Higgins M.D. Quantitative textural measurement in igneous and metamorphic petrology
Подождите немного. Документ загружается.

3
Grain and crystal sizes
3.1 Introduction
One of the most commonly quantified aspects of rock texture is the size
distribution of crystals, grains and, more rarely, blocks. Although the first
published study of crystal size in igneous rocks was in the early days of
petrology (Lane, 1898), the subject really only took off in 1988 after the
publication of seminal papers by Marsh and Cashman (Cashman & Marsh,
1988, Marsh, 1988b). In metamorphic petrology the study of size distributions
started a bit earlier with the study of garnets by Kretz (1966a). The importance
of grain size distribution in sedimentology completely outweighs that of other
branches of petrology and is covered in more detail in sedimentological texts
(e.g. Syvitski, 1991). Engineers have a considerable interest in the mechanical
and chemical properties of materials (Brandon & Kaplan, 1999): grain size and
composition play an important role in controlling these properties and hence
their quantification is very important (Jillavenkateas et al., 2001, Medley,
2002). Also of interest are pore and bubble sizes in rocks and these are
discussed separately in Chapter 7.
3.1.1 What are size and size distributions?
The size of a crystal or grain is a measure of the space occupied by the object. It
is a three-dimensional property and can be defined in several different ways,
according to the application of the parameter: if the shape of a crystal changes
during growth or solution then a volumetric measure of size may be useful.
Size based on volume is simple because every grain has a unique volume.
However, if we are interested in the growth or solution of crystals where the
shape is almost invariant then a linear measure of size is usually appropriate.
Hence, the change in size of a crystal can be expressed in terms of linear growth,
39
mm per second, for example. In most geological applications a linear measure of
size is used and various definitions are discussed in Section 3.1.2. From here on
the term size refers to a linear measure of the space occupied by a grain.
Petrologists commonly describe the mean size of crystals or grains in a rock,
without clearly defining what they mean. The definition of size is discussed in
Section 3.1.2, but here I want to examine the significance of mean sizes. If a
rock contains grains smaller than the resolution limit of the analytical method
then the value of the mean size will depend on the resolution (Figure 3.1). In
this situation the mean can be defined only if we can describe a size function for
that unknown interval. In any case although the mean is a useful parameter, it
has only one dimension. Another one-dimensional parameter that is some-
times measured is the crystal or grain number density. This is defined as the
total number of crystals or grains per unit volume, although some authors use
this incorrectly for number density by area. Again, there is a problem if a rock
contains grains smaller than the resolution limit of the method. Both these
parameters are sub-sets of a more information-rich parameter, the crystal
size distribution (CSD), grain size distribution or particle size distribution.
This has two dimensions; size and crystal or grain number. It is defined
between upper and lower size limits. Outside these limits it is indeterminate,
but changing the measurement limits will not change the CSD values within
the limits (Figure 3.1).
Finally, some workers loosely use the terms unimodal and bimodal size
distributions. The number of modes depends clearly on the variable examined
and how it is expressed. For instance, the size variable may be expressed in
terms of length or volume of grains: this will clearly change the distribution. In
many ‘bimodal’ rocks there is actually only one mode in the CSD. What is
frequently meant by this term is that there are two populations of crystals.
However, this should be verified by other means, such as chemical or isotopic
composition, as single populations may appear visually to be multiple
populations.
3.1.2 Linear size definitions
The linear size of a grain has many different definitions. This is compounded
by a common confusion between true size in three dimensions and size in
intersections or projections of grains. Three-dimensional linear size definitions
are listed here in order of typical decreasing numerical value.
