Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.

As mentioned before is the case for Cr in Fe-Cr alloys in the presence of O or S
in the gas phase. Cr behaves similarly in Ni-Cr alloys [83].
For Ni
75
Fe
75
, the composition of the sulfur-covered surface was observed to
be identical to the bulk composition for both the (100) and (110) faces [84]. This
is in agreement with the fact that the two metallic elements of the alloy have nearly
the same affinity for sulfur. In contrast, the (111) face of the same alloy behaves
differently. The sulfur-covered surface was shown to be enriched in Fe with a
maximum Fe/Ni ratio of 1. Such an effect was explained by a competition between
metal-metal and metal-sulfur interactions and an ordering of the adsorbed layer
that maximizes the Ni– Fe bonds at the composition of the maximum.
When the difference in affinity for the gas of the two components of a binary
alloy is large, only one element is preferentially incorporated into a two-dimensional
(2D) compound. This is the case when oxygen interacts with Pt
80
Co
20
to form a
single layer of CoO [85]. Similarly, when an Ag-Ni alloy containing a low Ni
concentration reacts with H
2
S, a 2D Ni-S compound is formed as identified by
radio-tracers for both S and Ni [86].
The nonmetal species that induces the surface enrichment of a metallic
compound of an alloy may also originate from the bulk material. A thermodynamic
treatment of the cosegregation process has been proposed [87,88], and various
models of segregation have been critically discussed [89].
Equilibrium segregation of nonmetal species—C, N, O, Si, P, and S—on
iron and iron-based alloy has been extensively studied [90]. Investigations of
single crystals showed the formation of epitaxially arranged 2D surface com-
pounds such as CrN on Fe-15%Cr-N(100) and VC on Fe-3% Si-0.04%V-
C(100) [91–93]. Most concepts of segregation at free surfaces can be applied
to grain boundaries [89].
WATER ADSORPTION
The main features of the interaction of water with solid surfaces have been
reviewed with special emphasis on studies performed in ultrahigh-vacuum (UHV)
systems on single crystals of metals [94]. Precise data on the binding energy, the
orientation, the tendency for hydrogen bonding of H
2
O molecules, and their ability
to dissociate are now available. The effect of coadsorbed species has also been
explored in some detail. The characterization by UHV spectroscopies of the
interaction of water molecules with ionic species is a promising attempt for
investigating the solid-electrolyte interface [95,96], The different steps of building
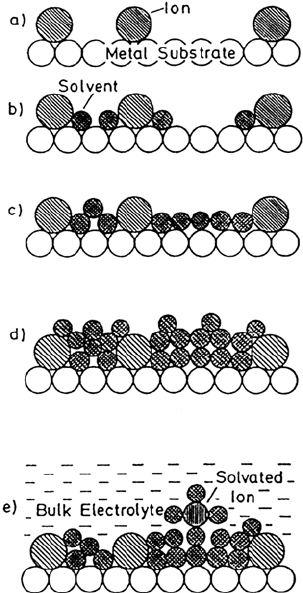
an electrochemical double layer in the presence of specific adsorbed ions are
shown in Figure 10. In UHV, the three first steps can be reached, allowing a study
of the properties of the inner layer. Direct comparison between gas-phase
experiments and electrochemical data is possible by measuring the work function
change and the electrode potential, by properly referencing these two values. The
potential drop across the interface can be measured and compared. It was shown
by this method that the double layer can in some favorable cases be emersed
essentially intact from solution [97].
Adsorption From Gas Phase 35
Copyright © 2002 Marcel Dekker, Inc.
Molecular Adsorption
The two lone pairs in the molecule enable the oxygen to interact with the substrate
and with neighboring molecules. The general characteristics of the interaction of
H
2
O molecules with metals can be summarized as follows [94]:
1. Water bonds through the oxygen atom to the surface by overlap between the
metal’s orbitals and water’s lone pair orbitals.
2. Water acts as an electron donor (Lewis base) to the substrate surface. As a
consequence, the work function of the metal decreases during H
2
O adsorption.
3. Preferred adsorption sites are the “on top” adsorption sites that are electron
depleted.
