Coker A.K. Fortran Programs for Chemical Process Design, Analysis, and Simulation
Подождите немного. Документ загружается.


Table 2-2
(continued)
8 1000.0 28.500
9 ii00.0 30.310
i0 1200.0 32.120
ii 1300.0 33.930
12 1400.0 35.740
13 1500.0 37.550
HEAT CAPACITY OF CARBON DIOXIDE
******************************************************************************
TEMPERATURE K HEAT CAPACITY cal/gm-mol.K
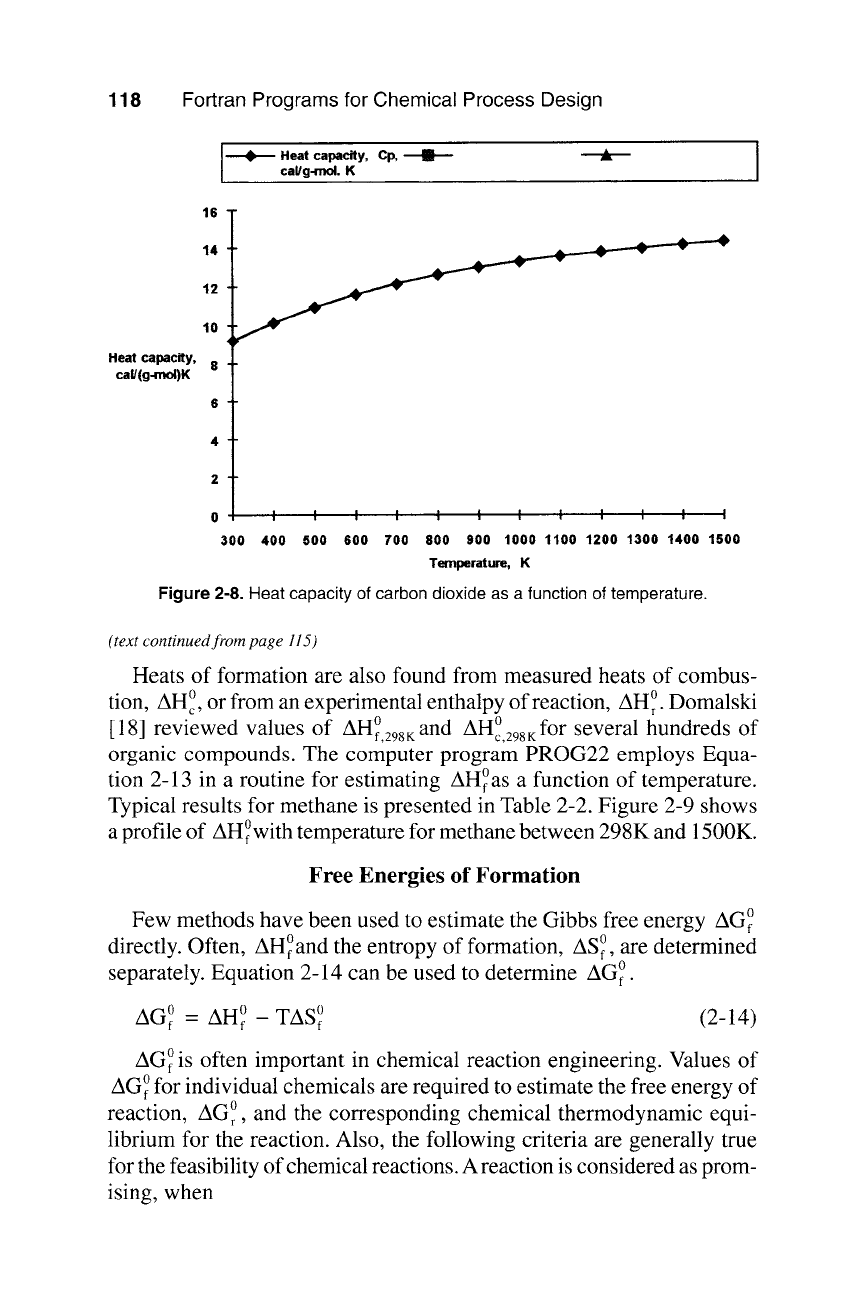
1 300.0 9.201
2 400.0 10.134
3 500.0 10.927
4 600.0 11.594
5 700.0 12.149
6 800.0 12.607
7
900.0 12.983
8 i000.0 13.290
9 1100.0 13.544
10 1200.0 13.758
Ii 1300.0 13.948
12 1400.0 14.128
13 1500.0 14.312
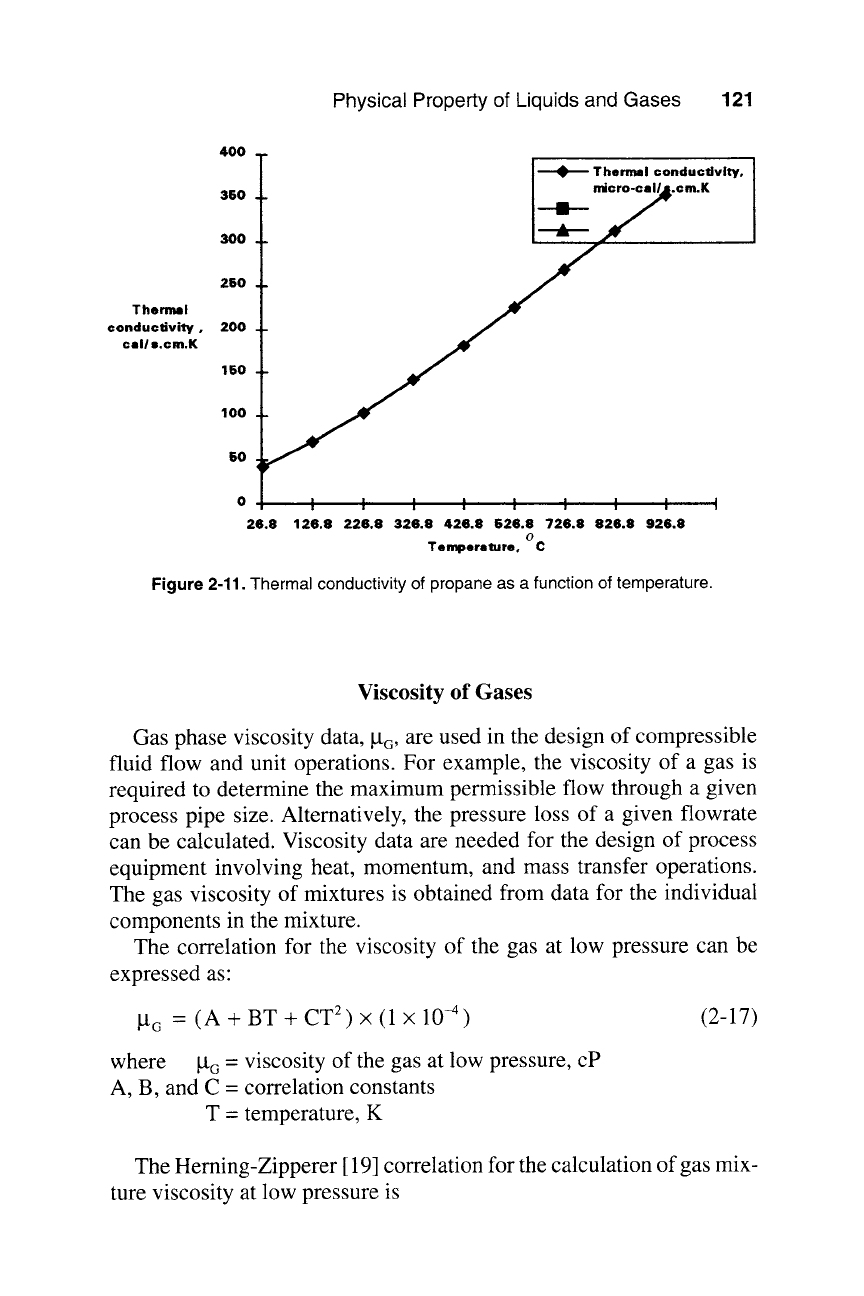
THERMAL CONDUCTIVITY OF PROPAN~:26.8oC-1026.8oC
******************************************************************************
TEMPERATURE oC THERMAL CONDUCTIVITY micro-cal/s.cm.K
1 26.8 42.432
2 126.8 70.267
3 226.8 103.678
4 326.8 141.461
5 426.8 182.412
6 526.8 225.324
7 626.8 268.995
8 726.8 312.218
9 826.8 353.789
10 926.8 392.504
11 1026.8 427.156
THE VISCOSITY OF ETHANE: 26.8oC-1026.8oC
******************************************************************************
TEMPERATURE oC VISCOSITY cP.
1 26.8 .009
2 126.8 .012
3 226.8 .015
4 326.8 .017
5 426.8 .019
8 726.8 .026
9 826.8 .028
10 926.8 .030
ii 1026.8 .031
117
118 Fortran Programs for Chemical Process Design
Heat capacity,
Cp,
cal/g-mol. K
16
14
12
10
Heat capacity, 8
cal/(g-rnol)K
6
o
300 400 500 600 700 800 90o 10o0 11o0 1200 13oo 1400 1500
Temperature,
K
Figure
2-8. Heat capacity of carbon dioxide as a function of temperature.
(text continued from page 115)
Heats of formation are also found from measured heats of combus-
tion, AH~, or from an experimental enthalpy of reaction, AH~. Domalski
0 0
[18] reviewed values of AHf,z98Kand AHc,z98Kfor several hundreds of
organic compounds. The computer program PROG22 employs Equa-
tion 2-13 in a routine for estimating AH~ a function of
temperature.
