Watts J.F., Wolstenholme J. An Introduction to Surface Analysis by XPS and AES
Подождите немного. Документ загружается.


X-RAY
ANALYSIS
IN THE
ELECTRON MICROSCOPE
j 57
LAMMS
laser ablation microprobe mass spectrometry
RBS
Rutherford backscattering spectrometry
STEM scanning transmission electron microscopy
SIMS
secondary
ion
mass spectrometry
TEM
transmission electron microscopy
6.1
X-ray
Analysis
in the
Electron
Microscope
The
addition
of an
X-ray analyser
(either
energy
or
wavelength disper-
sive,
EDX or
WDX)
to a
scanning
electron
microscope
provides
an
electron probe microanalysis
facility
widely used
in all
branches
of re-
search
and
development. This provides
a
very
flexible
means
of
micro-
analysis
and,
in the
conventional
EDX
mode, elements
from
beryllium
onwards
can be
detected; with
a WDX
spectrometer,
resolution
and
sensitivity
are
improved.
Recent
advances
in EDX
detectors have extended
the
range
to
much
lighter elements,
but the
vacuum requirements
are
more
stringent
to
prevent
icing
up of the
detector. However, although
it has now
become
possible
to
undertake light-element analysis
by the
consideration
of
emitted characteristic X-rays, such analyses
are
essentially probing bulk
composition.
The
'interaction
volume'
of the
electron beam
with
the
sample
determines both
the
lateral resolution
(in the
analytical mode)
and the
depth
of
analysis; this
is a
function
of
primary beam energy
but
will invariably
be of the
order
of a
micrometer.
By
reducing beam
en-
ergy,
the
depth
of
analysis
may be
reduced
to as
little
as 500 nm but
there
is a
limit
to
this approach; although
an
electron beam
of 1 keV
will
only
have
a
small penetration depth
it
will
not
excite X-rays
of
analyt-
ical
use in the
conventional sense.
The
characteristic electron
inelastic
mean
free
path
for
electrons
of
analytical
use in
electron spectroscopy
is
compared with
that
for the
higher energies used
as
primary radiation
in
the
electron
microscope
in
Figure 6.1. Thus
the
analysis
depth
in
X-ray
analysis
is
determined
by the
energy
of the
primary radiation (the elec-
tron
beam)
whereas
in
electron spectroscopy
it is the
energy
of the
secondary radiation (emitted electrons) which controls this parameter.

168
COMPARISON
OF XPS AND AES
WITH
OTHER
TECHNIQUES
100
10
LL!
0
(b)
1
10 100
Electron
energy
(eV)
1000
0 10 20 30 40 50
Electron
energy
(keV)
Figure
6.1
Electron mean
free
paths
of
energies used
in
electron spectroscopy (a),
and of the
primary beam energies used
in
electron microscopy
(b)
500
1000
Photon
energy
(eV)
Figure
6.2
Light-element
analysis
(the
sample
is
oxidized
boron)
in the TEM by EDX

X-RAY ANALYSIS
IN THE
ELECTRON MICROSCOPE
In
the
TEM,
the use of a
thin
foil
specimen immediately
defines
the
depth
of
analysis,
as the
pear-shaped interaction volume
is
abruptly
truncated. This dramatically improves spatial
resolution,
although there
will
be
some degradation
of
spatial resolution
as a
result
of
electron
beam
interaction with
the
sample.
An EDX
spectrum
of
pure boron
Figure
6.3 A
modern
Auger
electron
spectrometer
fitted
with
an
energy
dispersive
X-ray
detector

170
COMPARISON
OF XPS AND
AES
WITH
OTHER
TECHNIQUES
(with
a
thin oxide
film
present), obtained
in a
TEM,
is
shown
in
Figure 6.2;
the BKa
line
is
very clear
in the
spectrum.
