Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.


Isotherm of H Surface-Bulk Transfer The equation of the isotherm for the
surface-bulk transfer step at equilibrium [Eq. 8] is identical to that in Eq. (2)
except that ΔG
Hads
is replaced by ΔG
Hads(aq)
.
H
2
Pressure / Potential Correspondence for Adsorption / Absorption
Equilibria
Comparing Eqs. (10) and (11) with Eqs. (15) and (19) shows that the term
exp[–FE
(RHE1)
/RT] for isotherms in aqueous solution plays the role of the fugacity
term f
1/2
H2
in chemical absorption from ½H
2(g)
. So in solution, at equilibrium, the
fugacity of H
2
equivalent to a given potential versus RHE1 (reversible H electrode
at 1 atm) is equal to:
66 Protopopoff and Marcus
This equation provides the correspondence between adsorption / absorption equilibria
in the gas phase and electroadsorption / electroabsorption equilibria in solution [28,73]
(for example, the H coverage or bulk solubility at 0V / RHE1 is equal to the coverage
or solubility at an H
2
pressure of 1 atm). However, this relation applies only to
electrosorption processes in equilibrium or at least in quasi-equilibrium and is not
valid not at large overpotentials where coupled mechanisms prevail.
Gibbs Free Energy of Adsorption for UPD H and OPD H
For gas-phase adsorption, from Eq. (10), it is deduced that the fugacity f
H2
corresponding to θ
H
= ½ is equal to exp[2 ΔG
ads
(½) / RT]. Hence, adsorption sites with
ΔG
ads
(½) negative, i.e., in which the Gibbs partial molar free energy (chemical
potential) of the adsorbed H species at half-saturation is lower than ½G°
H2(g)
, attain
half-saturation for an H
2
pressure lower than 1 atm, whereas sites with ΔG
ads
(½)
positive reach half-saturation only for a pressure higher than 1 atm. Similarly,
analysis of Eq. (15) shows that H electroadsorption may occur significantly at
E>0V/(RHE1) (at underpotential) in sites with ΔG
ads
(½) negative [53,74],
whereas the sites with ΔG
ads
(½) positive reach half-saturation only for E
RHE1
< 0V
(at cathodic overpotential). On Pt, the value of the Tafel slope at low cathodic
overpotentials (~30 mV) indicates that the H intermediate of the HER is
electroadsorpbed under quasi-equilibrium at very low coverages around 0V/RHE1
and reaches significant coverages only cathodically; i.e., it is overpotentially
deposited (hence its name OPD H). So it is a weakly bonded H species that has a
positive ΔG
ads
(0.5) [53] and is adsorbed on or among a full monolayer of strongly
bonded UPD H atoms with negative ΔG
ads
(0.5) [29,35,51,53,75]. This simple
a priori analysis shows that at least on Pt and neighboring metals, H UP
electroadsorption can be correlated with low-pressure adsorption in the gas
phase [37,40,76] and OP electroadsorption with high H
2
pressure adsorption in
the gas phase [53].
Similarly, the fact that electroabsorption in Pd occurs at E > 0V / RHE1
[44,58,59] is correlated with the exothermic character of H absorption in Pd
(ΔH
diss
< 0), giving a value of ΔG
diss
close to zero at room temperature (see Figure 4).
Thermochemical Data for H Underpotential Electroadsorption on Pt
in Aqueous Electrolyte
As mentioned earlier, thermodynamic measurements of H adsorption in aqueous
electrolytes are possible only on the UPD H electroadsorbed reversibly above
Copyright © 2002 Marcel Dekker, Inc.
0V
/ RHE1 on some noble metals close to Pt [27,37,38,59,71] for which ΔG
ads
(½)
is negative. The analysis of the data obtained in aqueous electrolytes is complicated
by the fact that the potential region of UPD H adsorption / desorption often overlaps
the potential region of the specific adsorption / desorption of anions [39,59], in
which case the two processes are competitive and the overall charge density obtained
by integration of the voltammograms corresponds to a replacement reaction. The two
processes do not occur in the same potential range on Pt(111) in HClO
4
or in diluted
H
2
SO
4
electrolytes [39,59]. Thermodynamic measurements on Pt(111) single-
crystal electrodes show that the heat of adsorption of UPD H at zero H coverage
and the Pt-UPD H bond energy at zero coverage [ ≈ 260 kJ mol
–1
for Pt(111)]
[27,59] are close to those measured in low-pressure gas-phase experiments
(E
M–Hads
≈ 255 kJ mol
–1
) [8].