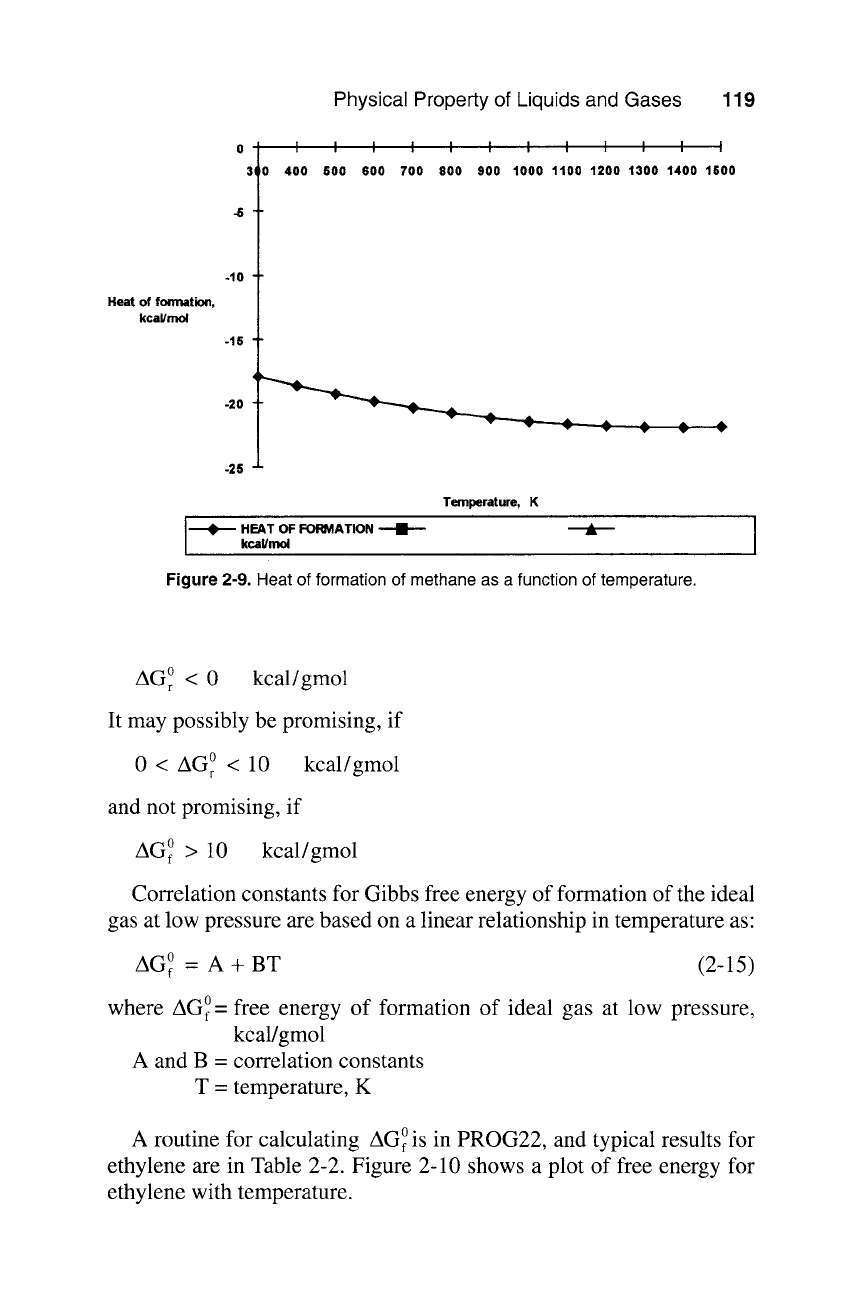
Typical results for methane is presented in Table 2-2. Figure 2-9 shows
a profile of AH~with temperature for methane between 298K and 1500K.
Free Energies of Formation
Few methods have been used to estimate the Gibbs free energy AG~
directly. Often, AH~and the entropy of formation, AS~, are determined
separately. Equation 2-14 can be used to determine AG~.
- - TAS
(2-14)
AG~ is often important in chemical reaction engineering. Values of
AG o for individual chemicals are required to estimate the free energy of
reaction, AG~, and the corresponding chemical thermodynamic equi-
librium for the reaction. Also, the following criteria are generally true
for the feasibility of chemical reactions. A reaction is considered as prom-
ising, when
Heat of formation,
kcal/mol
-10
-16
-20
-26
Physical Property of Liquids and Gases 119
I I I I I I I I I I I I
~0 400 600 600 700 800 900 1000 1100 1200 1300 1400 1600
A
v
A A A
,.
#
Temperature,
K
I ~, HEAT OF FORMATION --'1--- .
I
kcal/mol
I
Figure
2-9. Heat of formation of methane as a function of temperature.