In
the
scanning Auger microscope
the
analysis depth
is
determined
by
the
energy
of the
outgoing electrons
as
described
in
Chapter
1 and the
spatial resolution depends
on the
size
of the
electron
probe.
Thus,
the
spatial resolution attainable with
SAM can be an
order
of
magnitude
better than that recorded with SEM/EDX. With
the
development
of
sub-micron features in microelectronics SAM is finding a new use: as a
high-resolution chemical imaging facility,
the
emphasis
no
longer being
on the
need
for a
surface
analysis.
Although X-rays
do
show
a
small chemical
shift
with oxidation
state,
this feature
is not
employed
in
analytical X-ray analysis
of the
type used
in
electron microscopes; thus
EDX
only provides elemental information
unlike
the
additional chemical information provided
by XPS and
AES.
The
addition
of an EDX
facility
to a
surface analysis spectrometer
is
worthy
of
consideration.
In the SAM
mode
it is
possible
to
acquire both
Auger
(surface)
and
X-ray
(bulk)
chemical maps
of the
specimen.
A
typical set-up
is
shown
in
Figure 6.3.
In
conjunction with XPS,
an
X-ray detector
is
able
to
provide good
quality fluorescence
spectra
(XRF)
to
within about
2 keV of the
X-ray
source operating potential.
As
this will usually
be
around
15
keV, X-ray
spectra
up to 13 keV can be
obtained. This method
is
particularly
useful
for
insulating specimens
not
amenable
to
analysis
by
AES/EDX such
as
paint
films or
some catalysts.
XRF is
also
useful
in XPS
depth
profiling
where
a
global analysis
of
specimen chemistry
can be
achieved before
segregation
or
interfacial
effects
are
studied
in
detail.
6.2
Electron
Analysis