Relation between the Energetic and Structural Aspects of H
Adsorption
These thermodynamic measurements indicate that the M–H
ads
bonds involved at
the metal-electrolyte and the metal-gas interfaces at low pressure are of the same
nature, hence that UPD H atoms probably occupy the same adsorption sites as
those characterized by UHV techniques [24,40,53,59], i.e., the highly coordinated
or hollow sites [8].
With the use of a
35
S radiotracer allowing accurate measurement of the sulfur
coverage, the present authors studied quantitatively and compared on a Pt
single-crystal surface the blocking by chemisorbed sulfur of the H underpotential
(UP) adsorption and the HER. It was concluded that OPD H on Pt is not adsorbed in
the same sites as UPD H [53,75]. More direct evidence was obtained by in situ
infrared (IR) spectroscopy on a Pt(111) electrode at overpotential that the OPD H
atoms reacting in the HER on Pt(111) are adsorbed on top of substrate atoms [52],
while UPD H atoms are adsorbed in hollow sites.
Thus, a joint analysis of experiments in the gas phase and in aqueous electrolyte
gives evidence of the existence of two main kinds of H adsorption sites on a Pt
surface: high coordination sites (hollow sites) with a strong M–H
ads
bond
(ΔG
Hads
< 0) and low coordination / on-top sites with a weak M–H
ads
bond
(ΔG
ads
> 0); UPD H is adsorbed in the hollow sites as H adsorbed from H
2
in
low-pressure gas-phase adsorption, whereas OPD H is probably adsorbed at the
on-top sites, as the labile and highly reactive H involved at atmospheric or high
pressure in the catalytic reactions [53,74].
On non-noble transition metals, the existence of two H
ads
species with different
bond energies and site coordinations is probable. H UPD is not detected by cyclic
voltammetry on oxidable metals, although these metals readily adsorb H in the gas
phase (ΔG
Hads
for the hollow sites on these metals is more negative than on Pt),
because the UPD oxygen species (O
ads
or OH
ads
) are even more strongly chemi-
sorbed than H in the same sites. A thermodynamic calculation based on gas-phase
data shows that on nickel and iron H electroadsorption is in direct competition with
electroadsorption of oxygen species because O species are electroadsorbed near
0V / RHE1. So that H begins to electroadsorb in the hollow sites only at potentials
where most of the oxygen layer is desorbed, i.e., slightly under or over 0V / RHE1,
depending on the binding strength of the O species and their possible inhibiting effect
on the M–H
ads
bond [23]. It is likely that on non-noble transition metals, there is
Surface Effects on Hydrogen Entry into Metals 67
Copyright © 2002 Marcel Dekker, Inc.
coexistence of a stable monolayer of H species electroadsorbed in the hollow sites
around 0V / RHE1 by replacement of the O species and a mobile one adsorbed at
overpotential at on-top sites and reacting in the HER [23].
MECHANISMS OF THE H SURFACE-BULK TRANSFER
Structural Aspects
H Bulk Interstitial Sites
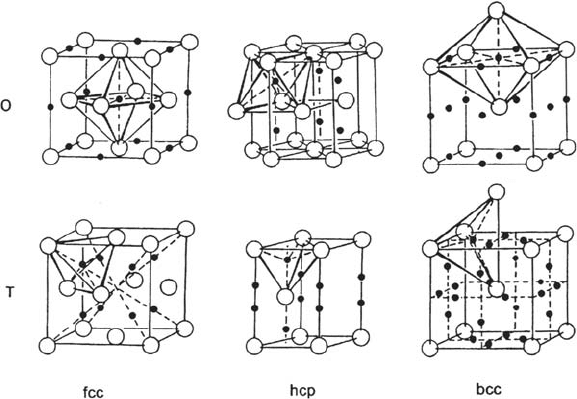
Figure 5 shows the different kinds of bulk interstitial sites in perfect face-centered
cubic (fcc), hexagonal close-packed (hcp), and body-centered cubic (bcc) lattice
structures. The sites populated by H atoms in the concentration range of solid
solution are known to be the octahedral sites for the fcc metals, whereas they are most
probably the tetrahedral sites for the bcc and hcp metals [66]; In fcc and hcp metals,
there are one octahedral site and two tetrahedral sites per metal atom, whereas in bcc
metals there are three octahedral and six tetrahedral sites per metal atom.
