Henini M. Handbook of Self Assembled Semiconductor Nanostructures for Novel devices in Photonics and Electronics
Подождите немного. Документ загружается.

Semiconductor Quantum Dots for Biological Applications 779
available using high-temperature crystal formation in organometallic solvents. Knowing that a
great number of divalent transition ions show strong coordination ability towards various solvent
molecules such as amine and phosphine containing molecules [43] , forming low-dimensional
structures referred to as chalco(genido)metalates [44] , researchers directed these reactions
towards the obtention of II–VI nanocrystals. In 1993 Bawendi and collaborators [37] introduced
the “ organometallic TOP/TOPO ” procedure by synthesizing highly luminescent CdSe quantum
dots with nearly perfect crystal structures and narrow-size variations (relative standard deviation
5%). This route was based on the pyrolysis of organometallic reagents (e.g. dimethylcadmium –
Cd(CH
3
)
2
– and bis(trimethylsilyl)selenium) by injection into high boiling point coordinating sol-
vents (like tri- n -octylphosphine oxide (TOPO) and tri- n -octylphosphine (TOP)). A series of orga-
nometallic routes was described aimed at the obtention of II–VI nanocrystals, mainly CdSe, but
also CdS, CdTe and ZnSe [45, 46] . CdSe nanoparticles showed narrower-size dispersion, conse-
quently a greater control on the amplitude of emission colours (ranging from violet to red).
The central point in this methodology is the control of the nucleation and growing steps of
the particles performed at high temperatures ( T 300°C) for an extended period of time (rang-
ing from minutes to hours, depending on the desired particle size) [47] . In this process, smaller
nanocrystals are broken down, and the dissolved ions are transferred to larger crystals. The rate
of this “ ripening ” process is dependent on both the temperature and the amount of the limiting
reagent [47, 48] . Continuous injection of precursor solutions into the CdSe reaction mixture at
300°C also produces larger nanocrystals. As a matter of fact, when using different TOP:TOPO
ratios for the preparation of ZnSe, Hines and Guyot-Sionnest commented that the reaction could
render nanocrystals so small that they could not be isolated by standard solvent/non-solvent pre-
cipitation techniques, neither could they precipitate as large aggregates [46] . The authors sug-
gested that these diffi culties arose from TOPO binding too strongly and TOP too weakly to Zn.
This new organometallic route succeeded in obtaining homogeneous nanocrystals with
a small size dispersion but the fl uorescence quantum yields were still relatively low ( 10%).
Moreover, it was observed that the TOP/TOPO system showed better growth control for CdSe
nanocrystals not larger than 4.5 nm (i.e. in the strong quantum-confi nement size regime, with
fi rst exciton absorption peak 600 nm) [49] .
Currently the organometallic procedures are still the most popular choice for the development
of a variety of colloidal nanocrystals in non-aqueous solutions although the main chemicals
used are highly toxic, expensive, pyrophoric and explosive (the systems are heated above TOPO’s
fl ash point: 350°C). To improve this methodology, some researchers tried to introduce other coor-
dinating systems in the reaction solution, such as amines which were a logical choice for ligands
of intermediate strength. Amines are slightly weaker bases than phosphine oxide and long-chain
alkylamines have much higher boiling points (hexadecylamine, bp: 330°C). In addition, the less
sterically hindered amine creates a larger capping density, which probably increases the surface
passivation. As such, dodecylamine (DDA) and hexadecylamine (HDA) have been successfully
used to cap the surface of CdSe nanocrystals [49, 50–53] . Another feature of these organo-
metallic methodologies is the possibility of the nanocrystal’s fi nal shape control (e.g. obtaining
nanorods) by altering the ratio of the surfactants (e.g. TOPO and hexylphosphonic acid, HPA)
[53–55] ).
Peng and Peng [49] , motivated by the instability and the low reproducibility of the optical
properties of the nanocrystals obtained by the organometallic route, developed a series of meth-
odologies based on CdO, CdCO
3
and Cd(acetate)
2
in fatty acid solutions aimed at the substitution
of the pyrophoric Cd(CH
3
)
2
. They argumented that this cadmium precursor actually decomposes
in hot TOPO and generates insoluble metallic cadmium precipitates. Studying a different set of
experimental conditions and precursors these authors suggested that the existence of any anion
of a strong acid, either in the form of the cadmium precursor or as an added cadmium ligand,
made it impossible to form high-quality CdSe nanocrystals in the current systems. Thiols, which
bind strongly to cadmium, were found to inhibit the nucleation process. Using this procedure
they reported a size range of nearly monodisperse CdSe crystals, from about 1.5 nm to above
25 nm, a much broader range than that achieved by the original organometallic method.