AG~ < 0 kcal/gmol
It may possibly be promising, if
0 < AG~ < 10 kcal/gmol
and not promising, if
AG~ > 10 kcal/gmol
Correlation constants for Gibbs free energy of formation of the ideal
gas at low pressure are based on a linear relationship in temperature as:
AG~ - A + BT (2-15)
where AG~-free energy of formation of ideal gas at low pressure,
kcal/gmol
A and B = correlation constants
T = temperature, K
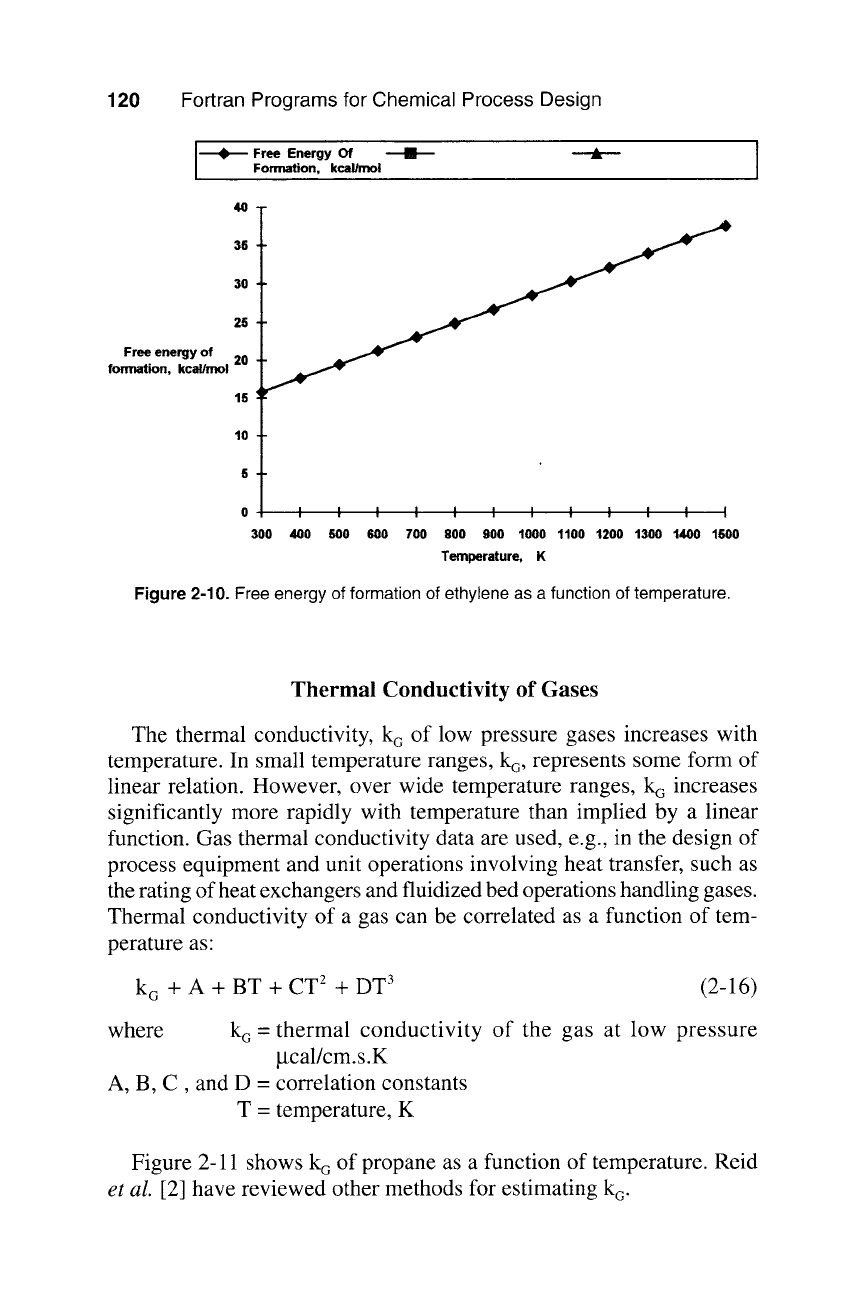
A routine for calculating AG~ is in PROG22, and typical results for
ethylene are in Table 2-2. Figure 2-10 shows a plot of free energy for
ethylene with temperature.
120 Fortran Programs for Chemical Process Design
~ Free Energy Of
Formation, kcal/mol
40
36
30
26
Free energy of 20
formation, kcallmol
300 400 800 600 700 800 900 1000 1100 1200 1300 1400 1600
Temperature,
K
Figure
2-10. Free energy of formation of ethylene as a function of temperature.
Thermal Conductivity of Gases
The thermal conductivity, k G of low pressure gases increases with
temperature. In small temperature ranges, k G, represents some form of
linear relation. However, over wide temperature ranges, 1% increases
significantly more rapidly with temperature than implied by a linear
function. Gas thermal conductivity data are used, e.g., in the design of
process equipment and unit operations involving heat transfer, such as
the rating of heat exchangers and fluidized bed operations handling gases.