1. The greatest dist ance between any points on the surface of the grain.
2. The length of the smallest parallelepiped that can be fitted around the grain.
40 Grain and crystal sizes

l mm
l mm
(a) Measurement of crystals larger than 0.02 mm
(b) Measurement of crystals larger than 0.10 mm
–2
0
2
4
6
8
10
0 0.5 1.0 1.5
ln (population density)
crystal size (length)
mean size = 0.205 mm
mean size = 0.067 mm
–2
0
2
4
6
8
10
0 0.5 1.0 1.5
ln (population density)
crystal size (length)
Figure 3.1 Effects of size limits on values of mean crystal size and crystal
size distributions: example of an andesite from Soufriere Hills, Montserrat
(Higgins & Roberge, 2003). The original data have been slightly modified for
this diagram. (a) All crystals larger than 0.02 mm have been counted. Crystals
smaller than this have been observed but not quantified. Hence, the CSD is
undefined below 0.02 mm. The mean size is 0.067 mm. (b) All crystals larger
than 0.10 mm have been measured. The CSD for sizes greater than 0.1 mm is
identical to the upper diagram, but the mean crystal size, 0.205 mm, is almost
three times greater. Hence, if a rock contains crystals of all sizes then the mean
size is meani ngless – it just depends on the measurement size limits.
3.1 Introduction 41
3. The mean projected height (mean calliper diameter) of the grain. This can be
visualised at the mean height of the shadow of the grain when it is illuminated
from all different angles. This is commonly used in stereology.
4. The length of the major axis of a best-fit ellipsoid. This ellipsoid has the same
volume and moment of inertia as the grain.
5. The diameter of a sphere of equivalent volume, or more rarely the edge of a cube of
equal volume.
Other size definitions used for unconsolidated sediments are described in
Section 3.3.1. For grains close in shape to a sphere all definitions give similar
sizes. The difference between the measures of size increases with the anisotropy
of the grain. Definitions 2, 3 or 4 are generally similar and should be used. The
definition of size used should always be specified in publications.
3.2 Brief review of theory
A wide range of processes and constraints can affect the crystal or grain size
distribution of rocks, hence the interest in this parameter. For igneous rocks
we must consider the kinetic process of nucleation and initial growth
(Figure 3.2). The driving force for kinetic processes can be expressed as the
undercooling or supersaturation of the system (see Section 3.2.1.2). The resul-
tant crystal populations may be modified by mechanical processes such as
sorting and mixing. Once the kinetic driving force is reduced, the initial or
modified crystal populations will start to equilibrate, although complete equi-
librium is never achieved. This process involves the minimisation of the total
energy of the crystal population. The importance of distinguishing kinetic and
Kinetic textures
Initial
magma
Mechanically-modified
textures
Equilibrium textures
Sorting and
compaction
Nucleation and
growth
Renewed nucleation
and growth
Equilibration Equilibration
Development of
igneous textures
Figure 3.2 Development of igneous textures – role of kinetic, mechanical
and equilibrium processes.
42 Grain and crystal sizes
equilibrium effects cannot be overstated – it is frequently a source of significant
confusion in the interpretation of CSDs. Melting of igneous rocks will probably
be more controlled by equilibrium textural processes, as it is easier to destroy a
crystal lattice than to make one. But again we must consider mechanical effects
on the crystal population. Kinetics and equilibrium are also important in
metamorphic rocks, but deformation clearly plays a more important role than
in igneous rocks. Finally, I will consider the most generally applicable constraint
on grain and crystal populations, closure: this is the fact that a rock cannot
contain more than 100% crystals or grains. As in geochemistry, this constraint is
commonly acknowledged but it is ignored (Higgins, 2002a).
3.2.1 Primary kinetic crystallisation processes
In many branches of geology the roles of kinetics and equilibrium are impor-
tant, and that is certainly the case here: kinetics will control the speed at which
crystals can nucleate and grow (or dissolve) and equilibrium will dictate the
final amount of the phases. The most common way of expressing the kinetic
effect is in terms of undercooling: a certain overstepping of equilibrium is
necessary to force the nucleation and/or growth of a new phase. However, the
amount of undercooling encountered during the crystallisation of rocks is
probably strongly dependent on the crystallisation environment and minerals
present, and is not well established so far.