Peng and Peng [49] further reported that the temporal evolution of the size and size distribu-
tion of CdSe nanocrystals in fatty acid systems were quite reproducible, although the reaction
CH026-I046325.indd 779CH026-I046325.indd 779 6/25/2008 3:33:48 PM6/25/2008 3:33:48 PM
780 Handbook of Self Assembled Semiconductor Nanostructures for Novel Devices in Photonics and Electronics
rates were fast. They considered this to be due probably to the more controllable nucleation step
initiated by cadmium carboxylates less active than Cd(CH
3
)
2
used in the traditional organometal-
lic approach. This phenomenon implies that the control of the nucleation process may be the key
step towards a fully controllable synthesis. Moreover, the authors state that in practice, the fatty
acid systems are not recommended to synthesize small nanocrystals because of their fast growth
rates.
In the late 1990s two rather different alternative methodologies were also described for the
synthesis of selenides and tellurides. In the fi rst one the chalcogenide precursors were produced
in situ by using reducing agents such as KBH
4
, which converts Se
2
and Te
2
to Se
2
and Te
2
,
respectively [56, 57] . Wang and collaborators proposed a simple solution synthesis for pure
quantum dots of M chalcogenides ( M Bi, Cu, Cd, Sn, Zn; chalcogenide S, Se) by providing
the in situ reduction of S or Se in the presence of KBH
4
and the corresponding metal salt at room
temperature in strong basic solvents. They showed that the solvent signifi cantly infl uenced the
quality of the fi nal product, yielding small uniform nanoparticles (4–6 nm) in the case of ethyl-
enediamine and a mixed metal/chalcogenide precipitate with poor crystallinity and low yield in
the case of pyridine. In fact, pyridine is known to provide stable capping through the N atom, but
its low boiling point suggests limitations as a growth solvent.
Recently, amine-capped PbSe nanoparticles of tunable sizes and shapes were also obtained
with this method [58] . As in the previous methods, the main limitation is the diffi culty to achieve
narrow-size distributions and high crystallinity.
The second methodology, proposed for large-scale production, involves the application of ultra-
sound (formation and implosive localized hot spots induced by acoustic cavitations) on chemical
reactions [59, 60] . Zhu et al. reported the preparation of spherical ZnSe nanoparticles of average
sizes of 3, 4, and 5 nm by reacting Zn(acetate)
2
and selenourea in water followed by sonication
with a high-intensity ultrasonic probe under inert atmosphere for a determined period of time
[60] . Pb and Cu selenide nanoparticles were also obtained by using the corresponding acetates.
A recent search for “ greener ” and simpler procedures that could produce semiconductor
nanocrystals directly in aqueous media, aimed at bioapplications, readapted the thiol stabi-
lized CdTe synthetic methods originally reported by Rogach et al. [33, 34] and the one reported
by Nosaka et al. for the synthesis of CdS [36] . By using different approaches Gaponik et al. [38] ,
Zhang et al. [61] , and Menezes et al. [17] prepared highly luminescent CdSe or CdTe nanoparti-
cles directly in water. Gaponik reported the dissolution of Al
2
Te
3
in an acidic solution to render
the Te
2
precursor ions as H
2
Te(g), while, Zhang et al. [61] and Menezes et al. [17] used NaBH
4
in aqueous solution to reduce Te to Te
2
. This reduction process generates Na–Te–Te–Na a sta-
ble intermediate complex which will be converted to CdTe after injection of the metal precursor
complexed with a thiol molecule in water under inert atmosphere. These colloidal systems render
particles in the 2–6 nm range and show luminescence after a certain period of time suggesting
a slow kinetic surface passivation process [17] . This observation will be discussed in the next
section.
Regarding the inherent toxicity of these systems for in vivo applications, for example, it was
recently reported by Pradhan et al. [62] that the synthesis of pure and doped ZnSe QDs as an
alternative for CdSe nanocrystal aimed at the obtention of a less toxic labelling material which
could be envisioned for a safe use in in vivo experiments and diagnostics.
On the other hand, to avoid the inconvenient autofl uorescence observed in biological systems
when these are excited in the UV blue region of the spectrum, lower band gap II–VI semicon-
ductor QDs are also being converted into biolabels. These systems when quantized present the
onset of the absorption and emission bands in the near-infrared region (700–1300 nm). As an
example, Kumar and Jahkmola reported on the RNA-mediated fl uorescent PbS nanoparticles
as novel tools for biophotonic applications [63] . Also a recent report of DNA-directed semi-
conductor quantum dot synthesis described highly optically emissive PbS nanocrystals [64] .
Furthermore, Hinds et al. [65] were able to synthesize infrared emitting PbS QDs (4 nm) stabi-
lized with Guanine-triphosphates (GTP). The authors systematically investigated how nucleotide
functionalities (base, sugar and phosphate) infl uenced nanoparticle growth. They proposed a set
of rules for using nucleic acids as ligands in order to profi t from the natural biorecognition prop-
erties of DNA and programmable templates for nanoparticle synthesis.
