Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.


Combining these assumptions, this leads to a comparative diffusion coefficient of
water D
*
, which can be calculated using:
714 de Wit et al.
In which RH1 and RH2 are the relative humidities of the atmospheres (of course,
this D
*
is not a real diffusion coefficient, but it can be used for comparison of the
results). When this equation is used to calculate the D
*
value of each step in the
cycles of Figures 15 and 16, it gives the results presented in Table 2.
From these data, despite the rough method, it is clear that there are quite
large variations in D
*
as a function of the RH. The higher the concentration of
water in the coating, the higher the D
*
. This can be expected because of the
plasticising effect of water on epoxies [94]. As expected, both grids do have
comparable values for D
*
.
Impedance Measurements and Delamination
EIS can also be used to measure and characterize defects in coating. This because
a defect will also have a specific impedance response. Not very defect is, however,
visible in measurements and care must therefore be taken in the interpretation of
the data. To show this, two essentially identical panels were tested and will be
referred to as cases A and B. On both panels a 100-μm-thick industrial coating was
applied over a salt film that was present on the metal surface. As a result of this,
both panels showed delamination and osmotic blistering as soon as they were
exposed to an electrolyte.
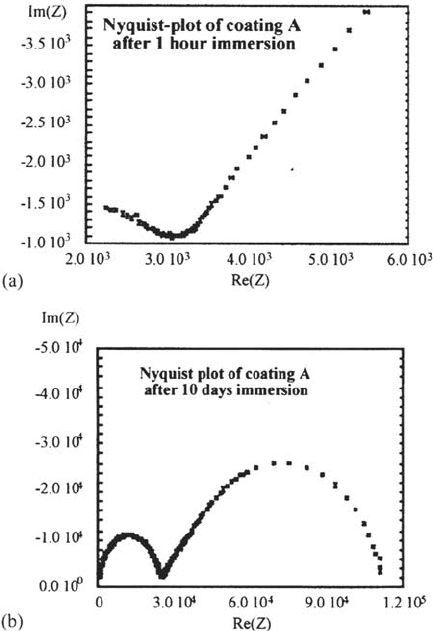
Directly after immersion, case A showed a Nyquist and Bode plot that deviated
from the expected plots for intact coatings. Representative measurements after 1 h
and 10 days are given by Figure 17a and b.
Analysis of the data with equivalent circuits showed that at least four
overlapping time constants were present. This implies that a rather complex
equivalent circuit should be used to explain the data and that it is not sufficient for
a fundamental study to monitor only one frequency or phase angle [95]. A complete
analysis of these data is beyond the scope of this chapter, but an important conclusion
is that the corrosion component in the equivalent circuit is in parallel with the
coating component. This implies a real defect that penetrates through the coating.
Prolonged exposure indeed showed corrosion products coming out of a small pore in
the coating.
Table 2 Comparative Diffusion Coefficient for Two Cycles and Different Changes in
Atmospheres as Calculated from the Results of Figures 15 and 16
a
a
Values can be used only for comparison within this table.
Copyright © 2002 Marcel Dekker, Inc.
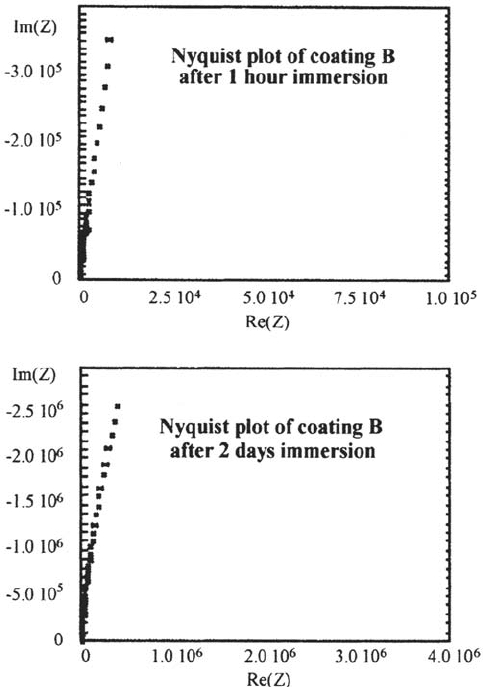
The almost identical panel B has a completely different start upon exposure. Here
we see a normal, capacitive behavior after 1 h of immersion as can be seen in Figure
18a. After 2 days this still has not changed, as can be seen in Nyquist plot of Figure
18b. This behavior is observed for several days. However, after 2 days delamination
is already clearly visible as a small blister above the contaminated area. So in this case
it is clear that, although a defect is present, it is not (easily) visible in the EIS data.