in the
Electron
Microscope
As
well
as the
possibility
of
X-ray analysis
in the TEM and
STEM,
it is
possible
to
analyse
the
energy
of the
transmitted beam which
is the
basis
of
electron energy-loss spectroscopy
(EELS).
As an
electron beam passes
through
an
electron transparent specimen,
it is
able
to
eject electrons
whose binding energies
are
less than that
of the
primary
beam
energy.
By
recording
the
depletion
of the
primary beam energy with
an
electron
spectrometer positioned below
the
specimen,
an
energy-loss spectrum
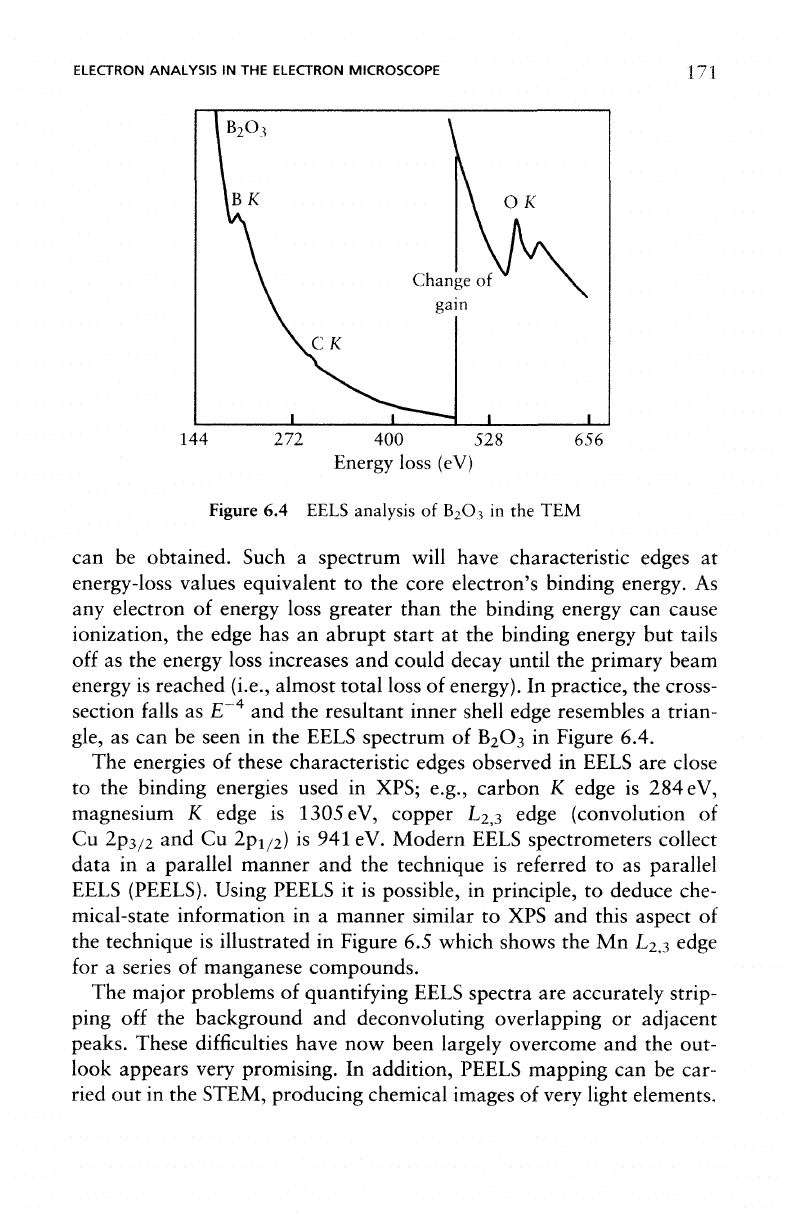
ELECTRON ANALYSIS
IN THE
ELECTRON
MICROSCOPE
144
272
400 528
Energy
loss
(eV)
Figure
6.4
EELS
analysis
of
B
2
O
3
in the TEM
can be
obtained. Such
a
spectrum will have characteristic edges
at
energy-loss values equivalent
to the
core electron's binding energy.
As
any
electron
of
energy loss greater than
the
binding energy
can
cause
ionization,
the
edge
has an
abrupt start
at the
binding energy
but
tails
off
as the
energy loss increases
and
could decay until
the
primary beam
energy
is
reached (i.e., almost
total
loss
of
energy).
In
practice,
the
cross-
section
falls
as E
-4
and the
resultant inner shell edge resembles
a
trian-
gle,
as can be
seen
in the
EELS spectrum
of
B
2
O
3
in
Figure 6.4.
The
energies
of
these characteristic edges observed
in
EELS
are
close
to the
binding energies used
in
XPS; e.g., carbon
K
edge
is 284 eV,
magnesium
K
edge
is
1305eV,
copper L
2,3
edge (convolution
of
Cu
2p
3/2
and Cu
2p
1/2
)
is 941 eV.
Modern
EELS spectrometers collect
data
in a
parallel manner
and the
technique
is
referred
to as
parallel
EELS
(PEELS).
