Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.

compressor with sufficient extra capacity to compensate for the drop in the long
hoses. The air is freed from oil, moisture, and dust by means of an oil-water separator.
Air spraying is a much faster application technique than brushing or rolling;
moreover, the layers obtained are more uniform in thickness. The disadvantage of
this technique is the larger waste. Part of the paint is lost in the so-called overspray:
missing the object to be coated. Another cause of paint loss is the rebound of the
air containing paint spray from the surface of the object to be painted. Paint losses
can be excessively high when spraying is done outdoors.
The directions for spraying distance, pressure, and nozzle type are rather
critical and should always be followed. Distance from surface should normally be
45–60 cm. If the spray gun is held too far from the substrate, the paint droplets are
already dry when they reach the surface and will not aggregate. This phenomenon,
called dry spray, gives a discontinuous, porous film.
Dry spray may also be expected in the case of highly pigmented products
(for example, zinc silicate) or paints containing very volatile solvents (for example,
vinyl paints). This phenomenon may also be expected when the paint material is
insufficiently thinned or spraying is done during hot or windy weather.
Airless Spraying
The paint is brought to a high pressure (75–300 bar) by means of a pump and forced
though a narrow nozzle, which atomizes the paint. In contrast to air spraying, in this
case the paint is not atomized by air and consequently the amounts of spray mist,
overspray, and rebound are much less than in air spraying. Because the paint is not
mixed with air, there is almost no risk of contamination with impurities such as oil or
water. Further advantages are:
Significantly higher production rates: faster spraying.
Paint can be applied at a higher viscosity, resulting in higher dry film thickness.
Loss of solvents and thinner by evaporation is much lower than in air spraying.
The technique is also suitable for more volatile solvents.
Airless spraying is in particular appropriate for painting surfaces, such as
buildings, ships, and offshore structures. It has the following disadvantages:
The worker has to work in a very fast and concentrated way because of the high
rate of deposition.
It is unsuitable for spraying small objects.
To obtain thin layers, very narrow nozzles are requited. Consequently, they
become warm (particularly with paints that contain hard pigments and
extenders) and malfunction.
The paint jet has such a high velocity that it can injure workers.
Electrostatic Spraying
In this process atomized paint and the object to be painted are respectively negatively
and positively charged. This results in a good adhesion and a uniform layer.
Further, it is an economical process because of the low waste, and application
of water-based paints is possible. Disadvantages are the restriction to coat objects
only on the outside and the occurrence of defects due to an incorrect viscosity/
solvent balance.
694 de Wit et al.
Copyright © 2002 Marcel Dekker, Inc.
Powder Coating
Powder coating can be considered as a variation of the previous technique. A constant
flow of powder, fluidized in an air stream, passes through a highly charged
(40–150V) region of ionizing discharge. The charged paint particles are directed
to the (grounded) substrate. Together with the electrostatic attraction, an enforced
airflow increases the velocity of the particles to the substrate. After deposition, the
layer is fused in an oven at 180° for 10 to 15 min, depending on the characteristics of
the powder. Coated thicknesses of up to 500 μm are possible with these coatings
without loss of flexibility; thickness between 300 and 450 μm are more common.
Electrodeposition
Charged paint particles in a solution of water-soluble resins are directed by a
potential field between cathode and anode (respectively the object to be painted
and bath tank or vice versa) to the surface of the object.
Advantages are that a uniform coating is obtained, penetration in narrow
places (for example, seams) is possible, the process is fast, and there is no fire risk.
The main disadvantage is the limitation of the thickness (due to the electrical
resistance of the formed coating). The limit is 25 μm and only one coat is possible.
