Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

3.2 An electron in a potential well with infinite barriers 73
E
2
E
1
x
L0
E
n
,U
U(0) = ∞
ψ
1
2
ψ
2
2
L/2
U(L) =
∞
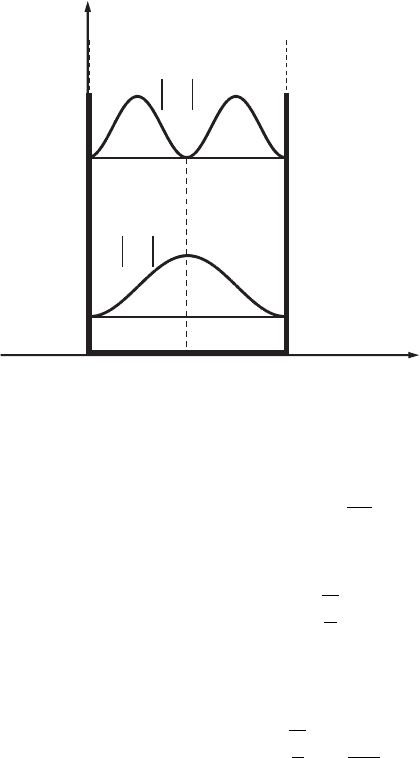
Figure 3.2 The probability
density distributions,
|ψ
1
(x)|
2
and |ψ
2
(x)|
2
,for
the two lowest energy
levels, E
1
and E
2
,inawell
with impenetrable walls.
Taking into account Eq. (3.42), this integral reduces to
L
0
sin
2
(k
n
x)dx =
1
2k
n
k
n
L. (3.49)
The parameter C is
C =
2
L
. (3.50)
Finally, the wavefunction of the electron in the potential well for the nth energy
state can be written as
ψ
n
(x ) =
2
L
sin
nπ x
L
. (3.51)
The graphs of the wavefunctions ψ
1
(x) and ψ
2
(x)forn = 1 and n = 2, are
shown in Fig. 3.1. The distribution of the probability density, |ψ
n
(x)|
2
, of finding
an electron in the region of the potential well whose number of maxima is
determined by the quantum number n is shown in Fig. 3.2. We see that in the
ground state (n = 1) the probability of finding an electron has a maximum at the
center of the potential well. In the first excited state (n = 2) the probability of
finding an electron at the center of the well is equal to zero, whereas the probability
of finding an electron has maxima at the points x = L/4 and x = 3L/4. This
behavior of the electron differs drastically from the behavior of a classical particle
in a potential well, for which the probability of finding a particle is the same at
any point in the well, for any value of the particle’s energy (see Appendix A,
Section A.2.6).

74 Layered nanostructures
Note that there are similarities between the mathematical description of elec-
tron motion in a potential well with infinite walls and the oscillations of a
string with fixed ends. In both cases the boundary conditions are such that
the length of the system (in our case it is the width of the potential well or
the distance between fixed ends in the case of the string) must be equal to an
integer multiple of half of the wavelength, which is described by the wavefunc-
tion (in the case of an electron) or by the transverse wave (in the case of a
string).
Example 3.2. An electron is located in a rectangular potential well with infinite
walls. The width of the well is L = 10 nm. Find the energies of stationary
states of the electron for which at the well’s boundary x = L the derivatives of
the wavefunction are negative and equal to −τ
n
(here τ
n
are arbitrary positive
constants).
