Kenneth E. Gonsalves, Craig R. Halberstadt, Cato T. Laurencin, Lakshmi S. Nair. Biomedical Nanostructures
Подождите немного. Документ загружается.

87. Mo XM, Xu CY, Kotaki M, Ramakrishna S. Electrospun P(LLA-CL) nanofiber:
a biomimetic extracellular matrix for smooth muscle cell and endothelial cell
proliferation. Biomaterials 2004;25:18831890.
88. Xu CY, Inai R, Kotaki M, Ramakrishna S. Aligned biodegradable nanofibrous
structure: a potential scaffold for blood vessel engineering. Biomaterials 2004;25:
877886.
89. Ma Z, He W, Yong T, Ramakrishna S. Grafting of gelatin on electrospun
poly(caprolactone) nanofibers to improve endothelial cell spreading and prolifera-
tion and to control cell orientation. Tissue Eng 2005;11:11491158.
90. He W, Yong T, Teo WE, Ma Z, Ramakrishna S. Fabrication and endothelializa-
tion of collagen-blended biodegradable polymer nanofibers: potential vascular
graft for blood vessel tissue engineering. Tissue Eng 2005;11:15741588.
91. Lee KW. Sustained release of vascular endothelial growth factor from calcium-
induced alginate hydrogels reinforced by heparin and chitosan. Transplant Proc
2004;36:24642465.
92. Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat
Med 2003;9:669676.
93. Schlessinger J, et al. Crystal structure of a ternary FGF FGFRheparin complex
reveals a dual role for heparin in FGFR binding and dimerization. Mol Cell
2000;6:743750.
94. Rajangam K, et al. Heparin binding nanostructures to promote growth of blood
vessels. Nano Lett 2006;6:20862090.
95. Fawcett JW and Asher RA. The glial scar and central nervous system repair. Brain
Res Bull 1999;49:377 391.
96. Holmes TC, et al. Extensive neurite outgrowth and active synapse formation on
self-assembling peptide scaffolds. Proc Natl Acad Sci USA 2000;97:67286733.
97. Ellis-Behnke RG, et al. Nano neuro knitting: peptide nanofiber scaffold for brain
repair and axon regeneration with functional return of vision. Proc Natl Acad Sci
USA 2006;103:50545059.
98. Chen ZJ, Ughrin Y, Levine JM. Inhibition of axon growth by oligodendrocyte
precursor cells. Mol Cell Neurosci 2002;20:125139.
99. Silva GA, et al. Selective differentiation of neural progenitor cells by high-epitope
density nanofibers. Science 2004;303:13521355.
100. Koh GY, Soonpaa MH, Klug MG, Field LJ. Strategies for myocardial repair. J
Interv Cardiol 1995;8:387393.
101. Malosky S and Kolansky DM. Gene therapy for ischemic heart disease. Curr Opin
Cardiol 1996;11:361368.
102. Hassink RJ, Dowell JD, Riviere BA, Doevendans PA, Field LJ. Stem cell therapy
for ischemic heart disease. Trends Mol Med 2003;9:436441.
103. Leor J and Cohen S. Myocardial tissue engineering: creating a muscle patch for a
wounded heart. Ann N Y Acad Sci 2004;1015:312319.
104. Akhyari P, et al. Mechanical stretch regimen enhances the formation of
bioengineered autologous cardiac muscle grafts. Circulation 2002;106:I137I142.
105. Kofidis T, et al. In vitro engineering of heart muscle: artificial myocardial tissue. J
Thorac Cardiovasc Surg 2002;124:6369.
406
BIOMEDICAL NANOSTRUCTURES
106. Zong X, et al. Electrospun fine-textured scaffolds for heart tissue constructs.
Biomaterials 2005;26:53305338.
107. Shin M, Ishii O, Sueda T, Vacanti JP. Contractile cardiac grafts using a novel
nanofibrous mesh. Biomaterials 2004;25:37173723.
108. Venugopal J and Ramakrishna S. Biocompatible nanofiber matrices for the
engineering of a dermal substitute for skin regeneration. Tissue Eng 2005;11:
847854.
109. Pattison MA, Wurster S, Webster TJ, Haberstroh KM. Three-dimensional, nano-
structured PLGA scaffolds for bladder tissue replacement applications.