PROTECTIVE MECHANISMS
The corrosion protection provided by organic coatings results either from the barrier
action of the layer, which may be supported by inert inorganic particles especially
if these are arranged like tiles (e.g., aluminum flakes or iron phosphates), or from
active corrosion inhibition provided by pigments in the coating. Ideally, a coating
provides protection by forming a physical barrier between the metal substrate and
the aqueous corrosive environment. In practice, however, these physical barrier
properties are limited, as all organic coatings are permeable to water and oxygen
to a certain extent. This, however, mostly does not influence the protective action
of the coating as long as the coating adheres well. In fact, adhesion is the primary
protection criterion. Permeation of the coating with water is not damaging as long
as no condensation of water at the metal/coating interface takes place.
Corrosion under a coating can take place only after an electrochemical double
layer has been established at the metal surface. For this to occur, the adhesion
between the coating and the substrate must be broken, after which a separate thin
water layer at the interface can be formed when the water permeates the coating.
Under normal, outdoors conditions an organic coating is saturated with water
at least half its service life. For the remainder of the time it contains a quantity
of water comparable in its behavior to an atmosphere of high humidity [50].
Furthermore, the average transmission rate of water through a coating is about 10
to 100 times larger than the water consumption rate of a freely corroding surface
[13]. Because it has also been established that in most cases the diffusion of oxygen
through the coating is large enough to facilitate unlimited corrosion, it is clear that
the physical barrier properties alone may not account for the protective action of
imperfectly adherent coatings.
Organic Coatings 695
Copyright © 2002 Marcel Dekker, Inc.
Resistance inhibition, which is also part of the barrier mechanism, may
supply additional protection. Inhibiting the charge transport between cathodic and
anodic sites retards the corrosion reaction. An increase in the electronic resistance
and/or the ionic resistance in the corrosion cycle may reduce the reaction rate. The
electronic resistance may, for example, be increased by the formation of an oxide
film on the metal. This is the case for aluminum substrates. The application of
organic coatings on a metal surface results in an increase of the ionic resistance.
One of the weak points of organic coatings in corrosion prevention is the fact
that these coatings are relatively easily damaged under mechanical and thermal
load. This may cause corrosion under the paint film at and near the damaged site.
The otherwise adequate barrier properties of the coating will no longer give sufficient
protection. Active pigments are then often incorporated in the polymer matrix of
the first coating layer near the substrate: the primer. These pigments (passivating,
blocking, or galvanic) provide protection through an active inhibitive mechanism
immediately when water and some corrosive agent reach the metal surface.
Again, protection can result only if the adhesion of the coating is good. Also, the
mechanical properties (e.g., the glass transition temperature) of the polymer reflect to
some extent the quality of the coating, as these determine the formability of coated
substrates (e.g., for coil-coated products) and their sensitivity to external damage.
Water Permeation
All organic coatings are to some extent permeable to water. The effective
permeability is closely related to the polymer structure and composition. The
permeability of a coating is often given in terms of the permeation coefficient P.
This is defined as the product of the solubility of water in the coating (S, kg/m
3
), the
diffusion coefficient of water in the coating (D, m
2
/s), and the specific mass of
water ρ (kg/m
3
). Different coatings can have the same permeation coefficient even
though the solubility and the diffusion coefficient, both being material constants, are
very different. Therefore the usefulness of the permeation coefficient is limited.
Water permeation occurs under the influence of various driving forces:
A concentration gradient. e.g., during immersion or during exposure to a humid
atmosphere, resulting in true diffusion through the polymer
Osmosis due to impurities or corrosion products at the interface between the metal
and the coating
Capillary forces in the coating due to poor curing, improper solvent evaporation,
bad interaction between binder and additives, or entrapment of air during
application
When a coated system is exposed to an aqueous solution or a humid atmosphere,
water molecules eventually reach the coating substrate interface. Normally, a coating
under immersion will be saturated after a relatively short time (of the order of 1 h),
depending on the values for D and S and the thickness of the layer. Typically values
for D and S are 10
13
m
2
s
–1
and 3% [7–9]. For atmospheric exposure the actual
cyclic behavior of the temperature and the humidity determines largely the periods
of saturation. In any case, situations will result in which water molecules reach the
coating-metal interface, where they can interfere with the bonding between the
two phases, eventually resulting in loss of adhesion and corrosion initiation if a
696 de Wit et al.
