Vij D.R. Handbook of Applied Solid State Spectroscopy
Подождите немного. Документ загружается.


16. Polarization Spectroscopy of Ordered Samples
While it is possible to determine any number of different K-values from the
two observed spectra, a separation of the observed absorbance into contributions
from different transition moment directions (a determination of the A
i
(ȞѺ) curves)
is much more complicated. This separation is particularly useful for UV spectra
where it, among other, may help reveal otherwise “hidden” transitions (usually
hidden due to overlap with other, stronger transitions). If only y- and z-polarized
intensity is present in a spectral region (A
x
(ȞѺ) = 0), we have for that region:
A
z
(ȞѺ) =
(1 ) ( ) 2 ( )
YU YV
ZY
KE KE
KK
(16.3)
A
y
(ȞѺ) =
2()(1)()
ZV Z U
ZY
KE K E
KK
(16.4)
We see that in a spectral region with intensity exclusively from transitions
polarized along the y- and z-axes, the two observed spectra E
U
(ȞѺ) and E
V
(ȞѺ)
provide sufficient information for a mathematical construction of A
y
(ȞѺ) and A
z
(ȞѺ),
the sums of transitions polarized along molecular axes y and z, respectively.
Construction of spectral functions like A
y
(ȞѺ) and A
z
(ȞѺ) is only possible in some
special cases. Even in symmetrical molecules, three different transition moment
directions (along x, y, z) will be present in the general case and the wanted three
spectral functions A
x
(ȞѺ), A
y
(ȞѺ), and A
z
(ȞѺ) cannot be constructed since only two
observed spectra are available.
In vibrational spectroscopy of symmetrical molecules, where all three
transition moment directions usually are present, a separation into A
x
(ȞѺ), A
y
(ȞѺ),
and A
z
(ȞѺ) is usually not considered necessary due to the very limited extent of
overlap between different IR transitions. However, as shown below in an
example, it is possible to produce spectral curves in which contributions from
any of the three transition moment directions have been removed, that is,
contributions from either A
x
(ȞѺ), A
y
(ȞѺ), or A
z
(ȞѺ). This is often a useful option,
although the construction of A
x
(ȞѺ), A
y
(ȞѺ), or A
z
(ȞѺ), whenever possible, is the best
option, especially in the case of the strongly overlapping UV transitions, as we
shall see later.
16.3.4 Transition Moment Directions and Reduced Spectra:
Symmetrical Molecules
The situation is particularly simple for molecules of C
2v
, D
2h
, and D
2
symmetry
(here labeled “symmetrical’’ cases, fortunately this group includes numerous
important molecules) for which only three different transition moment directions
are possible (along the symmetry-determined molecular axes x, y, and z). Thus
also only three different K
i
may be observed. The three K are not independent,
but (by definition) add up to 1
K
x
+ K
y
+ K
z
= 1.
.
704
Ȟ
Ȟ
Ȟ
Ȟ
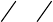
16.3 One-Photon Spectroscopy: Linear Dichroism
the high symmetry molecular axis, and another with transition moments
perpendicular to the axis. This is all the information that is available and all that
An interesting case is that of molecules which for reasons other than
symmetry, are either aligned like “rods’’ (the two smaller K are identical) or like
‘‘disks’’ (the two larger K are identical) [2]. For these molecules the absorption
may also be separated into two parts, one with transition moments along the
molecular axis with the unique K value and another with transition moments
perpendicular to this axis. For a rod-like aligned symmetrical molecule this
would mean that, for example, A
z
(ȞѺ) and A
x
(ȞѺ) + A
y
(ȞѺ) may be determined.
x
Ѻ
y
Ѻ
16.3.4.1 The Orientation Triangle
If we define the K so that K
x
d K
y
d K
z
, keeping in mind that K
x
+ K
y
+ K
z
= 1, a
plot of, e.g., (K
y
, K
z
), in this case for planar, symmetrical molecules (see below),
will all be located within a triangle with corners (K
y
, K
z
) = (0, 1), (
1
3
,
1
3
), and
(½, ½); see Figure 16.6. The sides of the triangle are defined by:
K
z
+ K
y
= 1
This side corresponds to K
x
= 0; in other words, the (y, z)-plane is parallel to
the sample U-axis:
K
z
= K
y
Points on this side have a rod-like alignment with an equal (but not
necessarily identical) alignment of the two least aligned molecular dimensions (x
and y):
K
x
= K
y
or K
z
= 1 – 2K
y
.