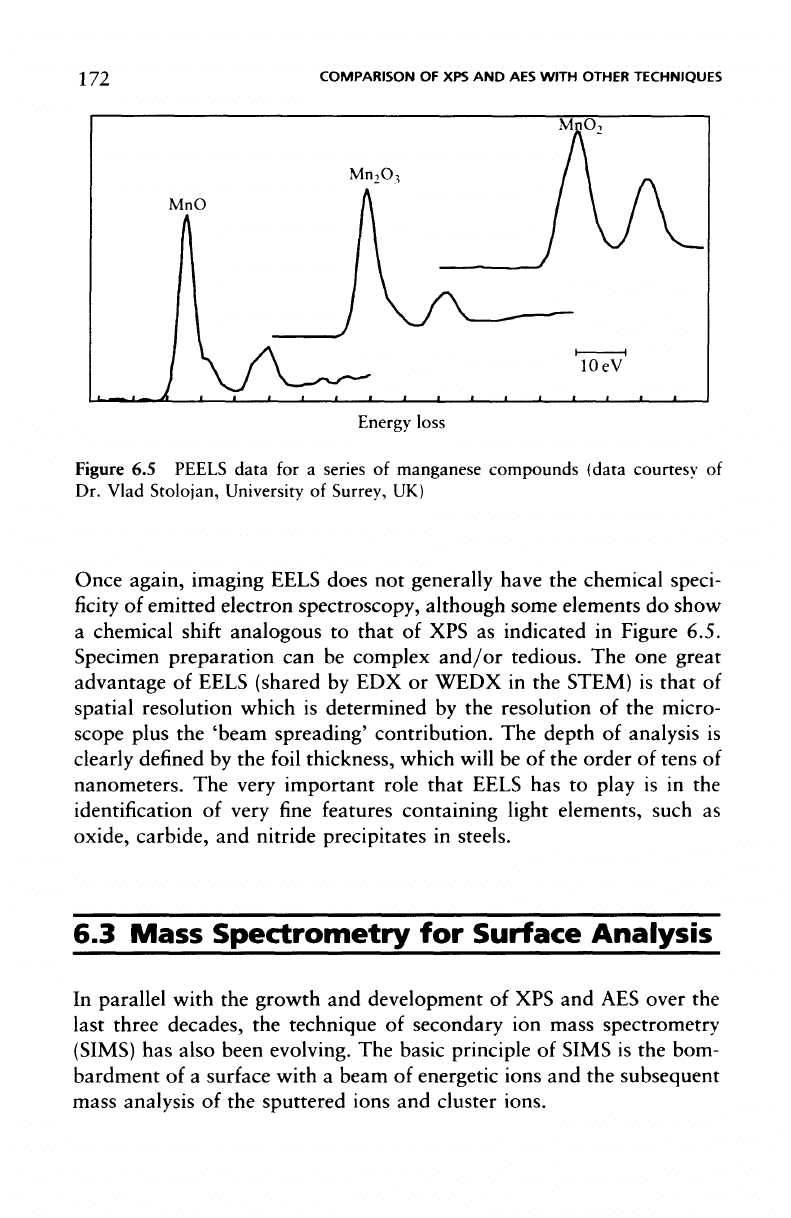
Using PEELS
it is
possible,
in
principle,
to
deduce che-
mical-state information
in a
manner similar
to XPS and
this aspect
of
the
technique
is
illustrated
in
Figure
6.5
which shows
the Mn
L
2
,
3
edge
for
a
series
of
manganese
compounds.
The
major problems
of
quantifying
EELS
spectra
are
accurately strip-
ping
off the
background
and
deconvoluting overlapping
or
adjacent
peaks. These
difficulties
have
now
been largely overcome
and the
out-
look appears very promising.
In
addition, PEELS mapping
can be
car-
ried
out in the
STEM, producing chemical images
of
very light elements.
172
COMPARISON
OF XPS AND AES
WITH
OTHER
TECHNIQUES
MnO,
MnO
Energy
loss
Figure
6.5
PEELS data
for a
series
of
manganese compounds
(data
courtesy
of
Dr.
Vlad Stolojan, University
of
Surrey,
UK)
Once again, imaging EELS does
not
generally have
the
chemical speci-
ficity
of
emitted
electron
spectroscopy,
although some elements
do
show
a
chemical
shift
analogous
to
that
of XPS as
indicated
in
Figure 6.5.
Specimen
preparation
can be
complex
and/or
tedious.
The one
great
advantage
of
EELS (shared
by EDX or
WEDX
in the
STEM)
is
that
of
spatial resolution which
is
determined
by the
resolution
of the
micro-
scope plus
the
'beam spreading' contribution.
The
depth
of
analysis
is
clearly
defined
by the
foil
thickness, which
will
be of the
order
of
tens
of
nanometers.
The
very important role that
EELS
has to
play
is in the
identification
of
very
fine
features containing light elements, such
as
oxide, carbide,
and
nitride precipitates
in
steels.
