
-
ω =
m
e
v
2
max
2
. (2.62)
In the X-ray range, where the energy of a quantum is h
-
ω ∼ 10
2
−10
3
eV, this
equation is valid with a high degree of precision since the work function is only
about A
wf
∼ 1eV.
Let us consider the process which is the reverse of the photoelectric effect, i.e.,
let us consider the transformation of the kinetic energy of electrons incident on a
metal into the energy of the photons that are created after a sudden deceleration
of electrons in the metal. If we take into account the fact that the electrons, in
order to gain the necessary kinetic energy, are accelerated by a corresponding
potential drop U, then Eq. (2.62) can be rewritten in the form
eU = h
-
ω. (2.63)
The process of acceleration of electrons takes place in the X-ray tube, where the
given potential drop is created between the cathode and the anode (anti-cathode).
After colliding with the anode the electrons abruptly begin to decelerate and,
because of this negative acceleration, the electrons emit electromagnetic waves
in the X-ray range. This radiation, which is called X-ray bremsstrahlung, has
a continuous electromagnetic spectrum. Figure 2.9 shows experimental depen-
dences of distributions of the radiation intensity on the wavelength, obtained for
a tungsten anode at several values of the accelerating potential drop U . The char-
acteristic feature of these curves is the existence of a short-wavelength cut-off
frequency. From the particle point of view this fact can be simply explained.
Since the radiation occurs because of the energy lost by an electron during its
deceleration, the energy of the photon cannot be greater than eU. Therefore,

2.2 Interaction of radiation with matter 35
there is a threshold frequency
ω
max
=
eU
h
-
, (2.64)
which is the highest for the given accelerating potential drop. Correspondingly,
the minimal wavelength in the spectrum of bremsstrahlung is defined as
λ
min
=
2π h
-
c
eU
. (2.65)
The existence of a short-wavelength cut-off frequency in the X-ray spectrum is
one of the most important quantum features of X-ray radiation. According to the
classical wave theory such a threshold in the radiation spectrum must not exist.
2.2.3 The Compton effect
In 1923, while studying scattering of hard X-ray radiation on samples consisting
of light atoms (graphite, paraffin, etc.), Arthur Compton discovered that the wave-
length λ
of the radiation scattered at an angle θ is greater than the wavelength λ
of the incident radiation. The wavelength of the scattered radiation depends on θ
according to the equation
λ
(θ) = λ + (1 −cos θ), (2.66)
where = 2.42 ×10
−12
m is a constant called the Compton wavelength of the
electron, which can be found experimentally.
Classical theory was unable to explain the appearance of the observed wave-
length shift of the radiation scattered at some angle. According to classical
concepts the scattering can be considered as a process during which an electron
undergoes forced motion under the influence of the electric field of an inci-
dent wave. At the same time the electron behaves as the source of secondary
(scattered) waves with the frequency of the incident radiation. The results of the
experiment become clear if the scattered radiation is considered as the conse-
quence of an elastic scattering of the photon on the free electron. For the light
atoms of the materials on which the experiments were carried out, the binding
energy of valence electrons with their atoms is smaller than the energy of the
X-ray photon. Therefore, these electrons can be considered as free electrons. In
such a case the formula (2.66) is a consequence of energy and momentum conser-
vation laws for the elastic collision of the photon (with energy h
-
ω = 2π h
-
c/λ and
momentum h
-
k) and the electron at rest with energy m
e
c
2
and zero momentum:
2π h
-
c
λ
+ m
e
c
2
=
2π h
-
c
λ
+ m(v)c
2
, (2.67)
h
-
k = h
-
k
+ m(v)v, (2.68)
where wavenumbers k and k
are equal to k = 2π/λ and k
= 2π/λ
, respectively.
