Kasper C., van Griensven M., P?rtner R. (Eds.) Bioreactor Systems for Tissue Engineering II: Strategies for the Expansion and Directed Differentiation of Stem Cells
Подождите немного. Документ загружается.

dominant in this case, by the cryopreservation of the cell beads prior to the
cultivation. It could be shown that the vitality, and thus the quality of the beads,
is gradable during the cultivation process.
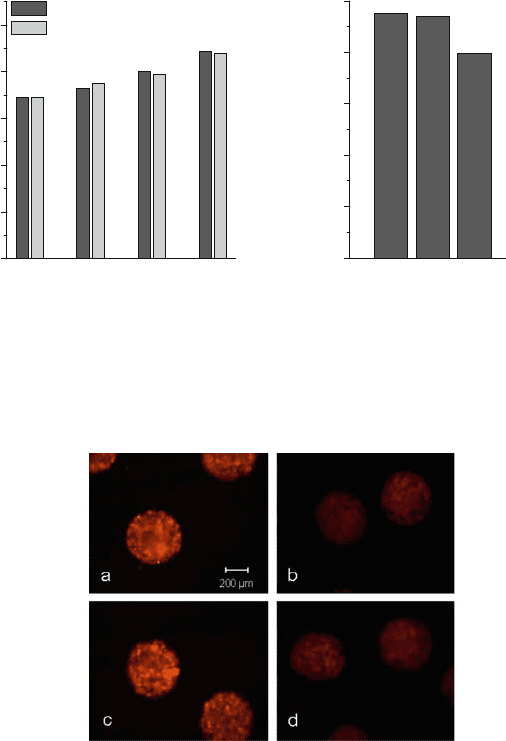
As an example, an adipogenic differentiation protocol was applied, whereby the
differentiation to adipocytes was verified by staining with the lipophilic fluorescence
dye Nile red (Fig. 15)[28]. Adipogenic cultured cell beads showed higher fluores-
cence intensity and thus are interpreted to be differentiated to adipocytes. No differ-
ences between the fixed-bed culture and the reference culture in T-flasks are detectable.
Table 6 shows the kinetics obtained by fitting the model parameter to the
experimental data which were used for a theoretical scale up of the system.
0
20
40
60
80
100
Fixed bed
Reference (T25-Flask)
Vitality [%]
Cell number N
x
[-]
0
2x10
6
4x10
6
6x10
6
8x10
6
1x10
7
Time [h]
500
200
1000
Time [h]
500
200
0
Fig. 14 Time dependent vitality and cell number of cell beads cultured in fixed-bed reactors and in
T-flasks (reference)
Adipogenic
cultivation
Non-adipogenic
cultivation
T25-Flask
(Reference)
Fixed bed
Fig. 15 Nile red staining of cell beads which were cultured under adipogenic and non-adipogenic
conditions in fixed-bed reactors or T25-flasks
Production Process for Stem Cell Based Therapeutic Implants 159
3.3 Theoretical Scale Up of the Cell Bead Cultivation Process
A calculational scale up was carried out for a cultivation of 200 single doses of cell
beads each of 5 mL (Fig. 16). The inlet oxygen concentration was assumed to be air
saturated. This can be realized, for example, by using membrane oxygenators.