Copyright © 2002 Marcel Dekker, Inc.
cathodic reaction can take place. For a corrosion reaction to proceed, a constant
supply of a cathodic species such as water or oxygen is required. Some authors
suggest that the transport rate of oxygen through the coating is higher using the
interconnected water phases in a saturated polymer [51,52], whereas other
experiments lead to the conclusion that the oxygen permeation is independent of
the presence of water [53].
Dickie [12] determined the quantities of water and oxygen consumed
during corrosion of a freely corroding unpainted steel substrate. Both were of
the same order of magnitude and also comparable to the average permeability
of coatings for oxygen. An overview of some data is given in Table I. This leads
to the conclusion that for some coatings with somewhat lower permeability for
oxygen the transport of oxygen may be rate determining. Water permeation
may also result in the buildup of high osmotic pressures, which are responsible
for blistering and delamination.
CORROSION UNDERNEATH ORGANIC COATINGS
The causes of proceeding delamination underneath organic coatings can be roughly
categorized in two types: delamination due to clustering of water at the interface and
delamination caused by specific corrosion processes that produce either low or high
Organic Coatings 697
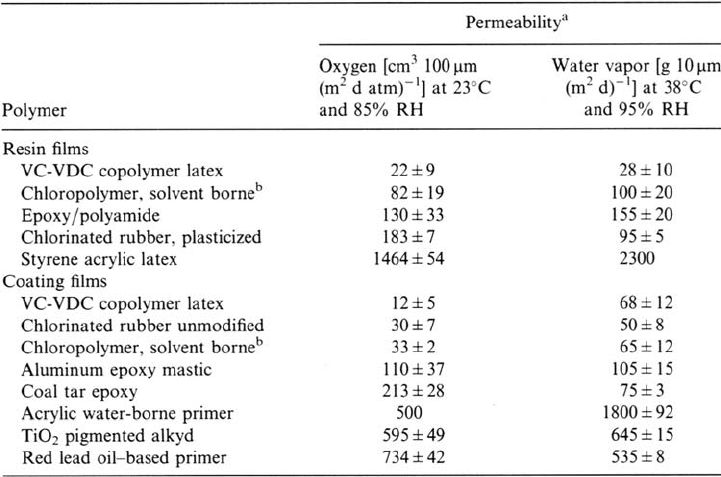
Table 1 Permeability of Oxygen and Water Vapor in Several Resin and Coating Films:
Results Obtained for Free Films That Were Cast on Glass
a
The permeability of oxygen is given as the number of cm
3
gas of 1 atm permeating through a coating
of 100 μm thickness per m
2
per day.
b
Product under development.
Source: Ref. 54.
Copyright © 2002 Marcel Dekker, Inc.
pH values at the interface. In the case of blisters underneath an intact paint layer, the
first mechanism will often be the starting point for the more “chemical” delamination
caused by processes of the second category. Within the first category wet adhesion
and osmotic blistering are the main mechanisms. The second category is mainly
referred to as cathodic or anodic delamination.
Clustering of Water
Clustering of water at the metal-polymer interface and the subsequent formation
of an electrochemical double layer can occur only if the adhesion between metal
and coating is weaker than the bond between metal and water or polymer and
water. For a proper evaluation, however, a distinction must be made between wet
adhesion and osmotic blistering.
Wet adhesion is the complete deadherence of the coating without actual
blistering. From a thermodynamic point of view this implies that the whole
interface must have very weak binding energy. Therefore the bonds will be easily
broken by water that can be transported through the coating at a sufficient rate.
Fast temperature cycling of saturated coatings may, however, also lead to this type
of delamination although the adherence under static conditions is sufficient.
Typical of wet adhesion is the absence of real blisters in the first stages. This is
due to the fact that the water at the interface is relatively pure and as long as corrosion
processes have not generated enough ions, osmotic mechanisms will not cause a further
growth of the water layer. Therefore the adhesion may also be partly restored when
coatings are completely dried after immersion. Only when ions are present due to
contamination or are formed by corrosion processes may further blistering occur.
