Holik H. (ed.) Handbook of Paper and Board
Подождите немного. Документ загружается.


obvious appearance of treatment, most notably cockling. Liquid techniques can
function economically over a broad treatment range. The approach of Church in
the late 19th century was updated by Baynes-Cope in the mid-1960s [25]. This
followed his experimentation in 1961 with a magnesium methoxide solution
which, while quite effective, was impractical in terms of preparation and storage.
Independently, Richard Smith from the University of Chicago developed a process
that utilized a magnesium methoxide compound which was dissolved in a mixture
of methanol and chlorofluorocarbons (CFC) [26]. This overcame many of the prob-
lems associated with earlier approaches regarding stability and ease of use.
Ethanolate and propanolate of magnesium which are dissolved in the appro-
priate alcohol and a nonpolar halogenated hydrocarbon can also be used as deaci-
dification agents. By transformation of the magnesium alcoholate in the appro-
priate carbonates (alkoxy magnesium carbonate) using (dry) carbon dioxide, a fur-
ther improvement of the solubility and permanence could be reached.
13.3.2
History of Commercial Mass Deacidification
In the last 25 years some mass deacidification processes have been developed, by
national libraries and companies, which could not gain acceptance. A short de-
scription of the most important follows:
In contrast to procedures working with solvents, a vapor-phase approach offers
prospects for minimal preselection and maintenance of document integrity be-
cause no solvents are involved. At least one researcher likened this approach to a
“fountain of youth” among deacidification methods [27]. Gas-phase processes work
best on a substantial scale because the facilities characteristically have high capital
intensity.
Early attempts at gaseous deacidification included the work reported in 1962 by
Kathpalia at the Nehru Library in New Delhi, involving exposure of books to high
concentrations of ammonia [28]. The compound proved to be too weak a base to
neutralize completely strong acid and the ammonia buffer was highly imperma-
nent. At about the same time, Barrow working in the United States and Baynes-
Cope in England combined ammonia and short-chain amines, including butyla-
mine, diethylamine and piperidine, and observed similar results – a short term
alkalization that was reversed as the amine left the treated paper.
A more promising approach was advanced by Langwell who placed cyclohex-
ylamine carbamate impregnated sheets in books [29]. The active compound dif-
fuses into the book paper to neutralize acidity, but no alkaline reserve is estab-
lished and the neutralization is reversible. Finally, morpholine treatment was pro-
moted by Kusterer and others working at the Barrow Laboratory in 1977. Besides
the lack of permanence, significant discoloration was observed in the paper of
treated books. Kelly and Williams at the Library of Congress designed a mass
deacidification process in 1977 that utilizes diethyl zinc (DEZ) [30]. The compound
easily diffuses between the pages of closed books, neutralizing the acidity and
establishing a uniform, controllable reserve of zinc oxide.
13 Book and Paper Preservation482

13.3.2.1 BPA Process
Book Preservation Associates (BPA) was formed in 1988 as a wholly owned subsid-
iary of ETO Sterilization, a processor of spices, medical products and cosmetics. In
the BPA process books or other library materials are placed in closed boxes in a
vacuum chamber. The pressure is reduced and the books are exposed to ammonia
for 6 h. The chamber is then purged and evacuated. Ethylene oxide is introduced
and the materials are held for 12 h at nearly atmospheric pressure. The chamber is
alternately purged and evacuated up to 25 times before treated materials are re-
moved. The total cycle time is 24 h [31]. Books are placed in an aeration room for
two days to permit off-gasing of the ethylene oxide before being returned to li-
braries.
The process was designed to create high molecular mass, primary, secondary,
and tertiary amines within the cellulose matrix of the book paper. The in situ
generation of amines, together with free ethylene oxide, neutralizes acidity and
establishes an alkaline reserve of ca. 1%. Ethanolamine generation during treat-
ment is described as follows:
NH
3
+ CH
2
–CH
2
W NH
2
CH
2
CH
2
OH
\/
O
NH
2
CH
2
CH
2
OH + CH
2
–CH
2
W NH(CH
2
CH
2
OH)
2
\/
O
NH(CH
2
CH
2
OH)
2
+ CH
2
–CH
2
W N(CH
2
CH
2
OH)
3
\/
O
Developers of the process reported that the mono-, di-, and triethanolamines are
formed in approximately equal amounts with a modest dominance by the mono-
ethanolamine. The reaction depends on the ratio between reactants and is adjusted
intentionally. BPA personnel report that the pH drops approximately 1.5 points in
the two months following the treatment.