Reasoning. The wavefunction, ψ
n
,forthenth stationary state in the potential
well with infinite walls is given by Eq. (3.51). Let us find its derivative:
dψ
n
dx
=
2
L
nπ
L
cos
nπ x
L
. (3.52)
For the point x = L,Eq.(3.52) gives the relation between the derivative of the
wavefunction and its quantum number, n:
2
L
nπ
L
cos(nπ) =−τ
n
. (3.53)
Since the quantum number n is an integer and positive, and because the left-hand
side of Eq. (3.53) must be negative, the quantum number n must be an odd
number:
cos(nπ) =−1, n = 1, 3, 5,... (3.54)
On substituting Eq. (3.54) into Eq. (3.53) we obtain the expression for n for a
given τ
n
:
n =
τ
n
L
π
L
2
. (3.55)
On substituting Eq. (3.55) into the formula for the energy of the nth state,
Eq. (3.44), we obtain
E
n
=
π
2
h
-
2
2m
e
n
L
2
=
h
-
2
Lτ
2
n
4m
e
. (3.56)
On substituting the value of L = 10 nm into Eq. (3.56) we obtain
E
n
= 3.1 × 10
−47
τ
2
n
J = 1.91 × 10
−28
τ
2
n
eV. (3.57)

3.3 An electron in a potential well with finite barriers 75
In reality we never deal with the case of given τ
n
, and rather relation (3.55) has
to be rewritten in the form
τ
n
=
nπ
L
2
L
, (3.58)
i.e., Eq. (3.58) defines the value of the derivative of the wavefunction at the
boundary of the potential well and the most important conclusion from Eq. (3.58)
is that the derivative τ
n
is a quantized quantity. Thus, from Eq. (3.58) we obtain
τ
n
≈ 4.4 × 10
12
n m
−3/2
, (3.59)
where n = 1, 3, 5,... By substituting the first two values of τ
n
into Eq. (3.57)
we obtain for the magnitudes of the first two odd energy levels E
1
= 3.7meV
and E
3
= 33 meV. Note that at boundaries with infinite walls the condition of
continuity of the derivative of the wavefunction is violated while the continuity
of the wavefunction is preserved since each wavefunction becomes zero at the
boundaries.
3.3 An electron in a potential well with finite barriers
The technology of fabrication of planar layered structures with given physical
properties allows the experimental realization of the confinement of electrons
inside potential wells of various profiles. Most important for practical realizations
are one-dimensional potential profiles of rectangular wells, (1) with one of the
potential barriers being finite and the other infinite, and (2) with both potential
barriers finite.
3.3.1 The potential well with one of the barriers finite
Let the dependence of the potential energy of an electron on the coordinate x
have the following form:
U (x ) =
∞, x ≤ 0,
0, 0 < x < L ,
U
0
, x ≥ L .
(3.60)
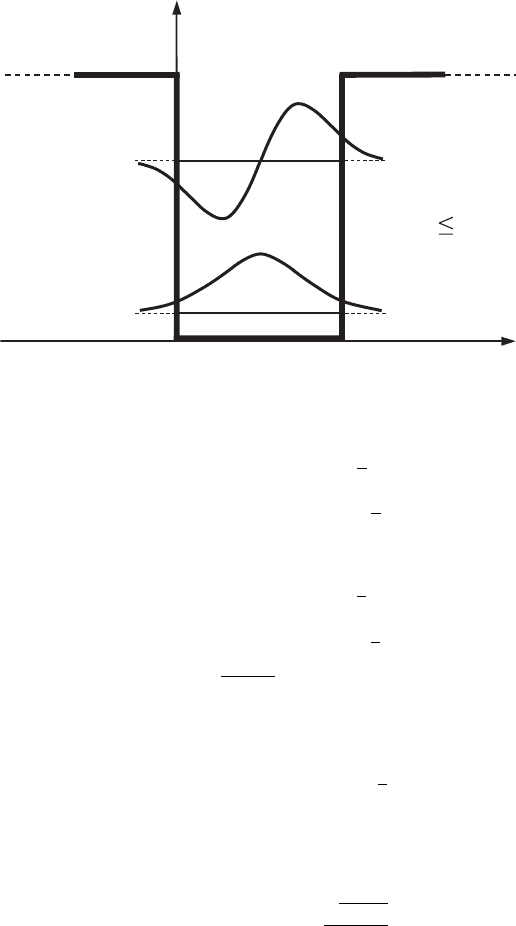
Such a potential profile is shown in Fig. 3.3. The finite value of the potential at
the right boundary, x = L, considerably changes the solutions of the Schr
¨
odinger
equation and correspondingly the behavior of an electron. It is now necessary
to distinguish between two different types of electron motion in the well. The
first type corresponds to E ≤ U
0
, and the electron in the potential well can have
bound states, which correspond to the discrete set of energy levels. The second
type of motion corresponds to E > U
0
, and the electron does not have bound
states and can be found at any point with coordinate x > 0. This type of motion
corresponds to a continuous energy spectrum. In this section we will consider
only the case when E ≤ U
0
and the electron is confined in the potential well.