Points located on the last side of the triangle correspond to a disk-like
alignment with an equal (but not necessarily identical) alignment of the two most
well-aligned molecular dimensions (y and z).
This orientation triangle provides a convenient overview of molecular
alignment in stretched polymer sheets. By inspection of the figure it is clear that
alignment is closely related to molecular shape, and it is actually possible to
in or near the vacuum UV region. The benefits of a separation of spectral
intensity according to the two in-plane transition moments have been
demonstrated numerous times (see, for example, [2, 15, 23, 24]), especially for
UV spectra with strongly overlapping bands.
In a few other cases the molecular alignment is such that only two different K
exist. This is true for molecules that have a C
3
or higher axis. For such molecules
the absorption may be separated in two parts, one with transition moments along
Spectral regions with only two different transition moment directions are
common for “symmetrical’’ planar, polycyclic hydrocarbons, for which the low-
energy transitions are dominated by the in-plane polarized S-S* transitions (see
below). For these molecules, out-of-plane polarized intensity usually only occurs
705
However, a separation of A (Ȟ) and A (Ȟ) is not possible.
is required for this kind of experiment.
16. Polarization Spectroscopy of Ordered Samples
Figure 16.6 The orientation aromatic molecules in stretched polyethylene at room temperature.
Reproduced from [25].
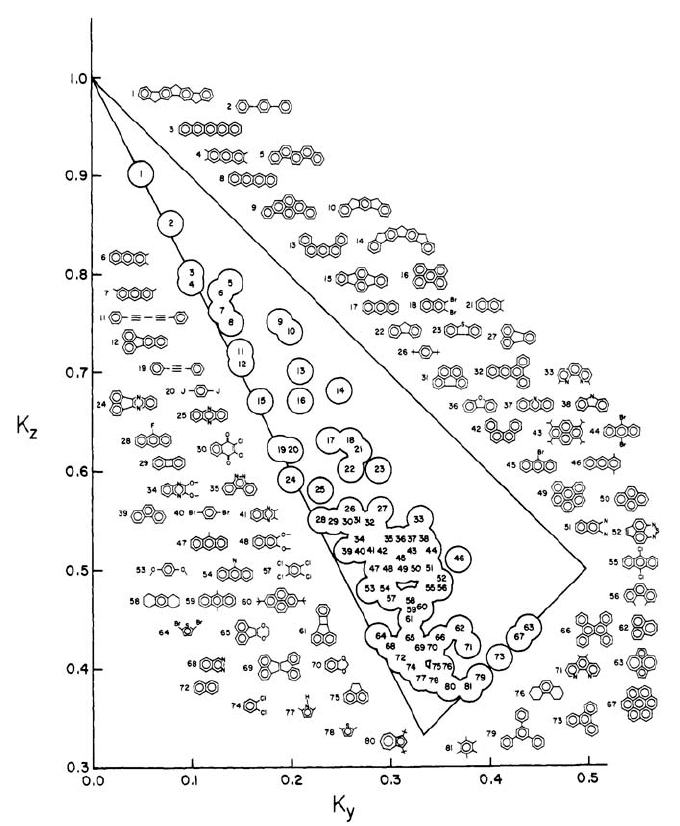
Example 1: The IR Spectrum of 9,10-Anthraquinone
In connection with a study of colored anthraquinone derivatives of interest in
liquid crystal displays, an IR spectrum of the parent compound in stretched
polyethylene sheets were recorded, mainly to check the out-of-plane axis (x)
alignment independently [26]. It turned out that this simple spectrum was able to
solve a few long-time problems within the assignment of the vibrational
transitions of 9,10-anthraquinone. 9,10-anthraquinone is a molecule of D
2h
make a reasonable guess on the K values for a new compound based on
inspection of the known points in the triangle.
706
16.3 One-Photon Spectroscopy: Linear Dichroism
transitions can only be directed along the three symmetry-determined
axes, x, y,
U
V
Figure 16.7 FT-IR LD spectra of 9,10-anthraquinone in stretched polyethylene with an
indication of the assignment of transition moment directions of individual bands. Reproduced
from [26].
