Vij D.R. Handbook of Applied Solid State Spectroscopy
Подождите немного. Документ загружается.

16. Polarization Spectroscopy of Ordered Samples
16.1.3 Spectroscopy with Linearly Polarized Light
One of the important applications of LD spectroscopy is now clear. It provides a
directional (or symmetry) criterion for spectral assignments, which often may be
crucial, as illustrated later. In the case of molecules with symmetry elements the
transition moment direction often acts like an absolute criterion, i.e., it specifies
the correct symmetry without any deviations, and in that sense it is better than,
e.g., the energy or intensity criteria.
Another important application of the directional information provided by LD
spectroscopy is the determination of sample structures, both intramolecular—for
example determination of the pyramidilization angle around N in aniline [8]—as
well as intermolecular—for example the bonding of carcinogenic molecules
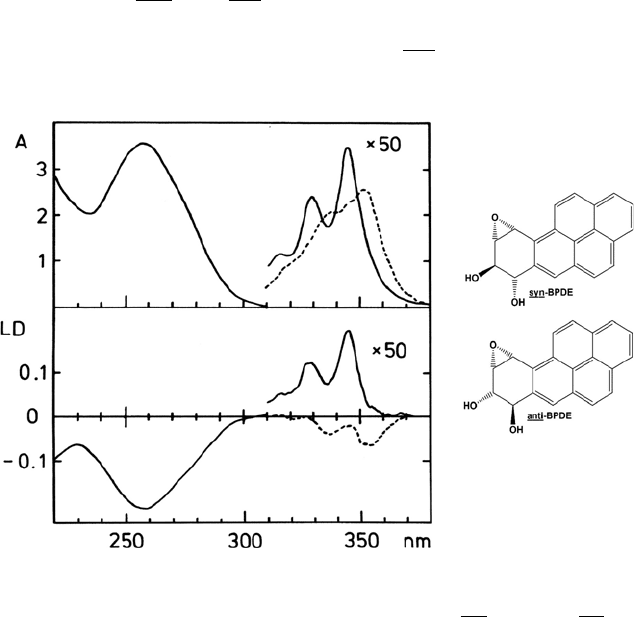
to DNA [9]. The latter case is illustrated in Figure 16.5, where the difference in
DNA binding of syn-
and anti- forms of benzo[a]pyrene-diol-epoxides is
reflected in the LD spectra. There exists a corresponding difference in
carcinogenicity of these two stereoisomers, the anti
-form being the stronger
carcinogen [9].
Figure 16.5 Absorbance (A) and linear dichroism (LD) spectra of anti(------)- and syn(------)-
BPDE-DNA in aqueous solution (10 mM cacodylate buffer, pH = 7.0).The strong negative LD
band at 258 nm can be attributed to DNA, while the longer wavelength signals correspond to
SS* transitions in the two benzo[a]pyrene-diol-epoxides. The data is reproduced from [9].
694

16.1 Introduction
Clearly, sample structure will influence the result of an LD experiment and
this gives an additional possibility for extracting information from an LD
experiment. When, for example, the directions of one or more transition
moments in the framework of a given sample molecule are known, they may be
used to identify the position (orientation) of the molecule in the sample. It is,
however, important to stress from the start that an LD experiment cannot provide
anywhere near complete information on the sample structure, such as a complete
orientation distribution function, except in the simple case where the alignment
is perfect.
The information that is available from LD spectroscopy on the molecular
orientation distribution in the sample is restricted to information about average
values over all sample molecules of cosine squares of the angles between
transition moments in molecules and the sample (laboratory) axes, as we shall
see later. In the general case, a given set of such averages will correspond to an
infinite number of possible orientation distribution functions for the sample
molecules.
It is interesting to note that often neither the transition moment directions, nor
the sample structure are known before the LD spectrum is recorded, but may be
uncovered through initial assumptions on both kinds of information, which later
may be verified.
In addition to absorption (LD) spectroscopy, other kinds of spectroscopy
with linearly polarized light may also be performed on aligned samples.
The most important of these involve two photons (see Table 16.1).