For a calculation of the oxygen or glucose concentration profile in the cell bead,
the following diffusion and diffusion-reaction equations were used:
D
CC
d
2
c
CC
dr
2
þ
2
r
dc
CC
dr
¼ q X
CC
(11)
D
AC
d
2
c
AC
dr
2
þ
2
r
dc
AC
dr
¼ 0 (12)
Table 6 Consumption kinetics of encapsulated hMSC-TERT (cell beads) which were obtained
by fitting of model parameters to the experimental data of the adipogenic cultivation in a 1-cm
3
fixed-bed scale (Fig. 14)
Growth rate m
max
0 (1 day
1
)
Maximal glucose consumption rate q
Glc;max
(7.3 – 9.4) 10
8
(mg h
1
cell
1
)
Monod constant k
M;q
Glc
0.06 (mg mL
1
)
Oxygen consumption rate q
Ox
5.5 10
9
(mg h
1
cell
1
)
0
1
2
3
4
5
Glucose concentration
cell bead center [mg/ml]
Glucose concentration
in medium [mg/ml]
10 20 30 40 50 60
0
10
20
30
40
50
Oxygen saturation
cell bead center [%]
Oxygen saturation
in medium [%]
012345
0 100 200 300 400 500
0
1
2
3
4
5
inlet outlet
Glucose concentration
in medium [mg/ml]
Time [h]
20
40
60
80
100
outlet
inlet
Oxygen saturation
in medium [%]
Fig. 16 Simulated adipogenic cultivation of 200 single doses (5 cm
3
) of cell beads as well as the
glucose and oxygen profile at the cell bead center. Medium volume per cycle: 40 L, superficial
velocity: 2.5 10
4
ms
1
160 C. Weber et al.
with the concentration in the cell containing core bead c
CC
as well as in the cell free
alginate capsule c
AC
, the effective diffusion coefficient in the core bead D
CC
and
alginate capsule D
AC
; and the cell density of the core bead X
CC
.
It could be shown that an oxygen saturation in the medium of 40% leads to an
oxygen saturation at the center of approximately 22% (Fig. 16). The differences in
glucose between the medium and the cell bead center is negligible. Thus, it can be
assumed that no limitations of the cells at the center of a cell bead are expectable.
3.4 Conclusion and Outlook
Two fixed-bed reactor systems for the production of stem cell based therapeutic
implants were introduced. One system was developed for the expansion of the
production cell line (hMSC-TERT) and a second for the cultivation of encapsulated
cells in order to increase their vitality and thus the quality of the implants.
The fixed-bed system for the expansion of the production cell line is based on
non-porous BSGS. Cells can be cultured and harvested with high yield and vitality.
The separation of the cells from the carrier can easily be performed by flushing
them out with the medium flow. This saves additional process steps.
The fixed-bed system for the cultivation of encapsulated cells is based on commer-
cially available syringes in which the cell beads represent the bed. The advantage of
this system is that it can be used as an implantation tool after the cultivation procedure.
It could be shown that the vitality is gradable by the cultivation process. Furthermore,
the application of an adipogenic differentiation protocol could be demonstrated.
Both systems can be automated and produced as disposable items due to their
simple design.
The next steps will concern the development of a GMP-conform cryopreser-
vation procedure for the cell beads and the implementation of the cultivation
systems to the overall GMP-process of cell bead production.
Acknowledgements The authors would like to thank the Federal Ministry of Economics and
Technology for financial support as well as the CellMed AG for providing the production cell line
hMSC-TERT and the CellBeads1.
References
1. Lanza RP, Hayes JL, Chick WL (1996) Encapsulated cell technology. Nature Biotech
14:1107–1111
2. Freimark D, Czermak P (2009) Cell-based regeneration of intervertebral disc defects: review
and concepts. Int J Artif Organs 32:197–203
3. Baksh D, Song L, Tuan RS (2004) Adult mesenchymal stem cells: characterization, differen-
tiation, and application in cell and gene therapy. J Cell Mol Med 8:301–316
4. Chiu RCJ (2003) Bone-marrow stem cells as a source for cell therapy. Heart Failure Rev
8:247–251
Production Process for Stem Cell Based Therapeutic Implants 161
5. Fraser JK et al (2004) Adult stem cell therapy for the heart. Int J Biochem Cell Biol
36:658–666
6. Mimeault M, Hauke R, Batra SK (2007) Stem cells: a revolution in therapeutics–recent
advances in stem cell biology and their therapeutic applications in regenerative medicine
and cancer therapies. Clin Pharmacol Ther 82:252–264
7. Simonsen JL et al (2002) Telomerase expression extends the proliferative life-span and
maintains the osteogenic potential of human bone marrow stromal cells. Nat Biotechnol
20:592–596
8. Heile AMB et al (2009) Cerebral Q1 transplantation of encapsulated mesenchymal stem cells
improves cellular pathology after experimental traumatic brain injury. Neurosci Lett
