Heidersbach R. Metallurgy and Corrosion Control in Oil and Gas Production
Подождите немного. Документ загружается.

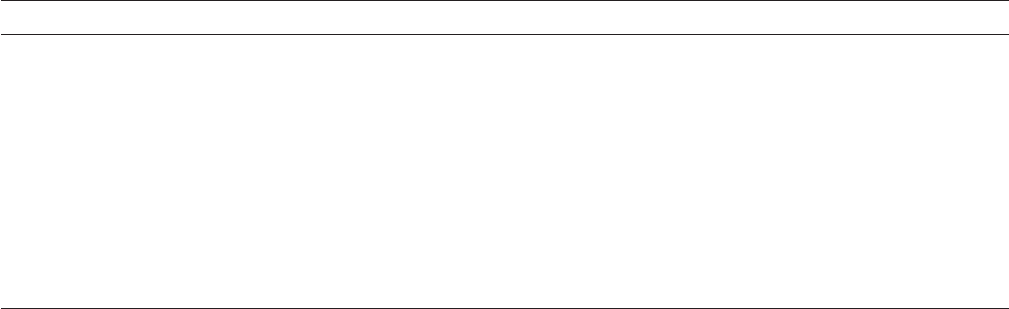
FORMS OF CORROSION 99
that cause this form of corrosion is the presence of
chlorides, although copper - based alloys stress corrode
in ammonia and other nitrogen - containing environ-
ments . Many environments that cause SCC are only
mildly corrosive insofar as other forms of corrosion are
concerned. It is not unusual to see SCC on surfaces that
otherwise seem to be uncorroded, or only mildly cor-
roded, at least to the unaided eye.
SCC initiation sites include pits, metallurgical defects,
surface discontinuities, intergranular corrosion, and
other stress raisers.
65
SCC is often unaccompanied by
visible corrosion products and may appear to be the
result of strictly mechanical causes. This is shown in
Figures 5.50 – 5.54 . Note the “ river branching pattern ” of
cracks in Figure 5.55 . This branched - crack appearance
is direct evidence of SCC, and no other metallurgical
failure mode produces this pattern of cracking. The
absence of this branching should not be taken as evi-
dence of a lack of SCC, because many metals, to include
high - strength steels, may reach a critical fl aw size and
fail by SCC before the crack has extended long enough
to start branching. Cracks as short as a fraction of
an inch (several millimeters) have been known to
produce SCC failures in high - strength steel and other
materials.
The growth of stress corrosion cracks is discontinu-
ous and is thought to usually involve initiation and fi rst -
stage propagation, secondary steady - state propagation,
and fi nal rapid failure. HE has been proposed as the
mechanism whereby these stages occur, although this is
disputed by some researchers. The strongest evidence
for hydrogen - related mechanisms is in high - strength
ferritic steels.
65
In the presence of active corrosion,
usually pitting or crevice corrosion, the cracking is gen-
erally called SCC, but many authorities suggest that this
should more properly be called hydrogen - assisted stress
corrosion cracking (HSCC). This form of corrosion
will treat environmental cracking in accordance with
the guidance available in NACE/ISO RP010156, the
most widely used oilfi eld standards on the subject.
33,62,63
There are a wide variety of terms used to describe envi-
ronmental cracking, including SCC, season cracking,
corrosion fatigue, HE, caustic embrittlement, and liquid
metal embrittlement. Many oilfi eld personnel will use
the term SCC as a synonym for all of the above terms.
One clear consensus is that most environmental
cracking, with the exception of corrosion fatigue, occurs
due to a combination of tensile stresses and specifi c
corrosive environments. Corrosion fatigue can occur in
any corrosive environment. A few oilfi eld environments
are listed in Table 5.5 . It is apparent from Table 5.5 that
H
2
S and chlorides are common to environmental crack-
ing in most oilfi eld environments. Despite all of the
research on environmental cracking, no screening tests
have been developed that identify new environments,
and all of the alloy – environment combinations have
been identifi ed due to fi eld failures.
At one time, environmental cracking was considered
to be a problem of certain alloys in certain environ-
ments. Unfortunately, every structural alloy system has
some environment where cracking is known to occur.
64
Note that in the following discussion, there are some
mechanisms where the stresses come from chemical
reactions either on the surface or within the metal.
This concept of environmental cracking is a major
departure from the idea of tensile stresses (applied or
residual) found in the general (as opposed to oilfi eld)
literature.