In Raman spectroscopy and luminescence spectroscopy (fluorescence and
phosphorescence), one photon is absorbed and another one, with different
energy, is created. Other two-photon experiments, in which both photons are
absorbed, are two-photon absorption and photoinduced dichroism. In Raman
scattering and two-photon absorption spectroscopy the two photons act
simultaneously, and new optical properties may be revealed: the Raman
polarizibility tensor and the two-photon absorption tensor. In luminescence
spectroscopy and photoinduced dichroism experiments the two photon events
are successive and the spectra may be described in terms of transition moments.
695
Table 16.1 Overview of two-photon events and the corresponding physical processes [2].
Photon 1 annihilated annihilated annihilated annihilated
Photon 2 annihilated annihilated created created
Timing successive simultaneous simultaneous successive
Physical
Process
photoinduced
dichroism
2-photon
absorption
Raman
scattering
photolumi-
pescence
16. Polarization Spectroscopy of Ordered Samples
16.2 OCCURRENCE, PRODUCTION, AND OPTICAL
PROPERTIES OF ALIGNED, SOLID SAMPLES
16.2.1 Perfectly and Partially Aligned Samples
While the molecular orientation distribution in liquid solutions is isotropic, many
naturally occurring samples have some kind of order (are aligned). This is not
only the case for crystals; many biological samples, for example, from plants or
animals, have alignment, although this alignment tends to be only partial, not
perfect. Also, many industrial products are aligned samples: the strength of
common plastic bags, for example, is due to the fact that they consist of two
crossed layers of stretched or extruded, in other words, aligned polyethylene.
Such polyethylene material cannot be stretched further in the direction of
stretching; it is therefore very strong in this direction, and by crossing two layers
a very sturdy material is produced. The alignment in the polyethylene bags and
in many naturally occurring or industrial samples is not perfect, but only partial.
However, it turns out that such samples, especially those produced by means of
aligning (solid) solvents, are often the most useful. Therefore, after a brief
discussion of other alignment techniques, we shall concentrate on solutes that are
partially aligned in solid solvents.
Intuitively, one might expect that perfectly aligned crystals would provide
ideal samples for LD spectroscopy. It is also true that both single crystals and
mixed crystals have been used for polarization spectroscopic purposes [2].
However, spectroscopic experiments with polarized light on crystalline samples
have often been difficult, for example, because very thin slices of single crystals
are required, especially for studies of electronic transitions. An alternative
possibility, to measure reflection spectra, leads to new complications. In
addition, interpretation of the spectra in terms of single molecular transitions
requires knowledge of the crystal structure, as well as of the often very strong
intermolecular interactions (Davydov splitting).
Instead, mixed crystal methods have sometimes been used. This requires that
a host crystal be found that does not absorb in the region of interest and in which
the molecule to be studied fits perfectly, without changing the crystal structure
696
In all two-photon experiments, new information may be obtained, both on
the sample structure and on the optical properties of individual molecules. While
the only information on the sample structure that may be provided by absorption
spectroscopy are average values over the sample molecules of cosine squares of
angles between transition moments and the sample axis, two-photon
spectroscopy may in addition provide averages of the fourth powers of such
angles. This is important information; one may say that while LD absorption
spectroscopy provides information on the average molecular alignment, the
addition knowledge from two-photon spectroscopy may provide information
about the spread of the molecular orientation distribution function around these
average alignments.
(or possibly changes it in a known fashion). The intermolecular interactions are
often quite limited in such mixed crystals, but it is frequently very difficult to
find suitable host crystals [2].
A very elegant way of orienting molecules, or rather of selecting an oriented
subsample is photo-orientation. For example, if a sample absorbs light, linearly
polarized along axis U, the subset that absorbs will have a symmetrical
orientation distribution of the absorbing transition moment M
i
around axis U,
with a probability of excitation that is proportional to cos
2
(M
i
, U)—assuming
that saturation effects are not important. The set of remaining molecules will also
be aligned; if the absorption experiment runs to near completion the alignment of
the remaining molecules may become extremely good (near perfect)—then only
molecules for which M
i
and U are nearly perpendicular will be left.