CH026-I046325.indd 780CH026-I046325.indd 780 6/25/2008 3:33:48 PM6/25/2008 3:33:48 PM
Semiconductor Quantum Dots for Biological Applications 781
In summary, much work has been done in the synthesis of II–VI nanostructured semiconduc-
tor compounds in the past three decades. Still, researchers are looking for the ideal preparation
methodology, aimed at particles with fewer surface defects and small size dispersion using less
expensive, non-toxic, less risky and simpler experimental conditions which can be extended to a
large-scale production. The need to improve their surfaces in order to increase their fl uorescence
quantum yields, for example, prompted a close look at the QDs surfaces. The next section com-
prises the processes used to overcome this problem.
26.2.2 Still some problems: quantum dots have imperfect surfaces! The passivation process
The observed fl uorescence in semiconductor nanoparticles is produced upon the recombina-
tion of the charge carriers which are generated by light absorption. The fi rst colloidally obtained
nanocrystals showed a very low fl uorescence quantum yield ( ϕ 1%). The non-radiative proc-
esses involved in semiconductor nanocrystals are said to have the same physico-chemical nature
as those observed in bulk semiconductor materials [66] . Taking into account the high number of
surface atoms compared to bulk atoms, it was suggested that the main contribution for this was
that the prepared colloidal particles had a lot of surface defects (shallow and deep traps) where
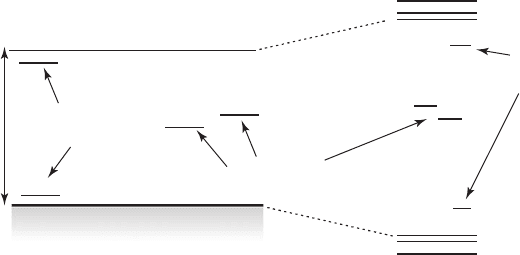
radiationless recombination of the charge carriers occurred. A schematic representation of these
defects is illustrated in Fig. 26.5 .
E
g
CB
VB
Shallow
traps
Shallow
traps
Deep traps
Bulk
QD
Figure 26.5 A schematic representation of deep and shallow traps (originated from structural defects) claimed to
be responsible for the radiationless processes in bulk and quantized semiconductor particles.
Soon it was realized that if the defect sites, most probably resulting from dangling bonds, were
located at the surface of the colloidal particles, there was a chance to chemically modify these sites.
Several studies on the chemical infl uence on the spectroscopic properties of semiconductor quan-
tum dots were reported. The fl uorescence intensity (and also the fl uorescence spectrum pattern)
of CdS nanocrystals was shown to be drastically increased by certain surface modifi cation pro-
cedures such as exchanging the aqueous solvent by alcohol [31] , adsorbing triethylamine in low
concentrations [31] , and covering the surface with cadmium hydroxide or silver sulphide [28] .
By coating the CdS nanocrystals with a layer of Cd(OH)
2
the resulting system is a core semi-
conductor coated by a shell of another semiconducting material possessing a higher band gap.
The resulting assembly was later called a core–shell system. The increased luminescence quan-
tum yields of the core–shell particles is explained by preventing from the photogenerated exci-
tons spreading over the entire particle, forcing them to recombine while being spatially confi ned
to the core. This luminescence enhancement was taken as an indication of the formation of the
proposed structure due to the diffi culty at that time to investigate such a thin chemical layer (just
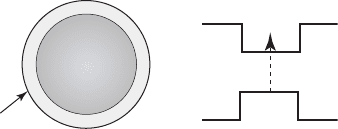
a few atomic monolayers). Figure 26.6 illustrates schematically the band gap offset created in the
interface of the CdSe core ( E
g
1.71 eV, wurtzite) and the ZnS shell ( E
g
3.68 eV, zinc-blende)
during the passivation process.
CH026-I046325.indd 781CH026-I046325.indd 781 6/25/2008 3:33:49 PM6/25/2008 3:33:49 PM
782 Handbook of Self Assembled Semiconductor Nanostructures for Novel Devices in Photonics and Electronics
Using the synthetic methods described in the previous section, several core–shell semiconduc-
tor nanoassemblies have been prepared since the CdS/Cd(OH)
2
CdS/Ag
2
S [67] , CdS/ZnS [68] ,
CdS/HgS [69] , CdSe/ZnS [70–73] , CdSe/CdS [50] , CdTe/CdS [17, 38, 61] . The capping of II–VI
nanocrystals with long chain organic surfactants was also utilized to passivate surface atoms,
but at room temperature the luminescence quantum yield was as high as 10% with a very long
fl uorescence lifetime and some non-band edge luminescence [74, 75] .