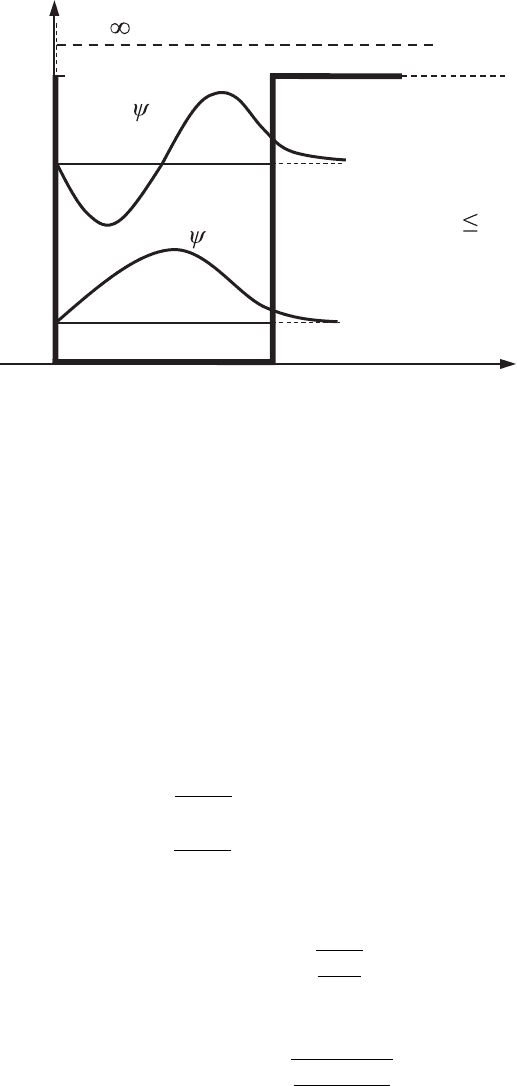
76 Layered nanostructures
E
1
xL0
E,U
U =
U
0
E
2
2
(x)
1
(x)
12
E >U
0
E U
0
Figure 3.3 The first two
energy states, E
1
and E
2
,
and the corresponding
wavefunctions, ψ
1
and ψ
2
,
foranelectronina
potential well with the
right-hand-side potential
barrier finite.
A classical particle cannot move out of the well because its kinetic energy
would be negative when it is outside of the well. On reflecting from the walls
of the well the particle would move only inside the well. The behavior of the
electron described by the solution of the corresponding Schr
¨
odinger equation is
fundamentally different from the behavior of a classical particle.
Because the potential profile U (x) is a step-like function, let us divide the
electron’s accessible region x > 0 into regions 1 and 2 with potential ener-
gies U
1
= 0 and U
2
= U
0
. In each of these regions we will obtain solutions of
the Schr
¨
odinger equation. Then, in correspondence to the boundary conditions
applied to the wavefunctions, we will join these solutions in such a way that these
wavefunctions and their derivatives are continuous at the boundaries.
Let us write the Schr
¨
odinger equation for each of these regions, 1 and 2:
d
2
ψ
1
(x )
dx
2
+ k
2
ψ
1
(x ) = 0, 0 ≤ x ≤ L , (3.61)
d
2
ψ
2
(x )
dx
2
− κ
2
ψ
2
(x ) = 0, x > L. (3.62)
Here we introduced for the wavenumbers of the electron wave in the potential
well
k =
2m
e
E
h
-
2
, (3.63)
and outside of the potential well
κ =
2m
e
(U
0
− E)
h
-
2
. (3.64)
3.3 An electron in a potential well with finite barriers 77
The solutions of Eqs. (3.61) and (3.62) can be written as follows:
ψ
1
(x ) = A
1
e
ikx
+ B
1
e
−ikx
, (3.65)
ψ
2
(x ) = A
2
e
κx
+ B
2
e
−κx
, (3.66)
where A
1
, A
2
, B
1
, and B
2
are constants, which can be found from the bound-
ary conditions. In our case the boundary conditions are the continuity of the
wavefunction and its derivative at the boundaries of the potential well. From the
condition of continuity of the wavefunction at x = 0 we obtain the equation for
unknowns A
1
and B
1
:
A
1
+ B
1
= 0, (3.67)
since in the region x ≤ 0 the function ψ(x) = 0. Taking into account the last
equation, Eq. (3.65) transforms into
ψ
1
(x ) = 2iA
1
sin(kx). (3.68)
The wavefunction must be finite everywhere, in region 2 as well. For this reason
A
2
= 0 because in region 2 the term A
2
e
κx
increases infinitely with increasing
x. At the boundary x = L the following conditions for the continuity of the
wavefunction and its derivative must be satisfied:
ψ
1
(L) = ψ
2
(L), (3.69)
dψ
1
dx
x=L
=
dψ
2
dx
x=L
. (3.70)
After substitution of the corresponding expressions for ψ
1
and ψ
2
, the boundary
conditions (3.69) and (3.70) can be rewritten in the form
2iA
1
sin(kL) = B
2
e
−κ L
, (3.71)
2ikA
1
cos(kL) =−κ B
2
e
−κ L
. (3.72)
Let us divide Eq. (3.71)byEq.(3.72). As a result we obtain the following
equation:
tan(kL) =−
k
κ
, (3.73)
which defines the energy spectrum of the electron in the potential well. This is
a transcendental equation, whose solution cannot be obtained analytically. For
the analysis of such equations very often graphical methods of solution are used,
together with numerical methods.
