Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.

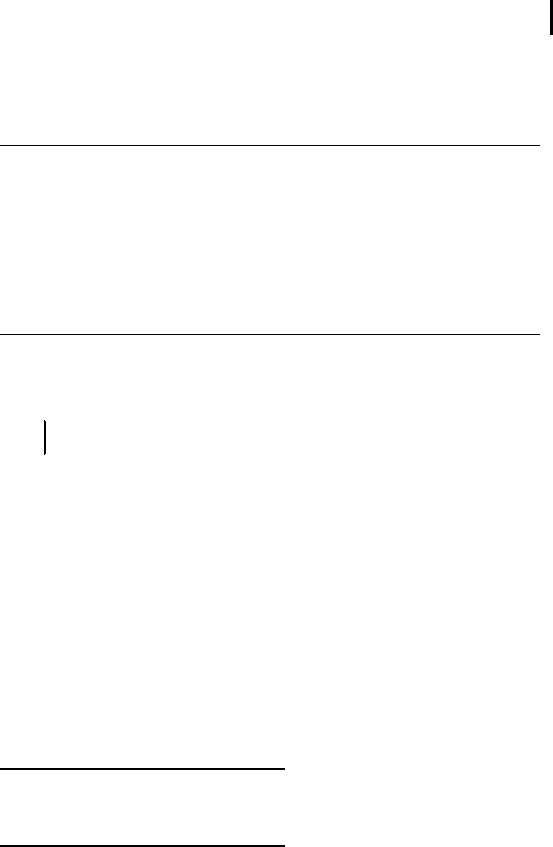
3.1 Reswelling Tray 311
vents the streaking caused depletion of the reductant. This measure
can also improve wide pH gradient separations.
Tab. 3.4: Basic gradients pre-rehydration solution.
7 mol/L urea 4.2 g
2 mol/L thiourea 1.5 g
15 mg/mL DeStreak reagent 120 mL
0.5% (w/v) CHAPS 50 mg
10% (v/v) glycerol 1.2 mL
1.25% (v/v) IPG buffer according to pH gradient 125 mL
0.002% Bromophenol blue*) 10 mL
Water, deionized, fill up to 10 mL
*) From 1% Bromophenol blue solution
& Note: Do not mix sample containing reductant
and rehydration solution containing DeStreak!
Be sure that the reswelling tray has been carefully cleaned and dried
before use.
.
Adjust the feet to level the tray horizontally,
using the inbuilt spirit level as a control (see Fig-
ure 3.1).
.
Pipette rehydration solution containing the sam-
ple according to Table 3.5 into the grooves as
streaks slightly shorter than the strips to be rehy-
drated.
Tab. 3.5: Rehydration volumes.
7 cm strip 125 mL
18 cm strip 340 mL
24 cm strip 450 mL
.
Remove the cover film from the IPG strip start-
ing at the acidic (+) end.
.
Place the strip into the slot with the dried gel
side down, avoiding air bubbles.
.
Check whether the serial number of the strip
can be read correctly.
.
Remove air bubbles by lifting the strip up again
with a forceps.
Such a rehydration solution is
available as ready-made
mixture (DeStreak solution),
however, without the IPG
buffer added; see page 79
Starting at the basic end might
damage the – usually softer –
basic gel surface.
If the number is mirror-
converted, the gel surface is
turned upside.
Step 3: Isoelectric Focusing312
If some of the sample flows onto the back of the film, it will be
pushed down around the edge of the strip when the paraffin oil is
pipetted onto it.
.
Pipette 2 mL paraffin oil over the strip, starting
at both ends of the strip and moving to the cen-
ter.
.
Repeat this procedure for all samples to be ana-
lyzed.
.
Close the tray with the sliding coverlid.
.
Leave to rehydrate at room temperature accord-
ing to Table 3.6.
Fig. 3.1: Rehydration of IPG strips in individual grooves in the reswelling tray.
Tab. 3.6: Rehydration times.
Without sample >6 hours
Including sample >12 hours or overnight *)
The grooves are numbered 1–12 to allow sample identification.
However, ideally the IPG strips have serial numbers printed on the
back of the film support, which are used for tracking the samples.
To avoid urea crystallization
and oxygen contact.
*) The large protein molecules
need a long time to diffuse into
the strip.

3.2 Rehydration Loading and IEF in IPGphor Strip Holders 313
After rehydration
.
Rinse the surface of the strips with distilled
water using a squeeze bottle and then place
them for a few seconds on their edges on a
damp filter paper to drain excess liquid, so that
the urea on the surface does not crystallize out.