The deposition of a second layer onto the particle’s surface represents the chemical growth
of another crystalline phase. Two main problems may arise during this process: (i) formation of
single particles in the colloidal suspension instead of the second layer growth and (ii) imper-
fect growth patterns or even alloying of the second layer due to a large or too small crystal mis-
match, respectively. This last structural problem either may lead to unstable chemical assemblies
presenting a large quantity of surface defects or may alter the original optical properties of the
core itself.
The fi rst diffi culty is overcome by controlling the experimental parameters during the forma-
tion of the second layer. Very low concentrations of the capping precursors and a fast crystal-
lization rate are recommended. Dabbousi et al. [71] reported that the growth of ZnS shell on
CdSe particles was accomplished, without the precipitation of ZnS, by dropwise adding diethyl-
zinc and hexamethyldisilathiane (as Zn and S precursors) in equimolar amounts to vigorously
stirred CdSe colloidal solutions held at temperatures between 140 and 220°C, depending on the
core sizes. ZnS has a wider band gap than CdSe ( E
g
3,91 eV and 1.71 eV, respectively [76, 77] )
and, in fact, the authors report an enhancement of 10 to 50% of the quantum yield of CdSe
nanocrystals after this passivation procedure. The thickness of the capping layer was monitored
by small angle X-ray scattering (SAXS) measurements indicating 0.65 monolayers to 5.3 mono-
layers. The authors also suggest, by correlating the structural and optical properties, that the ZnS
layer tends to generate structural defects when more than 1.3 monolayers are grown, decreasing
the luminescence quantum yield.
The second diffi culty is the possible chemical alloy between the core and shell layers which
would infl uence the core optical properties. By choosing capping layers with different crystal
structures or even with same crystal structure but possessing different bond lengths one pre-
vents this possibility. For example, the growth of wurtzite-type CdS on wurtzite CdSe nanocrys-
tals shows a lattice mismatch of 3.9% which is small enough to allow epitaxial growth while
still preventing alloying. The band gap of CdS ( E
g
2.50 eV [76, 77] ) is greater than the CdSe
band gap ( E
g
1.71 eV [76, 77] ) promoting a quantum yield enhancement [50] . On the other
hand, in CdSe/ZnS core–shell nanostructures the Zn–S bond length is about 12% larger than
Cd–Se [77] . This chemical mismatch that would prevent the growth of fl at heterostructures in
epitaxial growth is believed to be relaxed in nanocrystals with short facets [71] explaining the
chemical stability and excellent optical properties of these core–shell nanoparticles. Regarding
the optical properties of the core after passivation, several reports on different core–shell systems
show that there is no observable modifi cation of the original properties of the core semiconduc-
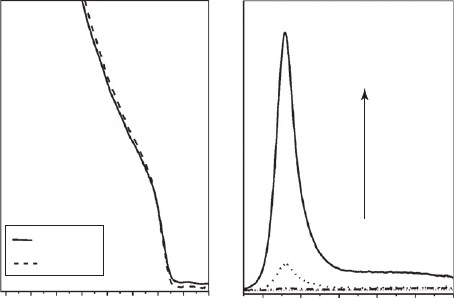
tor nanocrystal. As an example, Fig. 26.7a shows the absorption spectra of CdS nanoparticles of
d 6 nm prior and after passivation with a Cd(OH)
2
layer.
In this methodology the surface of the CdS nanocrystals is loaded with hydroxyl groups by
increasing the pH of the suspension up to 10.5 [28, 78] , followed by a dropwise addition of a
low-concentration solution of Cd
2
. The increase of the fl uorescence intensity may be monitored
CdSe
ZnS
ZnS
ZnS
h
CdSe
e
Figure 26.6 Schematic representation of the energy band gap offset between the CdSe core and the ZnS shell during
the surface passivation procedure.
CH026-I046325.indd 782CH026-I046325.indd 782 6/25/2008 3:33:49 PM6/25/2008 3:33:49 PM
Semiconductor Quantum Dots for Biological Applications 783
by emission spectroscopy (as observed in Fig. 26.7 ). The quantum yield increases to 20% and
the mechanism suggested is that the excess of OH
will convert SH
surface states into S
2
,
which in turn will bind to the excess Cd
2
ions. The cadmium-rich surface will then associate to
the polyphosphate anions present.
The passivation on the thiol-capped cubic CdTe nanocrystals prepared in aqueous solution
[61, 79–81] was brought up in a different form. It was mentioned in the previous section that
the photoluminescence of the colloidal suspensions of CdTe nanoparticles (2–6 nm) developed
slowly after its synthesis take up to several days to become readily observable even in daylight.