The corrosion potential of panel B also appeared to be unstable. Several times
the open-circuit potential drifted to –3000 mV versus SCE. Every time this happened
it was polarized at a normal potential for steel in the electrolyte. This unstable
behavior is due to the very high resistance of the coating, which in this case exceeds
the input impedance of equipment used.
These observations support the conclusion that the coating is completely
intact although partly delaminated due to the contamination underneath.
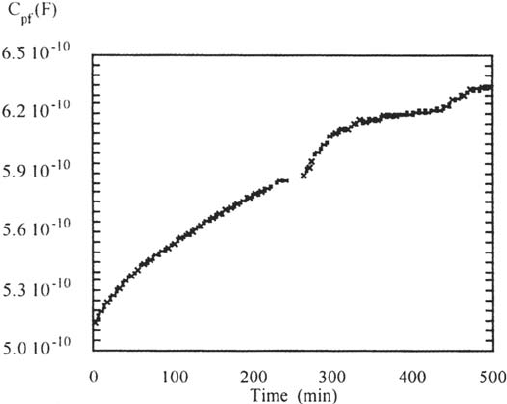
From the measurement of the water uptake in the first hours of immersion,
some qualitative information on this delamination can be obtained. From the
coating capacitance (C
pf
) against time plot of Figure 19 it is clear that the coating
does not show stable Fickian water uptake but shows deviations that are probably
Organic Coatings 715
Figure 17 Two Nyquist plots of measurement on coating A: (a) after 1 h and (b) after
10 days of exposure to water (plots are not isotropic).
Copyright © 2002 Marcel Dekker, Inc.
caused by the delamination. Because the delamination is forced and is already
starting when the coating is still taking up water, the water uptake and the deviation
from normal saturation behavior are not visible separately.
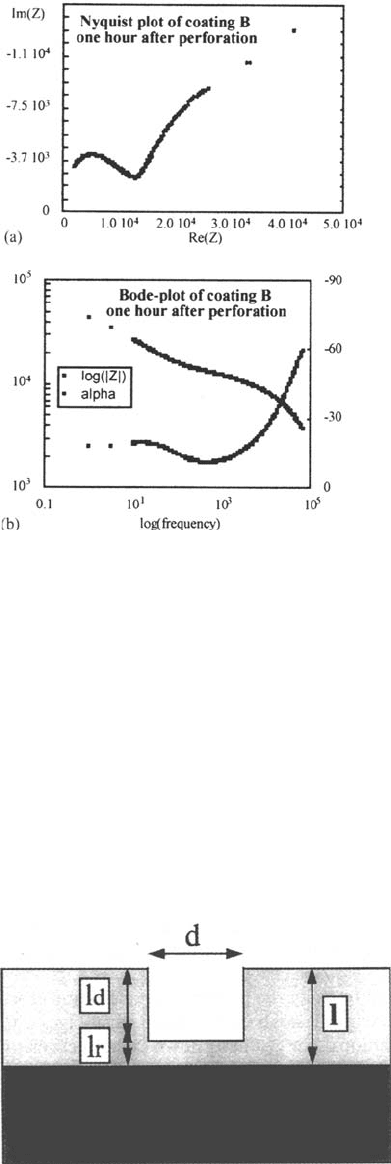
To further prove the similarity between panels A and B, the blister on panel
B was perforated deliberately with a needle. Directly after this the impedance
spectrum of Figure 20a and b was measured, which is rather similar to the data
measured on panel A. Because it is known that defect and delamination are in this
case combined, an equivalent circuit with a corrosion component in parallel to the
coating impedance must be used if further analysis is to be performed.
From the results of the measurements just described it is clear that quantitative
measurements on delamination under an intact coating are not possible with
impedance measurements as long as the impedance of the systems remains high,
which is often the case for thicker heavy-duty systems. In terms of equivalent circuits
that are used to explain impedance measurements, this implies that the corrosion
716 de Wit et al.
Figure 18 (a and b) Two Nyquist plots of measurement on coating B (plots are not
isotropic).
Copyright © 2002 Marcel Dekker, Inc.
component of the circuit has to be in series with the intact coating components. In
the literature this corrosion component is often placed in series with a relatively
low resistor, meaning that the properties of the coating have already changed
considerably (e.g., porosity and other flaws have developed).