4. Compared with the isolated molecule, the internal bond angle, bond lengths,
and vibrational frequency are only slightly perturbed by the interaction with
the surface.
36 Oudar
Figure 10 Schematic representation of the synthesis of the electrochemical double layer
in UHV. (a) Adsorption of specifically adsorbed ions without solvent; (b) addition of
hydration water; (c) completion of the inner layer; (d) addition of solvent multilayers;
(e) model for the double layer at an electrode surface in solution. (From Ref. 95.)
Copyright © 2002 Marcel Dekker, Inc.
5. The chemical bond is typically of the order of 40 to 65 kJ mol
–1
on the
borderline of physisorption.
6. The adsorption of H
2
O on metals is nonactivated—the probability of
adsorption is close to unity.
Special attention has been devoted to the structure of the H
2
O adsorbed
layer. The strength of attractive interactions by hydrogen bonds in this layer is
comparable to that with the substrate. As a result, H
2
O molecules tend to form
short- and long-range ordered structures. At fractional monolayer coverages,
well-oriented dimers have been observed on Ni(110) [98].
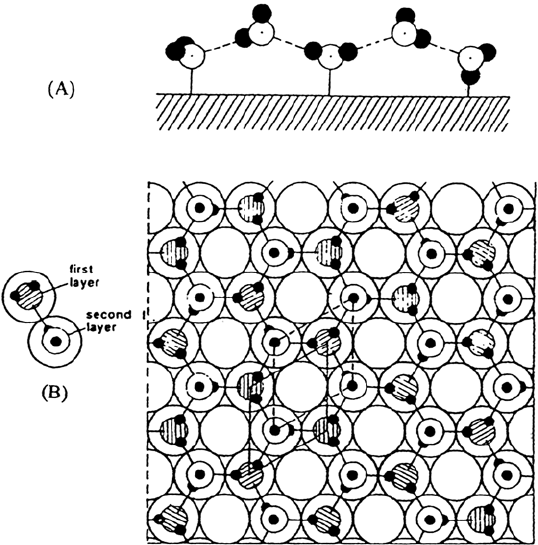
At higher coverages the adsorbed layer is characterized on the densest plane
of many fcc or hexagonal metals by an identical hexagonal low-energy electron
diffraction (LEED) pattern for which a structural model similar to the ice lattice
has been proposed [99,100] (Fig. 11). In this model, the first layer of water molecules
is bound by direct chemisorption bond to the metal. Molecules in the second layer
are linked by two or three hydrogen bonds to the first layer molecules. The stability
of this bilayer is strongly dependent on the lattice matching between the metal and
crystalline ice. The bilayer model may also be adapted with some distortions to
nonhexagonal planes: fcc (100) or (110) [101].
Adsorption From Gas Phase 37
Figure 11 Schematic diagram of the water bilayer. (A) Side view; (B) top view. (From
Ref. 99.)
Copyright © 2002 Marcel Dekker, Inc.
Water Dissociation
Water can be partially or totally dissociated on metals to form hydroxyl and
hydrogen or atomic oxygen and hydrogen. The driving force for dissociation is
provided by the heat of formation of the dissociation fragments (OH, H, O) relative
to the chemisorption bond of molecular water.
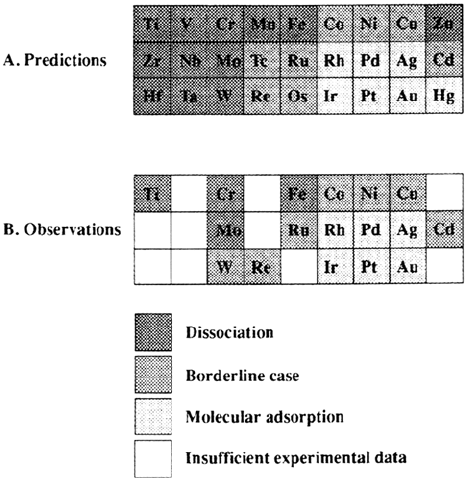
Based on the enthalpy change that accompanies molecular or dissociative
adsorption, metals for which dissociation is favored or not are listed in Figure 12.