For diffusion in fcc metals, because close-packed metal atoms row prevent
the direct jumps from one octahedral site to another in the 〈110〉 directions, the H
migration paths involve intermediate jumps in the 〈111〉 directions to nearest
neighbor tetrahedral sites. If a is the cell parameter, an octahedral site is surrounded
by 8 nearest neighbor tetrahedral sites at a distance of a / √3 in the 〈111〉 directions
and 12 octahedral sites at a / √2 in the 〈110〉 directions (see Fig. 5). A tetrahedral
site is surrounded by four octahedral sites.
For hcp metals, there exist possibilities of direct jumps from one tetrahedral
site to another in the [0001] direction. For the bcc metals, the possibilities of jumps
68 Protopopoff and Marcus
Figure 5 Interstitial sites: octahedral (O) and tetrahedral (T) sites in face-centered cubic
(fcc), hexagonal close-packed (hcp), and body-centered cubic (bcc) lattices. (From Fig. 1.14
in Ref. 5.)
Copyright © 2002 Marcel Dekker, Inc.
between tetrahedral sites are direct from one tetrahedral site to four nearest neighbor
tetrahedral sites along the 〈110〉 directions or across the octahedrals sites to two
next neighbors in the 〈100〉 directions. The distances between nearest neighbor
tetrahedral sites in bcc metals are smaller by a factor of 2 than those between nearest
neighbor octahedral sites in fcc metals [66], which leads to much lower activation
energies for H diffusion than in fcc metals [78,79]. This explains the relatively
high H diffusion coefficient in bcc iron (between 10
–5
and 10
–4
cm
2
s
–1
), whereas
in fcc metals it is about 10
–6
cm
2
s
–1
for Pd and between 10
–10
and 10
–9
cm
2
s
–1
for
Ni and Pt [66].
H Adsorption Sites
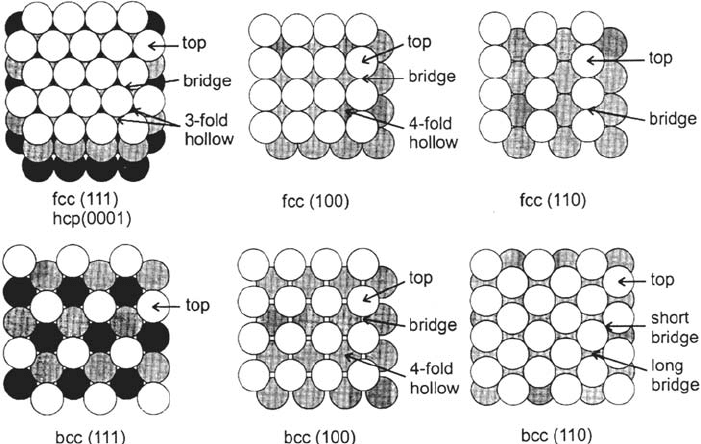
Figure 6 shows the atomic arrangement of the low-index faces of fcc, hcp, and bcc
metals, with the high-symmetry sites of H adsorption. There is general agreement
from UHV structural and vibrational measurements that the H adsorption sites
under low hydrogen pressure are the sites of highest coordination, hollow or
bridge sites [8,77]. On both the (111) face of fcc and the (0001) face of hcp, there
are two kinds of threefold hollow sites: sites over a second-layer metal atom,
called hcp type in surface science conventions, and sites over an interstice
between three second-layer atoms, called fcc-type sites. It has been reported that
H atoms adsorbed under low pressure tend to occupy the hcp sites on Pt(111) [80]
and also that D atoms occupy preferentially the fcc sites on Ni(111) [81].
Role of the Surface Structure in H Entry: Entry Sites
For the low-index faces of fcc crystals, the possible directions of H jumps from
the adsorption sites to the subsurface sites are represented in Figure 7.
Surface Effects on Hydrogen Entry into Metals 69
Figure 6 Top views of the atomic arrangement in the first two or three layers of the
low-Miller-index faces of fcc, hcp, and bcc crystals. High-symmetry adsorption sites are
indicated. (From Fig. 1 in Ref. 77.)