Impedance Measurements and Artificial Defects, Laser Ablation
Often organic coatings do not fail early even under severe laboratory conditions.
Therefore it is necessary to make artificial defects in a coating system to study the
corrosion initiation under a coating. Both standard testing by exposure of standard
panels with or without mechanically induced scribes and laboratory research into the
mechanisms of the reactions suffer from the rather uncontrolled application method
for these artificial defects. The width and the height of the scribe largely determine
the mechanisms. Sometimes, for complicated systems with more layers as in car
body panels, one wants to make sure that a certain layer is still intact, and for
mechanistic studies in the laboratory one wants to study the very early stages of the
delamination. In both cases, artificial defects made with the laser ablation technique
may provide the answer [96–98].
Defects in surface coatings are used to study the protective properties of these
coatings. Defects made by ablation with excimer lasers have important advantages
with respect to traditional techniques. A resolution of the geometry down to the
micrometer scale with high precision and good edge definition is feasible, yielding
little, if any, distortion of the remaining coating layer: no coloring, foaming, or
melting. Moreover, the results show no ruptures and no delamination of the coating
layer from the substrate material. Normally, subsequent laser pulses perform the
etching of a defect until the underlying metal substrate is reached. The etching is
automatically stopped at the metal surface, as the threshold for ablation of the metal
higher than the threshold for organic materials. The laser beam will not change the
remaining metal surface.
Organic Coatings 717
Figure 19 Coating capacitance as a function of time during water uptake of coating B.
Copyright © 2002 Marcel Dekker, Inc.
The fine control over the ablation process makes another feature possible. If the
etching is interrupted just before the metal is reached, a defect with a thin (several
μm) residual layer on the bottom can be produced, as illustrated schematically
in Figure 21. It is this defect that proved to have ideal characteristics for broad
characterization of organic coatings, including electrochemical impedance
spectroscopy.
718 de Wit et al.
Figure 20 Nyquist and Bode plots measured on coating B directly after perforation. (b)
Log Z is given on the left axis and alpha on the right axis.
Figure 21 Typical geometry of an artificial laser defect in an organic coating. d = defect
diameter, I
d
= defect depth, and I
r
= thickness of the residual coating layer in the defect.
Copyright © 2002 Marcel Dekker, Inc.
After impedance measurements have been performed, meaningful physical
information on the system may be obtained only from a correct analysis and
interpretation of the data as discussed before. Impedance analysis may be performed
at different levels of complexity. The easiest form is a visual inspection of the
measured impedance diagrams or changes in these diagrams with time. This may
give first insight into the performance of a specific system. More detailed information
on mechanisms and corrosion phenomena may be obtained from an elaborate
analysis procedure. In Figure 22 the different stages that may be distinguished in
the behavior of a coated system as a function of exposure time to water are
illustrated schematically. At different exposure times the coated system, with laser
defect, is shown with the typical shape of the impedance response and the
corresponding general equivalent circuits. The following stages are distinguished:
A. Water permeation
B. Corrosion initiation
C. Quasi-stationary corrosion
It should be stressed that the very start of the corrosion process is visible in the
changing appearance of the Nyquist plot as given in Figure 22, but no quantitative
interpretation of the impedance data is possible because of the relatively fast
changes of the system. When the quasi-stationary stage, C, has been reached,
detailed quantitative analysis is again possible, leading to a more detailed equivalent
circuit supported by a physical model. It must be stressed that data from different
techniques are always needed when searching for a detailed physical model. Also,
the changes in time during exposure of the various components of the equivalent
circuit can be used to check the practical value of a proposed model.
Local Electrochemical Test Methods
The local electrochemical techniques that are used for coatings research are mainly
used in the more fundamental studies of corrosion mechanisms. They can be divided
into techniques in immersion (SVET end SRET) and in atmospheric conditions
(Kelvin). All techniques are based on the observation that corrosion mechanisms
underneath coatings are generally mechanisms in which anodic and cathodic
processes take place on separated sites with a slightly different local “corrosion
potential.” The techniques are used to visualize these differences. SRET and
SVET can also be used on polarized samples to detect pinholes.
SRET is a technique in which a reference electrode is scanned over a surface.