9. Aris R (1975) The mathematical theory of diffusion and reaction impermeable catalysts.
Clarendon, Oxford
10. Bailey J, Ollis DF (1986) Biochemical engineering fundamentals. McGraw-Hill, New York
11. Froment GF, Bischoff KB (1979) Chemical reactor analysis and design. Wiley, New York
12. Fassnacht D (2001) Fixed-bed reactors for the cultivation of animal cells. Fortschritt-Berichte
VDI, vol. 17, VDI-Verlag, Du
¨
sseldorf
13. Willaert RG, Baron GV, de Backer L (1996) Modelling of immobilised bioprocesses. In:
Willaert RG, Baron GV, de Backer L (eds) Immobilised living cell systems. Wiley, New
York, pp 237–254
14. Perry RH, Green DW (2007) Perry’s chemical engineers’ handbook. McGraw-Hill
15. Weber C, Gokorsch S, Czermak P (2007) Expansion and chondrogenic differentiation of
human mesenchymal stem cells. Int J Artif Organs 30:611–618
16. Schop D et al (2009) Growth, metabolism, and growth inhibitors of mesenchymal stem cells.
Tissue Eng A 15:1877–1886
17. Higuera G et al (2009) Qvantifying in vitro growth and metabolism kinetics of human
mesenchymal stem cells using a mathematical model. Tissue Eng Part A 15:1–11
18. Schop D et al (2008) Expansion of mesenchymal stem cells using a microcarrier-based
cultivation system: growth and metabolism. J Tissue Eng Regen Med 2:126–135
19. Lonergan T, Brenner C, Bavister B (2006) Differentiation-related changes in mitochondrial
properties as indicators of stem cell competence. J Cell Phys 208:149–153
20. Conget P, Minguell JJ (1999) Phenotypical and functional properties of human bone marrow
mesenchymal progenitor cells. J Cell Phys 181:67–73
21. Guo Z et al (2001) Biological features of mesenchymal stem cells from human bone marrow.
Chin Med J 114:950–953
22. Soukup T et al (2006) Mesenchymal stem cells isolated from human bone marrow: cultiva-
tion, phenotypic analysis and changes in proliferation kinetics. Acta Med 49:27–33
23. Peng CA, Palson BA (1996) Determination of specific oxygen uptake rates in human
hematopoietic cultures and implications for bioreactor design. Ann Biomed Eng 24:373–381
24. Po
¨
rtner R et al (2005) Bioreactor design for tissue engineering. J Biosci Bioeng 100:
235–245
25. Acevedo CA et al (2008) A mathematical model for the design of fibrin microcapsules with
skin cells. Bioprocess Biosyst Eng 32(3):341–351
26. De Leon A, Mayani H, Ramırez OT (1998) Design, characterization and application of a
minibioreactor for the culture of human hematopoietic cells under controlled conditions.