If the absorbing molecules just returned to their ground state, little would be
achieved. They must undergo a transformation (chemical reaction) in order to
produce a useful sample. Similarly, they must also be fixed in their positions; if
they were allowed to rotate, the alignment of the subset would be lost. Therefore
the samples are typically fixed in a rare gas matrix at low temperatures, as we
shall see in an example given below.
Also, electric or magnetic (static) fields may be used to align samples in
which the molecules are not completely fixed. Molecules with a permanent
electric dipole or an anisotropic electric polarizability may be aligned in strong
electric fields. Similarly, molecules with a permanent magnetic dipole or an
anisotropic electric susceptibility may be aligned in magnetic fields, although
this latter effect is usually very weak and the methods are primarily relevant for
large molecules. The importance of these techniques for solid samples is,
however, limited. See, for example the book by Fredericq and Houssier [10] and
the work by Liptay [11], the latter deals primarily with smaller molecules. In
recent years, alignment by strong laser pulses have become of interest [12], but
again the methods are of very limited interest in the present context.
16.2.2 Solutes in Partially Aligning Solvents
In the following, we shall concentrate on the production of aligned samples by
means of solid solvents with an ability to align solutes. In particular we shall
look at stretched polymers, mostly polyethylene, but nematic liquid crystals are
also able to align solutes. However, they are hardly solids and are thus a
borderline case for this discussion. However, they may in many ways be used in
the same way as stretched polymers, as we shall see in a later example.
The main properties of aligning solvents to be considered for use in
spectroscopy are related to their baseline absorption (or scattering), their ability
to dissolve the solutes to be studied, and their ability to align them. Also, other
properties are of interest, for example, the ability to withstand low temperatures
and thus to be used in low temperature work. Partial alignment of solutes in
stretched polymers, especially polyethylene and to some extent also
16.2 Occurrence, Production, and Optical Properties
697
16. Polarization Spectroscopy of Ordered Samples
poly(vinylalcohol), has been studied in great detail [2] and the discussion below
will most specifically refer to these two solvents.
It is extremely important that the aligning solvent (e.g., the stretched polymer)
not absorb or scatter light excessively in the spectral region to be studied. Both
polyethylene and poly(vinylalcohol) have good transmission properties in the
visible region and down to around 200 nm in the ultraviolet (UV) region. In
addition, polyethylene has the added advantage of being quite transparent in
most of the infrared (IR) region. The main exceptions are small regions around
720 cm
–1
, 1450 cm
–1
, and 2900 cm
–1
[2]. These regions may even be included in
the investigation, through the recording of spectra with per-deuterated
polyethylene as the aligning solvent (see, for example, [13]). For the deuterated
solvent, the corresponding absorption bands are located at lower wavenumbers;
thus, the regions blocked in spectra of normal polyethylene are visible. However,
commercially available per-deuterated polyethylene is expensive and for some
qualities, the production of high quality stretched sheets is not always simple.
Poly(vinylalcohol) has a much more complicated IR spectrum than
polyethylene, the baseline problems are considerable in this region, and it is
usually avoided in IR LD spectroscopy. Nevertheless, a few examples exist of
successful use of poly(vinylalcohol) in IR spectroscopy (see Holmén [14]).
The reason for the wish to use poly(vinylalcohol) is that it is able to dissolve
and align quite large amounts of many important polar molecules, in contrast to
polyethylene. It is also possible to obtain high concentrations in a
poly(vinylalcohol) sheet by mixing poly(vinylalcohol) powder with the solute in
a water-alcohol solution, casting the thick solution on a glass plate and letting the
solvent evaporate [2, 14]. In general, spectral intensity in absorption is roughly
proportional to the transition energy (wavenumber) of the transition. This means
that in the infrared region, where transition energies are about one tenth of those
in the UV-visible regions, a much larger amount of solute is required. This
makes polyethylene less well suited for studies of strongly polar molecules,
especially in the IR region.