SCC
This term is used for most aqueous environmental
cracking that is not clearly associated with hydrogen or
H
2
S. The most common characteristic of environments
TABLE 5.5 Metals and Environmental Cracking Environments
46
Metal Environment Factors That Increase Risk of SCC
Carbon steels Hydrogen sulphide Increasing H
2
S, moderate temperatures, more acidic, higher strength/
hardness, higher stress levels
Carbon steels Carbonates Higher strength
Carbon steels Chlorides Higher strength, higher stress levels, more acidic
Copper alloys Ammonia Higher strength, higher stress levels
Martensitic stainless steels Hydrogen sulphide Increasing H
2
S, moderate temperatures, more acidic, higher strength/
hardness, higher stress levels
Austenitic stainless steels Chlorides Higher strength, higher chloride levels, higher stress levels, more acidic,
higher temperatures, presence of H
2
S
Duplex stainless steels Chlorides Higher strength, higher chloride levels, higher stress levels, more acidic,
higher temperatures, presence of H
2
S
Titanium Alcohol Higher stress levels, lower water content
c05.indd 99c05.indd 99 10/29/2010 10:35:28 AM10/29/2010 10:35:28 AM

100 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
possibility of hydrogen involvement in the SCC
mechanism(s) is an indication that corrosion control
methods based on cathodic protection must be used
with caution. This is one reason why pipelines, where
corrosion control is normally by a combination of pro-
tective coatings and cathodic protection, are seldom
constructed out of high - strength (yield stresses greater
than 80 ksi [550 MPa]) steel. Most cathodic reduction
reactions on carbon steel pipelines are due to oxygen
reduction, but the possibility of hydrogen reduction,
especially beneath disbonded protective coatings, is a
potentially serious concern.
Unlike the controversy associated with mechanisms
of SCC in steels, there is no dispute that SCC in tita-
nium alloys can involve hydrogen, and hydrides of tita-
nium have been detected in titanium after SCC events.
Stress corrosion cracks often appear in groups or
colonies on otherwise uncorroded surfaces. This is
shown in Figure 5.52 , which shows intersecting cracks
on the outside of a buried pipeline during external exca-
cracking can occur in almost any acidic environment
where hydrogen reduction is the cathodic reaction.
Increasing the strength and hardness levels of carbon
steel, high - strength low - alloy steels, and martensitic
stainless steels often increases susceptibility to this form
of cracking.
67
Carbon steels and other ferritic steels are the most
commonly used metals in oilfi eld environments, and the
Figure 5.50 Stress corrosion cracking underneath insulation
on a stainless steel condensate line. Photo courtesy of NACE
Basic Corrosion Course, reproduced with permission.
Figure 5.51 Multiple branching cracks typical of many stress
corrosion cracking failures.
55
Figure 5.52 Clustered “ colonies ” of stress corrosion cracks
on the outside of a carbon steel pipeline.
66
Figure 5.53 Small cracks joining together and intersecting
circumferential cracks on the exterior of a pipeline.
68
c05.indd 100c05.indd 100 10/29/2010 10:35:28 AM10/29/2010 10:35:28 AM

FORMS OF CORROSION 101
determine the causes of cracking, for example, in pipe-
line steels where the pH of the surface moisture may
infl uence whether cracking is intergranular or trans-
granular.
68
Figure 5.55 shows the transgranular nature
of cracks in the pipeline steel shown in Figures 5.53 and
5.54 . While this analysis has proven useful in analyzing
the causes of SCC in buried pipelines, many cracks will
change mode during the crack progression.
4
SCC is controlled by avoiding metal - environment
combinations that cause this problem. Additional
methods of SCC control include various methods of
stress relief, for example, post - weld heat treatment, pro-
tective coatings, corrosion inhibitors, and cathodic pro-
tection. Much of this control, for example, in pipelines,
is devoted to avoiding the formation of stress risers,
small defects on metal surfaces that serve as initiation
sites for SCC which, once started, may be diffi cult to
control.
Caustic Embrittlement Caustic embrittlement is one
of the fi rst forms of environmental cracking or SCC to
receive widespread industrial attention. Boiler water is
treated to a pH known to reduce corrosion rates (typi-
cally about pH = 8 – 9) by treatment with sodium hydrox-
ide. If water evaporation produces higher pH fl uids, for
example, in crevices or at points where tubes meet
restraints such as headers or baffl es, the pH can increase
to levels as high as 14, and this can cause intergranular
cracking of carbon steels. Caustic cracking is controlled
by the use of buffers, which prevent the buildup of high
pHs, and by the substitution of ammonium hydroxide
for sodium hydroxide. At high temperatures the
ammonia evaporates, which deprives the hydroxide
(OH
−
) ions of the necessary cation to balance electrical
charge and prevents the buildup of excessive pHs.