Asecond reference electrode is used to monitor the overall corrosion potential. In this
way, small differences in the potential can be visualized. An alternative type of SRET
has a tip with two platinum needles that are both placed close to the surface. If local
potential differences exist over the surface, a corrosion current will run through the
solution. In that case a small potential drop will occur in the solution between the
two platinum wires. Assuming an ohmic behavior for the solution resistance, this
potential difference is proportional to the potential differences on the surface.
Adrawback of the SRET is the fact that it has to measure very small differences
in a direct current mode. It is therefore not very sensitive and in most cases the
substrate has to be polarized to increase the current that is flowing through the
Organic Coatings 719
Copyright © 2002 Marcel Dekker, Inc.

720 de Wit et al.
Figure 22 Different stages in behavior of a coated system with progressing exposure time to water. (Left to right) System, impedance response,
and equivalent circuit Stages: (A) Water permeation; (B) corrosion initiation; (C) quasi-stationary corrosion. (R = resistance, C = capacitance,
Z = diffusion impedance, pf = paint film, cat = cathodic, an = anodic, dl = double layer, u = electrolyte.)
Copyright © 2002 Marcel Dekker, Inc.
solution. In this mode it can be used only to show the location and size of defects in
a coating.
A more sensitive technique is measuring the same IR drop in the solution by
the SVET. In the SVET the potential of a small vibrating tip is measured. If this
tip is vibrating in a potential gradient, the resulting potential vibration in the tip is
easily detected (AC mode) with sensitive electronics. Although more accurate,
this method is much slower than the SRET.
Adrawback of both methods is that complex corrosion mechanisms underneath
coatings including total delamination cannot be determined. Calculations with current
distribution models [99] can easily show that in the case of underfilm corrosion
almost no corrosion currents will be present in the outside electrolyte. Polarization
of the substrate is therefore necessary to show the presence of defects. This will,
however, at the same time influence the corrosion mechanisms.
The Kelvin probe is, unlike the other techniques, able to measure through an
insulating coating layer. Lord Kelvin used the principle of the Kelvin probe to show
that a Volta potential difference exists between metals. Apart from the obviously
modern electronics, the principle of the Kelvin probe is still the same: if two metal
plates are coupled through a wire and are brought close to each other, a potential
difference that is equal to the Volta potential difference between the two metals
exists over the narrow gap between the plates. As a result of this, the metal plates
will have a capacitive charge. If one of the plates is now vibrated, the capacity
between the plates is varied and as a result of this an AC current will flow through
the external circuit with a magnitude of
I
ac
= ΔVδC/δt
in which ΔV is the Volta potential difference between the two metals and δC/δt the
variation of the capacitance in time due to the vibration. If in this circuit an external
DC voltage source U is introduced, this equation changes to
I
ac
= (ΔV–U)δC/δt
From this equation it becomes clear that for the case in which the external DC
voltage U is equal to ΔV, the AC current I
ac
goes to zero, independent of δC/δt. This
implies that with this measurement it is even possible to measure through
vacuum, air, or an organic coating as this will influence only the term δC/δt.
Stratmann showed in several articles published around 1987 that for metals
with a thin moisture layer the Volta potential difference was proportional to the
corrosion potential of that system [100]. Using a calibration, it is therefore
possible to measure the corrosion potential of a corroding system through an air
gap and without making contact.
Although powerful, this technique is still under further development.
Especially the interpretation of the measured results is still a matter of discussion.
Without doubt it will in the future bring more and more results, especially on specific
corrosion mechanisms underneath coatings and on atmospheric corrosion. Recent
developments have shown that it is even possible to combine the extremely high
resolution of the AFM with the measuring principle of the Kelvin probe. For special
systems (uncoated), this may reveal even more details of corrosion mechanisms.
Organic Coatings 721
Copyright © 2002 Marcel Dekker, Inc.
Other Electrochemical Methods
Among the other electrochemical methods are the measurement of the corrosion
potential and the determination of the polarization resistance (R
p
). Both methods
are relatively simple but may give relevant information.
