Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

2.3 Wave properties of particles 43
51015200
0.5
1
I (a.u.)
U ( V )
1/2
n = 2
n = 3
n = 4
n = 5
n = 6
n = 7
Figure 2.13 The
dependence of the
intensity of the electron
beam on the square root
of the accelerating voltage
for its diffraction by a
monocrystal of nickel.
(
a
)(
b
)
Figure 2.14 The
distribution of diffracted
electrons over the surface
of a photographic plate
for (a) a short and (b) a
long time of exposure.
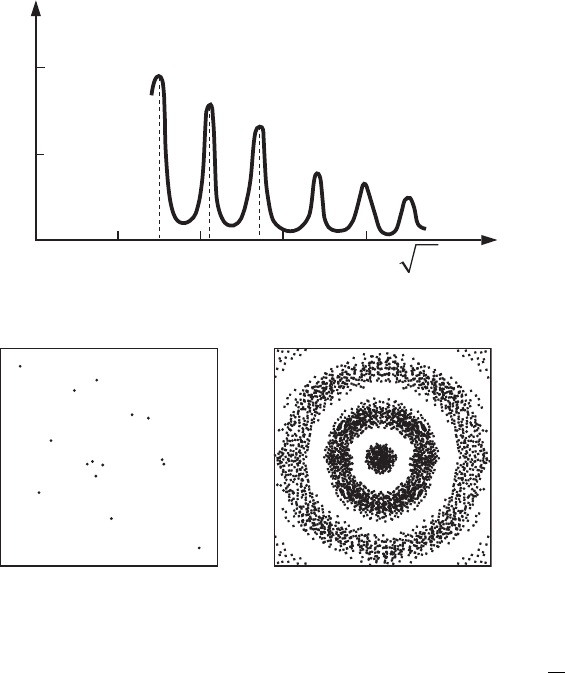
Since n is an integer number, the diffraction maxima on the axis
√
U must be
distributed equidistantly, and this is clearly seen from the results of the experiment
(see Fig. 2.13).
The de Broglie wavelength (at U = 54 V) calculated with the help of Eq. (2.95)
is equal to λ
Br
= 0.167 nm. At the same time the corresponding wavelength
found from Eq. (2.96) for the lattice parameter d = 0.215 nm and n = 2 is equal
to λ
Br
= 0.165 nm. The results obtained from this experiment convincingly
confirmed the de Broglie hypothesis.
Diffraction experiments studying the wave properties of individual electrons
are of great interest. The size of the electron beam in these experiments is
chosen to be so small that only one electron at a time is incident on the narrow
metallic film. The electrons transmitted through the film form diffraction patterns
in the form of concentric circles on a photographic plate after a sufficiently
long exposure time. At short exposure times the points on the photographic
plate, which correspond to incident electrons, are randomly distributed (see
Fig. 2.14(a)). However, with an increase in the exposure time the distribution of

44 Wave–particle duality and its manifestation
points acquires the characteristic form for the diffraction of X-ray radiation by a
polycrystalline sample (see Fig. 2.14(b)).
Note that not only electrons possess wave properties. In 1936 the diffraction
of neutrons by a crystal was observed for the first time. Neutrons are particles that
are found in atomic nuclei and have mass m
n
= 1.67 × 10
−27
kg. If the velocity
of the incident neutrons and the lattice constant of the crystal are known, then,
according to Bragg’s law, Eq. (2.96), the de Broglie wavelength of a neutron can
be found. For thermal neutrons, the energy is defined as
E =
3
2
k
B
T, (2.100)
and we obtain for the de Broglie wavelength of the neutron
λ
Br
=
2π h
-
√
3m
n
k
B
T
. (2.101)
At room temperature the de Broglie wavelength of a neutron is λ
Br
≈ 0.1nm.
The diffraction of neutrons has a wide range of applications in the study of solid-
state structures. Wave properties of atoms and molecules were also discovered.
For atomic or molecular beams incident on the surface of a crystal additional
diffraction maxima were observed in addition to specular reflection, for which
the angle of incidence is equal to the angle of reflection.
2.3.3 The criterion governing the classical and quantum
properties of a particle
The dimensionality of Planck’s constant, h, is [J s]. The same dimensionality,
[J s], can be obtained for the following products of two quantities: (1) energy and
time, (2) momentum and coordinate, and (3) angular momentum and angle. The
quantity with such a dimensionality, S, is called an action. Therefore, Planck’s
constant, which has the same dimensionality, can be called the quantum of action.