69
The
principals of boiler water treatment were developed in
the 1920s by the U.S. Bureau of Mines, and recent
advances have come from a variety of companies that
specialize in boiler water treatment chemicals. Most oil-
fi eld operators rely on guidance from these specialized
water treatment suppliers.
HE and H
2
S - Related Cracking
The small size of hydrogen atoms means that hydrogen
can readily dissolve in most metals. The dissolution of
hydrogen into metals comes from two common sources:
the reduction of hydrogen ions at cathodes in electro-
chemical cells due to corrosion or electroplating and the
entry of hydrogen into metals from environments having
hydrogen - entry promoters such as H
2
S and cyanides.
These environments produce small amounts of mona-
tomic hydrogen which usually combine to form molecu-
lar (diatomic) hydrogen molecules, which are too large
Figure 5.54 Typical cross section of secondary cracks
showing mid - wall bifurcation.
68
Figure 5.55 Transgranular SCC in pipeline steel.
68
100 μm
Mag. Approx. ×150
vation and examination. Cracks, either internal or exter-
nal, will often grow together until they reach a critical
fl aw size that may lead to fi nal rupture. This is shown in
Figure 5.53 , where small cracks have joined together
and a circumferential crack has grown around a
pipeline.
Cracks often branch as they progress into the metal.
There are a number of reasons for this branching, which
is shown in Figure 5.54 for the same pipeline that is
shown in Figure 5.53 . Note how the crack splits at
approximately the mid - wall location in the pipeline and
how the outside surface is generally roughened, even
though only one crack seems to have started at the
surface shown in this picture.
SCC can progress in an intergranular (between the
crystals) or a transgranular (across the crystals) manner.
This is one means that failure analysts have used to
c05.indd 101c05.indd 101 10/29/2010 10:35:29 AM10/29/2010 10:35:29 AM
102 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
some of which invariably dissolves into the steel. The
standard way of compensating for this inevitable intro-
duction of hydrogen into the metal substrate is to use
an elevated - temperature bake - out procedure of several
hours, depending on metal thickness, at temperatures
around 175 – 205 ° C (350 – 400 ° F).
Higher - strength steels are considered to be more sus-
ceptible than lower - strength alloys, and work hardening
seems to be preferable to heat treatment, for example,
in high - strength cables.
2
Concerns for HE are the
reasons that very high - strength wire, used in downhole
wireline applications, must be allowed to outgas for
days between downhole trips. Many authorities con-
sider the highly cold - worked metals used in these wires
to be less susceptible to hydrogen effects than thicker
metals, usually heat - treated for strength, at the same
strength (or hardness) levels. The presence of multiple
defects, primarily dislocations, is thought to serve to
minimize the accumulation of hydrogen in any one loca-
tion and to minimize formation of subsurface molecular
(diatomic) hydrogen considered to be associated with
HE.
Hydrogen charging, the introduction of monatomic
hydrogen into metals, can come from the breakdown of
water at elevated temperatures in welding processes.
This is the reason for protective, water - impermeable
coatings on most welding rod.
70
Charging can also occur on cathodes at defects expos-
ing steel underneath anodic protective coatings, for
example, zinc - plating. This is the reason that galvaniz-
ing (zinc coatings) is not allowed on high - strength
fasteners.
71
HE is considered to be a relatively low - temperature
phenomenon, and most failures seem to occur at tem-
peratures below 100 ° C (212 ° F). Prolonged exposure to
high temperatures can have counteracting effects. More
hydrogen may be generated on metal surfaces due to
accelerated corrosion or other chemical reactions, but
atomic diffusion and subsequent outgassing is also
enhanced.
Hydrogen Stress Cracking ( HSC ) This is a term used
in NACE RP0176/ISO 15156 to describe cracking in a
metal due to the presence of hydrogen in a metal along
with residual or applied tensile stresses.