The polarization resistance is determined as the slope of a polarization curve
at the corrosion potential at the point where the external current is zero. In this way
it is a linearization of the more complex polarization curve. The value of R
p
/cm
2
is
directly related to the reactivity of the exposed metal substrate through the Stern
and Geary relation. For a coated system the total value of R
p
is also determined by
the amount of metal that is exposed. Both a higher reactivity and a larger exposed
area lead to a lower R
p
. If for a given system the R
p
is rather constant in time, this
technique can be used to predict the corrosion rate. In closed metal containers as
used for beer and soft drinks only a minimum amount of oxygen in present in a
normally rather acidic electrolyte. In that situation, no solid corrosion products are
formed and the R
p
is constant over time. In these drinks the amount of iron after a
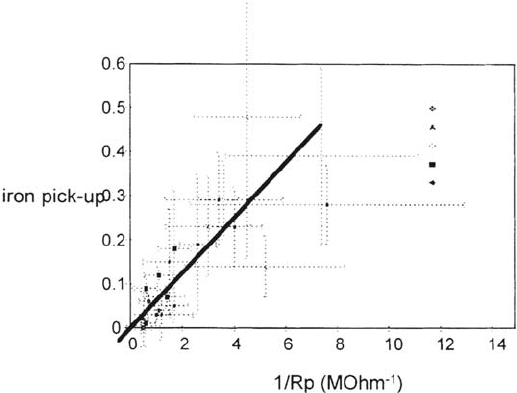
certain shelf life is an important quality check. Figure 23 shows the correlation
between 1/R
p
as measured after 2 weeks and the iron pickup of the drinks after 6
months. The R
p
was measured over a 20-mV range around the corrosion potential
of the system in the beverage itself (in this case cola, which has a pH of around 2.5).
In the case that no defect is present in the coating, an R
p
measurement is
measuring only the normally very high resistance of the polymer layer. In that case,
the obtained value should be similar (if measurable) to the values obtained from
fitting an impedance measurement.
722 de Wit et al.
Figure 23 Iron content of the electrolyte after 6 months of exposure versus polarization
resistance. Iron pickup in mg/L. Different points represent different series of filled cans
corresponding to various closing conditions.
Copyright © 2002 Marcel Dekker, Inc.
The Value of Enhanced Weathering Tests
The recent developments of electrochemical testing procedures, of which we have
discussed two, may result in better system lifetime prediction methods and monitoring
techniques for coating systems than are now available. Many new coating systems
are being developed as a result of the various environmental demands and legislation
together with demands for high quality for improved lifetime.
The industrial need for fast testing methods is therefore enormous because
testing by, e.g., atmospheric exposure of the systems takes too much time. Many
fast testing procedures exist. One of the first enhanced weathering tests was the
exposure of coated panels in Florida, where 3 years of exposure would be more or
less similar to 10 years of exposure in Europe because of the high temperature and
humidity in Florida. However, many new tests have been developed, and the exposure
in Florida is now not regarded as a fast test. Shortening the test period means that
various parameters determining the corrosion initiation and propagation processes
are considerably changed compared with natural conditions. These include:
The temperature
The composition of the corrosive including NaCl, pH, SO
2
, and additions such as
copper
The relative humidity
UV radiation
For modern cyclic testing some of these parameters are changed in time at regular
intervals, simulating more natural exposure conditions.
The aim of any testing procedure is to find a simple, perferably linear relation
between natural exposure and enhanced testing results. However, according to
many authors there is no such simple relation [101]. Some authors state that
enhanced weathering just gives some indication of the relative quality of coating
during testing, without any predictive value for the lifetime. Even standard tests
(e.g., ASTM B117) emphasize the lacking relation between, e.g., the salt spray test
and corrosion protection in different surroundings due to the enormous differences
in the reaction mechanisms. It has been stated that coated galvanized steel always
performs worse in a salt spray test than normal cold-rolled steel, whereas in reality
it always performs better [102]. Cyclic testing improves the relation between test
and practice [103].
It has been reported [104] that standard salt spray tests give reasonable results
for coatings in use in surroundings with a high salt concentration (seawater, deicing
salt) and high RH but not for other applications. The high salt concentration would
be responsible for decreased action of the anticorrosive pigments, which perform well
under natural low-salt conditions. Also, salt spray tests often give blistering, which
is often not relevant except for marine applications. Cyclic testing again results in less
blistering, which at least seems to indicate a better simulation of natural conditions.
A comparison of a cyclic test newly developed by Hoogovens for automotive
panels with the salt spray test, the SCAB (simulated corrosive atmospheric
breakdown) test, and the VDA(Verein der Deutschen Automobilindustrie) test showed
that some differences can be observed in the test results for the ranking as far as
undermining is concerned. A negative relation exists between the results of the new
cyclic test and the salt spray test. The new cyclic test shows reasonable agreement in
Organic Coatings 723
Copyright © 2002 Marcel Dekker, Inc.