The process could not gain acceptance on the market because of the inadequate
alkaline reserve (no depot effect of the deacidification) and further side effects.
Nevertheless new attempts are being made in Japan to try to use this process again
after some alterations.
13.3.2.2 The DEZ Process
The DEZ process was developed by the Library of Congress in Washington over
more than 15 years, progressing from simple laboratory experiments to an operat-
ing small-scale commercial unit. Much of the latter work was in cooperation with
Texas Alkyls, one of three major producers of compounds such as diethyl zinc
(DEZ). Akzo secured an exclusive license for the DEZ Process and tried to com-
mercialize the technology.
13.3 Development of Mass Deacidification Processes 483

As strong alkylating agents, metal alkyls react easily with acids to form the corre-
sponding neutral salts. DEZ also reacts with residual water in the paper to form a
stable, permanent buffer of zinc oxide. Unlike most other processes, the neutral-
ization of existing acidity is independent of the formation of the alkaline reserve.
Thus highly acidic and neutral paper are both provided with an optimum alkaline
reserve. The key reactions are summarized below [32]:
H
2
SO
4
+ (C
2
H
5
)
2
Zn W ZnSO
4
+ 2 C
2
H
6
2 CH
3
CO
2
H + (C
2
H
5
)
2
Zn W Zn(CH
3
CO
2
)
2
+ 2 C
2
H
6
H
2
O + (C
2
H
5
)
2
Zn W ZnO + 2 C
2
H
6
The buffer established by the DEZ process is more neutral (saturated solution with
a pH of 7.3–7.5) than that of magnesium or calcium salts used in other deacidifica-
tion methods.
The process consists of three steps: pretreatment, permeation, and post-con-
ditioning; the total cycle time is 55–60 h. Akzo operated a treatment facility near
Houston, Texas, with a capacity of about 40000 books per year. Owing to its high
reactivity, liquid DEZ is dangerous if mishandled. Poor design and operating pro-
cedures on the part of an earlier contractor threatened to halt further development
of the process because of the inoperative first pilot plant. The issue of safe han-
dling was fully resolved in the second facility. Nevertheless the process did not
become accepted and was stopped.
13.3.2.3 The Wei T’o Process.
Wei T’o Associates, developers of a nonaqueous liquid deacidification process,
derives its name from an ancient Chinese god regarded as the protector of books.
The National Archives of Canada operated a treatment facility in Ottawa with a
capacity of about 40000 books per year until 2002. The process is based on the
work conducted by Richard D. Smith, a graduate student at the University of
Chicago in the mid-1960s. In 1989, Union Carbide acquired the exclusive rights of
the process and intend to market the technique as part of a paper preservation
service unit.
The deacidification agent is methoxymagnesium methyl carbonate carried in a
solvent system composed of methanol, trichlorotrifluoroethane (Freon 113) and
dichlorodifluoromethane (Freon 12). The CFCs have been replaced by perfluor-
ocarbons. Books are placed in wire baskets and dried to a moisture content of
0.5%. The pretreated materials are immersed in the treatment solution for 1 h
under modest pressure. Drying is initiated by evacuating the chamber and recover-
ing the solvents. Final drying is performed under vacuum. During this stage the
deposition of the magnesium compounds occurs. Careful control of pressure reg-
ulates the evenness of deposition [33].