In this classification the most important factor is the metal-oxygen bond of atomic
oxygen, which varies more strongly from metal to metal than it does from the
metal-hydrogen bond strength. The same factor that stabilizes the electronegative
oxygen atom also tends to stabilize the electronegative hydroxyl fragments. As a
consequence, the same tendency is observed for total or partial water dissociation.
Experimental results are also included in Figure 12 [94]. Better agreement is
found between prediction for complete dissociation and experimental observations
despite the fact that these results usually refer to partial dissociation.
The upper limit for dissociation is about 110 kJ mol
–1
, far less than the barrier
for breaking an OH bond in the free water molecule (500 kJ mol
–1
). This illustrates
the ability of the surface to stabilize the dissociation fragments during the OH bond
breaking.
For borderline metals such as Co, Ni, Re, and Cu, water dissociation is favored
by the roughness of the surface on an atomic scale, whereas the adsorption remains
molecular on close-packed planes. A good example of a metal in this category is
38 Oudar
Figure 12 Comparison between predictions and experimental results for molecular
versus dissociative H
2
O adsorption on metals. Predictions refer to complete dissociation:
H
2
O
a
→ 2H
a
+O
a
. (From Ref. 94.)
Copyright © 2002 Marcel Dekker, Inc.
nickel. There is no evidence of water dissociation on the clean, atomically smooth
(100) and (111) planes. In contrast, on the atomically rough (110) face, water is
dissociated at about 100 K into OH
a
and H
a
. On Ni(111), steps increase significantly
the water binding energy and the tendency of water to dissociate [102]. This effect
has been attributed to the presence at steps of acid Ni
δ+
adsorption sites that
enhance the electron transfer from water molecules to the substrate. The increase
of the H
2
O–Ni bond strength is correlated with a decrease in the intramolecular
H
2
O bond strength that favors the H
2
O dissociation.
In addition to Ni(110), formation of a hydroxyl overlayer has been observed
on Fe(100) [103] and Fe(110) [104], on Ag(100) [105,106], and on Ag(111) [107].
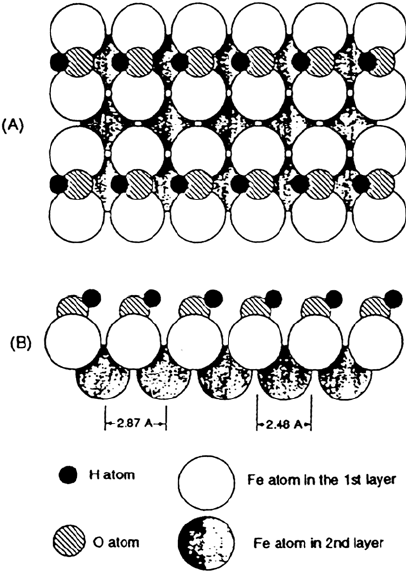
On Fe(100), mechanisms of water adsorption and dissociation have been studied
by LEED, thermal desorption, and high-resolution electron spectroscopy [103]. At
low temperature and low coverage, water is adsorbed by forming hydrogen-bonded
molecular clusters. As the surface is warmed, the clusters break apart, wetting
occurs, and water molecules begin to dissociate. Dissociation is complete at 250 K,
forming a well-ordered OH overlayer with the OH bond tilted from the surface
normal (Figure 13). Near 310 K the hydroxyl overlayer disproportionates or
dissociates with water or hydrogen desorption, The remaining oxygen is bound in
the fourfold hollow sites as for oxygen adsorbed from molecular oxygen.
Adsorption From Gas Phase 39
Figure 13 Hydroxyl species in p(1 × 2) structure of Fe(100) surface. (A) Top view; (B)
side view. (From Ref. 103.)
Copyright © 2002 Marcel Dekker, Inc.

Effect of Additives
Preadsorbed additives significantly affect the binding energy of water molecules.
Water molecules desorb at higher temperatures in the presence of an electronegative
(Br), an electropositive (Cs), or a more covalent adsorbate (H) [108]. This has
been attributed to the surface hydration of the ionic species, which creates stronger
attractive interactions than the water interactions in the pure water adsorbed layer.