Using the TEM method, the K values for a little more than a dozen peaks and
a single shoulder were easily determined. As expected, only three different K-
values were registered: K
x
= 0.15, K
y
= 0.25, and K
z
= 0.59, with a sum of 0.99,
in other words equal to 1 within the experimental accuracy, as they should be.
These K-values immediately provide symmetry assignments for the transitions,
those with K = 0.15 (out-of-plane, x) are b
3u
, those with K = 0.25 (short, in-plane
direction, y) are b
1u
, and those with K = 0.59 (long axis, z) are of b
2u
symmetry.
The transitions that were observed between 600 and 2000 cm
–1
are only about
half of the total number of allowed transitions; of the 66 (from 24 u 3 – 6)
fundamental vibrations, 28 are allowed (11 b
1u
, 11 b
2u
, and 6 b
3u
). The remaining
38 transitions of other symmetries cannot be observed in a simple absorption
experiment; they are symmetry forbidden.
One unexpected bonus was the information about the identical b
2u
symmetry
(and long-axis, z-, polarization) of the members of the quartet of peaks around
1300 cm
–1
. This quartet had never been properly assigned to z-polarized
707
vibrational
symmetry (see Fig. 16.4). Therefore, all transition moments for both electronic and
the mid-IR region and measured at room temperature, are shown in Figure 16.7.
and z. The two baseline corrected spectra, E (
v) and E (v ) covering
16. Polarization Spectroscopy of Ordered Samples
transitions. Now it became clear that only the outer two transitions in the quartet
are b
2u
fundamentals (Q
44
at 1284 cm
–1
and Q
43
at 1329 cm
–1
). The two center
peaks are combination bands of Q
10
+ Q
48
and Q
12
+ Q
47
(both a
g
+ b
2u
combinations of b
2u
symmetry) that gain unexpected strong intensity through
Fermi resonance caused by the two intense surrounding fundamentals.
In addition to the highly useful information about the z-polarized quartet, a
second new and valuable piece of information appeared. From the E
U
spectrum
in Figure 16.7 it is clear that the peak around 700 cm
–1
is really a double peak.
This is not obvious from the E
V
spectrum or from an ordinary solution spectrum.
The reason for the double peak is the occurrence at almost the same
wavenumber of both a strong b
3u
transition (Q
63
at 699 cm
–1
) and a weak b
2u
fundamental (Q
48
at 693 cm
–1
). The latter had never been observed before
because of the overlap with the stronger Q
63
, and various incorrect assignments
had been attempted. The reason for the observation of the double peak in E
U
, and
not in the other spectra, is that long axis polarized transitions are amplified in the
E
U
spectrum; similarly short axis polarized transitions are amplified in E
V
.
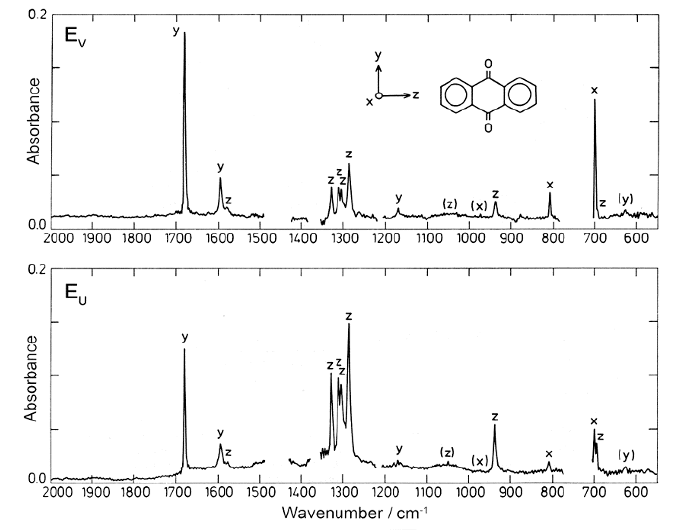
Example 2: The UV Spectrum of the Pyrene Molecule
As an example, let us look at the UV spectrum of the important, symmetrical
pyrene molecule [2, 27]. The low energy part of the electronic spectrum of this
conjugated, planar molecule is quite well described in a S-S* model, where
excitation only leads to changes in the orbitals of S-type; these are the molecular
orbitals that are antisymmetric with respect to the molecular plane. This
symmetry property restricts the transition moments for singlet-singlet S-S*
transitions to the molecular plane. Furthermore, the D
2h
symmetry of pyrene only
allows two transition moment directions in the molecular plane, along the
symmetry axes z (the long axis) and y (the short in-plane axis). Other transitions,
for example V-S*, will have transition moments perpendicular to the molecular
plane. But for the low energy transitions it is safe to assume that only in-plane
transition moments are of interest.
