Kenneth E. Gonsalves, Craig R. Halberstadt, Cato T. Laurencin, Lakshmi S. Nair. Biomedical Nanostructures
Подождите немного. Документ загружается.


CHAPTER 15
Nanostructures for Tissue
Engineering/Regenerative Medicine
SYAM P. NUKAVARAPU, SANGAMESH G. KUMBAR,
LAKSHMI S. NAIR, and CATO T. LAURENCIN
15.1 INTRODUCTION
15.1.1 Tissue Engineering/Regenerative Medicine
Tissue and organ failure has been a major health problem in the United Stastes
and worldwide. Autografting and allografting are the two main approaches
currently used to repair or replace damaged or lost tissue and organs. However,
limited availability and donor site morbidity are the problems associated with
autografting, while disease transmission and foreign body response are
associated with allografting. In addition, there is also a major concern about
the growing number of patients waiting for an organ replacement. According
to a recent report, in the United States alone 89,000 patients are waiting for an
organ transplant and 17 people die every day waiting for a transplant of a vital
organ such as a heart, liver, kidney, pancreas, lung, or bone marrow [1]. The
viable solution to this problem is tissue engineering/regenerative medicine,
which is defined as the application of biological, chemical, and engineering
principles toward the repair, restoration, or regeneration of living tissues using
biomaterials, cells, and factors, alone or in combination [2].
Researchers so far have identified three major strategies to regenerate tissues
using tissue engineering principles [3]. The first approach involves injecting
tissue-specific cells at the injury site. This is a noninvasive process and practiced
in the repair of minor injuries, however not useful for regenerating complex
tissues involving more than one cell type with a specific architecture. A second
approach is guided tissue engineering, where biodegradable scaffolds made of
BiomedicalNanostructures, EditedbyKennethE.Gonsalves,CraigR.Halberstadt,CatoT.Laurencin,
and Lakshmi S. Nair
Copyright # 2008 John Wiley & Sons, Inc.
377
poly(L-lactic acid) (PLLA), polyglycolic acid (PGA), poly(D,L-lactide-co-
glycolide) (PLGA), polycaprolactone (PCL), polyphosphazenes, and so on
are used for guiding cellular in growth. The material selection is based on the
desired application and is designed to permi t scaffold disintegration by the time
cells invade and completely repair the injured tissue.
Third and the most important strategy utilizes biodegradable scaffolds in
combination with growth factors and tissue-specific cells or stem cells that
ultimately differentiate into the required cell type. This approach is further
divided into two kinds, one is a closed system and the other is an open system.
In a closed system, cell-seeded scaffolds are encapsulated using a thin
semipermeable membrane that acts as a barrier an d protects cells from the
host immune response while allowing nutrient transport and waste removal
essential for cell survival. This approach is quite similar to extracorporeal
device technology where particular organ functionality is temporarily restored.
The open system is the most popular approach, where scaffolds are precultured
in vitro before implantation. Based on the requirement, growth factors that are
helpful in tissue regeneration are either released from the scaffold or delivered
through programming cells via gene therapy [4]. Biodegradable scaffolds in
combination with cells and growth factors have been used successfully to
regenerate various tissues like skin, bone, cartilage, blood vessels, and heart
valves. This method is further improved by replacing static culture methods
with dynamic culture systems using bioreactors. These advanced culture
systems not only mitigate nutrient transport limitations but also provide
mechanical stimula tion to seeded cells in the form of fluid shear stress [5].
Although tissue engineering approach has seen early success in the regenera-
tion of sim ple tissues like skin and cartilage, there are however biological and
engineering concerns to be addressed before the approach becomes a successful
clinical alternative for regenerating complex tissues. In order to become a
practical and successful procedure, scaffold selection, dosage of cells, and the
amount and the kind of growth factors have to be optimized based on the
particular application and the relative health status of the patient.