Biomaterials 2005;26:24912500.
110. Berthiaume F, Moghe PV, Toner M, Yarmush ML. Effect of extracellular matrix
topology on cell structure, function, and physiological responsiveness: hepatocytes
cultured in a sandwich configuration. FASEB J 1996;10:14711484.
111. Langer R. Tissue engineering: status and challenges. E-biomed: J Reg Med 2000;1:
56.
112. Langer R, et al. Tissue Engineering: biomedical applications. Tissue Eng 1995;1:
151162.
NANOSTRUCTURES FOR TISSUE ENGINEERING/REGENERATIVE MEDICINE 407

CHAPTER 16
Nanostructures for Cancer
Diagnostics and Therapy
KUMERASH S. SOPPIMATH and GURU V. BETAGERI
16.1 INTRODUCTION
In 2006, about 564,830 Americans were expected to die of cancer that is more
than 1500 people a day. Cancer is the second most common cause of death in
the United States, exceeded only by heart disease and accounts for one of every
four deaths [1]. According to a worldwide survey published for the year 2001,
there were 10.9 million new cases, 6.7 million deaths, and 24.6 million persons
alive with cancer (within 3 years of diagnosis). The most commonly diagnosed
cancers are lung (1.35 million), breast (1.15 million), and colorectal (1 million);
the most common causes of cancer death are lung cancer (1.18 million deaths),
stomach cancer (700,000 deaths), and liver cancer (598,000 deaths) [2]. The
5-year relative survival rate for all cancers diagnosed in the United States
between year 1995 and 2001 is 65%, up from 50% in 19741976. The
improvement in survival reflects progress in diagnosing certain kind of cancers
at the early stage and the use of new and or improved treatments [1].
16.1.1 Cancer and Early Diagnosis
Despite recent progress in the treatment of cancer, the majority of cases are still
diagnosed only after tumors have metastasized, leaving the patient with a grim
prognosis. However, there may be an opportunity to drastically reduce the
burden of cancer, if the disease can be detected early enough. Nanotechnology
is in a unique position to transform cancer diagnostics and to produce a new
generation of biosensors and medical imaging techniques with higher
sensitivity and precision of recognition [3].
BiomedicalNanostructures, EditedbyKennethE.Gonsalves,CraigR.Halberstadt,CatoT.Laurencin,
and Lakshmi S. Nair
Copyright # 2008 John Wiley & Sons, Inc.
409
Cancer patients undergo many different types of tests in order to accurate ly
diagnose their disease, determine their prognosis, and monitor their cancer for
progression or recurrence. The term ‘‘diagnostic test’’ can be misleading, as
these tests are used not only for diagnosing cancer, but also for monitoring
cancer progression. There are many reasons for employing diagnostic tests,
depending on whether the disease is active or progressing, being treat ed, or in
remission. Diagnostic tests may be used to
. Diagnose primary disease identify the disease the first time it occurs.
. Identify cancer subtypesome cancers are divided into subtypes that are
more or less aggressive; identification of a more aggressive subtype may
influence the type of treatment proposed.
. Predict prognosistest results may indicate chance of cure, based on
outcomes of other patients with similar results.
. Direct treatmentcancer is like many different diseases, all of which
respond differently to various treatments. A diagnosis that accurately
identifies the type of cancer and predicts prognosis will also help to
identify the type of treatment that maximizes chance of cure.
. Evaluate response to treatmentsome tests show whether the cancer is
responding to treatment.
. Detect minimal residual diseasecancer cells that remain after treatment
is completed are called MRD. Detection of MRD may indicate a higher
likelihood of recurrence.
. Monitor remission or progressionif a cancer is in remission, frequent
tests may help to detect the cancer if it returns and/or determine whether
it is progres sing.
. Screen at-risk individualsidentifying abn ormalities in cells or the DNA
of cells of asymptomatic (healthy) individuals may indicate an increased
risk (although not a certainty) of developing disease.
Pathology tests: Pathology tests involve microscopic evaluation of abnormal
cells.
Diagnostic imaging : Diagnostic imaging involves visualization of abnormal
masses using high tech machines that create images. Examples of diagnostic
imaging include X-rays, computed tomography (CT) scans, positron
emission tomography (PET) scans, magnetic resonance imaging (MRI), and
combined PET/CT scans.