For Eqs. (2.67) and (2.68) we used relativistic expressions for the energy and

36 Wave–particle duality and its manifestation
momentum of the electron because as a result of collision with an X-ray photon
the freed electron may become relativistic, i.e., it can acquire a velocity v that is
close to the speed of light, c. In these equations the mass of the electron depends
on its velocity in accordance with the relativistic equation
m(v) =
m
e
1 −v
2
/c
2
. (2.69)
Self-consistent solution of Eqs. (2.67)–(2.69) leads to the expression (2.66),
where
=
2π h
-
m
e
c
= 2.42 × 10
−12
m. (2.70)
By analyzing his results Compton proved that the photon is a particle that is
characterized not only by its energy but also by its momentum. This allows
application of the laws of conservation of energy and momentum to the processes
of interaction of photons with other particles.
Example 2.4. Show that the absorption of a photon in the case of its inelastic
collision with a free electron is not possible because it is prohibited by the laws
of conservation of energy and momentum.
Reasoning. Let us suggest that before collision with a photon the electron was
at rest. In this case the laws of conservation of energy and momentum can be
written in the form
h
-
ω + E
0
= E( p), (2.71)
h
-
ω
c
= p, (2.72)
where E
0
= m
e
c
2
is the energy of the electron at rest and the energy of the
moving electron is related to the momentum as
E( p) = c
m
2
e
c
2
+ p
2
. (2.73)
On squaring the left-hand and right-hand sides of Eqs. (2.71) and (2.72)we
obtain
h
-
2
ω
2
+ 2m
e
c
2
h
-
ω = c
2
p
2
, (2.74)
h
-
2
ω
2
= c
2
p
2
. (2.75)
At m
e
= 0 these two equalities contradict each other. From the above we can
draw the conclusion that an inelastic collision of a photon and a free electron is
impossible, i.e., a free electron cannot absorb a photon. Such a process can take
place in the case of the existence of a third particle, which will be able to take
part of the energy and/or momentum of the photon. In Compton’s experiment,
which we discussed previously, the third particle is the scattered photon.
2.2 Interaction of radiation with matter 37
2.2.4 Wave–particle duality
As a result of thorough experimental and theoretical analysis of the properties of
light its dual nature was established. In some processes light behaves as a wave
(interference, diffraction, etc.), whereas in others it behaves as a flux of particles
(blackbody radiation, the photoelectric effect, etc.). The equations
ε
ph
= h
-
ω, (2.76)
p
ph
= h
-
k (2.77)
relate the particle and wave properties of light. The left-hand sides of these
equations (with ε
ph
and p
ph
) characterize the photon as a particle and the right-
hand sides (with ω and k) characterize the photon as a wave.
There is an important trend in the observations of the dual nature of light. For
long-wavelength radiation (e.g., infrared radiation) its quantum properties are
not so obvious and mainly its wave properties are detected. However, on going to
the shorter wavelengths the quantum properties of light become more apparent.
Wave and quantum properties of light are connected, and they supplement each
other. The quantum properties of light become apparent by virtue of the fact
that the energy, momentum, and mass of radiation are concentrated in particles –
photons. The probability of finding photons at particular points of space is defined
by the amplitude of the light wave, i.e., by the wave properties of light.
The wave properties are inherent not only to large ensembles of photons,
but also to each individual photon. This is evident because it is not possible
to specify the location of a photon and the direction which it will have after a
collision with an obstacle. We can talk only about the probability of finding an
individual photon in one place or another. The description of the behavior of such
an object on the basis of classical laws is impossible. Nevertheless, experimental
facts allow us to state that this duality in light’s behavior is a law of nature. Light
was the first object that allowed physicists to observe and to interpret the wave–
particle duality of matter. The further development of physics greatly enhanced
the class of such objects. As will be discussed later, other particles, such as the
electron, proton, neutron, etc., may have wave properties too, which broadens
our knowledge about matter.
Example 2.5. An X-ray photon with frequency ν = 6 ×10
18
Hz scatters on a
free electron at angle θ = 90
◦
. Find the frequency of the scattered photon, as well
as the momentum, velocity, and energy of the electron after its collision with the
photon.