Let us transform Eq. (3.73) in order to carry out the graphical analysis. Taking
into account the trigonometric identity
sin ϕ =±
tan ϕ
1 +tan
2
ϕ
, (3.74)
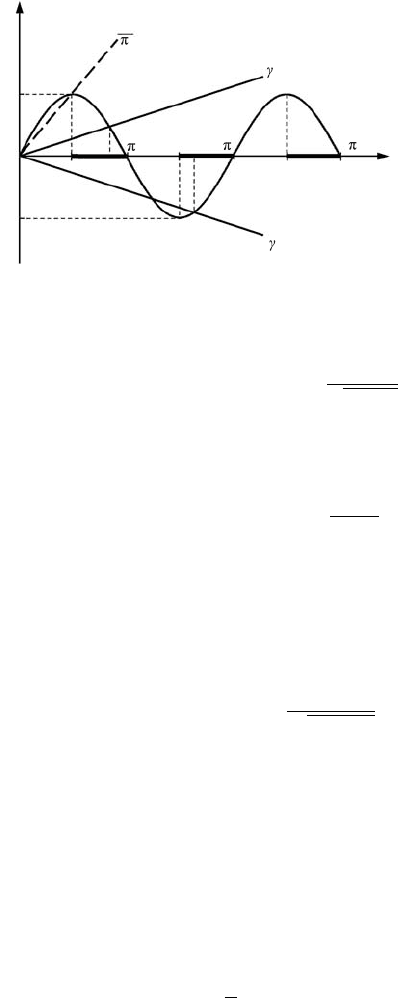
78 Layered nanostructures
k
2
L
3
k
1
L
2
1
−1
0
kL
f
f=
kL
f=−
kL
f= kL
2
f=sin(kL)
Figure 3.4 Graphical
solution of the
transcendental equation
(3.77).
we can transform Eq. (3.73) into the form
sin(kL) =±
k
√
k
2
+ κ
2
. (3.75)
According to Eqs. (3.63) and (3.64), for the terms under the square root in
Eq. (3.75) we obtain
k
2
+ κ
2
=
2m
e
U
0
h
-
2
. (3.76)
By substituting Eq. (3.76) into Eq. (3.75) we obtain
sin(kL) =±γ kL, (3.77)
where we introduced the parameter
γ =
h
-
2m
e
L
2
U
0
, (3.78)
which defines the slope f (kL) =±γ kL.
Let us plot graphs of the right-hand and left-hand sides of Eq. (3.77)as
functions of the parameter kL (see Fig. 3.4). The points of intersection of the
sine function with the lines f =±γ kL define the roots of Eq. (3.77). From all
the roots of Eq. (3.77) we separate the roots which are the roots of Eq. (3.73).
The chosen roots must satisfy the condition tan(kL) < 0. The tangent function
is negative only in the following intervals of values of kL where the roots can
exist:
π
2
+ nπ ≤ kL ≤ π + nπ. (3.79)
Thus, we have to choose the roots of Eq. (3.77) which exist in the interval (3.79).