Both polyethylene and poly(vinylalcohol) are able to align solutes sufficiently
for LD spectroscopy, unless these are near-spherical in shape. In particular, the
alignment of molecules in polyethylene has been studied in some detail [15–19].
very high crystallinity results in poor solubility for most solutes and that a very
low crystallinity reduces the aligning power of the stretched polymer. This is
understandable from LD IR measurements of the pure polyethylene sheet [18],
where molecules associated with the amorphous regions were shown to have
essentially no alignment (the transitions had K-values around 0.33,
corresponding to an isotropic sample, see below). In contrast, molecules
associated with crystalline regions turned out to be well aligned, although not
perfectly aligned (K-values between 0.60 and 0.90, see later) [18]. Based on
such measurements it has been concluded [15–19] that the most likely location
698
Polyethylene consists of crystalline and amorphous regions; it is known that a
16.3 One-Photon Spectroscopy: Linear Dichroism
of solute molecules in stretched polyethylene is the surface of lateral crystalline
regions.
It is important that the sample alignment be reasonably simple to handle in
terms of the mathematical treatment of the spectra. Thus, it is tempting to
assume that samples aligned in stretched polymers are uniaxial, i.e., that one
sample axis (U, the stretching direction for the polymer) is unique and all
directions perpendicular to U (including the other laboratory axes V and W) are
equivalent. This greatly simplifies the interpretation of the spectra. Uniaxiality
has been assumed without proof for many important samples. This has even
been done for solutes in thin stretched polyethylene sheets (with U as the
stretching direction, V as the other axis in the sheet plane, and W perpendicular
to the sheet), for which it may seem surprising that V and W are equivalent.
However, the assumption of uniaxiality has been studied in detail, both in the
case of undoped, thin stretched polyethylene sheets (by X-ray diffraction [15,
16]) and for solutes in stretched polyethylene (by the IR optical spectroscopic
methods mentioned above [18]), and has been shown to hold within
experimental accuracy. This is a major advantage in connection with the
mathematical treatment of the spectra.
16.3 ONE-PHOTON SPECTROSCOPY:
LINEAR
DICHROISM
16.3.1 Optical Spectroscopy with Linearly Polarized Light:
Experimental Needs
Linear dichroism absorption spectroscopy may be performed in the ultraviolet,
visible, near-infrared, infrared, and far-infrared spectral regions on the proper
ordinary spectroscopic instruments that have been equipped with an adjustable
linear polarizer or other polarizing element (see later) for the spectral region in
question. The only nontrivial instrumental demands exist in the infrared region
where the use of Fourier transform instruments in reality is necessary, but today
FTIR instruments have become both inexpensive and widely available. The light
polarization may be ensured by either a static polarizer or by means of a
polarization modulator. It may be added that measurements may even be
performed without polarizers by taking advantage of the fact the electric vectors
of natural light are not isotropic, but always perpendicular to the direction of the
light beam [20] (see later).
For the UV-visible region the most commonly used quality linear polarizers
Polaroid sunglasses or polarizers taken from discarded liquid crystal displays
(each contain two of such polarizers) may be used. For measurements around
699
sheet polarizers are available, in particular for the visible region, where even
have been calcite Glan prisms . They are efficient and transparent through-
out the visible region and down to about 215 nm. Much less expensive
16. Polarization Spectroscopy of Ordered Samples
200 nm and below more expensive and less convenient polarizers are required.
They may be of Rochon or Wollaston type and consist of magnesium fluoride or
quartz.
In the infrared region aluminum or gold grid polarizers have been widely
used, but today also higher quality sheet polarizers are available. Usually several
polarizers are required to cover the whole IR region.
In order to produce the two LD spectra either the sample or the polarizer must
be rotated. It is often most convenient to rotate the polarizer; however, if the
instrument in itself produces partially polarized light, as is often the case, the
polarizer must be rotated between positions that are equivalent with respect
the this polarization (e.g., be rotated from –45 to +45 degrees relative to the
vertical) in order to ensure identical light intensity for the two spectra. In the
light path, the polarizer may be placed either before or after the sample, but it
should not be separated from the sample by any other optical elements, if at all
possible, in order to avoid depolarization. However, both depolarization and
in the IR). For example, aligning solvents such as stretched polymers or liquid
crystals scatter UV light much more than most ordinary liquid solvents.
What is usually referred to as “baseline absorption” due to a stretched polymer
or liquid crystal should often really be called “baseline scattering.”
Many of the uniaxial samples used are birefringent; this means that the light
polarization will change when the beam passes the sample, unless it is polarized
either along or perpendicular to the sample axis U (or is progressing along U).