On metals such as Ag, Cu, Ni, Pt, and Pd on which H
2
O is molecularly adsorbed,
a fractional monolayer of oxygen can significantly increase the H
2
O-metal binding
energy and eventually initiate the H
2
O dissociation. The reaction proceeds via the
hydrogen abstraction reaction with the formation of OH
a
species:
40 Oudar
As shown on Cu(110) [109], this reaction occurs only for low oxygen coverage,
the oxygen-saturated surface being totally inactive. A typical curve in Figure 14
shows that, for θ
o
< 0.1 the slope of the curve is 2, indicating that, as expected
from reaction (1), 2OH
a
are formed per adsorbed O
a
.
Similarly, iron exposed to an equimolar gas mixture containing O
2
and H
2
O
reacts at room temperature according to [110]
Figure 14 Hydroxyl coverage as a function of initial oxygen coverage, Θ
Ox
, on Cu(110).
Maximum hydroxyl formation occurs for Θ
Ox
= l/8. (From Ref. 109.)
Copyright © 2002 Marcel Dekker, Inc.
ADSORPTION ON METAL OXIDES
Studies of chemisorption of gaseous molecules on oxides may provide new insights
into the various processes involved in the formation, aging, and degradation of
passive films. We will restrict ourselves to a brief description of the main features of
the chemisorption behavior on oxides. We will focus on results obtained on well-
defined surfaces of single crystals with O
2
and H
2
O as adsorbates. In the past 10
years, precise data on the structure and the electronic properties of surfaces of single
crystals of oxide have been obtained by LEED and surface-sensitive spectroscopies
(UPS, XPS, HEELS). Excellent introductions to this subject can be found in Refs.
111 and 112. A large part of the data presented here is extracted from these reviews.
For many different bulk oxides in which metal cations are octahedrally
coordinated, such as MgO, CoO, NiO, and TiO
2
, the most stable surface is
characterized by fivefold coordination of the surface cation. Such a surface, which
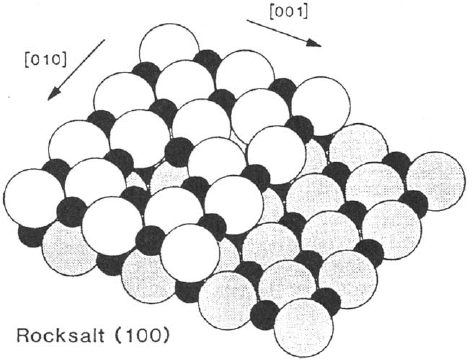
is usually the cleavage plane, is shown in Figure 15 for the rock salt lattice. Linear
defects such as step or point defects arising from anion or cation vacancies are also
represented in this figure. To a first approximation the structure of the cleavage
plane can be considered as the termination of the bulk lattice with minor relaxations
(rumpling of only few percent of the interionic spacing has been detected by
LEED). A good example showing the higher stability of the surface with fivefold
coordination of surface cations concerns TiO
2
(001). This plane, which contains
only fourfold-coordinated cations, decomposes spontaneously at moderate
temperature into facets containing fivefold coordination cations.
On many oxides the reduction of the ligand coordination of the surface cations
with respect to the bulk cations does not introduce specific band gap surface states
and the surface oxide exhibits an electronic structure undistinguishable from that of
Adsorption From Gas Phase 41
Figure 15 Model of the rock salt (100) surface. Large circles represent O anions; small
circles represent metal cations. Surface defects: step, anion vacancy, and cation vacancy
are also shown. (From Ref. 112.)
Copyright © 2002 Marcel Dekker, Inc.
the bulk. Further reduction of ligand coordination of cations due to their localization
at steps does not change significantly the electronic structure of oxides.
Mechanisms of Chemisorption
Chemisorption [114] on an oxide surface differs significantly from that on metals.
One of the main reasons for this difference is the ionic character of the solid, which
favors acid-base or donor-acceptor reactions. Lewis sites are localized on the cations
and basic sites on the anions. An example of this type of interaction is given by CO
2
,
which reacts with basic O
2–
to give a surface carbonate CO
3
2–
. Similarly, a donor
molecule such as H
2
O or NH
3
can be molecularly adsorbed via its lone-pair
electrons, which react with an acidic (cation) site. An alternative to the molecular
adsorption is that resulting from the heterolytic dissociation of the molecule. It may
occur by abstraction of H atom transferred to a basic site, producing a hydroxyl group.