62
It is used to describe cracking in metals that are not
sensitive to sulfi de stress cracking (SSC) but which are
embrittled when galvanically coupled as cathodes to
corroding anodes. The term galvanically induced HSC
is applied to this mechanism of cracking. The discussion
above on HE of galvanized bolts would be considered
to involve HSC by many oilfi eld authorities, although
this is not the terminology used outside the oilfi eld
industry.
to dissolve into metals. In the short time that mona-
tomic hydrogen atoms exist on surfaces, small amounts
of monatomic (nascent) hydrogen dissolve into the
metal substrate and follow diffusion paths from loca-
tions of high concentrations (the source surface) to
regions of lower concentration (the metal interior and
usually the opposite surface). This dissolution is usually
at interstitial sites (between the atomic locations of the
metal). Several forms of hydrogen degradation are asso-
ciated with the recombination of internal hydrogen
atoms to form hydrogen molecules, which are too large
for interstitial diffusion through the metal lattice. Other
forms of HE are due to mechanisms that are not pres-
ently understood and are the subject of research
controversies.
HE and other hydrogen - related problems can occur
in any H
2
S - containing environment and in electroplated
metals even in the absence of environmental hydrogen.
Another source of monatomic hydrogen is welding, and
improper welding procedures can introduce monatomic
hydrogen into metals.
There are a wide variety of hydrogen and H
2
S - related
cracking phenomena encountered in upstream environ-
ments. Many of the classifi cations discussed below
follow terminology in NACE MR0175/ISO 15156,
which emphasizes the choice of materials for use in H
2
S
environments.
33,62,65
HE Small quantities of hydrogen inside certain metals
make them susceptible to subcritical crack growth under
stress. Metals can also have major decreases yield
strength and undergo brittle failures in hydrogen -
containing environments. Both processes are commonly
called HE. Oilfi eld metals with HE problems include
high - strength steels, aluminum, and titanium, although
most problems occur in high - strength steels. The exact
mechanisms of HE in steels have not been established,
and no iron hydrides have ever been reported, but
brittle intermetallic hydrides have been found in tita-
nium and other hydrogen - embrittled metals. Ferritic
steels, for example, carbon steels and low - alloy steels,
are considered to be more susceptible than austenitic
alloys.
The initial 1975 version of NACE RP0175 limited
H
2
S exposures to metals having a hardness of HRC22
or less which, depending on the size and shape of the
metal, correlates to yield strengths of approximately
80 ksi (550 MPa).
The sudden propagation of brittle fractures may be
time delayed and occur months, even years, after expo-
sure to hydrogen. This is a characteristic failure mode
in plated metal components. It is thermodynamically
impossible to electroplate metals such as zinc or chro-
mium onto steel without also generating hydrogen gas,
c05.indd 102c05.indd 102 10/29/2010 10:35:30 AM10/29/2010 10:35:30 AM

FORMS OF CORROSION 103
they are used for determining environments where
HIC - resistant steels are necessary. Many operators
assume that virtually all oil fi elds will eventually sour
and require H
2
S - resistant materials, but some gas fi elds
are depleted so quickly that this may not be
necessary.
HIC susceptibility has been shown to be greatest in
high - sulfur steels and to be less likely in modern, low -
sulfur steels. However, in low - sulfur steels other consid-
erations, such as the presence of ferrite - pearlite banding,
also promote HIC. Condensate waters in gas systems
lack the mineral buffering available from formation
waters that accompany crude oil. For this reason, gas
Hydrogen - Induced Cracking ( HIC ) HIC, also known
as stepwise cracking, in carbon and low - alloy steels is
caused by atomic hydrogen diffusing into the steel and
forming hydrogen molecules internally at trap sites,
such as vacancies in the metal, grain boundaries, dislo-
cations, and second - phase particle boundaries, to
include inclusions.
70 – 73
HIC is a form of hydrogen -
related cracking that does not require tensile stresses,
either applied or residual, to produce cracking. This
lack of applied stresses is the main differentiation
between HIC and SCC. HIC is a major concern in the
H
2
S environments covered by NACE SP0176/ISO
15156 and in other environments, for example, strong
mineral acids (H
2
S is a weak mineral acid), known to
produce HE.
62
Figure 5.56 shows HIC in pipeline steel.
74
Note how
the cracks form parallel to the rolling direction of the
steel and how they tend to be planar — relatively long
and fl at. Modern pipeline steels are treated with calcium
that reduces the total volume of inclusions and tends to
form hard, rounded inclusions which should present
fewer locations for HIC to form.
75
Figure 5.57 shows environmental conditions where
HIC is considered important for pipeline steels. The
combinations of carbon dioxide (CO
2
) and H
2
S in the
environment shown in this fi gure are approximate, but
Figure 5.56 Hydrogen - induced cracking (HIC) in pipeline
steel.