Thermal conductivity of a gas can be correlated as a function of tem-
perature as:
k G + A + BT
+ CT 2 +
DT 3 (2-16)
where k G = thermal conductivity of the gas at low pressure
gcal/cm.s.K
A, B, C, and D = correlation constants
T = temperature, K
Figure 2-11 shows k G of propane as a function of temperature. Reid
et al.
[2] have reviewed other methods for estimating k G.
Physical Property of Liquids and
Gases
121
400
360
300
260
Thermal
conductivity,
200
calls.cm.K
160
100
60
, Av
Thermal conduclJvity,
micro-calLs.cm.K
--I--
26.8 126.8 226.8 326.8 426.8 626.8 726.8 826.8 926.8
0
Temperature, C
Figure 2-11. Thermal conductivity of propane as a function of temperature.
Viscosity of Gases
Gas phase viscosity data, ~t G, are used in the design of compressible
fluid flow and unit operations. For example, the viscosity of a gas is
required to determine the maximum permissible flow through a given
process pipe size. Alternatively, the pressure loss of a given flowrate
can be calculated. Viscosity data are needed for the design of process
equipment involving heat, momentum, and mass transfer operations.
The gas viscosity of mixtures is obtained from data for the individual
components in the mixture.
The correlation for the viscosity of the gas at low pressure can be
expressed as:
~t G -(A+BT+CT 2)x(lx10 -4 ) (2-17)
where let G - viscosity of the gas at low pressure, cP
A, B, and C - correlation constants
T- temperature, K
The Herning-Zipperer [ 19] correlation for the calculation of gas mix-
ture viscosity at low pressure is
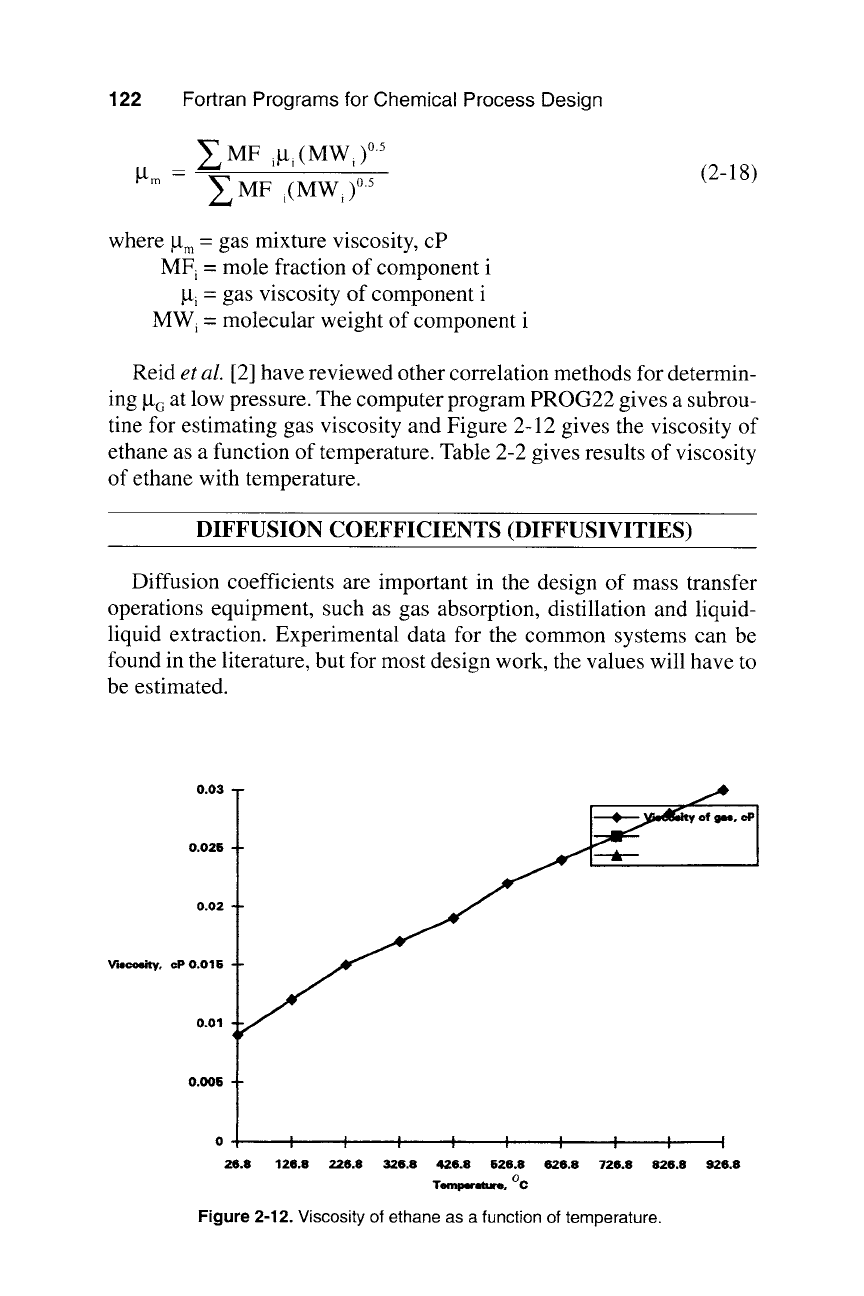
0.03
Fortran Programs for Chemical Process Design
Bm
~., MF
i~Lti
(MW i )0.5
Z MF
i(MWi )0.5
(2-18)
where la m = gas mixture viscosity, cP
MF i = mole fraction of component i
la~ = gas viscosity of component i
MWi = molecular weight of component i
Reid
et al.