Copyright © 2002 Marcel Dekker, Inc.
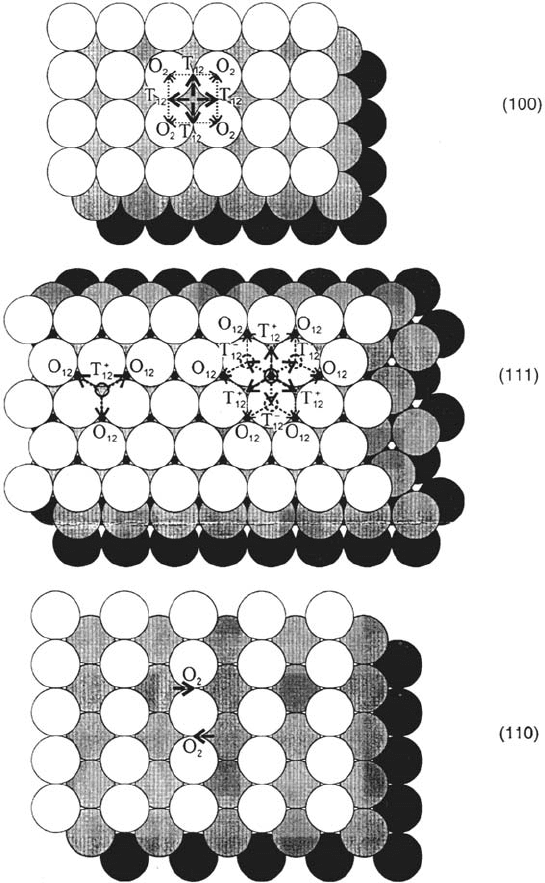
70 Protopopoff and Marcus
Figure 7 Possible paths of surface-bulk transfer on the low-index faces of fcc metals.
The full line arrows indicate the directions of first H atom jumps from surface sites; the
broken line arrows indicate second or third jumps from subsurface sites. On the (111) face,
first jumps occur vertically, either from an hcp surface site to a tetrahedral site T
+
12
(left) or
from a fcc site to an octahedral site O
12
(right).
Copyright © 2002 Marcel Dekker, Inc.
The lack of coordination above the surface leads to adsorbed H atoms closer
to the underlying metal atoms than in the bulk, so distances of jumps from the
adsorption sites to the nearest subsurface sites are shorter than the nearest neighbor
distances in the bulk (a /
√
3).
On the (111) face, each hcp surface site is located just above a subsurface
tetrahedral site T
+
12
(located between the first and the second metal layer, below
three surface atoms), from which three octahedral subsurface sites O
12
may be
reached. Each fcc surface site is just above such subsurface octahedral site O
12
,
from which, via three T
+
12
or three T
–
12
sites (below one surface atom), six O
12
sites
and three O
23
sites (below the second layer) may be reached, which makes nine
possibilities of jumps under the surface.
On the (100) face (if it is not reconstructed), an H atom adsorbed in a
fourfold hollow site (at a position close to that of an octahedral site) may jump to
four tetrahedral subsurface sites T
12
, from each of which two octahedral sites O
2
(at the second layer level, below surface atoms)may be reached.
On the (110) face (the more open face of the three low-index faces on fcc
metals) the adsorbed H atoms are alternatively adsorbed on each side of the
close-packed [110] rows, in the pseudo-threefold fcc sites close to the bridge sites
[82]. They may jump to one subsurface site O
2
at the second layer level, under the
bridge sites. On the (110) faces of Ni and Pd, above a coverage of one, hydrogen
induces reconstruction into a (110)-(1 × 2) structure, probably of the pairing-row
type [8,15,82]. This opens the surface more and allows accommodation of H on
the second metal layer, leading to a coverage of 1.5 H per first layer metal atom,
and makes it easier to reach subsurface sites [8,83,84].
Energetic Aspects
Gas Phase
Energetic analysis using temperature-programmed desorption (TPD), also called
thermal desorption spectroscopy (TDS), on a well-defined surface makes it possible
to determine the structure sensitivity and the heights of the energy barriers of the
surface processes. It was shown that the role of the substrate surface orientation in
the population of subsurface sites is crucial. On the reconstructed (110) faces of Ni
and Pd or on more open faces [8,83,84], subsurface sites are populated at
temperatures as low as 100 K and low H
2
pressures (10
–6
Pa), whereas higher
temperatures and pressures are necessary with the more densely packed planes [8].