In a simple MO picture, the first four singlet-singlet transitions of pyrene may
be described as transitions to excited electron configurations. These four
configurations correspond to a single electron moving from one of the two
highest occupied molecular orbitals (1 and 2) into one of the two lowest
unoccupied orbitals (–1 and –2), in other words:
|1 –1², |1 –2², |2 –1², |2 –2²
In the simple MO model the configurations |1 –2² and |2 –1² have the
same energy (are degenerate) and will be poor approximations to the excited
states. The + and – combinations of the two are much closer to reality. Thus we
end up with the following simple description of the four excited states (for a
discussion of the labeling that originally is due to Platt; see [2]):
708
o
o
o
o
oo
16.3 One-Photon Spectroscopy: Linear Dichroism
L
a
= |1 –1²,
L
b
= |1 –2² – |2 –1²,
B
b
= |1 –2² + |2 –1²,
B
a
= |2 –2²
In the case of pyrene, the singlet L
b
state turns out to have lower energy than
the singlet L
a
state. It also corresponds to an extremely weak transition, while the
other three are strong. The transition to L
b
is only seen because it appears at
slightly lower energy than the transition to L
a
. The transition moments to a-states
are all along the molecular z-axis (they go through atoms) while transition
moments to b-states are along the molecular y-axis (they go through bonds). It
may be added that the b-states correspond to molecular symmetry B
2u
, while the
1u
This picture is confirmed by the observed K-values of 0-0 transitions
corresponding to the three strong transitions: K(B
b
) = K
y
= 0.33, K(L
a
) = K(B
a
) =
z
may conclude that K
x
= 0.09. However, in the case of pyrene there is a rare
exception that has to do with the very weak L
b
transition, and the fact that it is
a
Figure 16.8 Polarized UV absorption of pyrene in stretched polyethylene at 77 K. Bottom:
Reduced absorption spectra A
z
= E
U
– 1.0E
V
and A
y
= 0.625(2.8E
V
– E
U
). Top: Baseline-corrected
polarized absorption spectra and the dichroic ratio E
U
/E
V
. The absorbance scale is in arbitrary units,
different for the different sections of the spectrum. Note the change in the horizontal scale in the left
section. Reproduced from [2].
709
o
o
o
o
o
o
we
K = 0.58; see Figure 16.8. Furthermore, because of the sum rule for the K- values
a-states correspond to B
located very close to L .
(Fig. 16.4).
16. Polarization Spectroscopy of Ordered Samples
A slight perturbation of the molecular structure and symmetry, e.g., from a
solvent, and even more from a substituent that destroys the symmetry, may result
in an excited state that is a mixture of two close-lying states. This happens
regularly for the L
b
and L
a
states of conjugated molecules. If one of the two
unperturbed transitions, say the L
a
transition, is much stronger than the L
b
transition, it may dominate the transition moment for a resulting transition, and
the transition moment will turn from the y-axis to a position closer to the z-axis.
Because of the large difference in the original intensities, even a small dose of L
a
mixed into L
b
will rotate the transition moment significantly.
A remarkable example of this solvent effect has been observed for pyrene,
aligned in stretched polyethylene [2, 28]. The low symmetry interaction with the
polymer solvent is able to mix a small amount of L
a
into L
b
. The result is that
the transition moment for the 0-0 transition of L
b
is located almost in the middle
between the y- and z-axes, and not along y as expected according to
the molecular symmetry. This is demonstrated by the observed K-value for the
0-0 transition that is neither 0.33, nor 0.58, but between the two values, actually
corresponding to a transition moment angle of around 37° with the y-axis.