15.1.2 Scaffolds for Tissue Engineering
Tissue engineering involves assembling relevant cells into the required three-
dimensional (3D) organs or tissue. Cells lack this ability to form a 3D tissue in
culture and end up forming 2D layers with no anatomical features. The three-
dimensionality can be achieved by seeding cells onto an artificial structure known
as a scaffold that is capable of supporting 3D tissue formation, which is an
important component in tissue engineering [6]. The required features of a
scaffold are biocompatibility and bioactivity, biodegradability or bioresorb-
ability, mechanical compatibility, interconnected porosity, and the nontoxic
nature of the degradation products. Biocompatibility is defined as supporting the
appropriate cellular activity without eliciting any undesirable effects in those cells
and the host [7, 8]. Bioactivity refers to having appropriate surface chemistry that
378 BIOMEDICAL NANOSTRUCTURES
favors cell attachment, proliferation, and differentiation [9, 10]. Controlled
biodegradability is crucial because the rate of degradation of the scaffold should
be in line with the neotissue formation, since scaffolds need to be absorbed by the
surrounding tissues without the necessity of a surgical removal. High pore
volume and an adequate pore size are necessary for cell infiltration and also for
effective nutrient transport and waste removal. For example, scaffolds with 30%
pore volume and 150 mm median pore size performed well for bone tissue
regeneration [2]. Matching mechanical properties of the intended tissue (high
modulus for bone and low modulus materials for vascular tissue engineering) is
also critical for successful tissue engineering.
Synthetic polymers such as PGA, PLA, PLGA, PCL, polyphosphazenes, and
polyanhydrides, and naturally derived proteins and carbohydrate polymers such
as chitosan, alginate, and hyluronic acid are the most commonly used
biodegradable polymers for tissue engineering scaffold applications [11, 12].
Over the years, several techniques have been developed to fabricate synthetic and
natural polymeric materials into 3D porous scaffolds. Some of the conventional
scaffold fabrication techniques include solvent casting and particulate leaching
[13], gas foaming [14], freeze drying [15], 3D printing (solid freeform fabrication)
[16], and microsphere sintering [17]. It has been demonstrated that most of the
3D porous scaffolds were capable of supporting cellular adhesion and
proliferation, but lack the ability to simulate the extracellular matrix (CM)
like environment that is always in close proximity with cells in all the tissues.
Therefore, the best alternative is nanofeatured scaffolds that not only mimic the
ECM conditions but also show higher reactivity for proteins and ultimately
enhances cell adhesion and thus regenerative capacity. Techniques like
electrospinning [18], temperature-induced phase separation [19], molecular self-
assembly [20], and surface patterning [21] are currently in practice for generating
scaffolds with nano- and micron-size features for better mimicking the native
ECM. In this chapter, we focus on the importance of nanofeatured scaffolds and
their fabrication using various techniques. Further emphasis is also made on in
vitro and in vivo studies where the tissue regenerative capacity is enhanced with
these novel scaffolds.
15.1.3 Nanofeatures of ECM
ECM is a self-assembled nanofibrillar network compo sed of complex
biomacromolecules that surround and support cells in tissues [22]. ECM is
present in interstitial complex (IC) and basement membrane (BM) along with
cells as shown in Fig. 15.1a. ECM is constructed with different classes of
biomolecules such as structural proteins (collagen and elastin) and specialized
proteins (fibrillin, fibronectin, and laminin) that help to structurally connect,
bind, integrate cells to form tissues, and also provide a surface for cellular
adhesion and migration. Proteoglycans are glycoaminoglycans like chondroitin
sulfate and heparin sulfate linked with serine like core proteins that provide
chemical cues and regulate cell growth, differentiation, and metabolic activity.