The fundamental functions that control growth textures are simply
expressed: during solidification the nucleation rate is a function of time J (t),
whereas the growth rate is a function of both time and crystal size G (t,l). If
both functions are known, then the evolution of the crystal size distribution
with time, n (t,l) can be determined. Generally speaking we are more interested
in the inverse problem: that is we have measured the final crystal size distribu-
tion and we want to know the nucleation and growth rate variations during
solidification. There is no unique solution to this problem, but many attempts
have been made by making assumptions about the form of G (t,l) and J (t)
(e.g. Brandeis & Jaupart, 1987, Marsh, 1988b, Lasaga, 1998, Marsh, 1998,
Zieg & Marsh, 2002).
It is commonly overlooked that significant undercooling is not necessary for
crystallisation: equilibrium requires that the total volume of crystals will
increase during cooling. The texture of the crystals responds to equilibrium
effects to minimise the total energy of the crystal population. This process is
common and has many names: the effect on crystal size is referred to here as
coarsening (see Section 3.2.4). Hence, the final texture of most igneous rocks
will reflect some combination of kinetic and equilibrium effects.
3.2 Brief review of theory 43
3.2.1.1 Nucleation of crystals
In a liquid, such as magma or an aqueous solution, atoms are continually being
added or removed from clusters that have a lattice arrangement similar to that
of major mineral phases. Addition of new atoms creates bonds that reduce the
energy of the cluster. However, bonds at the surface of the cluster are unsa-
tisfied and there is a net increase in energy. The sum of these two components
gives the total energy of the cluster. With increasing cluster size the total energy
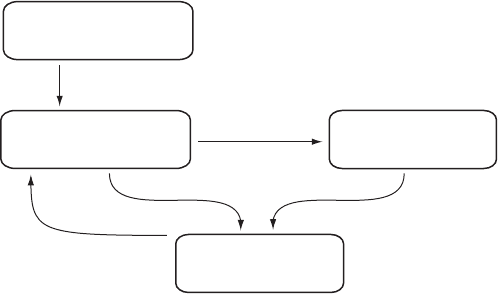
risestoapeakandthendescends(Figure3.3a;e.g.Dowty,1980,Markov,
1995). When one of these clusters exceeds a critical radius, r
, then it is
energetically stable and a crystal nucleus is formed. Addition of further
atoms to the nucleus is energetically favoured as it reduces the total energy.
This process is termed homogeneous nucleation. The driving force for nuclea-
tion is related to the undercooling of the system (see next section). At low
undercooling the energy barrier is not breached, no clusters reach the critical
sizeandthenucleationrateiszero(Figure3.3b).Withincreasingundercool-
ing, the nucleation rate increases rapidly to a peak and then diminishes.
Heterogeneous nucleation occurs on the surface of existing host grains or
bubbles, or on defect structures within crystals. The surface energy of the
cluster is lower, hence both the critical radius and energy barrier are lower
(Figure3.3a).Hence,formationofstablenucleioccursatlowerundercooling
than for homogeneous nucleation. Mismatches between the crystal lattices of
the new and existing grains will produce elastic stresses that store energy.
Hence, the actual undercooling will generally depend on the degree of crystal-
lographic similarity between the host and the new phase. This is expressed as
thedihedralangle,(seeSection4.2.3;Figure3.3a).
The necessary undercooling is obviously smallest when the new and host
crystals are mineralogically identical and in the same orientation. However,
in this case the greater ease of nucleation will be balanced by the lower
concentration of the mineral component in the magma surrounding the
growing host crystal.
Crystal nuclei are too small and unstable to be observed – we can record
them only if there has been an interval of growth. Hence, it has been difficult to
study nucleation. Many authors assume that magmas are never completely
liquid and that there is always the possibility of heterogeneous nucleation.
However, as was shown above, the energy difference as expressed by the
dihedral angle may be sufficiently high that homogenous nucleation occurs
even in the presence of solid phases.