This suggested a slow kinetic mechanism of surface defect supression. Rockenberger et al. [79]
studied these systems using extended X-ray absorption fi ne structure measurements (EXAFS)
and observed that the particles possessed CdS bonds although they were larger than the bulk
value for the CdS bond. Later Bouchert et al. [81] and Zhang et al. [61] investigated and reported
on the nature of the surface of these particles by photoelectron spectroscopy (XPS). Zhang et al.
suggested that the carboxyl groups of the mercapto-carboxylic acid stabilizers coordinated to
the surface of the CdTe particles have a great infl uence on both their photoluminescence and
stability by substituting some Te
2
ions with oxygen of the carboxyl group. They also observed
CdS bonds, which they suggested result from the decomposition of the thiol groups. On the other
hand, Bouchert et al. showed strong evidence for Cd–SR bonds near the surface and no indica-
tions for the presence of Te atoms at the surface. They suggested that this was the result of the
hydrolysis of the thiol molecules which ended up incorporated onto the particle’s surface, substi-
tuting Te
2
ions and satisfying Cd
2
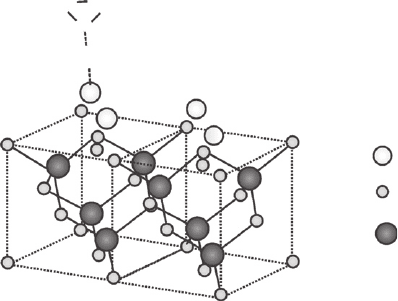
dangling bonds. Figure 26.8 shows a structure model of the
CdS layer growing on the surface of the cubic CdTe particle.
Thinking of a structural match one may observe that both the core and the shell possess cubic
crystalline structures. A reasonable extension of the thiol hydrolysis process would end up creat-
ing a CdS shell, which in turn possesses a higher band gap, and would be responsible for the pas-
sivation of the CdTe surface defects.
Once the particles are optimized to show a high fl uorescence quantum yield they may be
turned into fl uorescent biolabels by adapting their surface to be (i) water soluble and (ii) biocom-
patible. In the next sections further modifi cations of the nanocrystal’s surface are discussed.
26.2.3 How to render QDs soluble (hydrophilic surface) for biological applications.
Solubilization of the nanoparticles
In order to interact with biological systems there are two forms for the QDs to present them-
selves: solubilized in aqueous medium or encapsulated inside lipophylic carrying systems, such
200 300 400
Wavelength (nm) Wavelength (nm)
500 600 450 500 550 600 650 700
CdS
CdS-OH
Emission intensity
Absorbance (a.u.)
Incresing (Cd
2
)
Figure 26.7 (a) Absorption spectra of a polyphosphate stabilized CdS ( 6 nm) suspension prior to and after
the passivation procedure. (b) Monitoring the emission intensity increase as the cadmium hydroxide layer is being
deposited onto the particles’s surface.
CH026-I046325.indd 783CH026-I046325.indd 783 6/25/2008 3:33:50 PM6/25/2008 3:33:50 PM
784 Handbook of Self Assembled Semiconductor Nanostructures for Novel Devices in Photonics and Electronics
as micelles. Although the general properties of the nanocrystalline surface appear to be under-
stood, the exact surface chemistry involved in such processes remains to be elucidated.
While the QDs prepared in aqueous media such as the polyphosphate-and/or thiol-capped
CdS, CdSe and CdTe nanocrystals have inherent solubility, QDs obtained by the organometallic
route must overcome the aqueous incompatibility problem by altering the nature of their sur-
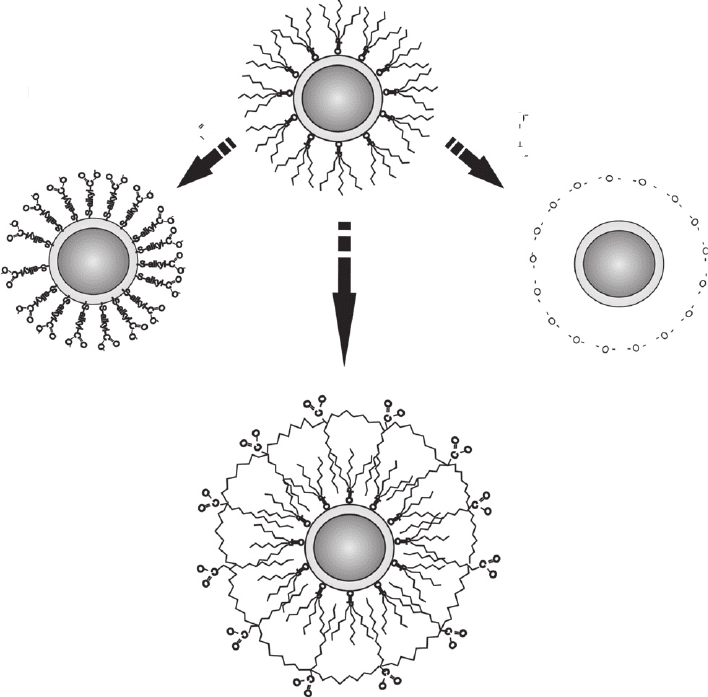
face. Among the different techniques developed for surface modifi cation, the four most general
methods described in the literature [82] are: the surface-exchange procedure, the silanization of
the nanocrystals, the formation of a hydrophilic interface with amphiphilic molecules and the
micellization of the nanocrystals. Figure 26.10 represents three of these methods.