NANOSTRUCTURES FOR TISSUE ENGINEERING/REGENERATIVE MEDICINE 379
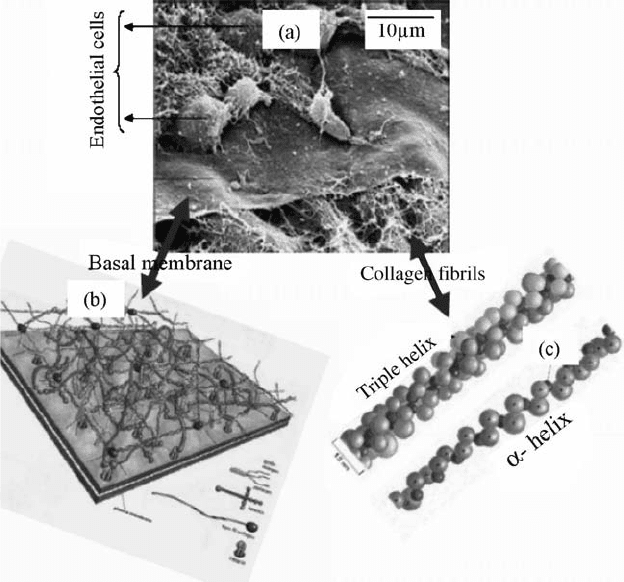
The interstitial complex is mostly present in connective tissues like bone,
cartilage, and ligaments , and present in association with a wide variety of cells
and the above-mentioned proteins. BM supports a single layer of endothelial,
epithelial, and some mesenchymal cells in many organs (Fig. 15.1b). BM’s
function is as an active barrier for cell infiltration and migration, and also acts
as a reservoir for a variety of growth factors.
ECM and BM are comprised of fibrils made of proteins such as collagen,
elastin, and fibronectin as shown in Fig. 15.1c. These fibrils are of nanosize and
much smal ler than micrometer-sized cells. The major component in the ECM is
collagen, whi ch is the fibrous backbone of the ECM an d is at least available in
28 forms in our body. The abundant collagen is mostly in a fibril structure
composed of 300-nm-long, 1.5-nm- diameter rodlike protei ns consisting of
three coiled chains. Each chain consists of 1050 amino acids that are wound
around in a characteristic right-handed triplex. Further, interaction of triple
helices of collagens results in the formation of fibrils that are roughly 50 nm in
FIGURE 15.1 (a) SEM image of a chick embryo cornea showing epithelial cells, basal
lamina, and network of collagen fibrils. Molecular structure of (b) basal lamina and
(c) collagen. (Reproduced from Reference [22] with permission from (a) Dr. Robert
Trelstad, (b) John Wiley & Sons Inc., and from (c) Garland Science Publishing).
380
BIOMEDICAL NANOSTRUCTURES

diameter. The fibril packing is such that adjacent triple helix is displaced
approximately 67 nm to yield a banded structure. Another important
component in the ECM is fibronectin that acts as a glue between cells and
the ECM by attaching cells to a variety of ECMs. Fibronectins (FNs) are
dimers of two similar peptides that are 6070 nm long and 23 nm thick. FN
contains nanodomains with an arginine glycineasparticserine sequence
that selectively exhibits high affinity for a particular substrate such as collagen,
fibrin, and cell surface receptors. In addition, other proteins that are present in
the ECM are also of nanosize and influence cell behavior at various levels.
15.2 NANOFIBROUS SCAFFOLDS
Cells within a tissue and organ are organized and encapsulated by the
nanofibrous ECM. For example, a SEM of rat cornea tissue is shown in
Fig. 15.2a that is composed of fibroblasts surrounded by ECM nanofibrils. A
similar cellular arrangement is observed when cells are cultured in vitro on
nanofibrous scaffolds (Fig. 15.2b). The tissue-like cellular organization observed
with nanofibrous scaffolds prompted researchers to look for more sophisticated
methods to fabricate nanofibrous structures that can better mimic the ECM and
serve as potential scaffolds for tissue regeneration. Electrospinning, phase
separation, and molecular self-assembly techniques that are currently popular
for fabricating nanofiber-based scaffolds are described below.
15.2.1 Electrospinning
Electrospinning is a facile, efficient, and inexpensive technique for fabricating
ultrafine fibers. A typical setup for electrospinning is shown in Fig. 15.3a.