Osmotic blistering is caused by the presence of soluble species underneath
the coating. Water that normally penetrates the coatings will dissolve these species
at the interface and form a highly concentrated solution. Also, some acid fumes
containing acetic on hydrochloric acid may penetrate through polymers and
condense together with water in small voids or places with minor adhesion. Due
to osmotic forces, more water will then be attracted to these solutions. This
causes an internal pressure within the blisters, which has been calculated to be
as high as 3 Pa [5,6]. These high internal pressures may even cause further
delamination. Because corrosion processes within the blister tend to produce their
own ions, the osmotic pressures may remain high for long times. In many cases
these blisters will eventually burst or gradually develop into the electrochemical
delamintation mechanisms described in the following.
Cathodic Delamination and Anodic Undermining
In the literature two mechanisms are proposed to describe the propagation of
underfilm corrosion in case of intact and defective coatings. These mechanisms are:
Cathodic delamination
Anodic undermining
In cathodic delamination the adhesion of the coating fails and causes lateral
blister growth as a result of a high pH at the delamination front [6,55,56]. The loss of
698 de Wit et al.
Copyright © 2002 Marcel Dekker, Inc.
adhesion in anodic undermining is caused by the dissolution of the metal or the
metal oxide at the interface with the coating [6].
Both cathodic delamination and anodic undermining are the result of a specific
type of electrochemical cell: the differential aeration cell. In this cell separation of
anodic and cathodic reaction sites takes place, but for both mechanisms this
happens in different ways depending on the path for oxygen transport to the metal
interface. This transport is, of course, also determined by whether the coating
is intact of defective. With intact coatings, oxygen is mainly supplied through
the coating or through both coating and formed solid corrosion products as
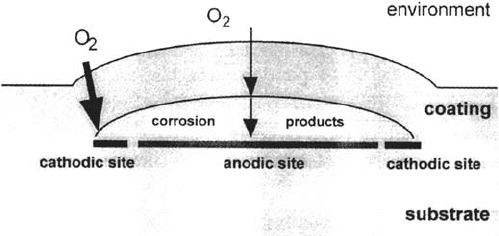
schematically shown in Figure 5. The length of the oxygen transport path to the
interface at the edge of the blister is shorter than that at other places in the blister
where oxygen also has to diffuse through the corrosion products. According to the
differential aeration cell theory, the cathodic reaction will occur at the edge of the
blister and the anodic reaction in the center of the blister as shown in Figure 5.
The separation of the anodic and cathodic reaction sites in Figure 5 can be
promoted by the nature of the corrosion products. When corrosion products consist
of species that can be further oxidized, oxygen may be reduced during the transport
through the corrosion products. The amount of oxygen that will reach the metal
will therefore collapse and in the center of the blister less oxygen will reach the
metal surface. An example is the formed corrosion product of iron, which initially
consists mainly of Fe(II). In the presence of oxygen, Fe(II) will be oxidized to
Fe(III) and will therefore consume the oxygen [7,12]. Because the solubility product
of Fe(III) (hydr)oxides is very low, a solid film is formed. The proceeding
corrosion process leads to film growth at the center of the blister, which hinders
further oxygen transport at the center effectively.
From the preceding discussions it is concluded that blisters under intact
coatings grow due to cathodic delamination.
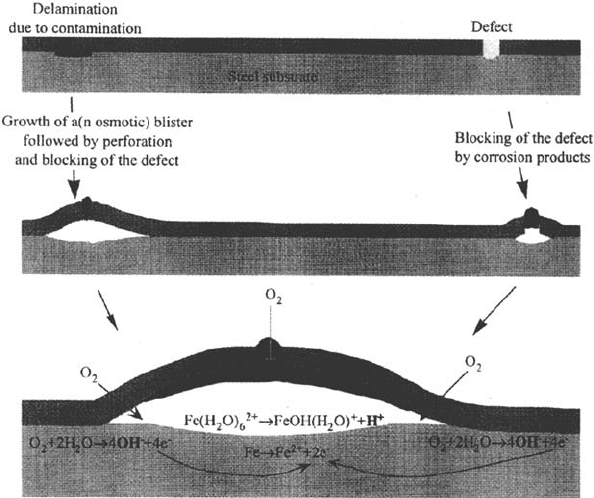
When defective coatings are considered, the situation is much more complex.