Cytotechnol 28:127–138
27. Youn BS, Sen A, Behie LA (2006) Scale-up of breast cancer stem cell aggregate cultures to
suspension bioreactors. Biotechnol Prog 22:801–810
28. Weber C et al (2007) Cultivation and differentiation of encapsulated hMSC-TERT in a
disposable small-scale syringe-like fixed bed reactor. Open Biomed Eng J 1:64–70
162 C. Weber et al.

Adv Biochem Engin/Biotechnol (2010) 123: 163–200
DOI: 10.1007/10_2010_67
#
Springer-Verlag Berlin Heidelberg 2010
Published online: 4 May 2010
Cartilage Engineering from Mesenchymal
Stem Cells
C. Goepfert, A. Slobodianski, A.F. Schilling, P. Adamietz, and R. Po¨rtner
Abstract Mesenchymal progenitor cells known as multipotent mesenchymal stro-
mal cells or mesenchymal stem cells (MSC) have been isolated from various
tissues. Since they are able to differentiate along the mesenchymal lineages of
cartilage and bone, they are regarded as promising sources for the treatment of
skeletal defects. Tissue regeneration in the adult organism and in vitro engineering
of tissues is hypothesized to follow the principles of embryogenesis. The embryonic
development of the skeleton has been studied extensively with respect to the
regulatory mechanisms governing morphogenesis, differentiation, and tissue for-
mation. Various concepts have been designed for engineering tissues in vitro based
on these developmental principles, most of them involving regulatory molecules
such as growth factors or cytokines known to be the key regulators in developmen-
tal processes. Growth factors most commonly used for in vitro cultivation of
cartilage tissue belong to the fibroblast growth factor (FGF) family, the transform-
ing growth factor-beta (TGF-b) super-family, and the insulin-like growth factor
(IGF) family. In this chapter, in vivo actions of members of these growth factors
described in the literature are compared with in vitro concepts of cartilage
engineering making use of these growth factors.
C. Goepfert (*) and R. Po
¨
rtner
Institute of Bioprocess and Biosystems Engineering, Hamburg University of Technology,
Hamburg, Germany
e-mail: c.goepfert@tuhh.de
A. Slobodianski
Kompetenzzentrum Tissue Engineering, Universita
¨
tzuLu
¨
beck, Lu
¨
beck, Germany
P. Adamietz
Department of Biochemistry and Molecular Biology II: Molecular Cell Biology, University
Medical Center Hamburg-Eppendorf, Hamburg, Germany
A.F. Schilling
Biomechanics Section, Hamburg University of Technology, Hamburg, Germany
Keywords Bone morphogenetic protein (BMP), Cartilage, Chondrocytes, Differ-
entiation, Fibroblast growth factor (FGF), Growth factors, Indian hedgehog (Ihh),
Insulin like growth factor (IGF), Mesenchymal stem cells (MSC), Multipotent
mesenchymal stromal cells (MSC), PTH related peptide (PTHrP), Sonic hedgehog
(Shh), Transforming growth factor-beta (TGF)