Following deposition, the active agent reacts with water from the air to form a
mixture of intermediate magnesium compounds [8]:
13 Book and Paper Preservation484

H
3
COMgOCOOCH
3
+ 2 H
2
OW Mg(OH)
2
+ 2 CH
3
OH + CO
2
H
2
O + CO
2
W H
2
CO
3
Mg(OH)
2
+ H
2
CO
3
W MgCO
3
+ 2 H
2
O
MgCO
3
W MgO + CO
2
These magnesium compounds react with strong acids to form the corresponding
neutral salts. For example, magnesium hydroxide converts sulfuric acid to magne-
sium sulfate:
Mg(OH)
2
+ H
2
SO
4
W MgSO
4
+ 2 H
2
O
Magnesium sulfate is also generated by the reaction of magnesium carbonate with
acid:
MgCO
3
+ H
2
SO
4
W MgSO
4
+ H
2
O + CO
2
Residual magnesium hydroxide, carbonate and oxide form the alkaline buffer that
protects paper from subsequent acid attacks.
Similar chemistry is practiced by the Bibliotheque Nationale at its treatment
facility in Sable sur Sarthe/France. The unit has an annual capacity for 60 000
volumes. However, the French process uses ethoxymagnesium carbonate as the
deacidification agent.
13.3.2.4 The British Library Process
The British Library has achieved excellent results in its effort to deacidify and
strengthen book paper in a bulk process that has been under development since
1980. The method involves graft copolymerization techniques initiated by low-
intensity g-radiation. This work has been performed by the Industrial Chemistry
Group of the University of Surrey under British Library supervision and spon-
sorship.
Short-chain monomers, in particular ethyl acrylate and methyl methacrylate in a
volume ratio of 5:1, are introduced in liquid form without solvents in a closed
system. Low-intensity g-radiation polymerizes the monomers under ambient con-
ditions. Additionally, the liquid monomer mixture has been modified to include an
amine-substituted alkyl methacrylate to neutralize acidity and establish a buffer
against future acid catalyzed hydrolysis [34]. The presence of a small amount of a
diacrylate leads to cross-linking and hence strengthening of the paper.
Paper strength, as measured by fold endurance, is enhanced by a factor of 5 to
10 and a gain in weight of 15–20% occurs. No dimensional changes in treated
books have been observed. Researchers have concluded that the polymer is de-
posited within the cellulose network of the paper as well as on and between the
fiber surfaces. The treated books retain their original appearance and pages do not
stick together at the prescribed polymer deposition levels. This process also could
not be realized, residual monomer contents and further problems brought this
project to a halt.
13.3 Development of Mass Deacidification Processes 485

13.3.2.5 The Lithco Process
In 1988 Lithco started developing a nonaqueous deacidification technique. Magne-
sium butoxytriglycolate, the active ingredient, is solubilized in Freon 113 as a ca.
10% solution. Later heptane was used as solvent instead of Freon 113. The com-
pany marketed the process for its ability to both deacidify and strengthen book
paper.
The process consists of three steps: preconditioning, impregnation, and post-
conditioning. Books or other library materials are dried rapidly by dielectric heat-
ing to a moisture content of ca. 2%. The documents are immersed in the deacidifi-
cation solution for deposition of the neutralization agent. Finally, excess solution is
drained, excess chemicals are rinsed out, the solvent is removed under vacuum,
and the books are removed. Total cycle time is less than 5 h.
The butoxytriglycolate ligands are intended to interact with cellulose to
strengthen the paper through hydrogen bonding. Similar chemistry based on zinc
compounds has also been explored by Lithco on a more limited basis. Additionally,
the company utilizes dielectric heating to speed the drying during both the first
and last steps [35].
Early work was conducted in a demonstration unit capable of processing 20
standard bound volumes per batch. Lithco commissioned a larger pilot unit with a
capacity for 1000 books per day in May 1990. In practice the stabilizing effect
through the monobutyl-triglycolether could not be observed. The opposite could be
observed more often: due to the oleagenous substance a loss in permanence oc-
curred. This process also did not become accepted and therefore was stopped.