Chemisorption may also proceed by a mechanism involving an electron
transfer between the adsorbate and the substrate (oxidation- reduction or redox
interaction). It is the case for adsorbates such as O
2
or Cl
2
that are strong electron
acceptors. O
2
can be molecularly or dissociatively adsorbed, Cl
2
is dissociatively
adsorbed. The redox reactions that involve electronic carriers are expected to
occur preferentially on semiconducting or metallic oxides. On wide-bang-gap
insulators these reactions are promoted by surface defects such as ion vacancies,
which may act as sources or sinks for electrons.
The role of coordinative unsaturation in determining the surface reactivity of
metal oxides has been discussed with special references to the dissociation of
Brönsted acids [115]. This reaction involves a metal-oxygen pair. The proton from
the acid is bound to the surface oxygen anion, which acts as a base, and the
conjugate base anion from the acid is bound to the surface metal cation, which
acts as a Lewis acid. The very different chemistry exhibited by the two polar
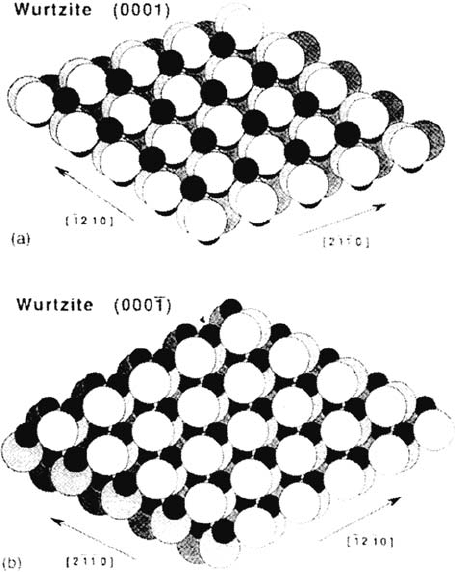
planes of wurtzite ZnO (Fig. 16) makes it possible to define the coordination
requirement of the surface ions for acid dissociation. In bulk wurtzite each anion
or cation is tetrahedrally coordinated to four cations or anions, respectively. At the
oxygen polar (0001) surface, an oxygen anion is coordinated to three Zn cations
in the plane immediately below. These cations are coordinatively saturated. At the
Zn polar (0001) surface the reverse situation is found; a vacant coordination site
is present on each surface cation, whereas oxygen anions in the layer below the
surface are coordinatively saturated. The Zn polar plane was observed to dissociate
various acids of the strength of carboxylic acid or less, whereas the O polar plane
remained totally inert with respect to the dissociation of these acids. This result
emphasizes the predominant role of coordination vacancy associated with the surface
cation for the dissociation of Brönsted acids. It was also shown that doubly
coordinatively unsaturated surface cations favor reactions involving the coupling
of the ligands originating from the dissociation of two acid molecules.
As regards chemisorption involving redox interactions, it is useful to make a
distinction between oxides whose cations are in their highest valence state (maximal
valence oxide) and oxides whose cations are in a reduced valence state (suboxides).
Among the most studied oxides, TiO
2
, SrTiO
3
, MgO, and Al
2
O
3
belong to the first
category and Ti
2
O
3
V
2
O
3
, and MnO to the second one. Nearly perfect surfaces of
42 Oudar
Copyright © 2002 Marcel Dekker, Inc.
maximal valence oxide are quite inert in chemisorption involving an electron
transfer from the oxide toward the adsorbed molecule because their surface cations
are not able to supply electrons by oxidation. For example, NiO(100) is inert to O
2
to
10
3
L exposure (sticking coefficient lower than 10
–9
). Conversely, CO, which is an
electron donor, reduces NiO at low exposure by abstracting O
2–
to form CO
2
and
leaving a vacancy behind. Perfect suboxide surfaces react much more easily with a
large number of molecules, particularly with O
2
. This can be ascribed to the ability
of surface cations to transfer their electron to adsorbed species. No general trends
have been found to characterize the chemisorption behavior of suboxides, which
varies significantly from one system to another.