If for an object the value of an action is comparable to the value of h, then the
behavior of such an object must be governed by the quantum laws. If the value
of an action, S, is much larger than Planck’s constant, h, then the behavior of
this object is described with high precision by classical physics. Let us note that
the smallness of an action does not always correspond to the non-applicability
of a classical approach. In many cases the classical approach gives a qualitative
picture of the behavior of the object studied, which can be refined further with
the help of a quantum approach.
Among particles the photon has the smallest possible action. The energy of
a photon is equal to ε
ph
= h
-
ω and the characteristic time is equal to the period
T
ph
= 2π/ω. Therefore, the value of an action for the photon is
S
ph
= 2π h
-
= h, (2.102)

2.3 Wave properties of particles 45
i.e., S
ph
is comparable to the quantum of action, h. It is obvious that photons
obey quantum laws.
For comparison let us calculate the action of an ion that is undergoing thermal
oscillations in a crystal at room temperature with a frequency ω
ion
∼ 10
8
s
−1
.The
average energy of thermal oscillations is E = k
B
T , and the period of oscillations
is equal to T
ion
= 2π/ω. Therefore, the ion’s action is
S
ion
= k
B
T
2π
ω
≈ 2.5 × 10
−28
Js. (2.103)
The ratio of the ion’s action to the quantum of action, h, in the case being
considered is S
ion
/ h ≈ 4 ×10
5
. Thus, the behavior of an ion in a crystal is
described sufficiently well by the laws of classical physics.
Example 2.6. A parallel beam of non-relativistic electrons accelerated by the
potential difference U is incident normally at a diaphragm with two narrow slits
with the distance between the slits being equal to d. Find the distance between the
neighboring maxima in the interference pattern on a screen located at a distance
l, which is much greater than the distance between the slits d, i.e., l d.
Reasoning. Using the analogy with wave optics (see Fig. B9(a)), we can state that
the interference maxima are formed in the place where the path-length difference
of rays contains an integer number of de Broglie waves, i.e.,
L ≈ r
2
−r
1
= mλ
Br
. (2.104)
Since the first interference maxima are observed at small angles (θ 1), the
path-length difference, L, can be presented as
L = d sin θ ≈ θ d. (2.105)
At the same time the coordinate of the mth interference maximum, x
m
, is much
smaller than the distance between the slits, d, and that to the screen, l. Therefore,
we can consider that θ ≈ x
m
/l. Thus, for the coordinate x
m
we obtain
x
m
= m
lλ
Br
d
. (2.106)
The maximum corresponding to zero path-length difference of the de Broglie
waves is located at the point x = 0. The number of the next interference maximum
changes by unity and its coordinate changes by a magnitude
x = x
m+1
− x
m
=
lλ
Br
d
, (2.107)
which is called the width of the interference fringe. On substituting the expression
for the de Broglie wavelength (2.95) into Eq. (2.107) we get
x =
2π h
-
l
d
√
2m
e
eU
. (2.108)

46 Wave–particle duality and its manifestation
From the last expression it follows that with increasing accelerating potential
difference, U , the distance between the interference maxima of electron waves
for two-slit interference decreases.
Example 2.7. Using the expression for the phase velocity of de Broglie waves
write the refraction constant of these waves passing through the inner potential
of a crystal U
0
and applied potential U.
Reasoning. An electron in a metal is affected by an inner crystalline field, which
is created by the positively charged lattice ions. This field changes periodically
from ion to ion. The magnitude of the potential of this field averaged over the
volume of a crystal is called the inner crystalline potential. To extract an electron
from a metal it is necessary to spend energy equal to the work function A
wf
= eU
0
(see Fig. 2.8). As we would do in optics, let us define the refractive index, n,of
the de Broglie waves as the ratio of the phase velocities of these waves in vacuum
and in a medium, i.e.,
n =
v
vac
v
med
=
ωk
med
ωk
vac
=
λ
vac
Br
λ
med
Br
. (2.109)
Taking into account Eq. (2.95), according to which the de Broglie wavelength is
inversely proportional to the square root of an electron’s kinetic energy, we can
write
n =
K + A
wf
K
=
1 +
A
wf
K
. (2.110)
On writing the energy through the potential, U
0
, we obtain
n =
1 +
U
0
U
. (2.111)
Since the work function of metals tends to be several electron-volts for electrons,
which are subjected to the high accelerating potential difference (U > 100 V and
U U
0
), the index of refraction is practically equal to unity.
Example 2.8. Find the de Broglie wavelength of an electron that is moving with
velocity v, at which the electron mass is equal to double the electron mass at rest.