Blood tests: Blood tests measure substances in the blood that may indicate how
advanced the cancer is or other problems related to the cancer.
Tumor markers: Tumor marker tests detect substances in blood, urine, or other
tissues that occur in higher than normal levels with certain cancers.
Genomics: Special laboratory evaluat ion of DNA involves the identification of
the genetic makeup, the DNA of the abnormal cells [4].
410 BIOMEDICAL NANOSTRUCTURES
Detection of cancer at early stages is a critical step in improving cancer
treatment. Currently, detection and diagnosis of cancer usually depend on
changes in cells and tissues that are detected by a doctor’s physical touch or
imaging expertise. Instead, scientists would like to make it possible to detect the
earliest molecular changes, long before a physical exam or imaging technology is
effective. To do this, they need a new set of tools. In order to successfully detect
cancer at its earliest stages, scientists must be able to detect molecular changes,
even when they occur only in a small percentage of cells. This means the
necessary tools must be extremely sensitive. The potential for nanostructures to
enter and analyze single cell suggests they could meet this need.
Nanomolecular diagnostics is the use of nanobiotechnology in molecular
diagnostics and can be termed nanodiagnostics [3]. Numerous nanodevices and
nanosystems for sequencing single molecules of DNA are feasible. Given the
inherent nanoscale of receptors, pores, and other functional components of
living cells, the detailed monitoring and analysis of these components will be
made possibl e by the development of a new class of nanoscale probes. Because
of the small dimension, most of the applications of nanobiotechnology in
molecular diagnostics fall under the broad category of biochips/microarrays
but are more correctly termed nanochips and nanoarrays. Nanotechnology-on-
a-chip is a general description that can be applied to severa l methods. Some of
these do not use nanotechnologies but merely have the capability to analyze
nanoliter amounts of fluids. Potential applications in molecular diagnostics are
listed in Table 16.1 [5]. In this chapter, we are discussing about some of the
successful nanomolecular diagnostics.
16.1.2 Cancer and Chemotherapy
The clinical arsenal in treating cancer has been greatly extended in recent years
with application of novel chemotherapy; however, the basic approaches
continue to be (in order of success) surgical resection, radiation, and
chemotherapy. Chemotherapy is the term broadly employed for the use of
pharmaceutical agents that possess cytotoxic or cytocidal activity. This
systemic form of cancer treatment has evolved over more than half a century
[6] and is undoubtedly the major weapon against most forms of cancer. It was
indeed the accidental observation with the chemical weapon mustard gas that
generated the idea that the systemic use of similar alkylating agents could be a
potential way to poison cancerous cells [7]. Since the first clinical trials on
nitrogen mustard by Gillman, there has been a continuous flow of novel
compounds belonging to a variety of subclasses with the sole intent of finding a
molecule or combination of molecules that could selectively knock out cancer
cells with minimal damage to their normal counterparts.
Although, these agents have been developed keeping in mind the subtle
differences between the biology of cancer cells and normal cells, a majority of the
existing compounds do not exhibit the level of desirable selectivity. In addition,
the probability of developing drug resistance is significantly higher if a single
NANOSTRUCTURES FOR CANCER THERAPY 411

agent is used over a period of time [8]. The latter is somewhat circumvented by the
routinely employed combination chemotherapy, the judicious cocktail of multiple
anticancer agents administered simultaneously or sequentially [9].
Anticancer drug therapy is fraught with systemic toxicities, resulting from
cytotoxicity to normal cells. Cancer cell s share many common features with the
TABLE 16.1 Nanotechnologies with Potential Applications in Molecular Diagnostics
Nanotechnology on a chip
Microfluidic chips for nanoliter volumes: NanoChip
Optical readout of nanoparticle labels
Nanoarrays
Protein nanoarrays
Nanoparticle technologies
Gold particles
Nanobarcodes
Magnetic nanoparticles: ferrofluids, supramagnetic particles combined with MRI
Quantum dot technology
Nanoparticle probes
Nanopore technology
Measuring length of DNA fragments in a high-throughput manner
DNA fingerprinting
Haplotyping
Cantilever arrays
Multiple combined tests (such as protein and DNA) to be performed on the same
disposable chip
Prostate specific antigen binding to antibody
DNA nanomachines for molecular diagnostics
Nanoparticle-based immunoassays
DNAprotein and nanoparticle conjugates
Nanochip-based single-molecular interaction force assays
Resonance light scattering technology
Nanosensors
Living spores as nanodetectors
Nanopore nanosensors
Nanosensor glucose monitor
Optical biosensors: surface plasmon resonance technology
Probes Encapsulated by Biologically Localized Embedding (PEBBLE) nanosensors
Photostimulated luminescence in nanoparticles
Quartz nanobalance DNA sensor
SEnsing of Phage-Triggered Ion Cascade
Reprinted from Jain KK. Nanobiotechnology. Switzerland: Jain PharmaBiotech Publications;
2005, reproduced with permission of the publisher.