Reasoning. After the collision of a photon with a motionless electron, the wave-
length of the scattered photon increases, in accordance with Eq. (2.66), by the
magnitude
λ = (1 − cos θ), (2.78)

38 Wave–particle duality and its manifestation
p
e
k
k
'
Figure 2.10 Scattering of a
photon and an electron.
where is the Compton wavelength of the electron. Taking into account that the
scattering angle is θ = 90
◦
, we obtain
λ = λ
− λ = . (2.79)
Therefore, for the frequencies of the incident, ν, and scattered, ν
, photons we
have
1
ν
−
1
ν
=
c
. (2.80)
Taking into account that = 2.42 ×10
−12
m and c = 3 ×10
8
ms
−1
, we find
the frequency of the scattered photon:
ν
=
cν
c +ν
= 5.72 × 10
10
Hz. (2.81)
Let us rewrite the momentum conservation law for the considered collision:
h
-
k = h
-
k
+ p
e
. (2.82)
You can see from Fig. 2.10 that the momentum of the electron after collision
with the photon is
p
e
=
(h
-
k)
2
+ (h
-
k
)
2
, (2.83)
where for the wavenumber, k, of the incident photon the following equality is
valid:
k =
ω
c
=
2πν
c
. (2.84)

2.3 Wave properties of particles 39
The same equality can be written for k
. Taking this into consideration, we obtain
for the electron’s momentum, p
e
,
p
e
=
2π h
-
c
ν
2
+ ν
2
= 1.83 × 10
−23
kgms
−1
. (2.85)
The velocity of the electron is
v
e
=
p
e
m
e
= 2 × 10
7
ms
−1
. (2.86)
The energy of the electron can be found from the expression
E =
p
2
e
2m
e
= 1.84 × 10
−16
J = 1.15 × 10
3
eV. (2.87)
2.3 Wave properties of particles
2.3.1 De Broglie’s hypothesis
The understanding of the dual nature of electromagnetic radiation that was estab-
lished in the first quarter of the twentieth century allowed scientists to explain
many phenomena on the basis of the particle picture, according to which elec-
tromagnetic radiation is a flux of photons – the particles which have zero mass
at rest. The success of such a description of electromagnetic radiation evoked
interest in wave–particle concepts and led to its extrapolation to particles that
have a finite mass at rest. In 1924 Louis de Broglie suggested the idea of the
universality of Eq. (2.38), which related the momentum of a photon to its wave-
length. Any particle that has momentum p corresponds to a wave for which the
following equality is valid:
λ
Br
=
2π h
-
p
=
h
p
. (2.88)
This physical quantity λ
Br
is called the de Broglie wavelength of a particle.The
second main equality that relates the parameters of the particle and wave is the
relation between the energy of the particle and the de Broglie frequency:
ω
Br
=
E
h
-
. (2.89)
A free particle of energy E and momentum p corresponds to a plane wave with
frequency ω
Br
and wavevector
k =
p
h
-
. (2.90)
This wave can be described by the scalar function
(t, r) = Ce
−i(ωt−k·r)
= Ce
−i(Et−p·r)/h
-
. (2.91)
The de Broglie waves are not electromagnetic waves. We will discuss later their
particular nature and the physical meaning of the function .