The surface exchange of the hydrophobic surfactant molecules (e.g. TOPO) for bifunctional
binding water-soluble molecules [9, 83] has been employed using a wide variety of species.
These bifunctional linkers (such as HS–alkyl–COOH molecules) act in similar fashions to solu-
bilize the phosphine-capped nanocrystals and to provide functional groups (e.g. carboxylic acid
and amine) which may be used, in a posterior step, to conjugate to biomolecules using well-
established protocols. This capping procedure yields gram quantities of water-soluble QDs, but
the thiol ligands are not completely stable. Slow desorption of thiol–alkyl–acid molecules and the
possibility of intermolecular reactions resulting in disulphides decreases the capping layer and
leads to the fl occulation of the particles [82] . Moreover, these particles show an overall decrease
in their fl uorescence quantum yields [84] .
Another approach is the silanization with organosilicone molecules containing NH
2
or SH
functional groups [8, 84–88] . A silica/siloxane shell is formed on the surface by the induced
hydrolysis of the silanol groups. Polymerizing silanol groups help to stabilize the nanocrystals
against fl occulation but only small amounts (milligram quantities) can be prepared per batch
[24] . Also, residual silanol groups on the nanocrystal’s surface often lead to precipitation and gel
formation at neutral pH.
A third possibility is to apply phase-transfer methods using amphiphilic molecules that act as
detergents for solubilizing the QDs coated with hydrophobic groups [17, 26] . This method has
been particularly advantageous in allowing the retention of the native surfactant molecules,
which appear to increase the stability and fl uorescence effi ciency over those samples where the
native layer has been replaced with a bifunctional binding molecule. Encapsulating QDs and their
initial ligands with macromolecules such as polymers or lipids can preserve the emission quan-
tum effi ciency, but generally adds a large volume to the nanocrystals, resulting in a fi nal size that
may be greater than desired. This may diminish imaging sensitivity by decreasing the number of
QDs that can be attached to a target. For in vivo imaging, bulky nanocrystals may have limited
accessibility to target systems.
Sulphur
Cadmium
Tellurium
alkyl
C
O
O
Figure 26.8 A schematic model of a cubic CdS layer growing on the surface of a cubic CdTe lattice from the
hydrolysis of the thiol alkyl carboxilate capping molecules.
CH026-I046325.indd 784CH026-I046325.indd 784 6/25/2008 3:33:50 PM6/25/2008 3:33:50 PM
Semiconductor Quantum Dots for Biological Applications 785
Two different solubilization approaches based on the micellization of the hydrophobic
nanostructures were recently reported by Dubertret and collaborators and also by Korgel and
Monbouquete. By encapsulating individual CdSe/CdS QDs in phospholipid block–copolymer
micelles Dubertret et al. overcame the solubility problems and also demonstrated a longer sta-
bility of the particles in a living body [89] . Moreover, by adapting DNA to the nanocrystal
micelles, these systems acted as in vitro fl uorescent probes to hybridize to specifi c complementary
sequences. Korgel and Monbouquete successfully used phosphatidylcholine vesicles to prepare
mixed core and layered (Zn,Cd)S and (Hg,Cd)S nanocrystals [90] .
26.2.4 How to target the QDs to biological systems. The functionalization step
The synthesis, passivation and stabilization steps for the obtention of core–shell fl uorescent
semiconductor quantum dots prepared for biological labelling are generally succeeded by a func-
tionalization procedure. The term functionalization (also known as organic capping and ligand
conjugation) refers to the chemical modifi cations of the quantum dot’s surface to render biocom-
patible systems feasible for binding with specifi c biomolecules which will target the QDs to spe-
cifi c sites in the biological systems. This process is thermodynamically favoured due to the highly
Silica encapsulation
Ligand exchange
Hydrophobic interaction
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-OH
-S-alkyl-SI-O-
-S-alkyl-C
O
O
O
O
Figure 26.9 Representation of three different experimental modifi cations described in the literature to produce
water-soluble QDs.
CH026-I046325.indd 785CH026-I046325.indd 785 6/25/2008 3:33:51 PM6/25/2008 3:33:51 PM

786 Handbook of Self Assembled Semiconductor Nanostructures for Novel Devices in Photonics and Electronics
active chemical surfaces of the nanocrystals, either presenting dangling bonds at the passivation
shell or active binding groups in the stabilizing species.