412 BIOMEDICAL NANOSTRUCTURES
normal host cells from which they originate, so finding unique targets against
which anticancer dru gs can be selectively directed is difficult. Many anticancer
drugs have a marginal selectivity for malignant cells because they target the
reproductive apparatus in cells having high proliferation rates. However,
anticancer drugs having this mechanism of action result in high toxicities
against rapidly dividing normal cells, for example, hair follicles, germ cells, and
hematopoeitic cells, leading to dose-limiting side effects like mucositis,
stomatitis, alopecia, and reproductive effects. The side effects associated with
chemotherapy limit the dose or cumulative doses that can be administered to
patients, which can lead to relapse of the tumor and often the development of
drug resistance. [10].
Pharmacokinetics of anticancer drugs play an important role in determining
the quantitative relationship between drug dose, exposure, and drug activity,
thereby allowing adjustment of the dose to achieve maximum benefit [11, 12].
Improving the pharmacokinetics may result in enhanced antitumor activity and
reduce toxicity (pharmacodynamics). Two important factors influencing the
pharmacokinetics of antitumor drugs are route of administration and type of
formulation [12]. Most anticancer drugs are administered intravenously (IV) due
to its advantages of complete and immediate bioavailability and accurate dosing.
However, due to large volumes of distribution and lower specificity, IV route
exposes whole body with anticancer drugs to obtain high concentration of these
drugs specialized parenteral routes such as intraarterial or intrathecal injection
and direct injection to tumor are practiced [12, 13]. However, these routes of
administrations have a limitation in accessing remote and deep cancer tissues.
The medical community has sought alternative therapies that improve
selective toxicities against cancer cells, that is, therapies that increase efficacy
and/or decrease toxicity, resulting in an increase in the therapeutic indices of
the anticancer drugs. Hence, various drug carriers are developed for delivery of
anticancer drugs and most of these carriers are in nanosize range.
16.1.3 Why Nanotechnology for Treating Cancer?
There are several reasons that nanotechno logy could help transform cancer
research and clinical approaches to cancer care [14]. Most biological processes,
including those processes leading to cancer, occur at the nanoscale. For cancer
researchers, the ability of nanoscale devices to easily access the interior of a
living cell affords the opportunity for unprecedented gains on both clinical and
basic research frontiers.
1. The ability to simultaneously interact with multiple critical proteins and
nucleic acids at the molecular level will provide a better understanding of
the complex regulatory and signaling patterns that govern the behavior of
cells in their normal state as well as the transformation into malignant cells.
2. Nanotechnology provides a platform for integrating research in proteo-
micsthe study of the structure and function of proteins, including the
NANOSTRUCTURES FOR CANCER THERAPY 413
way they work and interact with each other inside cellswith other
scientific investigations into the molecular nature of cancer.
There are many nanotechnologies that are explore d for delivery of
anticancer drugs and some of the successful tools are explained in this chapter.
16.2 NANOTOOLS FOR EARLY CANCER DETECTION
16.2.1 Quantum Dots
Quantum dots (QDs) are semicondu ctor nanocrystals or particles, by adjusting
their size and composition, QDs can be prepared to emit fluorescent light from
the ultra violet (UV) light, through the visible and into the IR spectrum
(4004000 nm). They are composed of an inorganic elemental core (e.g.,
cadmium, mercury) with a surrounding metal shell [15–18]. QDs pos sess
unique optical properties that not only allow them to be tunable to discreet
narrow frequencies but also are an order of magnitude more resistant to
photobleaching than their organic fluorophore counterparts [18, 19]. QDs find
application in biological science as probes; QDs can absorb and emit light very
efficiently allowing sensitive detection relative to conventionally used dyes.