13.4
Current Commercial Processes
13.4.1
Papersave Process
®
The Papersave process
®
was developed by Battelle (Frankfurt) and is often called
the Battelle process in older literature. The inventor was Jürgen Wittekind. In this
mass deacidification process, titanium magnesium ethanolate (METE) dissolved
in hexamethyldisiloxane (HMDO) is used as deacidification agent. Titanium
ethoxyl has the task of dissolving the magnesium ethoxyl in the form of a titanium
magnesium double ethoxyl in HMDO. The magnesium alcoholate performs the
deacidification. The advantage of this process is the use of the nonpolar solvent
HMDO in which nearly no component of the documents is soluble. The addition
of alcohol as a solubilizing agent, as in other processes, is not necessary because of
the production of the double alkoxide. Only the alcohols that are set free in the
hydrolysis of the deacidification agent can become a problem in some cases.
The Papersave Process
®
comprises three steps: (Fig. 13.6 and 13.7): First, the
books, which remain in a closed treatment chamber during the entire process,
need pre-drying. The normal moisture content of 5–7% is temporarily decreased
13 Book and Paper Preservation486
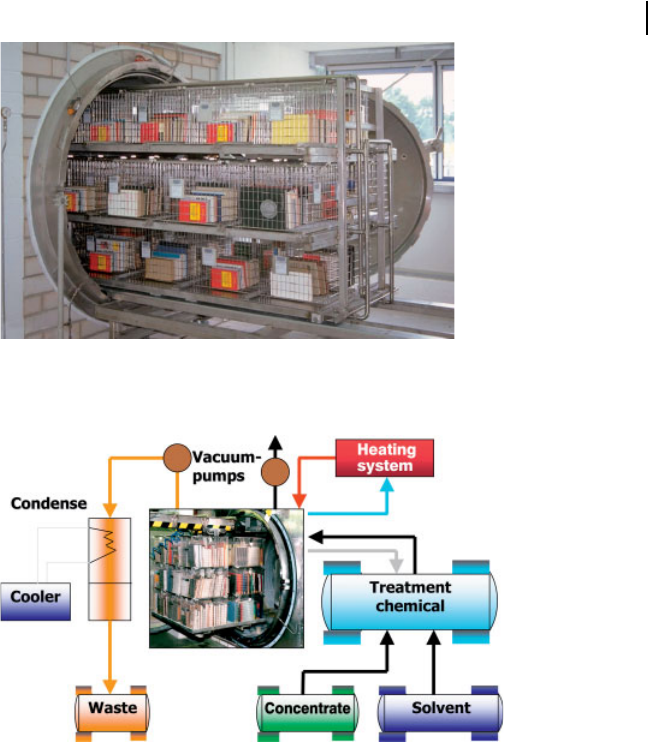
to less than 1%. The drying process takes place under vacuum conditions accom-
panied by a mild heating to not more than 120 °F (50 °C). Due to optimisation of
the drying technology, the previously used microwave technology has been re-
placed by a more favorable method.
Secondly, deacidification follows the pre-drying. The books are impregnated
with an alkali solution. The treatment chamber is completely flooded with the
treatment chemical. The deacidification chemicals are alcoholates of magnesium
and titanium dissolved in hexamethyldisiloxane. Due to the previous drying, the
closed books standing in the chamber absorb the solution like a sponge.
Thirdly, the books are again dried after the treatment solution has been drained.
The solvent absorbed during impregnation dries away while the deacidifying
chemicals (magnesium and titanium compounds) remain in the paper.
Fig. 13.6 Chamber of the Papersave Process
®
mass
deacidification plant with a capacity of 60 tons per year.
Fig. 13.7 Schematic of the Papersave Process
®
mass
deacidification plant.
13.4 Current Commercial Processes
487

The entire process takes three days. After the books have been removed from the
chamber, they regain lost humidity and return to their normal moisture content.
This process is finished after approximately three weeks. During this period the
books exude alcohol due to the reaction of the treatment chemicals with water.
Therefore, the treated books are stored in a well-ventilated room before being
returned to the customer.