74
Figure 5.57 Environmental conditions where HIC is important.
75
MODEST RISK OF HIC
Copper additions may be
effective
MODEST RISK
OF HIC
Increasing level of S
in the pipeline steel
SEVERE
RISK OF HIC
VERY SEVERE
RISK OF HIC
SEVERE RISK OF HIC
HIC
DOES
NOT
OCCUR
HIC
DOES
NOT
OCCUR
20
5
0.5
0.1 1 5
pH > 4.5 Oil Pipelines
a
b
pH < 4.5 Gas Pipelines
PP Carbon Dioxide (bar)PP Carbon Dioxide (bar)
Partial Pressure of Hydrogen Sulfide (bar)
Partial Pressure of Hydrogen Sulfide (bar)
110
c05.indd 103c05.indd 103 10/29/2010 10:35:30 AM10/29/2010 10:35:30 AM
104 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
way as the smaller cracks. Hydrogen diffuses into the
steel and then gets trapped at locations where migration
is hindered, and the recombination of thermodynami-
cally unstable monatomic hydrogen to thermodynami-
cally stable diatomic hydrogen molecules is most likely
to happen. The most likely sites for this to happen are
near large ceramic inclusions (impurities) within the
steel. Figure 5.59 shows schematically how these inclu-
sions are likely to be arranged in steel plate and other
thick sections. As a result of the hot rolling process,
ceramic inclusions near plate surfaces are more likely
to be broken up and become smaller. Near the center
of plate steel there has been less plastic deformation,
and both the steel crystals and the inclusion particles
tend to be larger. As hydrogen migrates through the
steel, the chances for monatomic atoms to meet and
become trapped are greater near the larger inclusions
near the center of the plate. This is the reason why plate
steel, especially the relatively low - quality, inexpensive
plate steel used for storage tanks and similar structures,
is more likely to produce hydrogen blistering. Many of
these blisters form near the center of plate steel and can
systems are likely to have acidic environments and are
usually more corrosive than crude oil systems. If the gas
contains H
2
S, then the gas systems will require H
2
S -
resistant materials of construction.
76
SSC This common form of environmental cracking
(SSC) is considered to be a form of HIC. It requires a
residual or applied tensile stress and the combined pres-
ence of water and H
2
S, which indicates that some orga-
nizations would consider this to be a form of SCC,
although this is not the interpretation in NACE RP0175/
ISO 15156.
62
Susceptible alloys, especially steels, react with H
2
S,
forming metallic sulfi des and monatomic nascent hydro-
gen. This monatomic hydrogen forms as a reduction
reaction product and diffuses into the metal matrix,
causing internal cracking. High nickel contents, which
limit surface corrosion and also cause the microstruc-
ture to have austenite, a more resistant phase than the
ferrite common in carbon and low - alloy steels, greatly
improve the resistance to SSC. SSC is worst around
80 ° C ± 20 ° C (176 ° F ± 36 ° F). Above this temperature,
hydrogen is more mobile and is likely to diffuse from
the metal before forming internal defects leading to
cracking. The term SSC is applied to cracking in liquid
water environments.
Stress - Oriented Hydrogen - Induced Cracking ( SOHIC )
Applying stresses to steel with HIC can result in inter-
connecting cracks between the hydrogen - induced
cracks. The result is relatively long cracks in the through -
thickness direction of a structure, for example, a pipe-
line.
62
This is shown in Figure 5.58 , where the small
hydrogen - induced cracks have been joined by connect-
ing cracks, resulting in considerably less resistance to
applied stresses. The linking of hydrogen - induced
cracks is referred to as SOHIC or as stepwise
cracking.
Susceptibility to SOHIC can be complicated, as low -
sulfur modern steels, thought to be resistant to this form
of damage, have been found to have additional prob-
lems associated with the metallurgical treatments of
newer steels used in repair and new construction. The
HIC shown in Figure 5.56 could lead to SOHIC in the
presence of applied stresses, for example, the hoop
stresses found in pressurized pipelines.
Hydrogen Blistering The small cracks discussed above
in sections on HIC, SSC, and SOHIC are sometimes
referred to as small blisters. Another use of the term
hydrogen blistering is for hydrogen gas blisters that
form inside structures that become so large that their
deformations can be seen macroscopically as surface
deformations. These larger blisters form in the same
Figure 5.58 Stress - oriented hydrogen - induced cracking
(SOHIC).