QDs have been found to be 1020 times brighter than organic dyes [18–20].
QDs surface can be coated with hydrophilic polymers and linked with
bioaffinity ligands such as monoclonal antibodies, peptides, oligonulceotides,
or small molecular inhibitors. Due to higher surfa ce area to volume ratio, QDs
can be conjugated to obtain multifunctionality [20].
QDs find application in both in vitro and in vivo cancer diagnostics. In vitro
screening of biological fluids for the presence of cancer markers is a commonly
used diagnostic test for cancer, as sensitive and a specific test for cancer
markers in biological fluids is laborious and time consuming by using
conventional methods. Conjugated QDs are not only sensitive but also can
detect multiple biomarkers [21]. Gerion et al. reported detection of specific
single nucleotide polymorphisms of the human p53 tumor suppression gene
using QDs in a microarray assay format and also demonstrated detection of
two DNA sequences using two different QDs [22]. Another potential in vitro
application of QDs is cellular imaging; recently water-soluble QDs for
immunofluorescent labeling of cancer cells were successfully reported with
very low nonspecific binding [23].
In vivo imaging of tumor tissue by tunable QDs is gaining lot of attention in
recent days. QDs can detect mult iple markers and increase the possibility of
early detection of tumors, which is not possible by using conventional image
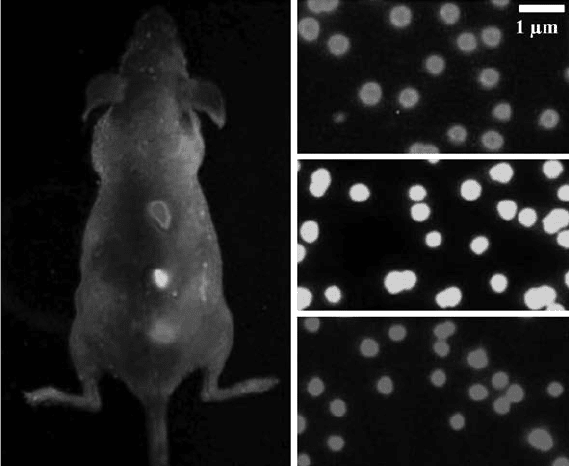
contrasting agents. Figure 16.1 shows capability of multicolor QDs imaging in
live animals [24, 25].
Qdot
1
, PLxBeads
1
and BioPixels
1
are the brand names of some of the
QDs that are commercially available for in vitroin vivo applications.
However, clinical success of QDs is impeded by their cytotoxicity. Besides
414 BIOMEDICAL NANOSTRUCTURES
cytotoxicity, the degradation and metabolism of these QDs in the body remains
to be investigated and there are reports that injected nanocrystals can
accumulate in kidney, liver, and spleen [26]. Whether or not QDs can
ultimately be cleared from the body is not known. More research in this area
must be completed before they can be used as probes for diagnostic
applications. However, advances in surface engineering and conjugation
certain to aid in progress of QDs as clinical labels.
16.2.2 Nanoshells
Nanoshells are composed of a dielectric core, usually silica, surrounded by a thin
metal shell, typically gold and in the size range of 10300 nm [27, 28]. Nanoshells
rely on the plasmon-mediated conversion of electrical energy into light. Similar
to quantum dots, nanoshells have the ability to be tunable optically and have
mission/absorption properties that range from the ultraviolet to the infrared [28].
Nanoshells are attractive because of their imaging potential and no heavy metal
toxicity unlike QDs. However, large size compared with quantum dots, is the
major disadvantage [28]. Polyethylene glycol (PEG)-coated long circulating [29,
30] nanoshells have been used in vivo as a contrast agent for imaging with optical
FIGURE 16.1 Multicolor quantum dot (QD) capability of QD imaging in live
animals. Approximately one to two million in each color were injected subcutaneously
at three adjacent locations on a host animal. Images were obtained with tungsten or
mercury lamp excitation. (Reprinted from Reference [25], with permission from
Macmillan Publisher Ltd.)
NANOSTRUCTURES FOR CANCER THERAPY 415