New crystals may also form within existing crystals, for instance during
exsolution related to cooling of feldspars. Such crystals can form
44 Grain and crystal sizes
discontinuously by nucleation and growth, in the same way as for nucleation
in fluids or on surfaces. However, heterogeneities in crystals may also arise
continuously by a process called spinoidal decomposition (McConnell,
1975). Compositional waves form spontaneously in the mineral lattice: the
θ
=
90°
θ
= 30
°
Critical
radius, r
∗
ΔG
High
High
Low
Low
Medium
Medium
New phase
energy
cluster
radius
(a)
(b)
Homogeneous
(
θ
=180
°
)
Homogeneous
(
θ =180°)
θ
= 30
°
undercoolin
g
nucleation rate normalised
to maximum
0
Surface
energy
Surface
energy
θ
=
90°
Total
energy
Figure 3.3 (a) Total energy of an atomic cluster against cluster radius. The
total energy of an atomic cluster is the sum of the surface energy and the new
phase (lattice) energy. For homogeneous nucleation the surface energy is high
and the critical radius is large. For heterogeneous nucleation the surface energy
is lower giving a lower critical radius. Two cases are shown here which are
expressed by dihedral angles of 908 and 308. Although homogeneous and
heterogeneous nucleation processes are commonly presented as opposites, it
is clear that there is a continuum between these two situations. (b) For
homogeneous nucleation at small degrees of undercooling the normalised
nucleation rate is zero. As undercooling increases, it increases rapidly to a
peak and then diminishes. In geological situations most nucleation probably
occurs to the left of the peak. Heterogeneous nucleation reduces the
undercooling necessary for nucleation: a lower energy barrier (DG)leadsto
lower dihedral angles and lower undercooling.
3.2 Brief review of theory 45

wavelengths are short at low temperatures, but become longer with increasing
temperatures. Such zones may later transform into separate phases which can
be observed as regular exsolution patterns of contrasting phases. The whole
process is not dissimilar to geochemical self-organisation (Ortoleva, 1994).
3.2.1.2 Growth and solution
At its simplest, crystals grow if this results in a reduction in the total energy
of the crystallising system, that is the population of crystals and the material
from which they are crystallising. Similarly, crystals will dissolve also if
this results in a reduction in the total energy. We are principally interested in
crystal growth rates: that is the change in size of the crystal with time.
Unfortunately, although time is the essence of geology, the timing of crystal
growth is generally difficult to measure in the earth. Therefore, experiments
are an important complement to natural studies for determining growth rates.
Crystal growth rates are very important in many industrial processes and
hence there is an immense amount of work in this subject. The problem for
geologists is to determine what aspect of this work is applicable to our field.
Some have seen the range in possibilities, found them wanting and created
their own arbitrary growth laws (e.g. Eberl et al., 1998, Eberl et al., 2002).
Here, I will present a very brief overview of what I consider to be important.
More complete treatment of the subject is available in a number of books and
articles (e.g. Markov, 1995, Marsh, 1998, Jamtveit & Meakin, 1999).
Crystal growth rates depend on the magnitude of the driving force, but the
exact relationship is controlled by the growth mechanism. The driving force
for crystal growth is equal to Dm/kT, where Dm is the chemical potential
difference between the crystallising phase and the host fluid (magma, liquid
or gas), k is the Boltzmann constant and T is the temperature. Dm/kT may be
expressed in many different ways, but the most useful for us is for solutions in
terms of concentrations or enthalpy and temperature (e.g. Lasaga, 1998):
Dm
kT
¼ ln
C
C
0
¼
DH
sol
ðT
sat
TÞ
kTT
sat
where C and C
0
are the real and equilibrium concentrations of the solute;
DH
sol
is the positive enthalpy of solution; T
sat
is the saturation temperature
(melting point; mineral liquidus temperature). It is assumed that the system is
ideal and that the enthalpy of the transition is independent of temperature. In
most applications it is easiest to use the undercooling as a measure of driving
force. However, if we want to look at the relationship between crystal size and
growth rate then it is more convenient to use concentration terms.