73
c05.indd 104c05.indd 104 10/29/2010 10:35:30 AM10/29/2010 10:35:30 AM

FORMS OF CORROSION 105
steels are not immune to this problem, but it is much
less common in the highly processed and refi ned low -
alloy steels used for most process pressure vessels.
Typical treatments for hydrogen blistering depend
on the application. For storage tank bottoms, which are
loaded in compression, it is fairly common to locate the
boundaries of the blisters with ultrasonic inspection.
Then, at a predetermined distance from the boundaries,
a plate is welded over the blister, and the blister is left
in place. Other organizations carefully pierce the blister
using non - sparking drills to relieve the pressure before
welding a patch over the surface. Similar procedures are
sometimes used on tank walls, and monitoring the
growth of blisters has been reported.
55
In pressure vessels, it is common to grind the blisters
out of the metal. This grinding to a predetermined dis-
tance beyond the detected limits of the blister is intended
to remove both the overlying “ blister ” steel, which
serves no structural purpose, and also to eliminate any
microcracking that has not been detected by the inspec-
tion process. The remaining wall thickness is then deter-
mined. While welding repairs are sometimes performed
to replace the missing metal, it is more common to
determine the maximum allowable operating pressure
of the equipment and to downgrade the service if neces-
sary. If reductions in operating pressure are inadvisable,
then external repairs (sleeves, clamps, etc.) are used to
increase the effective wall thickness in the area of
concern. The same downgrading or external reinforce-
ment procedures are used for pipelines, but it is seldom
feasible to grind out the blisters, especially if they are
internal.
The best way to avoid hydrogen blistering is by the
use of higher - quality, lower - inclusion steel, and this is
the reason that most hydrogen blisters are encountered
in above - ground storage tanks and other large struc-
tures where the economics of construction from higher -
grade steel are not justifi ed and inspection and repair
of occasional blisters is an accepted alternative.
Hydrogen Attack
High - temperature hydrogen environments can cause
hydrogen to diffuse into the metal and react with the
carbon in the iron carbides (cementite) in steel. The
resulting formation of gaseous methane can cause blis-
tering similar to HIC or hydrogen blistering. This form
of attack, while a concern in refi ning, is seldom a concern
in upstream operations.
Liquid Metal Embrittlement ( LME )
Liquid metals can attack solid metals and produce
fractures similar to those found in other forms of
become several inches (centimeters) in horizontal
dimension.
Common locations for hydrogen blisters include the
bottoms of above - ground storage tanks, where acidic
waters can concentrate, and the walls of tanks and
process equipment. Figure 5.60 shows blisters in the
wall of a CO
2
scrubber, and Figure 5.61 shows blisters
in a crude oil pipeline that collected sour crude for
many years.
The quality of steel determines the resistance to
hydrogen cracks and blisters of all types. Pressure vessel
Figure 5.59 Schematic representation of ceramic inclusions
(dark marks) and hydrogen blister formation in plate steel.
Figure 5.60 Hydrogen blister formed on wall plate of a CO
2
scrubbing tower.
55
Figure 5.61 Hydrogen blister on the inside of a crude oil
pipeline.
c05.indd 105c05.indd 105 10/29/2010 10:35:30 AM10/29/2010 10:35:30 AM
106 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
5.62 assume that the cyclic stresses are the same on each
repetition. This is approximately true for many rotating
shafts, sucker rods, and so on, but it is far from true for
wave loading on offshore structures and for many other
oilfi eld applications.
Fatigue cracks, like many other forms of environ-
mental cracks, often start at stress raisers such as corro-
sion pits, machined notches, or surface scratches. Once
corrosion starts, localized corrosion fatigue cracks
develop occluded cell concentration differences com-
pared to the external environment, and the interaction
between chemistry and mechanical loading becomes
very complicated and virtually impossible to predict or
model. It thus becomes important to monitor likely
fatigue sites for crack initiation and propagation in
attempts to detect damage and correct the situation
before fracture occurs.
Figure 5.63 shows a typical corrosion fatigue fracture
surface. The origin of the fracture surface is at the upper
center of the picture and the crack grew radially from
this origin, leaving indications of the crack progression,
called beach marks or clamshell markings, which are
indications that the equipment tarnished to different
levels, possibly due to intermittent operation of the
equipment or changes in the cyclic loading level. Once
the crack reached a critical fl aw size, the part broke,
leaving a shiny fracture surface at the bottom of the
picture. It is apparent that other surface defects started
additional cracks in the lower right of this picture. The
appearance of beach marks is a characteristic feature of
many corrosion fatigue failures. Note how the fatigue
fracture spread over most of the metal surface before
the fi nal tensile overload indicated by the shiny surfaces
at the bottom of the picture.