During electrospinning, a high electric potential in the order of few kilovolts is
FIGURE 15.2 SEM of (a) the rat cornea tissue where fibroblasts are surrounded
by extracellular matrix fibrils (reproduced from Reference [22] with permission, from
Garland Science, publishing) and (b) human dermal fibroblasts cultured in vitro on PCL
nanofibers (reproduced from Appl Biochem Biotechnol 2005;125:147158 with
permission, Humana Press Inc.)
NANOSTRUCTURES FOR TISSUE ENGINEERING/REGENERATIVE MEDICINE 381
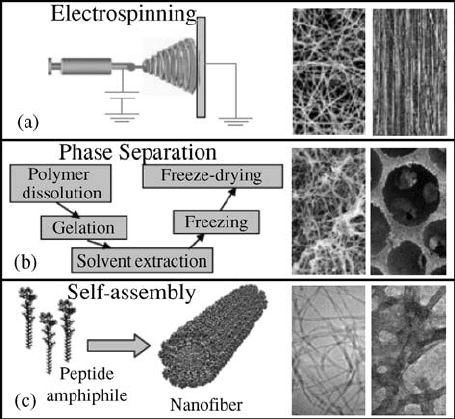
applied on the pendent drop of the polymer solution or melt held by surface
tension. The applied field causes charge separation and hence causes repulsion
within the polymer drop sitting at the nozzle of the spinneret. When the
opposing electrostatic force overcomes the surface tension of the polymer
solution, a thin polymer jet ejection initiates from the spinneret. The emerging
jet undergoes a series of bending and stretching instabilities and becomes
elongated until it is deposited on the grounded collector. Solvent evaporation
occurs during the process, and eventually a polymer jet is deposited into long
and ultrathin fibers with diameters ranging from tens of nanometers to a few
micrometers. Recently, this approach has draw n much interest within the tissue
engineering community. A number of synthetic and natural polymers have
been spun into a variety of structures having fibers randomly oriented,
oriented, coreshell structured, porous, and drug loaded that are suitable for
tissue regeneration and drug delivery applications (Fig. 15.3a) [23–27].
Electrospun nonwoven nanofiber matrices are promi sing as scaffolds for
tissue engineering since the fiber diameters are in line with most of the natural
ECMs, and the resulting high surface area might allow a high percentage of
FIGURE 15.3 Nanofibrous scaffolding methodologies; (a) Typical setup for electro-
spinning and the resulting nonoriented and oriented nanofiber scaffolds (reproduced
from Reference [33] with permission, from Elsevier). (b) Steps involved in the
temperature-induced phase separation process, and the obtained nanofibrous and
foams with spherical pore shape (reproduced from Reference [19] with permission, from
Elsevier). (c) Self-assembly process and the derived individual nanofibers and the
consolidated nanofibrous scaffold (reproduced from Reference [49] with permission
from AAAS).
382
BIOMEDICAL NANOSTRUCTURES
cellular attachment. In addition to the nanofibrous structure, the matrices also
exhibit porosities greater than 90 % with adequate pore size for cell migration
[27]. Thus, nanofiber matrices provide more structural space for cell
accommodation and make the nutrient and metabolic was te exchange process
efficient between the scaffold and the surroundings.
Electrospinning technique for creating tissue engineering scaffolds has been
demonstrated and patented by Laurencin and Ko [28]. These authors
demonstrated for the first time the formation of electrospun PLGA ultrafine
fibers with morphological similarity to the ECM of natural tissue with a
diameter ranging from 500 to 800 nm with porosity greater than 90% [27]. In
vitro studies with mouse fibroblasts further indicated good cellular adherence
and spreading onto PLGA nanofibers. Based on these results, it was
hypothesized that 25100 mm pore diameter that was observed is adequate
for human mesenchymal stem cell migration and tissue ingrowth.