Contents
1 Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 165
1.1 The Term “Mesenchymal Stem Cells” . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 165
1.2 Concepts for Cartilage Cultivation In Vitro . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 167
2 Development of Cartilage Tissue In Vivo . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 168
2.1 Migration and Proliferation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 178
2.2 Cell Condensation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 178
2.3 Chondrogenic Differentiation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 178
2.4 Endochondral Ossification . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 179
3 The Influence of Growth Factors on Cartilage
Development In Vivo . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 180
3.1 Fibroblast Growth Factors in Early Skeletal Development . . . . . . . . . . . . . . . . . . . . . . . . . 180
3.2 The TGF-b Superfamily of Growth and Differentiation Factors . . . . . . . . . . . . . . . . . . . . 181
3.3 The Role of IGFs in the Development of Cartilage Tissue . . . . . . . . . . . . . . . . . . . . . . . . . 182
3.4 Terminal Differentiation or Development of Articular Cartilage . . . . . . . . . . . . . . . . . . . 182
4 Growth Factors in Adult Cartilage Tissue . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 183
4.1 The Role of FGF in Adult Articular Cartilage . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 183
4.2 Members of the TGF-b Superfamily in Adult Articular Cartilage . . . . . . . . . . . . . . . . . . 184
4.3 The Role of IGF in Adult Articular Cartilage . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 184
5 Engineering of Cartilage Tissue In Vitro . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 185
5.1 Expansion of Progenitor Cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 185
5.2 Chondrogenic Differentiation In Vitro . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 186
5.3 Maintenance of the Hyaline Phenotype in Cultivated Cartilage Tissue . . . . . . . . . . . . . 188
6 Conclusion . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 188
References . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 189
Abbreviations
AER Apical ectodermal ridge
BMP Bone morphogenetic protein
ECM Extracellular matrix
FGF Fibroblast growth factor
FGFR FGF receptor
GF Growth factor
HA Hyaluronic acid
IGF Insulin-like growth factor
IGFBP IGF binding protein
IGFR IGF receptor
Ihh Indian hedgehog
164 C. Goepfert et al.
MSC Mesenchymal stem cells
OA Osteoarthritis
PTHrP PTH related peptide
RA Rheumatoid arthritis
Shh Sonic hedgehog
TGF-b Transforming growth factor-b
ZPA Zone of polarizing activity
1 Introduction
1.1 The Term “Mesenchymal Stem Cells”
For cartilage regeneration in vitro and in vivo, various strategies have been pursued
regarding the appropriate cell source, chemical and physical factors, and culture
conditions. It is widely agreed that tissue regeneration from autologous cells can be
achieved taking advantage of the natural course and progression of embryonic
development [1–4]. Therefore, autologous mesenchymal stem cells (MSCs) are
extensively investigated for their ability to regenerate articular cartilage tissue in
situ or in vitro.
Tissues forming the skeleton of the limbs originate from stem cells of the later al
plate and the somitic mesenchyme [2]. These embryonal MSCs undergo a series of
differentiation steps, finally producing differentiated skeletal tissues such as bone
and carti lage. In the adult organism, there are a limited number of mesenchymal
progenitor cells residing in the bone marrow which give rise to the repair of
damaged tissue, for instance bone, and which can easily be obtained by marrow
aspiration and selected by their ability to adhere to culture vessels. To date, it is not
clear whether they represent cells remaining from the embryonic mesenchyme or
whether they are a heterogenous population of mesenchymal precursor cells [4, 5],
possibly originating from the invading blood vessels populating the newly formed
bone marrow space during endochondral ossification [6].
First evidence for a precursor pool within the bone marrow was given by Frieden-
stein et al. [7], who described ectopic osteogenic differentiation originating from
whole bone marrow. Based on these results, they hypothesized a population of cells
occurring in the bone marrow which are able to differentiate along the osteogenic
lineage. These cells were characterized in vitro as colony forming fibroblastic cells
(CFU-f), isolated by their adherence to culture vessels [8]. MSC were hypothesized
as progenitor cells of mesenchymal tissues residing in the bone marrow and perios-
teum, persisting throughout lifetime as a pool for tissue regeneration, and which
might be isolated, expanded, and used for autologous treatment of damaged tissues
[9]. In 1999, Pittenger et al. [10] demonstrated the ability of clonally expanded human
bone marrow cells to differentiate towards the osteogenic, chondrogenic, and adipo-
genic lineages and thereby making it possible to develop treatments with human
Cartilage Engineering from Mesenchymal Stem Cells 165
autologous cells for the repair of mesenchymal tissues such as bone, cartilage and
adipose tissue. Since that time, these so-called MSCs have been isolated from various
tissues including adipose tissue [11], muscle and brain [12], bone [13], synovium [14,
15], umbilical cord [16], and blood [17]. Cells with even higher differentiation
potential have been isolated from cord blood [18, 19] and from bone marrow [20].
In order to characterize the MSCs, several markers have been described, but none
of them proved to be unique and exclusively present on MSCs. Enrichment of MSCs
has been carried out using the Stro-1 monoclonal antibody [21, 22]. The resulting cell
population was shown to be able to differentiate into the mesenchymal lineages of
osteoblasts, chondrocytes, adipocytes, and stromal cells supporting hematopoiesis
[23]. Other antigens specific for undifferentiated precursor cells and absent on
differentiated cells have been identified using the monoclonal antibodies SH-2 (endo-
glin, co-receptor for TGF-b3) [24], SH-3, and SH-4, respectively [25]. But there are
no unique surface antigens so far defining “the” MSC.