There are several different experimental routes for this functionalization [91–94] . The func-
tionalizing agents may be small organic molecules (e.g. thiol molecules [38] , nucleotides [95, 96] ,
carbohydrates [97] , polyphosphates [98] ), or larger biomolecules (e.g. proteins [7, 99] , biotin
[100] , avidin [101] , polyethylene-glycol [102] , streptavidin, RNA [63] , DNA [103] , glucose
oxidase and IgG). Usually the combination of small and large molecules will promote adequate
modifi cations on the inorganic QDs ’ surfaces, providing functional groups that will intermediate
the QD/cell interaction [104] . For a schematic illustration of a functionalized core–shell semicon-
ductor quantum dot, see Fig. 26.10 . Figure 26.11 represents, as an example, the reaction steps
involved in the obtention of fl uorescent CdS/Cd(OH)
2
QDs, functionalized with polyethylene-
glycol (PEG), a biocompatible polymer.
H
2
S
Stirring
PEG
CdS/Cd(OH)
2
/PEG
CdS/Cd(OH)
2
CdS
CD
3
2
(NaPO
3
)n
OH
pH 10.5
Figure 26.11 Synthesis, surface passivation and functionalization: reaction steps involved in the obtention of
fl uorescent CdS/Cd(OH)
2
QDs, functionalized with polyethylene-glycol (PEG).
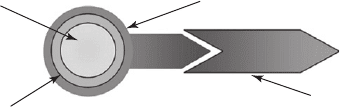
Functionalization shell
Passivation shell
QD (core)
Figure 26.10 A functionalized core–shell quantum dot is schematically represented here.
In order to promote an adequate functionalization procedure, every single modifi cation per-
formed in the colloidal suspension conditions (e.g. temperature, pH, composition) has to take into
account the fact that the colloidal suspension must remain stable. Another important feature
which cannot be neglected is the maintenance of the QDs characteristic luminescence. Moreover,
the choice of the adequate functionalizing agent is someway driven by the desired QD function/
application in the bio-system.
More complex functionalization procedures may employ conjugation protocols adapted from
those used in the binding of biomolecules and organic fl uorophores. The functionalization of
proteins (which represents the chemical nature of a great amount of biofunctional molecules,
such as antibodies, antigens, enzymes, growth factor molecules, etc.) to QDs may be performed
CH026-I046325.indd 786CH026-I046325.indd 786 6/25/2008 3:33:52 PM6/25/2008 3:33:52 PM
Semiconductor Quantum Dots for Biological Applications 787
in different forms, which will depend on the protein type and the nature of the nanocrystal’s cap-
ping surface. Some described methods are: (i) the condensation of amine groups of the protein
to carboxyl groups of the QDs ’ surface by using EDC, 1-ethyl-3-(3-dimethylaminopropyl) carbo-
diimide as a chemical activator; (ii) direct binding to the QD surface using thiolated peptides or
polyhistidine (HIS) residues; and (iii) adsorption or non-covalent self-assembly using engineered
proteins [91, 105] .
Optimal functionalization techniques should also be able to keep the quantum dots ’ funda-
mental properties for periods of time longer than several weeks. An example described in the lit-
erature refers to “ engineered-protein ” functionalized quantum dots which retain their quantum
effi ciency and offer longer shelf life [91] . These systems were also further functionalized with
multiple functional groups without decreasing their quantum effi ciency [93] .
It has been shown that the optical properties of semiconductor nanocrystals are very sensitive
to certain capping molecules attached to their surfaces. Some stabilizing agents or hydrophobic
surfactant molecules used in the solubilization procedure decrease the overall fl uorescence quan-
tum yield and the explanation for this effect remains in the “ electronic blinding capacity ” of the
passivation shell [61, 92, 106, 107] . This has been demonstrated in the case of mercaptoacetic
acid-treated QDs where the quantum effi ciency was drastically reduced [93, 94] . However, for
some probing applications this feature is taken as an advantage, especially if the change in fl uo-
rescence is generated by resonance energy transfer (FRET) to target analyte molecules [91] . This
opens up other applications in biosensing, where the extension of this FRET process can be con-
trolled by the thickness of the passivation shell and the distance to the binding species.
The choice of the functionalizers will provide low or high specifi city concerning the associa-
tion of the functionalized QDs with biomolecules. As already mentioned in the previous section,
thiol–alkyl–COOH ligands (such as mercapto-acetic acid) used as stabilizing agents in the syn-
thesis of CdTe QDs in aqueous medium also act as functionalizing agents and provide surface
carboxylate groups, making the QDs feasible to react with primary amines expressed by cell sur-
face proteins. This kind of reaction produces relatively stable amides [107] . Glutaraldehyde (at
very low concentrations), thiol-containing molecules and polyethylene-glycol can be cited as effi -
cient functionalizing agents, in an increasing order concerning specifi city in the bioconjugation
process [91] .