46 Grain and crystal sizes
The mechanism of crystal growth is by addition of atoms to the surface. The
rate of addition can be controlled in two ways (e.g. Baronnet, 1984, Lasaga,
1998);
*
By the processes of addition of atoms to the surface: interface-controlled growth.
*
By the transport of the crystal components towards the surface and unwanted
material and latent heat away from the surface: transport-controlled growth. The
actual movement is controlled by diffusion and movement of the fluid with respect
to the crystal (advection).
The growth shape of a phase depends partly on its physical isotropy or
anisotropy: a physically isotropic material has the same distribution of
atoms in every direction. Isometric or cubic minerals are optically isotropic,
but not physically isotropic. If crystals were physically isotropic then they
would grow in ideal circumstances as spheres: liquids are isotropic and do
grow in this way. However, all crystals are physically anisotropic: hence, the
nature of the crystal–host interface is different in every direction. Of course,
the amount of anisotropy in a crystal is variable and is expressed relative to the
contrast between the crystal and its host. This contrast varies with tempera-
ture: below the ‘roughening transition’ temperature crystal faces grow by
spiral growth or two-dimensional nucleation (Figure 3.4). They keep their
orientation and continue to grow as macroscopically flat faces. Above the
roughening transition the energy difference between the crystal and the host
dominates: the crystals grow with rounded surfaces, without facets. In the
roughening transition
temperature
temperature
Figure 3.4 Roughening transition (e.g. Baronnet, 1984, Lasaga, 1998,
Bennema et al., 1999). Below this temperature crystals grow with facets;
above it they grow with rounded surfaces. This transition resembles a phase
change. It should be remembered that the roughening transition temperature
may be higher than the saturation temperature (liquidus) of a mineral phase,
in which case the miner al will always crystallise with faces where it does not
impinge on other surfaces.
3.2 Brief review of theory 47
geological environment most crystals grow with facets, although these may be
lost where adjacent crystals impinge. Curved faces do occur (e.g. Heyraud &
Metois, 1987), but those produced by solution can generally be distinguished
from those produced during growth by examination of internal growth zones.
Supply of crystal materials depends on several factors: the chemical contrast
between the crystal and its environment and rate of transport of atoms.
Transport includes both diffusion and the relative physical movement of
crystal with respect to the host. The first term is easier to treat: if a crystal
has exactly the same composition as the ‘mother’ material then no diffusive
transport is necessary. An example would be the crystallisation of a pure
substance, such as water. Pure substances are rare in geology: we are normally
concerned with solutions such as magmas.
1
Another example is the enlarge-
ment of a grain by grain-boundary migration: here, the original and final
materials have the same composition and the transport distance is short.
In more complex situations transport of atoms is controlled by numerous
factors:
*
Physical state of the host material: solid or liquid. Diffusion is much faster in a fluid
phase
2
than a solid phase.
*
Composition of the host material. The overall composition is expressed as the
viscosity. Atoms move more slowly in viscous materials. However, all atoms do
not move at the same rate: generally, small ions move faster than large atoms.
Addition of material to chemi cally complex minerals requires diffusion of many
different ions, and the overall process is controlled by the slowest atomic species.
Addition of water to magma will reduce the viscosity and increase the diffusion
rates of all species.
*
The temperature of the system is extremely important: higher temperatures increase
diffusion rates. However, pressure has little effect as solids are almost incompressible.
*
Relative movement of fluid (advection) from which the crystal is growing with
respect to the crystal. This replenishes the boundary layer that was depleted in the
crystal components by crystallisation and enriched in the rejected components.
3.2.2 Kinetic textures
Crystal populations produced by kinetic processes alone are most commonly
modelled using the Avrami equations (e.g. Lasaga, 1998). However, the
solution of this equation requires knowledge of the variation of the nucleation
rate with time, J (t), the growth rate with time and crystal size, G(t,l). Both of
these are generally unknown and various models of their behaviour have
been proposed. The most common assumption is that the growth rate is
independent of size and hence this variable can be simplified, although Eberl
48 Grain and crystal sizes