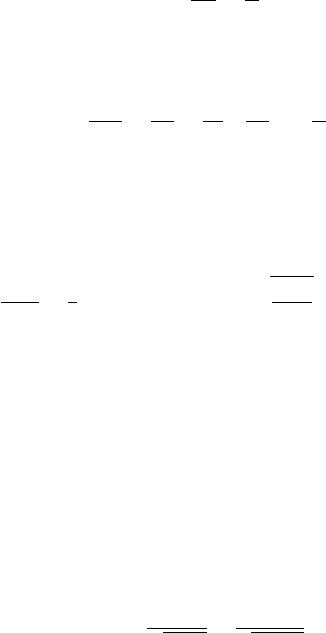
40 Wave–particle duality and its manifestation
The frequency and wavenumber of the de Broglie wave are related to the phase
and group velocities as follows. For the phase velocity we obtain
v
ph
=
ω
Br
k
=
E
p
. (2.92)
For the group velocity, taking into account that for a free particle E = p
2
/(2m),
we obtain
v
gr
=
dω
Br
dk
=
dE
d p
=
d
d p
p
2
2m
=
p
m
= v. (2.93)
Thus, the group velocity of the de Broglie wave coincides with the velocity of a
particle. Let us estimate the wavelength of the de Broglie wave of an electron for
various cases. For a free electron moving inside of a metal at room temperature
with thermal velocity, v
T
,
m
e
v
2
T
2
=
3
2
k
B
T and v
T
=
3k
B
T
m
e
≈ 10
5
ms
−1
. (2.94)
The de Broglie wavelength according to Eq. (2.88) is equal to λ
Br
≈ 7 × 10
−9
m,
while the distance between neighboring atoms in a crystal lattice is about a ≈
3 × 10
−10
m, λ
Br
a. The velocity of an electron rotating around a proton
in a hydrogen atom according to Bohr’s theory is v = 2 ×10
6
ms
−1
. Then,
λ
Br
≈ 4 × 10
−10
m, while the extent of the atom is about 10
−10
m, i.e., λ
Br
is of
the order of the atom’s size.
In an electric field with potential difference U an electron accelerated to
velocities less than the speed of light in vacuum gains kinetic energy K = eU .
Taking into account Eq. (2.88), we obtain for the de Broglie wavelength of the
electron
λ
Br
=
2π h
-
√
2m
e
K
=
2π h
-
√
2m
e
eU
. (2.95)
At U = 150 V the de Broglie wavelength is equal to λ
Br
≈ 10
−10
m ≡ 1
˚
A.
Let us estimate now λ
Br
for heavy particles, using the same procedure as for
the electron (see Eq. (2.94)). For a copper ion located at the site of the metal
crystal lattice (the mass of the ion is about m
Cu
≈ 10
−25
kg), at room temperature
the de Broglie wavelength is λ
Br
≈ 2 × 10
−11
m, which is one order of magnitude
less than the dimension of the ion and the distance between ions. If we consider
a macroscopic object even with a small mass, then its de Broglie wavelength
appears to be much less than the dimensions of this object. For example, for a
speck of dust with mass m ≈ 10
−9
kg and size 10
−5
m moving with the velocity
v ≈ 10
−2
ms
−1
, the de Broglie wavelength is λ
Br
≈ 10
−22
m.
Since there is no important difference between microobjects and macroob-
jects, the following question arises: when are the wave properties of particles
pronounced and when do the particle properties dominate? To answer this
question let us use the analogy with optics. The wave nature of light becomes
2.3 Wave properties of particles 41
S
C
D
Figure 2.11 A schematic
diagram of the
Davisson–Germer
experiment on the
diffraction of electrons by
a monocrystal of nickel.
The source of electrons is
denoted as S, the mobile
detector of electrons as D,
and the monocrystal of
nickel as C.
maximally apparent at wavelengths comparable to the characteristic dimensions
of obstacles, i.e., λ ∼ L (for example the phenomena of interference and diffrac-
tion). If λ L, then the wave properties become negligible and we can use
geometrical optics (rectilinear propagation of rays). If we extrapolate this idea
to particle waves, i.e., to de Broglie waves, then the wave properties of particles
will be more apparent if their dimensions are less than or of the order of the de
Broglie wavelength λ
Br
. If the dimensions of a particle are much larger than λ
Br
,
then the wave properties are weakly pronounced and they can be neglected. The
first situation (L ≤ λ
Br
) occurs for electrons in metals and in atoms. The second
situation (L λ
Br
) occurs for an electron in an electron-beam tube, for a copper
ion, and for a speck of dust.