By this treatment the following results are obtained: The acids contained in the
paper are completely neutralised by the magnesium compounds. The pH of the
paper after treatment is raised to between 7 and 9. The pH of some papers can
exceed 9 for a short period after treatment. In the following months, the pH of the
paper decreases to levels below 9 due to the transformation of magnesium alcoho-
lates and magnesium hydroxides in magnesium carbonate. After neutralisation
of the acids, 0.5–2 wt.% of superfluous magnesium carbonate remains in the
paper and serves as the alkaline buffer. Thus, acids subsequently formed or in-
troduced into the paper by environmental influences can be neutralised within the
paper.
This process has proven itself in practice during the last 10 years and is offered
by ZFB GmbH (Center for Book Conservation) in Leipzig/Germany which works
on the conservation and preservation of library and archive goods. The company
was founded in 1997 by hiving off the section Center for Book Conservation from
the German Library in Leipzig. Battelle (Frankfurt) was taken over by ZFB in 2001.
In Switzerland a Papersave deacidification plant with a capacity of 120 tons per
year was also opened in 2000, operated by Wimmis Nitrochemie under license
from ZFB.
13.4.2
The Bookkeeper Process
This is a liquid, nonaqueous process that employs a perfluorocarbon carrier to
deposit a submicron dispersion of magnesium oxide. No alcohol is involved and a
surfactant is employed to keep the neutralization agent evenly distributed in the
fluorocarbon solvent. The technique was developed by Koppers Chemical Com-
pany in 1981, who transferred the rights of the process to Richard Spatz in 1987.
Book Preservation Technologies, in which Spatz is a principal, continues the devel-
opment of the method.
Books are placed open on a supporting grate which is loaded into a treatment
chamber. No predrying is required. The chamber is flooded with the treatment
solution and the grate is mechanically agitated so that the pages are exposed and
completely wetted. Books are subsequently removed from the chamber and placed
in an air dryer, from which the solvent is condensed and recycled [36]. The total
process time is 5 h or less. The process can be operated in batch or continuous
mode. The Bookkeeper process relies on the migration of paper acid to the magne-
sium oxide for neutralization to occur.
The treated books show partially a dusty layer of magnesium oxide and damage
because of the intense mechanical strain during the deacidification process. Today
13 Book and Paper Preservation488

this process is used in the Library of Congress (Washington) in America and is also
offered commercially in the Netherlands.
13.4.3
Magnesium Oxide Dust
An automated process for the conservation of single sheets was evolved in the
Federal Archive of Lower Saxony in Bückeburg as a consequence of a development
of the PTS (Papiertechnische Stiftung) in Munich/Germany. The aqueous process
consists of three treatment stages:
1. Fixation of the writing and printing substance by means of a polyamine con-
densation product and a poly-condensed aromatic sulfonate (development of
this system by works in Stuttgart [37]).
2. Deacidification and generation of an alkaline reserve with an aqueous magne-
sium bicarbonate solution.
3. Sizing with cellulose ether (MC/CMC) for the increase of permanence.
All processes are carried out in dippings. The most progressive plant works with
only one dipping which combines all three chemical systems. The system is run
today by the company Neschen AG.
13.4.4
Aqueous Processes
13.4.4.1 Bückeburger Process
The companies Libertec and SOBU from the area of Nuremberg (Germany) offer a
paper deacidification by means of a nonaqueous process. In this process fine mag-
nesium oxide particles are blown into the opened books as dust which is then fixed
in the paper by humid air. The books are clamped individually into a gadget and
then treated. The treated books show an irregular deacidification and a dusty coat-
ing on the paper, especially in the book fold.
13.4.4.2 The Austrian National Library Process
Work on an effective deacidification process for bound newspapers began in the
Austrian Library in 1973. The technique, which has been in regular operation in
Vienna since 1987, involves immersion in an aqueous solution of calcium hydrox-
ide and methyl cellulose. Borax is then used as deacidification agent.
The process steps include: preparation, treatment, drying and rebinding. Covers
are removed from bound newspapers, and the materials are placed in a vacuum
chamber. Following immersion in the treatment solution, the paper is shock fro-
zen at –30 °C to prevent the formation of large crystals that would damage the
paper web. Final drying consists of freeze-drying to avoid the pages sticking to-
gether. Treated papers show no significant change in dimensions and permit the
use of the original covers [38]. Although testing and preselection is required, the
13.4 Current Commercial Processes 489

Library has successfully applied the technique to various other archival materials.