For this reason it is recommended that all spectra be recorded with light
polarized along U, V, or W. For a survey of actual experimental setups, with both
static polarization and modulated polarization, see [2].
16.3.2 Mathematical Descriptions of Aligned, Uniaxial Samples
Over the years, it has often been assumed that a full interpretation of LD spectra
of a partially aligned molecular sample requires a complete knowledge of the
orientation distribution function of the molecules in the sample. Fortunately this
is not so. Only those properties of the orientation distribution function that
influences the absorption probability are required.
Let us look at a single molecule in a uniaxial sample with sample axis U and
a transition in this molecule between states 0 and i caused by absorption of light
i
transition probability P
i
is proportional to (M
i
· İ)
2
or to
cos
2
(M
i
, İ), where
(M
i
, İ) is the angle between M
i
and İ. For a study of the
same transition in the
P
i
= ¢cos
2
(M
i
, İ)²
700
scattering within the sample is quite common in the UV region (but is much less so
polarized along İ transition moment M. We have already seen that the
the
is
whole sample we have to average over all sample molecules
and we
in a single molecule
obtain:
16.3 One-Photon Spectroscopy: Linear Dichroism
This quantity contains all the orientational information required to describe
the orientational dependence of the light absorption by the transition between
states 0 and i.
The interpretation of the LD spectra will be greatly facilitated if the light
polarization (İ) is chosen either along the sample axis U or perpendicular to it,
along laboratory axes V and W. Therefore, we will introduce the following short
notation for the quantities needed in former case:
¢cos
2
(M
i
, U)² = K
i.
Basic geometry tells us that for each individual molecule we have
cos
2
(M
i
, U) + cos
2
(M
i
, V) + cos
2
(M
i
, W) = 1.
This is thus also true for the averages:
¢cos
2
(M
i
, U)² + ¢cos
2
(M
i
,V)² + ¢cos
2
(M
i
,W)² = 1.
Since the sample is uniaxial the absorption probability for light polarized along
V
and W will be the same, and we obtain for the averages:
¢cos
2
(M
i
, V)² = ¢cos
2
(M
i
, W)² = (1–K
i
)/2.
In other words, all orientational information required for a description of the
relevant experiments that may be performed on the transition from the ground
state to state i is contained in the quantity K
i
. Note that if the sample has no
alignment (is isotropic) all K
i
will be equal to
1
3
. This description contains no
assumptions at all, except that the sample is uniaxial. It does not define the
orientation distribution function (except in the special case of perfect
alignment—K
i
being equal to 1), only aspects of it, but it contains exactly that
(minimum) information which is required to describe the orientational effects on
spectroscopic observations of transition i.
The literature on mathematical descriptions of partially aligned samples used
for LD spectroscopy has over the years demonstrated some unfortunate
approximations, or even mistakes; some of these led to incorrect conclusions [2].
Most of these mathematical models contain (unfortunately usually implicit)
assumptions about the orientation distribution of molecules in the sample. This
is, for example, the case for one of the early models, the Tanizaki model [21].
Tanizaki’s model is mathematically quite complicated. It produces a complete
orientation distribution function for stretched polymer sheets and solutes in it. It
was originally primarily applied for UV transitions in samples of stretched
poly(vinylalcohol) sheets and solutes in these; it was assumed that, at an infinite
stretch ratio, all molecules would be aligned similarly, with a specific molecular
axis (not necessarily the long axis) perfectly aligned with the stretching
direction, and that the orientation for actual stretch ratios would follow a specific
(and quite complicated) formula. A detailed analysis of the assumptions
contained in the Tanizaki model, when they are valid, and when they fail, may
be found in [2].
701

16. Polarization Spectroscopy of Ordered Samples
Fraser-Beer model it is simply assumed that that a fraction f of the sample
molecules are perfectly aligned and that orientation of the remaining fraction, 1 –
f, is perfectly isotropic. Although this orientation distribution is hardly ever
realistic, the Fraser-Beer model may still yield correct results for the description
of absorption experiments, but only as long as the molecules studied are aligned
like rods (see later). For other alignments than this very special case the model
will fail; see [2] for further details.