In Fig. 3.4 intervals of kL (Eq. (3.79)) are shown by thick lines. Note that from the
expressions derived we can identify wells for which the solutions of Eq. (3.73)do

3.3 An electron in a potential well with finite barriers 79
not exist. So there would be no roots and no bound energy states of the electron in
those potential wells. We show by the dashed line the maximal slope of f = γ kL
for the parameter γ = 2/π when the roots for Eq. (3.73) exist. For higher values
of the slope, γ , there are no bound states. Thus, the condition for which there is
at least one energy state with E ≤ U
0
reduces to the inequality
γ ≤ 2/π. (3.80)
Taking into account relation (3.78), inequality (3.80) takes the form
h
-
√
2m
e
U
0
≤
2L
π
. (3.81)
This condition can be rewritten in more convenient form:
U
0
L
2
≥
π
2
h
-
2
8m
e
, (3.82)
where the left-hand side of the inequality is defined by the potential-well param-
eters only, and the right-hand side by the type of the particle confined in the
potential well. In relatively shallow and/or narrow potential wells, there may be
no bound states for an electron, i.e., the electron cannot be captured by such a well
and the electron has energy E > U
0
, which is greater than U
0
. That corresponds
to the case of an unbound particle, which will be considered in Section 3.4.1.
The second energy level in the well can exist at γ ≤ 2/(3π ), i.e.,
U
0
L
2
≥
9π
2
h
-
2
8m
e
. (3.83)
As the parameter U
0
L
2
increases from the value at which equality is achieved
in Eq. (3.80) to the value at which equality is achieved in Eq. (3.83), the first
level appears at E
1
= U
0
and goes down to E
1
= 0.3 × U
0
, and the second level
appears at E
2
= U
0
. Further increase of U
0
L
2
brings both levels, E
1
and E
2
,
down and, at γ ≤ 2/(5π), the third energy level appears. Thus, the spectrum of
eigenvalues of the electron energy is discrete.
Let us now write expressions for the wavefunctions in each of the two regions
(Eqs. (3.65) and (3.66)), taking into account the relations between the coefficients
which we have obtained:
ψ
1
= A sin(kx), (3.84)
ψ
2
= A sin(kL)e
κ L
e
−κx
, (3.85)
where we introduced the constant A = 2iA
1
. From these two expressions it
follows that inside the potential well the wavefunction is an oscillatory function,
as it was in the case of the potential well with infinite barriers. Outside of the
well, as the electron moves away from the potential barrier, its wavefunction
decreases exponentially. This means that in a bound state there is a non-zero
probability of the electron being outside of the potential well where its energy
80 Layered nanostructures
is E < U
0
and therefore the electron’s kinetic energy, K , would be negative.
This can be explained as follows: in quantum mechanics the total energy, E ,
cannot be presented as a sum of kinetic and potential energy because, according
to Heisenberg’s uncertainty principle, kinetic and potential energies (as well as
momentum and coordinate) cannot be both exactly measured simultaneously.
The uncertainty relations Eq. (2.117) relate the particle’s coordinates and its
corresponding momentum projections, which define potential (coordinates) and
kinetic (momenta) energies. Thus, the situation with E < U
0
is possible for a
quantized particle, and it is very common in various theoretical and applied
problems, as we will see in later chapters. The qualitative dependence of the
wavefunctions for the first two energy states on the coordinate x is shown in
Fig. 3.3.
Example 3.3. Find the ratio E
1
/U
0
for an electron in a well with the poten-
tial profile described by Eq. (3.60) when only one bound state exists, which
corresponds to the parameter kL = 3π/4.
Reasoning. The parameter k is defined by Eq. (3.63),
k =
2m
e
E
1
h
-
2
.
Then, from the relation kL = 3π/4 we obtain for the energy, E
1
,thefollowing
equation:
E
1
=
9π
2
h
-
2
32m
e
L
2
. (3.86)
The parameter γ , which defines the slope of the function f = γ kL,asshownin
Fig. 3.4, can be found from Eq. (3.77):
γ =
sin(kL)
kL
=
√
2/2
3π/4
=
2
√
2
3π
. (3.87)
Using the definition of the parameter γ (Eq. (3.78)), we obtain the expression
for the height of the potential barrier U
0
:
U
0
=
9π
2
h
-
2
16m
e
L
2
. (3.88)
Taking into account Eq. (3.86), we obtain
E
1
U
0
= 0.5.