The overall energetics of the hydrogen-metal reactions are illustrated in the
schematic drawing of Figure 8a showing the one-dimensional potential energy
versus distance curves of the various H states at the metal-gas interface. Two H
adsorbed states (corresponding to the hollow and on-top sites) are represented.
Actually, the energy levels indicate the chemical potentials of these states, referred to
the standard chemical potential of ½H
2(g)
. The standard free energy differences
between the various states are indicated. The diagram show that at 1 atm
nonactivated adsorption occurs spontaneously in the deep energy wells of the hollow
sites, whereas adsorption is activated in the shallow wells of the on-top sites, which
need a higher pressure to be filled. The diagram shows a case of endothermic H
absorption, with a high activation energy barrier for the transition between strongly
bonded H
ads
and H
diss
. The depths, of the wells and the height of the energy barrier,
Surface Effects on Hydrogen Entry into Metals 71
Copyright © 2002 Marcel Dekker, Inc.
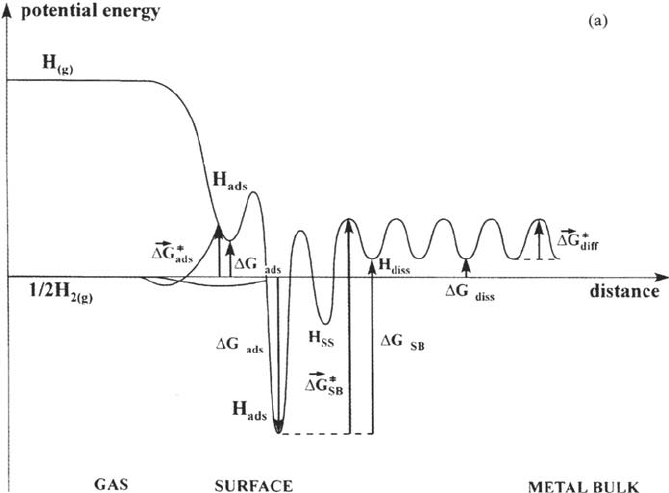
which determines the rate of the H uptake, depend on the nature of the metal, the
surface structure, and the H coverage. The barrier separating the chemisorbed and the
subsurface H states may in addition be lowered by a surface reconstruction [8,13].
Aqueous Electrolyte
An equivalent energy diagram is shown in Figure 8b for the metal-aqueous solution
interface [74]. It illustrates a case such as Pt(111) in HClO
4
, where the free energy of
adsorption of water from the liquid state can be neglected, and where specific
adsorption of anions and electroadsorption of oxygen species occur at higher anodic
potentials and do not perturb UP H adsorption [23]. The specificity of the metal-
solution interface, apart from the competition of H adsorption with adsorption of water,
anions, and oxygen species, is that the potential versus RHE1 is an external variable
72 Protopopoff and Marcus
Figure 8 (a) Schematic diagram of the potential energy vs. distance curves for the
various H states at the metal-gas interface, namely the two H adsorption states (weakly
bonded, corresponding to on-top sites, strongly bonded, corresponding to hollow sites),
the subsurface state (H
ss
), and the bulk dissolved (absorbed) state (H
diss
). The free
energy changes and activation free energies for the different H processes are indicated
(SB denotes surface-bulk transfer). (b) Same diagram as (a) for the metal–aqueous solution
interface in an electrolyte where competition with adsorption of other species is negligible.
The states are the same as for (a) except the specific state (H
+
(aq)
+ e
–
), initial state of
electroadsorption (EA), whose energy level with respect to the H
2(g)
level depends on
the potential referred to RHE1. If E is positive (anodic), electroadsorption occurs in the
strongly bonded state, hence denoted underpotential (UP) H
ads
. If E is negative (cathodic),
electroadsorption occurs in the weakly bonded state, hence denoted overpotential (OP)
H
ads
, and H
2
evolution (HER) occurs from this intermediate state.
Copyright © 2002 Marcel Dekker, Inc.