16.3.3 LD Spectra of Aligned, Uniaxial Samples
In the following, all the spectra are assumed to be corrected for baseline effects,
especially absorption and light scattering by the solvent (see later). We can now
write the observed solute absorbance in a spectroscopic absorption experiment
with light polarized along the sample axis U:
E
U
(ȞѺ) = Ȉ¢cos
2
(M
i
, U)²A
i
(ȞѺ) = Ȉ K
i
A
i
(ȞѺ) (16.1)
where the sum goes over all relevant transitions to states i and A
i
(ȞѺ) is equal to 3
times the contribution from transition i to the absorbance that would be observed
if the sample were isotropic, in other words, the absorbance that would be
observed if all transition moments M
i
were perfectly aligned with U.
Similarly we have for the observed absorbance when the light is polarized
perpendicularly to U (along V or W):
E
V
(ȞѺ) = E
W
(ȞѺ) = Ȉ ½(1 – K
i
)A
i
(
Ȟ
Ѻ). (16.2)
Now, K
j
for a nonoverlapping transition j at wavenumber Ȟ˾
j
may easily be
determined. We first define the dichroic ratio d (ȞѺ)
()
()
()
U
V
E
v
dv
E
v
and we obtain for the non-overlapping region of transition j:
()
.
2()
j
j
j
dv
K
dv
Spectral regions with only a single transition (nonoverlapping regions) are
common in IR spectroscopy, but much rarer in UV spectroscopy. However,
usually K
j
values may be determined quite accurately, also in the case of
overlapping transitions. The condition is that transition j may be recognized in
A simpler model that also attempts a determination of the complete
orientation distribution function is the Fraser-Beer model [22, 23]. It was
originally designed to describe the alignment of stretched polymers. However, it
has frequently been applied also to solutes in stretched polymers. In the
702

16.3 One-Photon Spectroscopy: Linear Dichroism
For a given value of x, x
j
, the spectral feature that is due to transition j, will
disappear. We now have, according to equations (16.1) and (16.2):
K
j
= x
j
1
2
j
K
and thus
K
j
=
2
j
j
x
x
.
In some cases it is not E
U
(ȞѺ) and E
V
(ȞѺ) that are measured, but related
quantities, such as E
U
(ȞѺ) or E
V
(ȞѺ) and E
iso
(ȞѺ) measured on an isotropic sample
[2], or E
U
(ȞѺ) or E
V
(ȞѺ) and E
U,V
(ȞѺ), where E
U,V
(ȞѺ) is measured with natural light
on the aligned sample (electric vectors along U and V ).
In instruments where the polarization may be modulated, e.g., by a
photoelastic modulator one may obtain the linear dichroism LD directly:
LD(ȞѺ) = E
U
(ȞѺ) – E
V
(ȞѺ).
The observed LD(ȞѺ) may then be combined with E
U,V
(ȞѺ) (the absorbance of
the aligned sample measured with natural light) in order to obtain the available
orientational information. In particular, direct measurement of LD(ȞѺ) may
provide a significantly better accuracy when the difference between E
U
(ȞѺ) and
E
V
(ȞѺ)—the linear dichroism—is very small and baseline inaccuracy becomes
significant.
Finally, as a low cost solution, since especially good IR polarizers are
expensive, it may be mentioned that similar information may be obtained
without polarizers. The fact that the electric vectors of a light beam are not
isotropic, but located in only two dimensions, perpendicular to the direction of
the beam, may be used for “polarizer-free polarization spectroscopy” [20].
Spectra measured with natural light, first on an aligned sample, then on an
isotropic sample of the same molecule, reveal a similar difference, although less
pronounced, as ordinary LD spectra. The two samples may be a squeezed
polyethylene tablet and an unstretched polyethylene sheet. This technique also
makes a distinction between different transition moment directions possible, at
least for somewhat elongated molecules.
method (the TEM method [2, 24, 25]) may be used. In this highly efficient
method, linear combinations of the observed spectra E
U
(ȞѺ) and E
V
(ȞѺ) are
formed:
E
U
(ȞѺ) – xE
V
(ȞѺ).
7 0 3
the LD spectra as a spectral feature (peak or shoulder). If so, the trial and error