2.3.2 Experimental verification
The de Broglie hypothesis about the existence of wave properties of particles
received experimental confirmation in 1927 in the Davisson–Germer experiment,
which involved scattering of electrons on a nickel monocrystal. The scheme of
the experiment is shown in Fig. 2.11. In the electron gun S there was produced a
beam of electrons, whose energy and velocity were defined by the accelerating
voltage. A narrow beam of monoenergetic electrons was incident on the surface
of a nickel crystal C, and was reflected from it. The crystal could be rotated,
changing the angle of incidence of the electron beam. The detector D registered
the number of electrons scattered by the crystal in different directions. Intensity
measurements were carried out at a fixed angle of incidence of the electron beam
by changing the magnitude of the accelerating voltage.
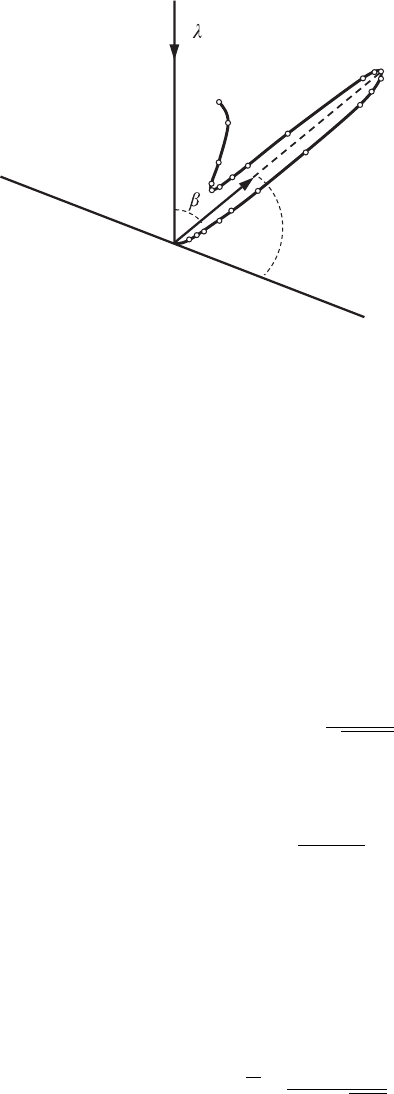
42 Wave–particle duality and its manifestation
q = 65
°
Crystallographic plane
U = 54 V
= 0.167 nm
Figure 2.12 The angle
distribution of reflected
electrons in one of the
diffraction maxima.
If electrons behaved as classical particles, they would have had to reflect
from the crystal in accordance with the laws of geometrical optics, i.e., the
incidence angle would have had to be equal to the reflection angle. However,
the experiments showed that the intensities of scattered electrons in different
directions differ. There was also alternation of the maxima and minima of the
number of electrons scattered at different angles, i.e., in short the phenomenon
of electron diffraction was verified. Analogously to the diffraction of X-rays, the
theoretical analysis of the diffraction of electrons by a crystal can be carried out
using Bragg’s law of diffraction Eq. (B.177),
2d sin θ = nλ
Br
, (2.96)
where, instead of X-rays of wavelength λ, the de Broglie wavelength of the
electron, λ
Br
, is used. Taking into account Eq. (2.95), Eq. (2.96) can be rewritten
as
2d sin θ = n
2π h
-
√
2m
e
eU
. (2.97)
The angle β between incident and reflected rays is related to the angle θ in
Eq. (2.96) via the equation
θ =
180
◦
− β
2
. (2.98)
The measurements were carried out at a fixed voltage U and various angles
θ, as well as at a fixed angle θ and different voltages U .InFig.2.12 the results
of measurements that correspond to a fixed voltage U = 54 V are shown. The
diffraction maximum becomes clearly pronounced at an angle θ = 65
◦
.The
results of measurements for the fixed angle θ = 65
◦
and various values of U are
showninFig.2.13.IfwesolveEq.(2.97) for the square root of the accelerating
voltage we get the equation
√
U =
π h
-
d sin θ
√
2em
e
n. (2.99)