Incorporation of drugs into these nanofibers has also been explored by these
authors for wound healing applications [29]. In a recent study, Nair et al. from
the same group developed polyphosphazene-based nanofiber scaffolds for
tissue engineering applications. These novel scaffolds support the adhesion and
proliferation of coronary artery endothelial cells and osteoblast-like MC3T3-
E1 cells [30]. Currently, we are focusing on developing various polypho-
sphazene fiber scaffolds alone or in combination with PLGA for bone tissue
engineering and wound dressing applications.
Many other resear ch groups have also successfully electrospun biodegrad-
able polyesters such as PLA, PGA, and their copolymer PLGA into nonwoven
fiber scaffolds [31, 32]. PLLA is electrospun into aligned and random nanofiber
mats with varied fiber diameters ranging from 300 to 700 nm. Ramakrishna
et al. studied the cellular compatibility of PLLA nanofibers using neural stem
cells [33]. It was noticed that cells adhered and proliferated on aligned and
randomly oriented PLLA nanofibrous scaffolds . Other synthetic biodegradable
polymers like PCL and PCL blends with polyorthoesters were also studied for
their spinnability and cell interaction. Fetal bovine chondrocytes seeded on
PCL nanofiber scaffolds, with an average fiber size of 700 nm, were able to
maintain the chondrocytic phenotype during 3 weeks of culture period [34].
Geng et al. demonstrated better cell attachment and proliferation with human
umbilical vein endothelial cells onto 50 : 50 poly(
L-lactic acid-co-e-caprolac-
tone) (PLCL) fibers of 300 nm diameter when compared to the matrices with
7-mm fibers [35]. The observed better cellular performance on these nanofiber
matrices is presumably due to the nanofiber structure that mimics the ECM.
Various natural polymers have also been considered for electrospinning
because of their prov en biocompatibility and biofunctionality. Natural
polymers such as collagen, fibrinogen, alginate, hyaluronic acid, chitosan,
and starch were electrospun alone or in combination with synthetic polymers
to improve scaffold performance [36, 37]. Nanofiber scaffolds of ECM
polymers may have added advantage of resembling not only the size scale but
also the chemical and biological functions of the ECM. Collagen I and III were
NANOSTRUCTURES FOR TISSUE ENGINEERING/REGENERATIVE MEDICINE 383
electrospun using 1,1,1,3,3,3-hexafluro-2-propanol (HFT) as a solvent that
created type I collagen nanofibers with diameters in the range of 100 nm and
type III collagen in the range of 250 nm [38]. The fabricated nanofiber scaffolds
exhibited good cytocompatibility and proliferation with smooth muscle cells,
and also showed cell infiltration into the collagen network. Efforts were made
to improve the mechanical properties of the collagen nanofi ber scaffolds by
PCL coating. Coating imparted the right mechanical strength while retaining
the collagen regions on the surface that maintained cell attachment and
proliferation of human dermal fibroblasts for the purpose of dermal tissue
engineering [39]. Even though electrospinning is a proven mild process,
biological integrity of some polymers has been found to be lost during
electrospinning. Such an adverse affect is presumably due to the harsh organic
solvents like HFT used for electrospinning that could potentially denature the
protein structures. Therefore, electrospinning of natural polymers is limited
and still not versatile as compared to synthetic polymers.
15.2.2 Phase Separation
Polymer phase separation has been known for many years and is widely used
for developing membranes for nonmedical applications [40]. Recently,
thermally induced phase separation (TIPS) has become a popular technique
for fabricating porous and nanofeatured 3D scaffolds for tissue engineering
applications [41]. In this method, a polymer is first dissolved in a suitable
solvent at high temperature. Lowering the solution temperature induces
liquidliquid or solidliquid phase separation into two phases, one with a
high polymer concentration and the other with a low polymer co ncentration.
Subsequent solvent removal by exchange, evaporation, or sublimation from
low polymer phase results in pores, whereas the high polymer phase solidifies
into 3D interconnected fibrous networks as seen in Fig. 15.3b. Scaffold
parameters such as porosity and fiber size can be controlled by varying the
solvent, the polymer concentration, and the temperature for any polymer
system.