Point defects are mainly involved in chemisorption on cleaved surfaces. On
such surfaces the density of point defects is usually very small. They can be created
by heating in vacuum and rapid quenching or by electron or ion bombardment. The
predominant point defects are O vacancies (F centers) with two electrons at each
vacancy in order to maintain the local electroneutrality. This localized charge
partially overlaps with the adjacent cations, increasing the population of their lowest
empty orbitals. The charge distribution does not affect significantly the O
2–
anions
near the vacancy defect because their already filled 2p shell cannot accept additional
charge. The O vacancies affect predominantly the electronic structure in maximal
Adsorption From Gas Phase 43
Figure 16 Models of wurtzite surfaces (a) (0001) and (b) (000–1) [From Ref. 113].
Copyright © 2002 Marcel Dekker, Inc.
valence oxides of transition metals. On most of these oxides, as observed for TiO
2
or SrTiO
3
, new surface states appear in the band gap and usually increase the surface
conductivity of the oxide. The only oxide that has been observed to become more
insulating in the presence of O vacancies is CoO.
Oxygen vacancy defects on most oxide surfaces, maximal valence or
suboxides, display a strong interaction with molecules such as O
2
, H
2
O, CO, or
SO
2
. O
2
molecules are dissociatively adsorbed to form O
2–
ions. After saturation of
the defect sites, a second species, tentatively identified as a doubly ionized mole-
cular species, adsorbs much more weakly on the surface. O
2–
ions adsorbed at the
defect sites interact differently from lattice ions with H
2
O or CO. A possible explana-
tion is that these adsorbed O species, which are probably less ionic than oxygen
ions, do not relieve completely the relaxation of the lattice around the defect site.
Water Adsorption
H
2
O adsorption from water vapor has been extensively studied, first on powdered
samples, more recently on single crystals. Most experiments on powders were
carried out at high vapor pressures (> 1 torr). Experiments on single crystals are
usually performed at low gas pressures (< 10
–6
torr) because of the limitations of
the modern surface science techniques. Powdered samples are highly defective
and contain various crystalline orientations and structures. They are more
representative of the behavior of passive films formed in aqueous solution. Studies
on well-characterized surfaces of single crystals make it possible to investigate the
role of surface defects and surface orientation on the mechanisms of chemisorption.
Results on powders can be summarized as follows:
1) H
2
O is dissociatively adsorbed on metal oxides at room temperature at a
pressure of a few torr to form hydroxyl groups. This reaction of acid-base type is
schematically represented in Figure 17 for a surface oxide exposing both anions
and cations. Two types of OH groups are shown. Those bound to two cations are
strongly polarized, thus loosening the bond to hydrogen. These groups are acidic
in character. The other OH groups bound to one cation are predominantly basic
and easily exchangeable for other anions [116]. The high reactivity of defective
oxides is confirmed by the external hydroxylation in aqueous solution of the films
of NiO formed by Ni passivation [117] or by sputtering deposition [118].
2) Zurface OH may react with water molecules through hydrogen bonds.
One may expect that the same type of interactions occurs at the passive film-aqueous
solution interface and influence the structure of the double layer. The ratio
H
2
O/OH was found to be close to ½ on metal oxides such as MgO, TiO
2
, Fe
2
O
3
,
and Al
2
O
3
[119]. Higher values close to one were observed on ZnO, SnO
2
, and
Cr
2
O
3
[119]. The behavior of hydroxylated powder of Cr
2
O
3
studied by infrared
spectroscopy, adsorption isotherms from water vapor, and thermal adsorption
[120] illustrates the complexity of the interactions between OH groups and H
2
O
molecules. Two kinds of adsorption were observed: (a) adsorption of H
2
O
molecules on localized OH groups with an adsorption energy of about 55 kJ mol
–1
and (b) adsorption of H
2
O on special OH groups leading to a phase transformation.
This phase transformation is clearly seen on the adsorption isotherms at around
300 K (Fig. 18). It seems to be favored by the strong structural similarity between
the hydroxylated (001) surface of Cr
2
O
3
and the basal plane of ice. A large
44 Oudar
Copyright © 2002 Marcel Dekker, Inc.