6.3
Mass
Spectrometry
for
Surface
Analysis
In
parallel with
the
growth
and
development
of XPS and AES
over
the
last three decades,
the
technique
of
secondary
ion
mass spectrometry
(SIMS)
has
also been evolving.
The
basic principle
of
SIMS
is the
bom-
bardment
of a
surface with
a
beam
of
energetic ions
and the
subsequent
mass
analysis
of the
sputtered ions
and
cluster ions.

MASS SPECTROMETRY
FOR
SURFACE ANALYSIS
173
The
strengths
of
this method are:
• all
elements including hydrogen
can be
detected,
• as the
analysis
is
based
on the
mass
separation
of the
secondary
particles,
it has
both isotopic
and
molecular
specificity,
• it has
much higher elemental sensitivity than either
XPS or
AES.
The
major
disadvantage
of
SIMS
is
that
quantification
of the
data
is
much
more
difficult
than
it is
with
the
electron
spectroscopies.
There
are
three modes
of
operation
in
SIMS.
1.
Static SIMS
(SSIMS)
is
where
the low ion flux (
<10
12
ions cm
-2
per
analysis)
ensures
that
the
original surface
is
insignificantly damaged
during
the
analysis. SSIMS
can be
applied
to
polymers
and
other
insulating
specimens with
a
great deal
of
success.
A
typical static SIMS
spectrum,
in the
range
m/z
0-200
for
both positive
and
negative ions
of
the
polymer poly(ethylene terephthalate),
is
shown
in
Figure 6.6,
2.
Dynamic SIMS
(DSIMS)
is
when sputtering proceeds
at a
high rate
(as
in
AES/XPS compositional depth
profiling)
and
only those mass
fragments
of
interest
are
monitored
and
plotted
as a
function
of
sputter
time.
An
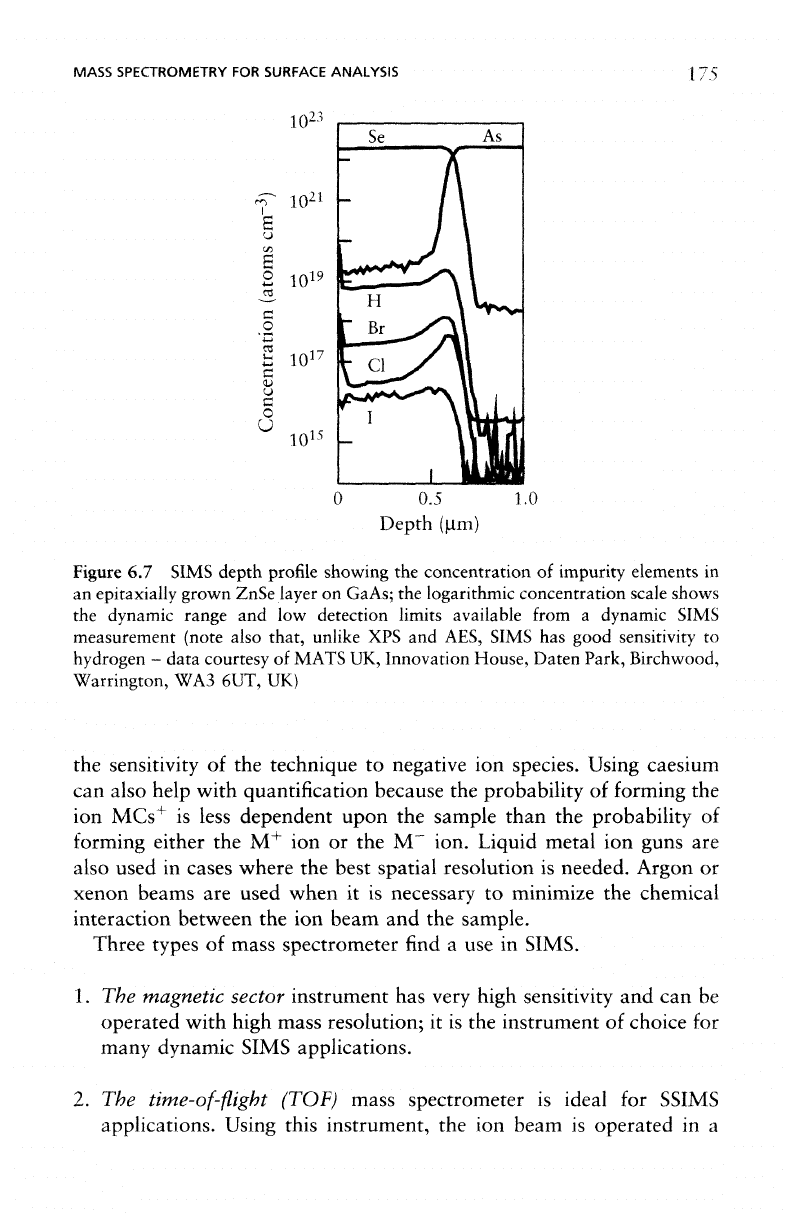
example
of a
DSIMS depth
profile
is
shown
in
Figure 6.7.
3.
Imaging SIMS, which
can be
achieved
in two
different ways, relies
on