Polymer phase separation has gained immense interest in recent days for
developing tissue engineering scaffolds for two reasons. The first reason is that
controlling scaffold porosity is ideal for supporting cell proliferation and 3D
tissue formation. Second is the ability to form nanofibrous 3D structure,
which mimics the fibrous 3D structure of natural collagen. Figure 15.3b shows
SEM of highly porous scaffolds from aliphatic polyesters such as PLLA,
poly(
D-L-lactic acid) (PDLLA), and PLGA produced by phase separation
[42–44]. However, the na nofibrillar structure is only observed with the highly
crystalline polymer PLLA . Ma et al. extensively studied the effect of gelation
temperature, polymer concentration, and solvent s used on porosity and fiber
diameters of scaffolds produced using PLLA. Combining the polymer with
porogens further modified this process and achieved macroporous networks in
scaffolds with nanofibrous pore walls [41].
384 BIOMEDICAL NANOSTRUCTURES
15.2.3 Molecular Self-Assembly
Molecular self-assembly is the spontaneou s organization of molecules into
structurally well-defined and stable arrangements through weak and non-
covalent interactions. Such interactions include hydrogen bonding, ionic
bonding, hydrophobic interactions, van der Waals interactions, and water-
mediated hydrogen bonding. Recently, molecular self-assembly has become
one of the powerful methods to produce nanofibrous scaffolds for tissue
engineering with properties similar in scale and chemistry to that of the natural
ECM.
Proteins, peptides, and lipid molecules are commonly used as building
blocks for forming nanofeatured scaffolds for tissue engineering applications.
Nature has already used these molecules to produce various struc tures like
collagen and keratin through molecular self-assembly [45]. For the first time,
Zhang et al. produced 3D peptide scaffolds utilizing naturally occurring amino
acids by exposing the self-assembling peptide ((Ala-Glu-Ala-Glu-Ala-Lys-Ala-
Lys)
2
(EAK16)) to physiological media or a salt solution [46]. Peptides with
alternating hydrophilic and hydrophobic amino acid moieties aggregated
depending upon salt concentration and pH to form stable scaffolds consisting
of interwov en nanofibers. This method has opened a new area of research
where the peptide solution forms into a scaffold in vivo with ECM
functionalities that would help in repair and regeneration of damaged or lost
tissues. Based on this concept BD biosciences has commercialized a product
‘‘PuraMatrix peptide hydrogel’’ with fiber diameters of 10 nm and pores of
50200 nm. This matrix has shown in vitro differentiation of hepatocyte
progenitor cells into hepatocytes [47], neurons from hippocampal slices [48],
and neurite out growth from PC12 cells [96].
Recently, Stupp et al. designed peptide-amphiphile (PA) nanocylinders
adopting a composite approach with nonpolar hydrocarbon tails and
hydrophilic peptide heads as seen from Fig. 15.3c [49]. The nanocylinder
formation is induced through a pH change from 8 to 4. The produced fibers
(diameter of 7.6 1 nm) were cross-linked by oxidation to achieve structural
integrity. Phosphorylated serine incorporation within the peptide end leads to
the calciophilic nature of the scaffold with the ability to form hydroxyapatite
mineral coating that promise a new approach for bone tissue engineering [50].
In vitro studies with different cell types on 3D peptide scaffolds exhibited
interesting functional behavior such as good proliferation, functional
differentiation, and active migration [51]. In a separate study with bovine
chondrocytes encapsulated in a peptide scaffold, Kisiday et al. showed the
extensive production of their own ECM (different types of collagen with
glycosaminoglycans) while maintaini ng their phenotype [52]. The self-assembly
process has also demonst rated the ability to present bioactive epitopes to cells.
For example, the nanofiber scaffold containing the neurite-promoting laminin
epitope isolucinelysinevalinealaninevaline (IKVAV) pentapeptide selec-
tively induced rapid differentiation of neural progenitor cells into neurons [99].
NANOSTRUCTURES FOR TISSUE ENGINEERING/REGENERATIVE MEDICINE 385