Part of the substrate is now directly exposed to the corrosive solution. Corrosion
will initiate at the defect and subsequently the formed corrosion products will
block the pore. Corrosion propagation depends on the nature of the corrosion
products in the pore. These stages are shown in Figure 6a and b.
Organic Coatings 699
Figure 5 Oxygen transport paths in a blister in case of an intact coating. The resulting
anodic and cathodic sites are shown. The transport paths for oxygen are shown.
Copyright © 2002 Marcel Dekker, Inc.

Whether anodic undermining or cathodic delamination will develop at
defective coatings depends on the ratio of the transport rates of oxygen through
the coating and that through the formed corrosion products. When the rate of
oxygen transport through the coating exceeds that through the corrosion products,
the mechanism is cathodic delamination; the opposite leads to anodic undermining.
The Model as Presented in the Literature
In more detail, the model of cathodic delamination can be described and understood
as follows [3,57]: the delamination starts with randomly distributed anodes and
cathodes in a defect or at a dalaminated area. When iron is taken as an example, the
dissolved Fe
2+
at the anode is further oxidized to Fe
3+
by oxygen and forms
insoluble corrosion products in the defect that often adhere to the polymer (in the case
of a blister) and at the edges of the defect (where oxygen enters the defect). In what
way the defect is blocked and a cap of corrosion products is formed on the top of the
blister; see Figure 7. In this cap oxygen is consumed through oxidation of Fe
2
(the
cap is sometimes said to be impermeable to oxygen [7]). Because of this, the area
underneath the cap will have a lower oxygen concentration than the regions at the
edges where no cap is present and where oxygen “easily” penetrates the coating. This
stimulates the separation of anodic and cathodic regions, which finally leads to large
pH differences due to the different reactions that take place at anode and cathods
(½ O
2
+ H
2
O + 2e
–
→ 2OH
–
at the cathode and Fe → Fe
2+
+ 2e
–
followed by
subsequent hydrolysis at the anode). This process is shown schematically in
Figure 7 for a delaminated and a defective coating. In both cases the same end situ-
ation results.
Taking a closer look at this model, some questions arise that are not directly
answered by the model as presented normally:
What is the mechanism of growth of these blisters?
How can these large pH differences that are reported in literature exist for
prolonged periods of time in such a confined space (differences of 10 pH
units over less than a few mm)?
Despite these remaining questions, the model could explain recent model
experiments on test panels that were deliberately contaminated with sodium chloride.
On most panels a large (set of) blister(s) was present at the contaminated site, but
700 de Wit et al.
Figure 6 (a) Corrosion initiation at defective coatings. (b) Blocking of the pore. Initial
loss of coating adhesion at the edges of the pinhole is shown.
Copyright © 2002 Marcel Dekker, Inc.
also other, small blister were present at other places on the panel. In between these
blisters, the polished metal underneath the transparent coating still has a mirror-like
surface. Closer examination showed, however, that even on these apparently
undamaged sites there was a thin layer of liquid present between coating and metal.
When the completely deadhered coating was lifted off, the pH of this thin layer of
liquid was “measured” with a pH indicator liquid. It was approximately 12.
Underneath the larger blisters the metal was etched as can be seen in Figure 8.
This can only be the result of the attack by a quite acid solution. Below pH 3,
however, hydrogen evolution may occur. Sometimes the presence of hydrogen
bubbles on steel is reported in the literature [58], but in all cases the cathodic
disbondment is caused by cathodic protection. As very little solutions be available
within the center of the blister, it is extremely difficult to measure the pH in this area.