either
the
resolution
of the ion
beam itself
or the
ability
of
the
spectrometer
to
retain spatial information
as the
secondary ions
pass through
a
magnetic analyser.
In the
former approach
a fine,
sub-
micron beam, ions, (e.g. gallium),
is
rastered across
the
surface
and a
particular fragment
is
monitored
and
used
to
build
up an
elemental
map of the
surface
in a
sequential manner,
as in
scanning Auger. This
mode
of
acquisition
is
referred
to as the
scanning
ion
microprobe.
The
alternative method
is
known
as ion
microscopy,
in
which
the
entire
field of
view
is
imaged simultaneously
in the
chosen
ion
species, analogous
to
parallel imaging
in
XPS.
In
this type
of
imaging
SIMS,
the
lateral resolution attainable
is
defined
by the
size
of the
apertures
in the ion
optics and,
at the
ultimate resolution (0.5 um),
by
the
optical aberrations
of the
spectrometer
itself.

174
COMPARISON
OF XPS AND AES
WITH
OTHER TECHNIQUES
J
1
0 20
1
D
20
[_J
111
40
JL
40
-o-
1(
1
1
klJL.j
)4
HC^C=0.
149
1
0 0
193
I,
_,.j|
ll.
,..!..
,.
,,.. .!..
60 80 100 120 140 160 180 20
m/z
^ _ (b)
,M
In
\=z/~
ii
o
1
,,.
.,l,,ll!l
I...J
.
Zl
-OC-^-COCH
2
CH
2
0 0
HOC-O-CO"
it
*zy H
0 165 0
1
1
191
1.
, I,. .1. 1 , . 1. ,
60 80 100 120 140 160 180 2
Of
m/z
Figure
6.6
SSIMS
of
poly(ethylene
terephthalate):
(a) the
positive
ion
spectrum,
(b)
the
negative
ion
spectrum (the structures
of the
characteristic cations
at m/z =
104,
149,
193 and
anions
at m/z =
121,
165,
191 are
also
shown)
All
SIMS instruments consist
of a
vacuum system
fitted
with
at
least
one ion gun and a
mass
spectrometer.
To
optimize performance
in
the
analysis
of a
wide range
of
materials
a
selection
of ion
sources
is
required.