The current capacity is 3000 bound volumes per year.
13.5
Strengthening Old and Brittle Paper
13.5.1
Overview
There are some successful new possibilities in the deacidification of paper up to
mass deacidification processes by nonpolar solvents (for example the Papersave
Process
®
) whereas no new achievements have been reached in the strengthening
of paper, especially not in nonaqueous treatments. The experiences of paper mak-
ers are only of limited use in the strengthening of old papers because the papers
cannot be changed in structure and compound during restoration or preserva-
tion.
The chain deterioration of cellulose during paper aging is irreversible and can-
not be cancelled. A renewing of the fibers is not possible but there are possibilities
to stabilize brittle paper and the following measures are worthy of consideration:
1. Elastic “conglutination” (sizing) of the paper texture with macromolecular sub-
stances (cellulose ether, gelatin).
2. Formation of macromolecules in the paper texture by polymerization, for exam-
ple the Parylen Process. Here the most commonly discussed method to accom-
plish only strengthening involves the gas-phase deposition of poly-p-xylylene.
Paralyne is the generic name for the various members of the polymer series of
p-xylylenes developed by the Nova Tran Corporation. The technique is capable
of establishing a thin, durable coating of the polymer on the paper. No deacidifi-
cation is accomplished, however, and the high cost of treatment has discour-
aged widespread use.
3. Inserting or applying a stabilizing matrix, normally a thin stabilizing paper.
13.5.2
Preservation of Originals by (Mechanical) Paper Splitting (Leipzig Paper Splitting
Technique)
If a work has to be maintained as an original because of its value, even if the paper
is seriously damaged, then deacidification is not sufficient. In these cases a stabili-
zation of the paper is necessary. This can only be achieved be preservational tech-
niques like wet treatment, leaf casting (if parts are missing), application of thin
stabilizing material [39] or paper splitting.
Paper splitting [40] was developed at first as a manual technique for preservation
and new permanence of highly damaged paper for valuable works by Günter
Müller, Jena/Germany. The Center for Book Conservation GmbH in Leipzig devel-
oped a paper-splitting machine (Fig. 13.8) The machine includes a laminating
13 Book and Paper Preservation490

unit, a splitting unit and a removing unit. First the binding has to be removed
from the book i.e. the single sheets have to be separated. In the laminating ma-
chine the sheets are embedded in gelatin between two carrier sheets. The carrier
sheets in roll form are spread with gelatin by means of sizing machines. In the
splitting machine the composite of original sheet and carrier material produced in
the laminating machine is pulled apart, i.e. it is split. Then, a thin but sturdy core
paper (often with an alkaline buffer) is brought in between the two sides and glued
together with the halves of the original paper with cellulose ether (for example
methyl cellulose) mixed with calcium carbonate as deacidification agent and alka-
line reserve. After that the split sheets are supplied to the removing unit where the
carrier sheets are removed by means of an enzyme (protease). Finally the stabilized
sheets are bound again.
Even despite occasional ethical doubts the paper-splitting process is often the
only practicable method for highly damaged paper, for example paper that is an
advanced stage of damage by micro-organisms or ink corrosion.
13.6
Commercial Prospects
The need for paper deacidification is enormous. It is reckoned that between 70%
and 80% of the library and archive collections consist of acid paper and are there-
fore exposed to a dynamic deterioration process. The reaction is self-accelerating
because the acid deterioration is an autocatalytic process. This means that in the
course of the deterioration process more and more acids are produced. The acids
in the paper can be neutralized and an alkaline reserve can be deposited. With that
the acid conditioned deterioration reaction in paper can be stopped effectively but
cannot be cancelled. If the deterioration is too far advanced, more complex preser-
Fig. 13.8 Paper splitting machine in the Center for Book
Conservation in Leipzig.
13.6 Commercial Prospects
491