26.2.5 Bioconjugation of the QDs
The term bioconjugation is used to denominate the interaction between the functionalized quan-
tum dots and the molecules present in a biological system. A more general defi nition includes
also the conjugation of functionalized QDs with biomolecules produced by the bio-system
and dispersed in a liquid medium (e.g. the conjugation of circulating antibodies in the human
serum). As mentioned previously, QDs are prepared to bind chemically (covalent bonds) or physi-
cally (adsorption phenomena) to molecules present in the bio-system. The nature of the func-
tionalizing groups will, in theory, defi ne the nature of the interaction with the biological system.
For instance, if the target biomolecule is expressed in the cell membrane then most probably a
superfi cial labelling will be observed.
Depending on the kind of interaction observed one may identify different internalization
mechanisms related to the bioconjugation process. In specifi c biomolecules recognition the QDs
will bind specifi cally to the target molecule [94, 95] . Biotinylated antigens, for example, interact
strongly with QDs functionalized with avidin or streptavidin molecules [94, 99] rendering great
specifi city labelling.
The QDs may be functionalized with molecules that interact specifi cally to species presented
in distinct cellular organelles/structures , this is exemplifi ed by the cell cytoskeleton microtubules
labelled with CdSe/ZnS quantum dots functionalized with anti-tubulin [92] ; another example is
the CdS/Cd(OH)
2
polyphosphate stabilized QDs which were directed to high consuming energy
internal sites of living leishmania parasites [98] .
When the functionalizing groups are not specifi c, the interaction may occur indistinctively
with all the similar molecules and in this case the QDs are internalized by a natural process
known as endocytosis and end up bound to internal cell compartments [13, 109] . Endocytosis is
CH026-I046325.indd 787CH026-I046325.indd 787 6/25/2008 3:33:52 PM6/25/2008 3:33:52 PM
788 Handbook of Self Assembled Semiconductor Nanostructures for Novel Devices in Photonics and Electronics
the process whereby cells engulf vitamins and nutrients from their outside surroundings. The
nanocrystals can also be artifi cially internalized by inserting them using micromanipulators
[91] . In fi xed cells the membrane pores are increased and the QDs can pass through these chan-
nels easily without any other kind of internalization.
Various covalent and non-covalent strategies employing crosslinkers [7, 13, 91–94, 109–
115] have been developed for conjugating biomolecules to the QDs ’ surfaces. Biomolecules can
be linked to the QDs via functional groups such as –COOH, –NH
2
or –SH, which may be present
at their modifi ed surface. These groups may be provided by functionalizing compounds such as
protein A, avidin, streptavidin, glucose oxidase horseradish peroxidase, and IgG [13, 114–116] .
Figure 26.12 summarizes the steps discussed up to this section, concerning the preparation and
use of fl uorescent semiconductor quantum dots for biological labelling purposes.
Quantum dot core Functionalizing agent
Passivation shell
Conjugated biomolecule
Figure 26.12 A schematic representation of a passivated, functionalized and bioconjugated quantum dot.
For living cells labelled with quantum dots, the bioconjugation step may be processed into the
cell culture medium [7, 13] or even in another appropriate medium, such as saline solution. The
resulting systems to be used for bioconjugation must be compatible with cellular/tissular condi-
tions, i.e. they must be used in physiological pH, present low toxicity, be isotonic with the intra-
cellular (or intra-tissular) medium, and the resulting QDs must remain hydrophilic and retain
their characteristic fl uorescence.
In the next section some applications of QDs in biolabelling procedures are presented.
26.3 Applications
Since the fi rst reports published in 1998 [8, 94] concerning the application of quantum dots as
biolabels, much progress has appeared in this fi eld. There are several reports which indicate the
fast improvement on conjugating these nanostructures to biological molecules, cells, microor-
ganisms and tissues. It is worthwhile mentioning some representative examples:
● Multiplexed biological detection and imaging [24, 117, 118] .
● Sensing trace analytes in biological samples [119] .
● Tools for rapid and sensitive diagnosis of viruses [120] .
● Molecular characterization in combination with optical imaging to detect the progression
of precancerous lesions [121] .
● Nanoparticles for targeting in vivo [122] .
● Cervical, breast and brain cancer diagnostics [13, 109] , as well as many other applications
in life sciences.
Multiplexed biological detection and imaging, using the unique properties of photostabil-
ity and especially the narrower emission spectra of QDs, have been applied for fl uorescence
in situ hybridization (FISH) to study molecular biology. For instance, Chan et al. used FISH and QDs
for monitoring mRNA transcripts [107] . This work demonstrated an increased sensitivity of
FISH using QDs in comparison with organic fl uorophores which can facilitate the ultrasensitive
CH026-I046325.indd 788CH026-I046325.indd 788 6/25/2008 3:33:53 PM6/25/2008 3:33:53 PM