Clonally derived MSCs have been extensively analyzed for their ability to
proliferate in vitro, retaining their multi-lineage differentiation potential upon
prolonged cultivation. It was shown that these clonally derived cells lost their
multi-lineage potential upon extended cultivation in vitro and thus behave like
plastic progenitor cells rather than stem cells [5, 26–28]. On the other hand, Jiang
et al. [20] were able to describe a subset of pluripotent cells in the mouse and the rat
bone marrow, virtually proliferating for up to 60 passages without losing their
characteristic growth rates and their potential even to transdifferentiate into the
endodermal and ectoderma l lineages displaying some features of hepatocytes or
neuronal cell types [20, 29]. Thus it is likely that MSCs are a heterogenous
population of rare cells in the bone marrow compartment and other tissues, dis-
playing various stages of predifferentiation. Due to the heterogeneity of the cell
preparations commonly referred to as MSCs, Dominici et al. defined mesenchymal
stem cells as plastic adherent cells positive for CD105, CD73, CD90 lacking the
expression of hematopoetic markers CD45, CD34, CD14 or CD11, CD79 alpha or
CD19 and HLA-DR surface markers [30]. Finally, according to this definition,
MSCs must be shown to differentiate into osteoblasts, adipocytes and chondrocytes
in vitro. According to Horwitz et al. [31], fibroblast-like plastic adherent mesen-
chymal proge nitor cells isolated from various tissues are termed multipotent mes-
enchymal stromal cells (also referred to as MSCs).
Recently, it has been proposed to define “stemness” as a state of cells which are
able to differentiate into various cell types rather than the cells themselves [32].
According to this view, plasticity would be the most prominent characteristic of
stem cells whereas self-rene wal and hierarchical different iation are regarded as
subordinate features of these cell populations. Therefore, dedifferentiated cells
capable of differentiating into more than their original cell type could also be
regarded as stem cells. Since in vitro-expanded and thus dedifferentiated articular
chondrocytes were shown to differentiate similarly to MSCs along the mesenchy-
mal lineages into osteoblasts, chondrocytes, includin g hypertrophic chondrocytes,
and adipocytes [33, 34], the term “secondary progenitor cells” could be appropriate
for dedifferentiated chondrocytes [35].
166 C. Goepfert et al.

1.2 Concepts for Cartilage Cultivation In Vitro
The regeneration of damaged tissues in the adult organism as well as in vitro
engineering of tissues is hypothesized to follow the principles of embryogenesis
[1–4]. Therefore it is assumed that cartilage formation by means of cell therapy or
tissue engineering is bound to recapitulate, at least in several aspects, the stages of
in vivo development. The development of the appendicular skeleton is initiated by
migration of the early mesenchymal progenitors of the skeletal tissue towards the
prospective limb regions and by proliferation of these undifferentiated progenitors.
The accumulation of high cell densities is the prerequisite of cell condensat ion as
the key event of cartilage tissue formation. Thus, the factors governing the migra-
tion and proliferation of the undifferentiated mesenchymal cells hold potential for
the expansion of undifferentiated precursor cells in vitro.
The morphogenesis of skeletal elements requires precartilage condensation
leading to chondrogenic differentiation and thus to the formation of cartilaginous
models of the skeletal elements. During chondrogenic differentiation, the cells
adopt a rounded morphology and start to synthesize cartilage specific matrix
molecules. Further shaping of the skeletal primordia involves the separation of
the digital rows by apoptosis of the cells within the interdigital mesenchyme. The
concomitant formation of the joints requires dedifferentiation of the cells in the
prospective joint regions transiently leading to high cell densities and finally to
apoptotic cell death and the development of the joint cavity.