[2] have reviewed other correlation methods for determin-
ing let C at low pressure. The computer program PROG22 gives a subrou-
tine for estimating gas viscosity and Figure 2-12 gives the viscosity of
ethane as a function of temperature. Table 2-2 gives results of viscosity
of ethane with temperature.
DIFFUSION COEFFICIENTS (DIFFUSIVITIES)
Diffusion coefficients are important in the design of mass transfer
operations equipment, such as gas absorption, distillation and liquid-
liquid extraction. Experimental data for the common systems can be
found in the literature, but for most design work, the values will have to
be estimated.
of
~m, r
0.026
0.02
Viecoeity, cP
0.016
0.01
0.006
122
Figure
2-12. Viscosity of ethane as a function of temperature.
o ! I I I I , I i I I
26.8 126.8 226.8 326.8 426.8 626.8 626.8 726.8 826.8 926.8
Temperature,
~

Physical Property of Liquids and
Gases 123
Liquid-Phase Diffusion Coefficients
Diffusion coefficients in liquid phases depend on concentration and
are valid for dilute solutions, that is, solute concentrations no greater
than 10%. Also, the lower the solute concentration, the more accurate
the calculated coefficients. For a binary mixture of solute A in solvent
B, the diffusion coefficient can be represented as D~ concentra-
tions of A up to 5 or 10 mole percent [20]. A number of correlations
have been proposed for predicting D~ dilute liquid solutions
[21,22,23]. Here, the Wilke-Chang method is employed for estimating
D o This can be expressed as:
AB"
_
)~
DOau 7.4 • 10 -8 (r (2-19)
0.6
~'~B 9 VA
where D~ - Diffusion coefficient of solute A at very low concentra-
tions in solvent B, cm2/s
MwB - molecular weight of solvent B
T- absolute temperature, K
~t B - viscosity of solvent B, cP
v A - molal volume of solute A at its normal boiling tempera-
ture, cm3/g-mol
- association factor of solvent B, dimensionless
= 2.6 for water (some workers recommend 2.26)
= 1.9 for methanol
= 1.5 for ethanol
= 1.0 for unassociated solvents
The Le Bas additive volumes given by Reid
et al.