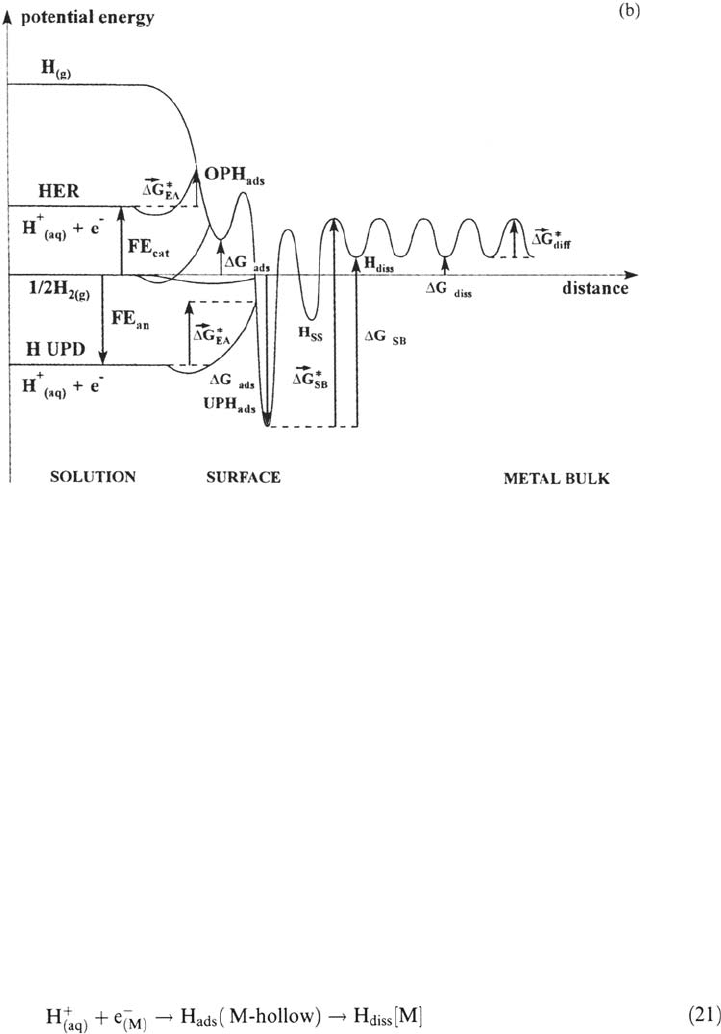
that controls the energy level (more exactly the standard electrochemical potential) of
the initial state (H
+
(aq)
+ e
–
), with respect to the reference level of ½ H
2(g)
, and hence
the standard activation free energy of electroadsorption (ΔG
≠
ea
) in the two kinds of
sites. Thus two main situations occur, according to the potential versus RHE1:
1. The potential E
(RHE1)
is positive (anodic).
This is the range of underpotential (UP) H adsorption on metals of the Pt group.
The energy levels of the initial state (H
+
(aq)
+ e
–
) and the final state (strongly bonded
H
ads
, with ΔG
ads
< 0) are of the same order of magnitude and far below the energy
levels of ½ H
2(g)
and the weakly bonded H
ads
; this explains why at these potentials UP
H electroadsorption is in equilibrium and there is negligible H electroadsorption in
the weakly bonded state and H
2
evolution. In the case of an exothermic absorber, such
as palladium, the energy level of H
diss
is located near the level of ½ H
2(g)
(ΔG
diss
≈ 0),
so H electroabsorption may occur at positive E
(RHE1)
[44,45,58,59] directly from
UPD H, without H
2
evolution, according to the following path:
Surface Effects on Hydrogen Entry into Metals 73
Figure 8b
This possibility of absorption from UPD H on Pd electrodes is consistent with what
was improperly called the “direct entry” mechanism, as explained earlier.
2. The potential E
(RHE1)
is negative (cathodic).
This is the range of overpotential (OP) H adsorption. The H electroadsorption
occurs irreversibly in the sites with negative ΔG
ads
, so the sites of UP H
ads
are
Copyright © 2002 Marcel Dekker, Inc.
saturated, and OP H
ads
is adsorbed in the sites with positive ΔG
ads
. The energy level
of OP H
ads
is of the same order of magnitude or higher than the energy level of ½ H
2(g)
so H
2
evolution occurs. The energy level of the initial state (H
+
(aq)
+ e
–
) is raised when
the potential increases cathodically, so OP H electroadsorption may be in
quasi-equilibrium only at low cathodic overpotentials and then becomes irreversible,
giving rise to the coupled mechanisms. Only the chemical combination step is
described in this simplified diagram, but the principle of the analysis may easily be
extended to the electrocombination (Heyrovsky) step. In the represented case of
endothermic absorption, cathodic overpotentials are needed to overcome the overall
activation barrier for H penetration into the bulk.