Since the precursor cells are usually expanded in 2D systems in vitro where they
display a flattened spindle-shaped morphology, 3D culture systems are supposed to
be appropriate for chondrogenic differentiation. The naturally occurring events are
mimicked by cell–cell-contacts established in high density pellet culture [36]by
cultivation in hydrogels [37–41] or 3D matrices of biocompatible materials [42, 43].
A comparison between the in vivo stages of cartilage development and the strate-
gies used for transferring these stages into in vitro concepts and the inherent
challenges is given in Table 1.
Usually these culture systems taken alone are not sufficient to induce chondro-
genesis of precursor cells. Therefore, growth factors are included into the culture
medium and combined with physical factors such as reduction of oxygen supply
Table 1 Strategies for transferring in vivo development into in vitro concepts
In vivo development of cartilage tissue In vitro cultivation of cartilage tissue
Migration and proliferation Expansion of MSC in vitro (2D cultivation)
Prechondrogenic condensation 3D aggregate culture imitating cellular condensation
Chondrogenic differentiation Cultivation in 3D biomaterials mimicking the 3D
structure of the ECM
Transient growth plate cartilage:
hypertrophy and apoptosis
Permanent articular cartilage: no
hypertrophy, no apoptosis
Prevention of hypertrophy and apoptosis in articular
cartilage regeneration
Cartilage Engineering from Mesenchymal Stem Cells 167
which occurs naturally by exclusion of the blood vessels from forming limb buds.
Chondrogenic differentiation in vitro is achieved after cultivation for several weeks
resulting in the formation of collagen type II and proteoglycans along with other
cartilage specific tissue components.
In vivo, the articular cartilage is maintained in a stage in which progression
towards hypertrophy naturally occurring in the growth plate is prevented by defined
mechanisms. In the growth plate, hypertrophy and calcification of the cartilage
tissue precede vascular invasion, finally leading to tissue replacement by bone. The
inactivation of the differentiation program towards hypertrophy is particularly
important for in vitro cultivation of cartilage tissue when MSCs are used as the
cell source since terminally differentiated cells will undergo apoptosis. Unlike
MSCs, articular chondrocytes usually do not undergo lineage progression upon
extended cultivation [44] or when implanted into ectopic sites [ 45]. Maintenance of
the phenotype of articular carti lage therefore comprises the inactivation of the
developmental pathway leading to terminal differentiation, and a switch to sus-
tained maintenance of funct ional extracellular matrix. For this reason, the appro-
priate stage of in vitro cultivated cartilage tissue needs to be carefully evaluated
regarding the state of cellular phenotype of chondrocytes and the characteristics of
the extracellular matrix formed.
In this overview, regimes of growth factor treatments currently applied in
cartilage tissue engineering in vitro are compared to stage specific actions of growth
factors in vivo during cartilage differentiation. Since growth factors of the fibroblast
growth factor (FGF)-family, transforming growth factor-b (TGF-b) superfamily
and the insulin-like growth factor (IGF) family are widely used in cartilage engi-
neering in vitro, the overview will be narrowed to these major groups of growth
factors. An overview of in vivo actions with in vitro applications of growth factors
is given in Table 2 .
2 Development of Cartilage Tissue In Vivo
The development of the skeleton in vivo is a multistep process tightly regulated
regarding the temporal and spatial distribution of the appropriate signals. Morpho-
genesis is evoked by gradients of specific signaling molecules along the axes of the
developing limbs. Following steps are triggered by the achievement of distinct
differentiation stages of the cells and by stage specific extracellular matrix synthe-
sis. Regulatory mechanisms such as feedback terminate a distinct developmental
stage and allow for further differentiation. Thus, tissue development depends on the
differentiation stage of the cells as well as on signals provided by paracrine,
autocrine or systemic factors, and the extracellular matrix (Fig. 1).
Taking into account the multi-sta ge process leading to functional tissues in vivo,
histogenesis in vitro also needs to be tightly regulated regarding the time sequence
of induction and the resulting consecutive sta ges of extracellular matrix synthesis
(Fig. 2).
168 C. Goepfert et al.