Another corrosion fatigue fracture surface is shown
in Figure 5.64 . This oilfi eld sucker rod has a clearly
defi ned origin, and beach marks indicate how the frac-
ture spread from this origin due to cyclic loading. After
environmental attack. Both intergranular and trans-
granular attack is known to occur. This form of attack
can happen during welding and other processes, but
careful removal of low - melting coatings, for example,
zinc coatings, prior to welding normally solves this
problem.
Most concerns with LME in upstream operations are
offshore and relate to safety in fi res and to cracking of
brazed aluminum heat exchangers, which are used in
cooling natural gas to reduce the volume before inject-
ing it into a pipeline for transmission to shore.
Many operators have banned the use of aluminum
or galvanizing for offshore platform structures, for
example, hand rails and ladders, because of a concern
that these liquid metals could cause embrittlement of
carbon steels and stainless steel if the lower - melting zinc
or aluminum were to melt in a fi re. The counter argu-
ment to this thinking is that by the time temperatures
have reached the melting point of these metals, it is too
late for the equipment concerned. No fi rm guidelines
on this question exist, and many operators are now
using aluminum, for example, for decks, ladders, hand-
rails, and helidecks. The thin layers of zinc on galva-
nized structures are not considered adequate corrosion
protection for most offshore structural members.
Galvanizing is commonly used for corrosion resistance
in bolts and similar fasteners, often with an overlay of
an organic coating for ease of disconnect. In some loca-
tions, cadmium, which is banned for toxicity reasons by
some authorities, is the preferred anodic coating mate-
rial for fasteners, although it is seldom applied to larger
components.
Brazed aluminum heat exchangers are used for off-
shore cooling of natural gas, because of their weight
savings over other metals. To avoid cracking by liquid
mercury, which is eventually found in virtually all
natural gas formations, it is standard practice to place
mercury removal processes in the gas treatment stream
prior to the brazed aluminum heat exchangers used for
cryogenic cooling.
76
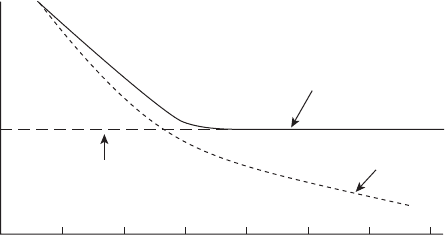
Corrosion Fatigue
Corrosive environments lower the fatigue life of many
oilfi eld components and structures. This is shown sche-
matically in Figure 5.62 , which shows the elimination of
the fatigue or endurance limit due to the presence of a
corrosive environment. It is possible to cyclically stress
a component so quickly that the effects of corrosion are
minimal, and it is also possible to introduce components
so corrosive that they corrode to failure before the
effects of cyclic stresses become signifi cant. It is very
hard to predict the cumulative effects of corrosion and
fatigue. The simplistic ideas shown in Figures 4.16 and
Figure 5.62 The reduction in fatigue resistance due to a cor-
rosive environment.
Cycles to Failure
Endurance Limit
Noncorrosive Service
Corrosive Service
Breaking Stress →
10
3
10
4
10
5
10
6
10
7
10
8
10
9
10
10
c05.indd 106c05.indd 106 10/29/2010 10:35:31 AM10/29/2010 10:35:31 AM

FORMS OF CORROSION 107
detect and monitor fatigue cracking is an important part
of corrosion fatigue control. Methods of preventing cor-
rosion, such as the use of more corrosion - resistant
alloys, corrosion inhibitors, and cathodic protection are
also helpful.
OTHER FORMS OF CORROSION IMPORTANT
TO OILFIELD OPERATIONS
There is no universally accepted terminology for corro-
sion, and the oil and gas industry has adopted a number
of terms for corrosion that frequently overlap with the
terms discussed in previous sections of this chapter. This
section discusses some forms of corrosion commonly
discussed in the oilfi eld literature and some other forms
of corrosion likely to be encountered in oil and gas
production.
Oxygen Attack
Oxygen attack normally refers to pitting corrosion due
to the presence of dissolved oxygen in production fl uids.