Reasoning. The relativistic mass of a moving electron is defined by the expression
m(v) =
m
e
1 −v
2
/c
2
. (2.112)
Let us introduce the variable η = m
e
/m(v). Then, the velocity of an electron is
equal to
1 − v
2
/c
2
=
m
e
m(v)
= η, (2.113)
v = c
1 − η
2
. (2.114)
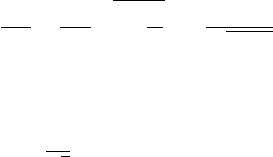
2.4 The uncertainty relations 47
The de Broglie wavelength of an electron moving with velocity comparable to
the speed of light in vacuum is equal to
λ
Br
=
2π h
-
p
=
2π h
-
m
e
v
1 −
v
2
c
2
=
η
1 −η
2
, (2.115)
where we introduced the Compton wavelength (Eq. (2.70)): = 2π h
-
/(m
e
c) =
2.42 × 10
−12
m. In our case η = 1/2, and
λ
Br
=
√
3
= 1.4 × 10
−12
m. (2.116)
Thus, the wave properties of relativistic (v ≤ c) electrons can be revealed when
they are in a volume with linear dimensions of about 10
−12
m (e.g., in the vicinity
of an atomic nucleus).
2.4 The uncertainty relations
In classical physics a complete description of a particle’s state is defined by
its dynamic parameters (coordinate, momentum, torque, energy, and so on).
However, the real behavior of microscopic particles shows that there is a limit
in principle to the precision with which the above-mentioned parameters can be
measured. The limits of application to the classical description of the behavior
of microscopic particles are determined by the uncertainty relations, which were
suggested by Werner Heisenberg in 1927.
The most important among them are the following two relations. The first
one relates the uncertainty in the values of the particle’s coordinate, r
α
,tothe
uncertainty in the corresponding component of momentum, p
α
,atthesame
instant in time:
r
α
p
α
≥ h, (2.117)
where α = x, y, and z. Because of the importance of these relations we will
rewrite them for each projection on the Cartesian coordinate axes:
x p
x
≥ h,y p
y
≥ h,z p
z
≥ h. (2.118)
The second expression, which relates the uncertainty in a change of the energy
of a particle to the uncertainty in time during which this change takes place, is
E t ≥ h. (2.119)
The impossibility of simultaneous exact measurement of the quantities which are
involved in the uncertainty relations is the manifestation of the dual wave–particle
nature of microscopic objects. The uncertainty relations (2.117)–(2.119)are
fundamental laws of nature and they are confirmed by all available experimental
data. Application of the uncertainty relations to specific physical systems allows
us not only to establish the limits of application of classical theory, but also to

48 Wave–particle duality and its manifestation
obtain numerical estimates of parameters of systems under study without using
complicated mathematical analysis and draw important conclusions about the
character of the processes involved.
2.4.1 The uncertainty relations of coordinate and momentum
for a particle
In the case of macroscopic objects the applicability of classical concepts is
clear. Let us consider for example a flying speck of dust (sufficiently small
from an everyday point of view) with mass m ∼ 10
−7
kg and velocity in the
x-direction v
x
∼ 0.1ms
−1
. If we define this velocity by available standard optical
methods with a precision v
x
≈ 10
−5
ms
−1
, then the unavoidable uncertainty
in measurements of the particle’s coordinate according to relation (2.118)is
x ≈
h
p
x
=
h
m v
x
≈ 10
−21
m. (2.120)
The size of a speck of dust with mass 0.1 mg depends on the material composing
this speck and is of the order of 10
−5
m. The uncertainty of measurement of its
location is 16 orders of magnitude less than its dimensions, and it lies far beyond
our possibility to make measurements of a coordinate with such precision. In
this case the behavior of the particle is entirely described by the classical laws of
physics.
Let us study the applicability of the different theories for the description
of the behavior of an electron. It turns out that there is no definite answer in
advance. The answer solely depends on the conditions of the electron’s state. For
example, in an electron tube with an accelerating potential difference U ∼ 10
4
V,
an electron acquires momentum
p =
2m
e
eU ≈ 5.4 ×10
−23
kg m s
−1
. (2.121)
This momentum is directed along the tube’s axis. The diameter of the focussed
beam can reach values of d ≈ 10
−5
m. This magnitude defines the uncertainty
in the electron’s position on the screen. According to the uncertainty relations
(2.117) the electron has non-controllable momentum in the transverse direction
p
⊥
≈
h
d
≈ 10
−29
kg m s
−1
. (2.122)
Since p
⊥
/ p 1 the motion of the electron follows a specific trajectory and
can be described by the laws of classical physics.