Such mechanisms involving two adsorbed H species with different binding
energies and sites probably apply to all transition metals. On oxidable metals, H
electroadsorption in the hollow sites followed by absorption is possible only at
potentials below the limit of desorption of the oxygen species, calculated for Ni and
Fe to be close to 0V
(RHE1)
[23]. As discussed earlier, it is likely that the sites for the
HER adsorbed intermediate (OP H
ads
) are the on-top sites on all transition metals. It
is admitted that on iron and ferrous alloys the HER and the HAR share a common
H
ads
intermediate and that the surface-bulk transfer step is in quasi-equilibrium (see
earlier) [31]. The mechanism of H absorption into the bulk of all transition metals at
overpotential must then involve a sequence of two adsorption steps:
74 Protopopoff and Marcus
The weakly bonded OP H
ads
in on-top sites simultaneously participates in the
HER and the HAR. This mechanism distinguishes two adsorption steps instead of
one in the surface-bulk transfer. Of course, this has no implications if the overall
transfer step is in quasi-equilibrium, but if it is not—a situation occurring, for
example, for H permeation in thin membranes [53,72]— the treatment of the
kinetics of H absorption will become more complex.
KINETICS OF THE HER AND HAR
Expressions of the Absolute Rates of the Elementary Surface Steps
The absolute rates of all the H elementary surface reactions occurring during the
HER and the HAR at the surface of an electrode M in aqueous solution may be
expressed as functions of the potential and the Gibbs free energy of H adsorption,
using the theory of absolute reaction rates [85] and the Brönsted-Polanyi relations
[86,87]. As usual, a model of regular localized solution is assumed for adsorbed H
(Frumkin-Fowler model) and dissolved H, i.e., random distribution of these atoms
among their respective sites and pair interactions between nearest neighbor H atoms
[88]. It is assumed that the activation free energies depend on θ
H
or X
H
only via their
linear dependence on ΔG
ads(aq)
[60]. ΔG
ads(aq)
varies linearly with θ
H
, according to
where ΔG°
ads(aq)
is the initial adsorption free energy or free energy at zero H coverage
and g
HH
is a pair-energy parameter describing the lateral interactions between
adsorbed H atoms [88]. Similarly,
Copyright © 2002 Marcel Dekker, Inc.
where ΔG°
diss
is the H dissolution free energy at infinite dilution and h
HH
a parameter
for interactions between dissolved H atoms [88].
Electroadsorption and Electrodesorption
Only adsorption by proton discharge in acid medium is considered here [Eq. (4)];
the expressions of the rates of adsorption from water are similar. The rates for the
forward and reverse reactions are
Surface Effects on Hydrogen Entry into Metals 75
with k
ea
=
κ
ea
kT / h Γ
s/2
exp – ΔG
≠
ea
/ RT. (For definition of the terms used, see the
following).
Chemical Combination and Dissociative Adsorption [Eq. (3)]
Note: The reactions of chemical and electrochemical combination [Eqs. (3) and
(6)/(7)] lead to H
2(aq)
dissolved in the electrolyte, so the rate equations for the reverse
adsorption reactions involve the activity of H
2
dissolved in water, a
H2
. However, as
the reaction of dissolution of H
2(g)
is in equilibrium, a
H2
= f
H2
exp – ΔG°
sol
/ RT,
where ΔG°
sol
= μ°
H2(aq)
– μ°
H2(g)
. So the expressions for the rates may be rearranged to
involve the fugacity f
H2
and ΔG
ads(aq)
, the free energy for adsorption from H
2(g)
.
Electrocombination and Electrodissociation
[in Acid Medium—Eq. (6)]
Surface-Bulk Transfer
For simplicity, in the absence of detailed energetic characterization of the subsurface
state and knowledge of the transition between the two adsorbed H states (see
preceding section), the overall surface-bulk transfer reaction is considered here as an
elementary step from one single adsorption state, as is usually done.
Copyright © 2002 Marcel Dekker, Inc.