This seldom occurs in downhole environments, and
when it does, it is usually due to inadequate treatment
of injection waters that were exposed to oxygen -
containing air in topside processing and storage. The
most common treatments for oxygen attack are mechan-
ical deaeration treatment of the water plus the use of
oxygen scavengers.
Sweet Corrosion
This term, which is becoming outdated, refers to corro-
sion in environments where the corrosion is due to the
presence of dissolved CO
2
. Most production fl uid cor-
rosion, especially in natural gas production, is due to
CO
2
. The most common treatments for sweet or
CO
2
corrosion is the use of corrosion inhibitors. These
Figure 5.63 Beach marks on the surface of a corrosion -
fatigue surface from a marine propeller.
Beach Marks
Origin
Figure 5.64 Corrosion fatigue and shear lip formation on a
sucker rod fracture surface. Photo courtesy of NACE Basic
Corrosion Course, reproduced with permission.
Figure 5.65 Schematic showing the side view of a typical
corrosion fatigue rod failure.
Origin
Polished
Surface
Shear
Lip
the crack progressed across approximately one - half
of the rod, the surface became rough, due to the fact
that the crack remained open during compression
strokes and did not polish the surface as much as near
the origin. Once the crack reached a critical fl aw size,
the rod broke, forming shear lips at approximately 45 °
to the fracture surface. The shadows formed by the
shear lips on the lower left and right of the picture
appear as dark triangles in Figure 5.64 . The bottom
center of the picture shows where the metal pulled apart
by shear, but the shear lip is down and away from the
viewer. Most overload failures, to include fatigue fail-
ures, will form shear lips on the fi nal fracture surface.
A side view of this phenomenon is shown in Figure 5.65 .
Most fatigue failures progress for signifi cant dis-
tances before fi nal overload failures, and inspection to
c05.indd 107c05.indd 107 10/29/2010 10:35:31 AM10/29/2010 10:35:31 AM

108 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
inhibitor can be maintained in the water phase at the
bottom of the line, it is diffi cult to apply this inhibitor
to the top of the line. Much effort concentrates on
developments in modeling of where TOL corrosion is
likely to occur and how delivery of corrosion inhibitors
can be maintained. Research efforts include modeling
the temperature profi les of subsea pipelines. This is
shown in Figure 5.70 , which shows the water dropout
profi le for a subsea pipeline.
Efforts to apply corrosion inhibitor to the inside top
of multiphase pipelines include periodic pigging with
Figure 5.66 Mesa corrosion of a 9 - chrome sucker rod.
Figure 5.67 Mesa corrosion on carbon steel plate.
55
Figure 5.68 Mesa corrosion at a threaded connection on
downhole tubing.
55
inhibitors become ineffective as the downhole tempera-
tures and pressures increase.
73
Sour Corrosion
The term sour corrosion refers to corrosion that occurs
in production fl uids due to the presence of H
2
S. H
2
S is
a weak mineral acid, and most corrosion reactions are
relatively minor in these environments.
Figure 3.3 compares the effects of oxygen, CO
2
, and
H
2
S on corrosion. It is obvious that H
2
S is the least cor-
rosive of the three gases. Most problems with H
2
S are
associated with hydrogen - related cracking and, while
the environments associated with H
2
S cracking are con-
sidered to be “ sour, ” the cracking is seldom referred to
as sour corrosion.
Mesa Corrosion
Mesa corrosion refers to corrosion in CO
2
environments
that produce relatively fl at surfaces where the metal is
protected by carbonate fi lms, usually siderite (FeCO
3
).
This corrosion is characterized as fairly deep pits in the
form of sharp - edged holes that are considered to look
like the fl at - topped “ mesa ” (table topped) mountains
found in the southwestern United States.
5,77
Mesa cor-
rosion is shown in Figures 5.66 – 5.68 . All three pictures
show relatively fl at surfaces with localized pitting where
the partially protective carbonate fi lms break down.
Most control of CO
2
(sweet) corrosion is by the use of
corrosion inhibitors.
Top - of - the - Line (TOL) Corrosion
The increased use of multiphase offshore pipelines and
gathering lines has led to interest in top - of - the - line cor-
rosion. This form of corrosion is shown schematically in
Figure 5.69 . Condensate, containing water high in CO
2
and possibly acetic acid or other organic acids, aggres-
sively attacks the top of horizontal pipelines where no
corrosion inhibitor is present.
This condensation happens as the pipeline cools with
distance from the compressor station. While corrosion
c05.indd 108c05.indd 108 10/29/2010 10:35:31 AM10/29/2010 10:35:31 AM