For the electron in a hydrogen atom the radius of the first Bohr orbit is equal
to r ≈ 5 ×10
−11
m. In accordance with Newton’s second law of motion the
acceleration, a, of the electron along the circular orbit is defined by the Coulomb
force:
m
e
a =
k
e
e
2
r
2
. (2.123)
2.4 The uncertainty relations 49
For a circular orbit,
a =
v
2
r
. (2.124)
By combining Eqs. (2.123) and (2.124) we obtain the magnitude of the electron’s
momentum:
p = e
k
e
m
e
r
≈ 2 × 10
−24
kg m s
−1
. (2.125)
Let us use now the uncertainty relations. Since the electron is localized in an
atom, the uncertainty of its coordinate is equal to the dimension of an atom, i.e.,
r ≈ 2r. The corresponding uncertainty of momentum is
p ≈
h
r
≈
h
2r
≈ 6.6 × 10
−24
kg m s
−1
. (2.126)
The uncertainty of momentum, p, is comparable to the magnitude of the
momentum, p. Thus, the laws of classical physics cannot describe the behavior
of such a particle and it is necessary to apply the quantum approach instead.
2.4.2 The uncertainty relations of energy and time
for a particle
The detailed consideration of the process for the measurement of energy shows
that the energy of a system can be measured only with a finite uncertainty, E,
i.e., E cannot be equal to zero. It follows from relation (2.119) that the energy
conservation principle for a system can be verified by two measurements with
accuracy h/t, where t is the time interval of measurement. The process of
measurement with t →∞is unrealistic; this is why E = 0.
Let us apply the energy–time uncertainty relation to a system that can be in
a state with energy E for a finite time t. After this time the system makes
a transition to another energy state. In this case, taking into account relation
(2.119), we can write
E t ≥ h, (2.127)
where E is the unavoidable uncertainty of the energy for that state. This form
of the uncertainty relation allows us to estimate the finite width of the energy
state of the atomic system, which remains in that state for a finite time t.
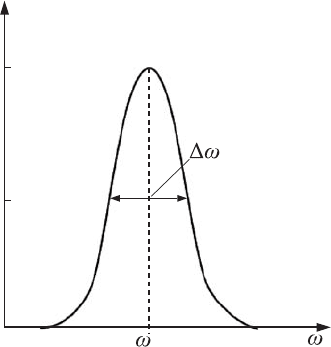
It is well known that the atomic emission spectral lines are not infinitely
narrow – this would correspond to an uncertainty in the radiation of a quantum
of energy of E = 0. The spectral lines that are observed in experiments have a
so-called natural linewidth ω (see Fig. 2.15). The width of this spectral line is
determined by the spread of the photon’s energy values E = h
-
ω with respect
to the mean value that characterizes the center of the line E = h
-
ω. This width
50 Wave–particle duality and its manifestation
i
0
0.5
1.0
I
Figure 2.15 The form of
an atom’s emission
spectral line.
is related to the lifetime of an atom in the excited state, t, by the uncertainty
relation
h
-
ω t ≈ h, or ω t ≈ 2π. (2.128)
By experimentally measuring the natural linewidth we can find, with the help of
relation (2.127), the lifetime of an atom in the corresponding excited state. The
natural linewidth which corresponds to the visible range of emission of atoms is
of the order of ω ∼ 10
8
s
−1
. Therefore, the atomic lifetime in the excited state
is about t ∼ 10
−8
s. Taking into account that the frequency in the visible range
is about ω ≈ 4 × 10
15
s
−1
, we can find the relative width of the emission spectral
line: ω/ω ≈ 2.5 ×10
−8
. Note that in the ground state, i.e., in the non-excited
state, stable (light) atoms can have indefinitely long lifetimes, i.e., in this state the
lifetime of an atom τ →∞, while the width of the energy level that corresponds
to the ground state E → 0.
The wave properties of particles and the probabilistic character of de Broglie
waves indicate that the description of a particle’s motion has to be very different
from the classical description. In Newton’s classical mechanics the motion of a
particle under the influence of a force is completely determined by the initial
position and velocity of the particle. By solving Newton’s equation we can find
the trajectory, coordinates, and velocity of the particle at any instant with any
degree of accuracy, which is limited only by the accuracy of the measuring
devices used. From the uncertainty relations it follows that, in cases in which
the uncertainty of a coordinate is comparable to the dimension of the region
where the particle is moving, the notion of the trajectory loses its meaning. The
simultaneous determination of a particle’s position and its velocity cannot be
done with an arbitrary degree of accuracy. This is true for the pairs of physical
quantities whose product has the dimension of Planck’s constant. From this
discussion follows the necessity of creating a more complete description of

2.4 The uncertainty relations 51
physical phenomena, in which particle and wave properties of the objects under
study can be revealed. We can consider the year 1926 as the beginning of the
emergence of such a theory, when Erwin Schr
¨
odinger discovered the equation
for the description of microscopic particle behavior.