To
maximize
the
sensitivity
of the
instrument
to
positive ions
it is
customary
to use
oxygen ions. Similarly, caesium ions will improve
MASS SPECTROMETRY
FOR
SURFACE ANALYSIS
10
23
175
c
<u
u
C
O
U
10
15
-
0.5 1.0
Depth
(urn)
Figure
6.7
SIMS
depth
profile
showing
the
concentration
of
impurity elements
in
an
epitaxially
grown
ZnSe
layer
on
GaAs;
the
logarithmic
concentration
scale
shows
the
dynamic
range
and low
detection
limits
available
from
a
dynamic
SIMS
measurement
(note
also that,
unlike
XPS and
AES,
SIMS
has
good
sensitivity
to
hydrogen
-
data
courtesy
of
MATS
UK,
Innovation
House,
Daten
Park,
Birchwood,
Warrington,
WA3
6UT,
UK)
the
sensitivity
of the
technique
to
negative
ion
species. Using caesium
can
also help with quantification because
the
probability
of
forming
the
ion
MCs
+
is
less dependent upon
the
sample than
the
probability
of
forming
either
the M
+
ion or the M
_
ion. Liquid metal
ion
guns
are
also used
in
cases where
the
best spatial
resolution
is
needed. Argon
or
xenon beams
are
used when
it is
necessary
to
minimize
the
chemical
interaction between
the ion
beam
and the
sample.
Three types
of
mass
spectrometer
find a use in
SIMS.
1. The
magnetic sector instrument
has
very high sensitivity
and can be
operated
with high mass resolution;
it is the
instrument
of
choice
for
many
dynamic SIMS applications.
2. The
time-of-flight
(TOF)mass spectrometer
is
ideal
for
SSIMS
applications.
Using this instrument,
the ion
beam
is
operated
in a

176
COMPARISON
OF XPS AND
AES
WITH
OTHER TECHNIQUES
pulsed
mode
and,
following every pulse,
the
complete SIMS spectrum
is
collected.
This
minimizes
the
number
of
incident ions
and
therefore
maximizes
the
information available while remaining within
the so-
called
static SIMS regime.
The
other aspects
of TOF
spectrometers,
which make them ideal
for
SSIMS,
are
their very large
(in
principle,
infinite)
mass range
and
their high mass resolution. This type
of
spec-
trometer
is now
becoming important
for the
dynamic SIMS analysis
of
ultra-thin
films or
semiconductor materials implanted with dopants
at
very
low
energy.
3. The
quadrupole mass spectrometer
is
compact, versatile
and
relatively
inexpensive.
In the
past,
its
versatility
led to it
being used
on
stand-alone SIMS instruments because
it can
produce reasonable
data
in
both
static
and
dynamic SIMS measurements. However,
with
the
advent
of TOF
SIMS, users
of the
technique have polarized
preferring
either magnetic sector instruments
for
dynamic SIMS
or
TOF
SIMS
for
static SIMS.
The
quadrupole instrument
is now
used
only
as a
secondary technique
on
other surface analysis instruments
or for
highly
specific
applications
as a
stand-alone instrument
for
dynamic
SIMS.
A
SIMS analysis
is
extremely surface sensitive, having
an
analysis
depth
in the
static SIMS mode
of
around
1 nm.
With dynamic SIMS,
the
material
is
being sputtered
at
such
a
rate that
the
actual
surface
is
constantly being eroded
and the
term
'depth
of
analysis' becomes less
important;
it is now the
depth resolution that
is the
parameter
by
which
the
technique
is
judged.
The
detection limit
for
elemental species
is
probably
one of the
best
obtainable with
the
methods discussed
in
this chapter, being
of the
order
of
p.p.b
in
favourable
cases,
such
as
boron implanted into silicon.
Quantification
can be
carried
out
quite accurately
by
comparison
of
the
specimen being examined with standards
of
very
close composition
but,
for
routine analysis
of
unknown specimens,
quantification
will
not
usually
be
attempted.
One
method which
has
been used
to
improve quantification, albeit
with much
inferior
detection limits,
is
secondary neutral mass spec-
troscopy (SNMS). Using this technique,
the
secondary ions
are
rejected from
the
secondary beam
and a
proportion
of the
neutrals
are
ionized
and
detected using
the
mass
spectrometer.
This removes