[2] can be used for
the value of V A, if no experimental data are available.
Gas-Phase Diffusion Coefficients
Diffusion coefficients for non-polar gases may be estimated from
Chapman and Cowling [24], Wilke and Lee [25] and Fuller, Schettler,
and Giddings [26]. Here, the Fuller, Schettler, and Giddings's method
can be expressed as:
f
t0.5
10 -3T 1"75
MwA +
MwB
DoB MwA 9 MwB
- 2 (2-20)
P{(ZV)~ 3 -[(ZV)~ 3}
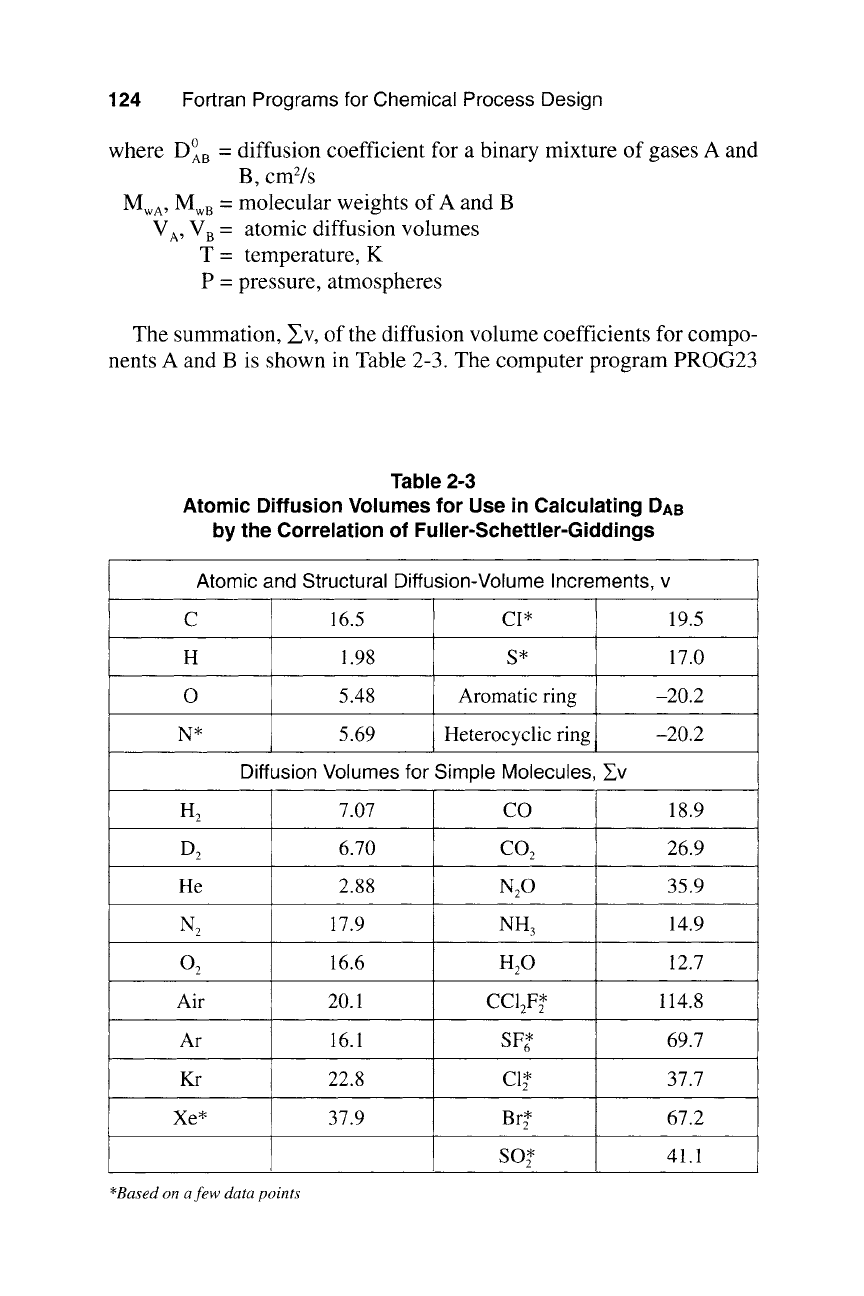
124 Fortran Programs for Chemical Process Design
where DOAB -- diffusion coefficient for a binary mixture of gases A and
B,
cm2/s
Mwa , MwB = molecular weights of A and B
VA, VB = atomic diffusion volumes
T = temperature, K
P = pressure, atmospheres
The summation, ~v, of the diffusion volume coefficients for compo-
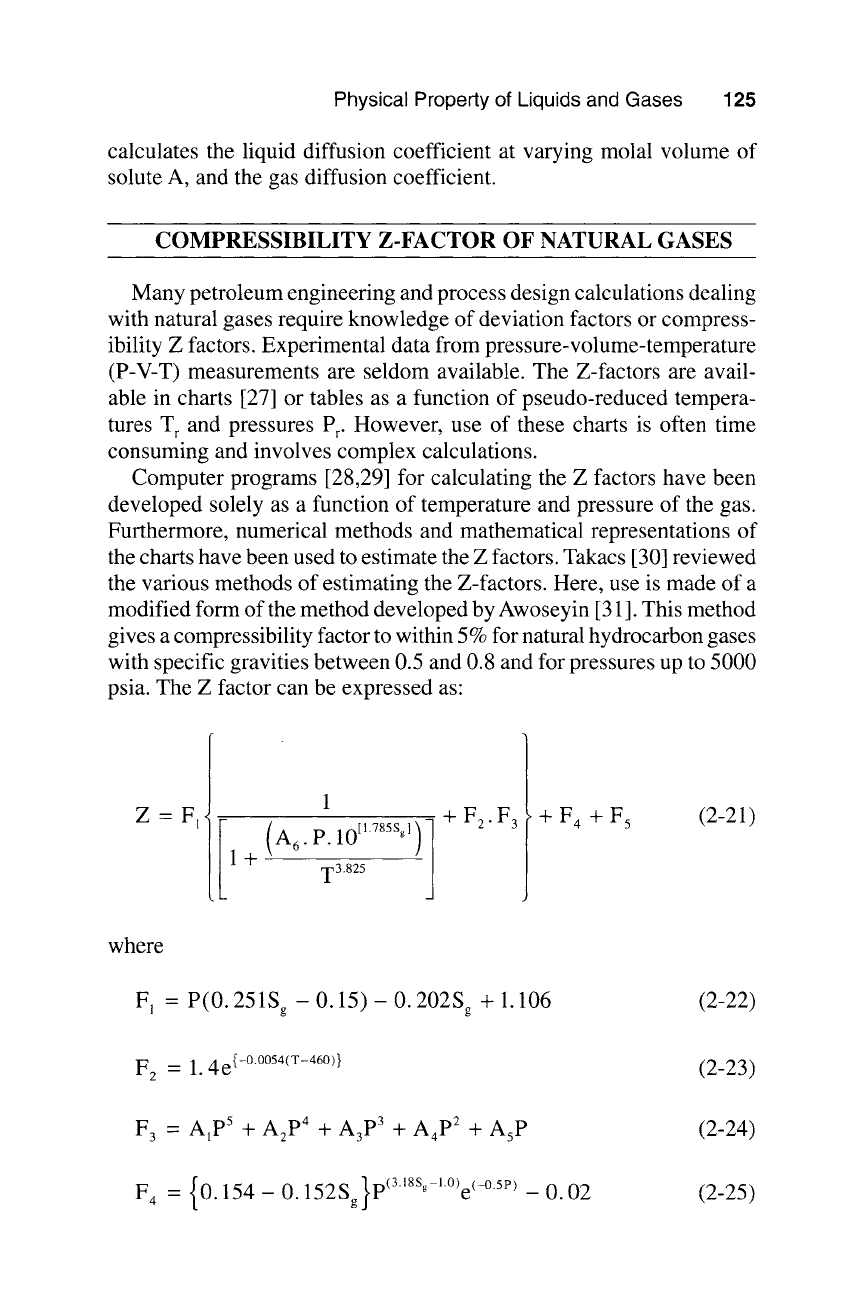
nents A and B is shown in Table 2-3. The computer program PROG23
Table 2-3
Atomic Diffusion Volumes for Use in Calculating
DAB
by the Correlation of Fuller-Schettler-Giddings
Atomic and Structural Diffusion-Volume Increments, v
C
H
O
N*
16.5
1.98
5.48
5.69
CI*
S*
Aromatic ring
Heterocyclic ring
19.5
17.0
20.2
20.2
Diffusion Volumes for Simple Molecules, Y_,v
H2
D2
He
N2
O2
Air
Ar
Kr
Xe*
7.07
6.70
2.88
17.9
16.6
20.1
16.1
22.8
37.9
CO
CO 2
N20
NH3
H20
CClzF*
SF *
CI*
Br*
so*
18.9
26.9
35.9
14.9
12.7
114.8
69.7
37.7
67.2
41.1
*Based on a few data points
Physical Property of Liquids and Gases 125
calculates the liquid diffusion coefficient at varying molal volume of
solute A, and the gas diffusion coefficient.
COMPRESSIBILITY Z-FACTOR OF NATURAL GASES
Many petroleum engineering and process design calculations dealing
with natural gases require knowledge of deviation factors or compress-
ibility Z factors. Experimental data from pressure-volume-temperature
(P-V-T) measurements are seldom available. The Z-factors are avail-
able in charts [27] or tables as a function of pseudo-reduced tempera-
tures T r and pressures
Pr.
However, use of these charts is often time
consuming and involves complex calculations.