Example 2.9. Using the uncertainty relations, we can relate a particle’s coordi-
nate and its de Broglie wavelength. Find the condition under which we can neglect
quantum effects if the particle occupies a region with linear dimension equal
to L.
Reasoning. According to the uncertainty relations (2.118) for the uncertainty of
the coordinate we have
x ≥
h
p
x
. (2.129)
The smallest value of x corresponds to the largest value of p
x
according to
the above uncertainty relation. Since p
x
cannot exceed the total momentum of
a particle p,
x ≥ h/ p. (2.130)
Taking into account the relation between the momentum and the de Broglie
wavelength (Eq. (2.88)), we can obtain from Eq. (2.130)
x ≥ λ
Br
. (2.131)
The notion of a particle’s trajectory can be used only when the uncertainty of
its coordinate is small compared with the characteristic dimension of the region
where it moves, i.e., x L. Therefore, the condition for correctness of the
classical description of a particle’s behavior is
λ
Br
L. (2.132)
When the size of the region, L, becomes of the order of λ
Br
the quantum effects
cannot be neglected, as shown in the example discussed earlier.
Example 2.10. An electron is in a spherical cavity with impenetrable walls (a
quantum dot). The dimension (diameter) of this confined region is d = 3nm.
Estimate the minimum energy of the electron.
Reasoning. In this case the uncertainty of the electron’s coordinate r ≈ d
and the uncertainty of the momentum is comparable to the magnitude of the
momentum itself: p ≈ p. The electron’s momentum is related to its energy as
p =
2m
e
E. (2.133)
Taking into account the uncertainty relations, we have
r p = d
2m
e
E ≥ h. (2.134)

52 Wave–particle duality and its manifestation
From the last inequality for the minimal energy of the electron we obtain
E
min
=
h
2
2m
e
d
2
, (2.135)
which gives E
min
≈ 2.6 × 10
−20
J ≈ 0.16 eV, i.e., an electron cannot have energy
smaller than E
min
. Because E
min
is finite, the electron cannot be in the state of
rest that corresponds to E = 0 and p = 0.
2.5 The world of the nanoscale and the wavefunction
The study of processes and phenomena involving microscopic particles, where
the wave properties are taken into account, is carried out in the framework of the
so-called wave mechanics, which is also frequently called quantum mechanics.
For the description of a particle’s motion well-known classical quantities such as
the position vector r, momentum p, energy E, etc. are used. The state of a particle
at a given instant is defined by a number of physical quantities, i.e., by a set of
variables. The significant peculiarity of the quantum-mechanical description is
that this set includes a smaller number of variables than is necessary for a classical
description.
The quantum-mechanical description uses three variables. These variables
can be either the three projections of r(t) or the three projections of p(t ), but
not both. In classical physics all six variables, r(t) and p(t), are required for
the description of a particle’s state. Usually the coordinates r(t ) are chosen for
the quantum-mechanical description as the total set of variables. Then, such a
description of a particle’s state is called the coordinate representation.
According to the uncertainty principle (2.118), in quantum mechanics it is
impossible to measure simultaneously a particle’s coordinate and its momentum.
From the physical point of view this is equivalent to the absence of a trajectory
for the particle. Indeed, a trajectory is a series of sequential close positions of
a particle, which are defined by the position vectors at close instants in time:
r(t
0
), r(t
1
), r(t
2
), . . .. To define the position of a particle at an instant in time t
k
it is necessary to know its position at the previous instant in time, t
k−1
, and its
momentum at that time, p(t
k−1
), i.e.,
r(t
k
) = r(t
k−1
) + (t
k
− t
k−1
)
p(t
k−1
)
m
. (2.136)
The impossibility of simultaneous definition of position and momentum leads to
the impossibility of indicating the exact position of the particle at any given time.
Therefore, the motion of a particle cannot be described by its position vector r
as a function of time. For any microscopic process it makes sense to talk only
about the probability of finding a certain value of a physical quantity, i.e., the
quantum-mechanical description is a probabilistic description.
Another feature of the description on a microscopic scale lies in the fact that
some values of physical quantities cannot be measured because these values are