Judging from the metal attack is should, however, be around pH 3.
Figure 8 also shows the undamaged cathodic areas around the blisters. These
observations lead to the conclusion that the large pH differences do indeed exit.
Observations on smaller blisters give the answer to the question how these
large pH differences can exist in such small areas. It appeared that in the case of
very small blisters a closed sphere of corrosion products remained when the coating
was removed. So there is not only a “cap” as presented in Figure 7 but also “walls”
between anodic and cathodic sites. Together with the cap, the walls form a closed
sphere of iron (hydr)oxides. This result can be observed only on very small blisters
because larger ones are damaged when the coating is removed. Aphotograph of such
Organic Coatings 701
Figure 7 Schematic representation of the mechanism of cathodic delamination starting
from a defect and from an osmotic blister.
Copyright © 2002 Marcel Dekker, Inc.

a small blister is shown in Figure 9. In this picture one can also see the beginning of
the etched surface that is also present in Figure 8. To look inside the blister, the top
cap was in this case removed with a needle (it is still visible at the top of the picture).
Around the blister again a fully reflective surface is visible; only some debris from
the opening of the sphere of corrosion products is present.
The formation of the walls of the sphere can be understood considering the ions
that are present in the system: at the cathode OH
–
will be present in rather high
concentrations. In the center, the anodic region, Fe ions are present in rather high
702 de Wit et al.
Figure 8 Photograph of a large blister after removal of the coating and the corrosion
product (enlargement 20 times).
Figure 9 Photograph of a small blister. The top cap is visible at the top (enlargement 50
times).
Copyright © 2002 Marcel Dekker, Inc.

concentrations. Low soluble iron (hydr)oxides will precipitate after interdiffusion
of OH
–
and Fe
2+
at the boundary region [59]. These precipitates form a wall that
is connected with the cap, thus forming a closed sphere.
Extended Model for Cathodic Delamination
From the preceding observations the model for cathodic delamination can be
extended in the following way:
1. Initially, the normal model as presented by Funke [3,57] describes the
delamination process.
2. After some time the anodic and cathodic areas become separated as a barrier
of corrosion products is formed in between them. This wall stabilizes the pH
differences that occur due to the separation of the anodic and cathodic
reactions. Wall and cap together form the observed sphere
3. The wall slowly dissolves from the inside in the increasingly acid
environment within the sphere. Fe ions diffuse through the wall, resulting in
the formation of new Fe(OH)
2
-like products on the outside. In this way the
anodic area of the blister is able to grow. The high pH on the cathodic
edges causes the delamination of the organic coating to proceed (as was
already assumed in the existing model)
The mechanism of growth implies that the center of the anodic area has been
exposed for a longer period to the acid corrosive environment than the edges of
the anodic area. As a consequence of this a shallow “crater” must be formed under
every blister. Figure 10 shows that this is indeed the case.
Another importance consequence of the introduction of the wall is that
contaminating ions such as Na
+
and Cl
–
are necessary in both the anodic and the
cathodic region for cathodic delamination to occur: the Fe ions cannot compensate
the charge of the OH
–
; they would directly produce a solid product in the cathodic
region. On the other hand, in the anodic areas the H
+
and Fe
n+
should be
compensated by a negative ion such as Cl
–
(OH
–
would, of course, react directly with
H
+
). So the positive and negative ions of the contaminating salt will be separated.
This further implies that the final pH values are also determined by the amount of
Organic Coatings 703
Figure 10 Cross section of a metal with a shallow “crater” due to cathodic delamination
of a coating. The coating was pulled off before the sample was embedded and the cross
section was made. The black areas are formed by a crevice between the metal and the
embedding. The white line parallel to the metal surface indicates the (exaggerated)
formation of a crater. The cross section was embedded as a metallographic specimen and
photographed with an optical microscope.
Copyright © 2002 Marcel Dekker, Inc.