Computer programs [28,29] for calculating the Z factors have been
developed solely as a function of temperature and pressure of the gas.
Furthermore, numerical methods and mathematical representations of
the charts have been used to estimate the Z factors. Takacs [30] reviewed
the various methods of estimating the Z-factors. Here, use is made of a
modified form of the method developed by Awoseyin [31 ]. This method
gives a compressibility factor to within 5 % for natural hydrocarbon gases
with specific gravities between 0.5 and 0.8 and for pressures up to 5000
psia. The Z factor can be expressed as:
Z=F l
I
, + (A6 P 785s ) 1
+ F 2 . F 3
+ F 4 +
F 5 (2-21)
where
F~ - P(0.251Sg - 0.15) - 0.2028g + 1.106
(2-22)
F 2 - 1.4e {-~176176162176
(2-23)
F 3 -
A~p 5
+ A2 p4 + A3 P3 + A4 p2 +
AsP
(2-24)
F 4 -
{0. 154 - 0. 152Sg}p~3"ss~-"~ ~-~ - 0.02
(2-25)

126
Fortran Programs for Chemical Process Design
F 5 - 0.35{(0.6- Sg)e [-1~ }
(2-26)
The values of the constants A~,
A2, A3, A4,
As, and
A 6 are:
A 1 = 0. 001946
A 2 = -0.027635
A 3 = 0. 136315
A 4 = -0.23849
A 5 =0.105168
A 6 -
3.44 x 10 s
The specific gravity,
S g,
of natural gas can be calculated from its
density or molecular weight. This is expressed as the ratio of the gas
density at 60~ and 1 atmosphere (14.7psia),
Pgas,60OF, to
the density of
air, Pair,60OF,
under the same conditions.
Sg -
density of gas
density of air
Pgas,60~
Pair,60~
(2-27)
Using the molecular weight of the gas, Sg, can be expressed as"
Sg =
molecular weight of gas
molecular weight of air
Mw, gas
M
w, air
(2-28)
Appendix A2 shows the computer outputs of the Z-factor as a func-
tion of temperature, pressure and specific gravity. The computer pro-
gram PROG24 calculates the compressibility Z-factor of natural gases
as a function of specific gravity and temperature. Figure 2-13 gives a
plot of the compressibility Z-factor of natural gases at 60~ as a func-
tion of pressure and specific gravity. Tables 2-1 to 2-25 in Appendix B
give results of Z-factors at varying specific gravity and temperature.