It may be added that the complicated picture of vibronic peaks between the L
b
0-0 transition and L
a
are quite dominated by z-polarized intensity, gained
through vibronic mixing of the two states through non-totally symmetric
vibrations of B
3g
symmetry. It is said that the L
b
transition “borrows” or “steals”
intensity from L
a
. This is a common phenomenon in such spectra, and LD
spectroscopy is particularly well suited to study these processes. However, the
twisting of a transition moment for the (L
b
) 0-0 transition due to solvent effects
is very rare. The best known other case of this kind is the appearance of the 0-0
band of the symmetry forbidden first singlet-singlet transition (to B
2u
) in benzene
in various solvents (it is not seen in the gas phase).
For the overall analysis of the pyrene UV spectrum, the TEM procedure is
required because of the extensive spectral overlap between two or more
transitions. The TEM treatment results in the expected K-values for the other 0-0
peaks, as indicated above. The case of symmetrical S-electron systems in
spectral regions with only in-plane polarized S-S* transitions makes a very
detailed analysis possible, as discussed above.
With only intensity from transitions polarized along the y- and z-axes, the two
observed spectra
()
U
E and ()
V
E
provide sufficient information for a
mathematical construction of A
y ()
and A
z ()
, the sums of transitions polarized
along molecular axes y and z, respectively. Equations (16.3) and (16.4) provide
the results that are shown in the bottom of Figure 16.8.
Q
Q
Q
Q
710
16.3 One-Photon Spectroscopy: Linear Dichroism
We immediately notice that there is a medium-strong z-polarized transition
around 38,000 cm
–1
and one or more y-polarized transitions under the strong B
a
band. These correspond to excited states with large contributions from other
excited configurations than those above, involving only the two highest occupied
and the two lowest unoccupied molecular orbitals. For a test of theoretical
models such information is of considerable interest and LD spectroscopy has
been widely used in this connection; see [27, 29, 30].
In the general case of symmetrical molecules, the LD spectra will reveal three
different K: K
x
, K
y
, and K
z
. In regions with little overlap (as in IR spectra) this
usually allows a simple interpretation, in particular assignment of transitions to
specific symmetry classes. In the case of spectral overlap by two differently
polarized transitions, the situation may often be handled as shown above. But in
the case of spectral overlap by three differently polarized transitions, the
interpretation is usually not simple.
Example 3: A Comparison of LD Spectra with Stretched Polymers and Liquid
Crystals as the Aligning Solvent
The infrared spectrum of fluorene has been studied by LD spectroscopy, using
both a stretched polymer (polyethylene) and a nematic liquid crystal (4-cyano-
4’-alkylbicyclohexyl mixture) as the aligning solvent [31]. Fluorene is a
symmetrical molecule, point group C
2v
; it has 63 normal modes: 22a
1
+ 10a
2
+
11b
1
+ 20b
2
. Of these, the 10 a
2
vibrations are symmetry-forbidden and cannot
be observed in absorption spectra.
The LD spectra, measured at room temperature in stretched polyethylene, are
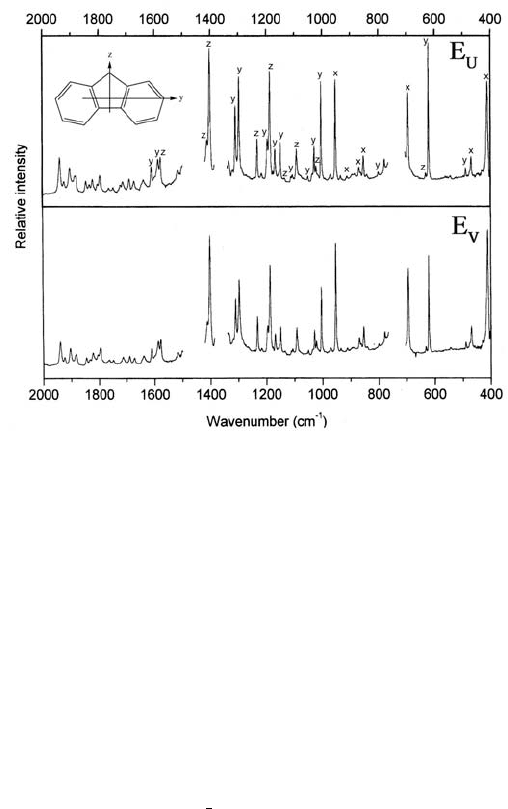
shown in Figure 16.9. The Figure shows the assignments that are revealed
almost instantly, by inspection: A peak with E
U
>> E
V
corresponds to a long axis
(y) polarized transition, one with E
U
| E
V
to a short in-plane axis (z) polarized
transition, and a peak with E
U
<< E
V
corresponds to an out-of-plane (x) polarized
transition.