Example 3.4. Find the normalization constant A for the wavefunction described
by Eqs. (3.84) and (3.85).
Reasoning. Since the electron’s behavior in regions 1 and 2 is described by its
own wavefunction, the normalization condition must be written in the form
∞
0
|ψ|
2
dx =
L
0
|ψ
1
|
2
dx +
∞
L
|ψ
2
|
2
dx = 1. (3.89)

3.3 An electron in a potential well with finite barriers 81
Let us substitute into this condition the expressions for the wavefunctions ψ
1
and
ψ
2
:
|A|
2
L
0
sin
2
(kx)dx + sin
2
(kL)e
2κ L
∞
L
e
−2κx
dx
= 1. (3.90)
By carrying out the integration we obtain
|A|
2
L
2
−
sin(2kL)
4k
+
sin
2
(kL)
2κ
= 1. (3.91)
Let us assume that the constant A is real. Then
A =
2
L − [sin(2kL)]/(2k) + [sin
2
(kL)]/κ
. (3.92)
At U
0
→∞the parameters κ →∞and kL → nπ . Then
A =
2
L
,
which coincides with the normalization constant from Eq. (3.50).
3.3.2 A symmetric potential well with finite
potential barriers
The dependence of the electron potential energy in a symmetric potential well
with finite potential barriers is defined as
U (x ) =
U
0
, x ≤ 0,
0 , 0 < x < L,
U
0
, x ≥ L .
(3.93)
Such a potential well is shown in Fig. 3.5. Two cases, E ≤ U
0
and E > U
0
,as
in Section 3.3.1 are possible. Let us consider here one of these cases, in particular
the case with E ≤ U
0
. By analogy to Eqs. (3.65), (3.66), and (3.68) the solutions
of the Schr
¨
odinger equation for the three regions 1, 2, and 3 (see Fig. 3.5), taking
into account that wavefunctions at x →±∞must be finite, can be written in the
following form:
ψ
1
(x ) = C
1
e
κx
, x ≤ 0,
ψ
2
(x ) = C
2
sin(kx + φ), 0 < x < L ,
ψ
3
(x ) = C
3
e
−κx
, x ≥ L.
(3.94)
In Eqs. (3.63) and (3.64) we have already introduced the parameters k =
√
2m
e
E/h
-
and κ =
√
2m
e
(U
0
− E)/h
-
. The boundary conditions of continu-
ity of the wavefunctions and their derivatives at points x = 0 and x = L give us
a system of four equations to find the constants C
1
, C
2
, C
3
, and φ as well as the
82 Layered nanostructures
E
2
E
1
U
0
xL0
E
n
,U
ψ
2
ψ
1
123
E > U
0
E U
0
Figure 3.5 A symmetric
potential well with
potential barriers of finite
height. The first two
lowest energy levels, E
1
and E
2
,andtheir
corresponding
wavefunctions, ψ
1
(x)and
ψ
2
(x), are shown.
relationships between the parameters, which replace Eq. (3.73):
tan φ =
k
κ
, (3.95)
tan(kL +φ) =−
k
κ
. (3.96)
These equations can be reduced to
sin φ =
k
b
, (3.97)
sin(kL +φ) =−
k
b
, (3.98)
where the parameter b =
√
2m
e
U
0
/h
-
. If we eliminate from these equations the
parameter φ we obtain an equation that defines the energy spectrum of an electron
in the symmetric potential well with finite barrier heights:
kL = nπ − 2arcsin
k
b
, (3.99)
where the quantum number n = 1, 2, 3,... defines the electron energy-level
number in the well. Since the positive values of the argument of the arcsine
function cannot exceed unity, i.e., k/b ≤ 1,
k
max
= b =
√
2m
e
U
0
h
-
. (3.100)
The energy of an electron in the potential well is quantized in accordance with
Eq. (3.99), i.e., its energy spectrum is discrete. Let us show this graphically.
Figure 3.6 shows the graphs of f (k) = kL and f
n
(k) = nπ − 2arcsin(k/b),
which correspond to the left-hand and right-hand sides of Eq. (3.99). The index
n of the function f
n
(k) coincides with the quantum number n. The points of
intersection of the line f (k) with the curves f
n
(k) define the roots of Eq. (3.99).
We see from Fig. 3.6 that the spectrum of values of k and their related values