The K-values obtained are (K
x
, K
y
, K
z
) = (0.17, 0.49, 0.31) with polyethylene
as the aligning solvent and (0.11, 0.64, 0.25) with the nematic liquid crystal as
the aligning force. It may be added that in an earlier experiment [2], performed
in the UV region and with a lower solute concentration in a thin polyethylene
sheet, the K were measured to be (0.14, 0.60, 0.26). Still, the fluorene molecules
in the nematic liquid crystal demonstrate the best alignment of all. However, the
spectra obtained with polyethylene as solvent are sharper and show more details.
711
16. Polarization Spectroscopy of Ordered Samples
Figure 16.9 LD absorbance spectra of fluorene in stretched polyethylene recorded at room
temperature. Reproduced with permission from [31].
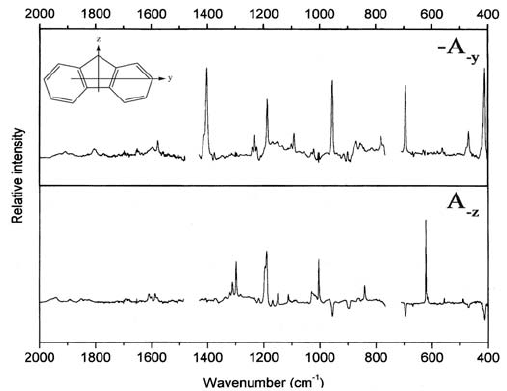
In Figure 16.10 the spectra with the nematic liquid crystal as the aligning solvent
are shown in a format that even more directly reveals the transition moment
directions.
Although individual curves A
x
, A
y
, and A
z
cannot be constructed from only
two observed spectra, it is possible to construct curves in which either A
x
, A
y
, or
A
z
is removed! If we want to remove all contributions from y-polarized
transitions, A
y ()
, we have to form the linear combination:
A
–y
() = (1 – K
y
) E
U
() – 2K
y
E
V
() = (1 – K
y
) 6K
i
A
i
() – 2K
y
(½)6(1 – K
i
)A()
= (K
x
– K
y
)A
x
() + (K
z
– K
y
)A
z
().
The curve shown in the top part of Figure 16.10 is equal to –A
–y
() (minus
is used because both coefficients to the two A
i
()
; are negative) and is thus a
linear combination of only A
x
(); and A
z
();. Note that the two contributions
occur with different coefficients (unless K
z
= K
x
—a less interesting case of
isotropic-like alignment). In other words, the contributions from transitions
Q
Q
Q
Q
Q
Q
Q
Q
Q
Q
Q
712
Q
16.3 One-Photon Spectroscopy: Linear Dichroism
Figure 16.10 Linear combinations of absorbance spectra of fluorene at room temperature,
aligned in a nematic liquid crystal. Top: –A
–y ()
; bottom A
–z ()
. See text. Figure adapted by
permission from [31].
polarized along different axes are not necessarily on the same scale! This is
even clearer from the bottom of Figure 16.10, in which all z-polarized
intensity has been removed:
A
-z
() = (1– K
z
) E
U
() – 2K
z
E
V
()
= (K
x
– K
z
)A
x
()
+ (K
y
– K
z
)A
y
()
Here, y-polarized intensity occurs with a positive sign (the coefficient on
A
y ()
is positive), while x-polarized intensity appears with a negative sign. This
may easily be seen from a comparison with Figure 16.9. The removal of spectral
intensity with a given transition moment direction may be applied to any set of
LD spectra, and is commonly used also for spectra obtained from stretched
polymer samples.
From the information obtained on transition moment directions and excited
state symmetries it was possible to correct several mistakes in earlier
assignments of the vibrational transitions of fluorene [31].
Example 4: Photoinduced Dichroism in E-Thioxoketones in a Frozen Matrix
Photoinduced dichroism can be observed when a photochemical reaction is
made to occur through a polarized light source on an immobilized group of
Q
Q
Q
Q
Q
Q
Q
Q
713