The strips can be run on the Multiphor flatbed chamber in the Immo-
biline DryStrip Kit (tray for up to 12 strips) or on the IPGphor in the
manifold.
Cleaning of the reswelling tray The reswelling tray is thoroughly
cleaned with detergent, using a toothbrush or Q-tips for the slots.
After rinsing it with deionized water it must be completely dry before
the next use.
3.2
Rehydration Loading and IEF in IPGphor Strip Holders
The IPGphor chamber must be horizontally leveled on the bench.
Rehydration and IEF separation are carried out at 20 C.
It is highly recommended to connect the IPGphor to an external
computer via the serial port. In this way the electrical conditions can
be monitored. This allows one to judge from the shape of the graphs,
whether the separation will give good or bad 2-D results.
.
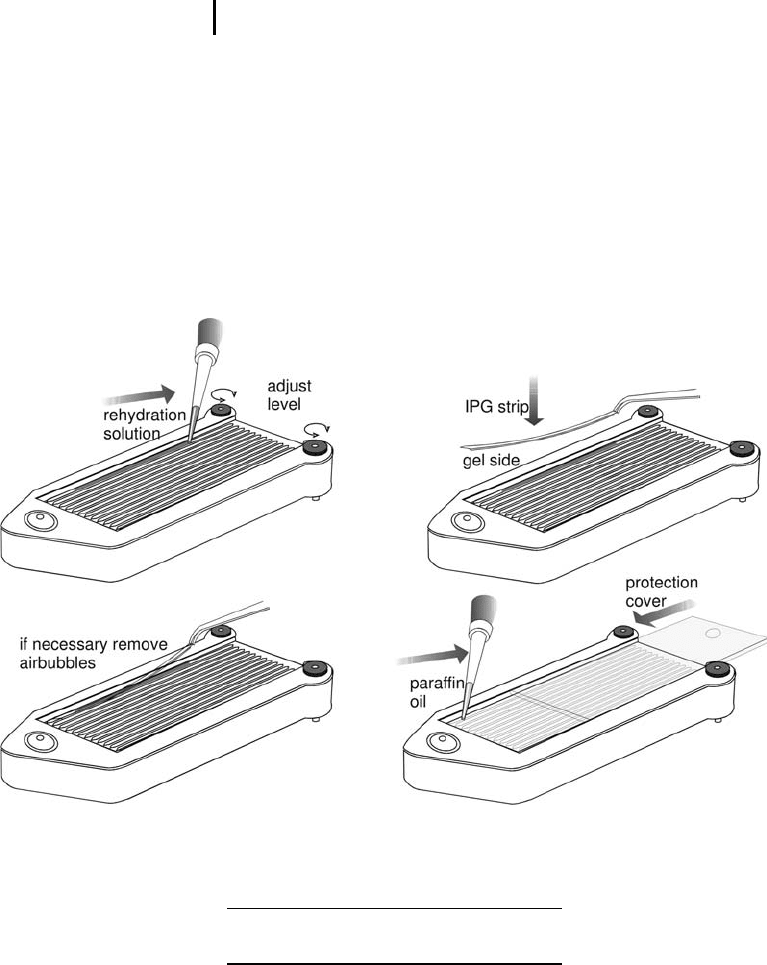
Be sure that the strip holders are carefully
cleaned and dried.
.
Place strip holders on the cooled electrode con-
tact areas of the power supply with the pointed
end on the anodal contact area.
.
Pipette rehydration solution mixed with sample
into the strip holder as a streak from electrode
contact to electrode contact (see Figure 3.2).
.
Remove the cover film from the IPG-strip start-
ing at the acidic (+) end.
.
Starting at the anodal side, place the IPG strip
with the acidic end into the strip holder – dried
gel side down. Slowly lower the basic end into
the strip holder.
Should an air bubble be caught, lift the strip up with forceps and
slowly lower it down again. You might need to repeat this procedure.
See Section 1.5.3.4 on page
91 f for further explanations.
Never use new strip holders
without cleaning them before
the first run.
Starting at the basic end might
damage the – usually softer –
basic gel surface.
The distance from the platinum
contact to the end of the tray is
shorter at the anodal side.
Step 3: Isoelectric Focusing314
Fig. 3.2: Rehydration loading of the sample solution in the
strip holder of the IPGphor.
When some of the sample flows onto the back of the film, it will be
pushed down around the edge of the strip when the paraffin oil is
pipetted on it.
.
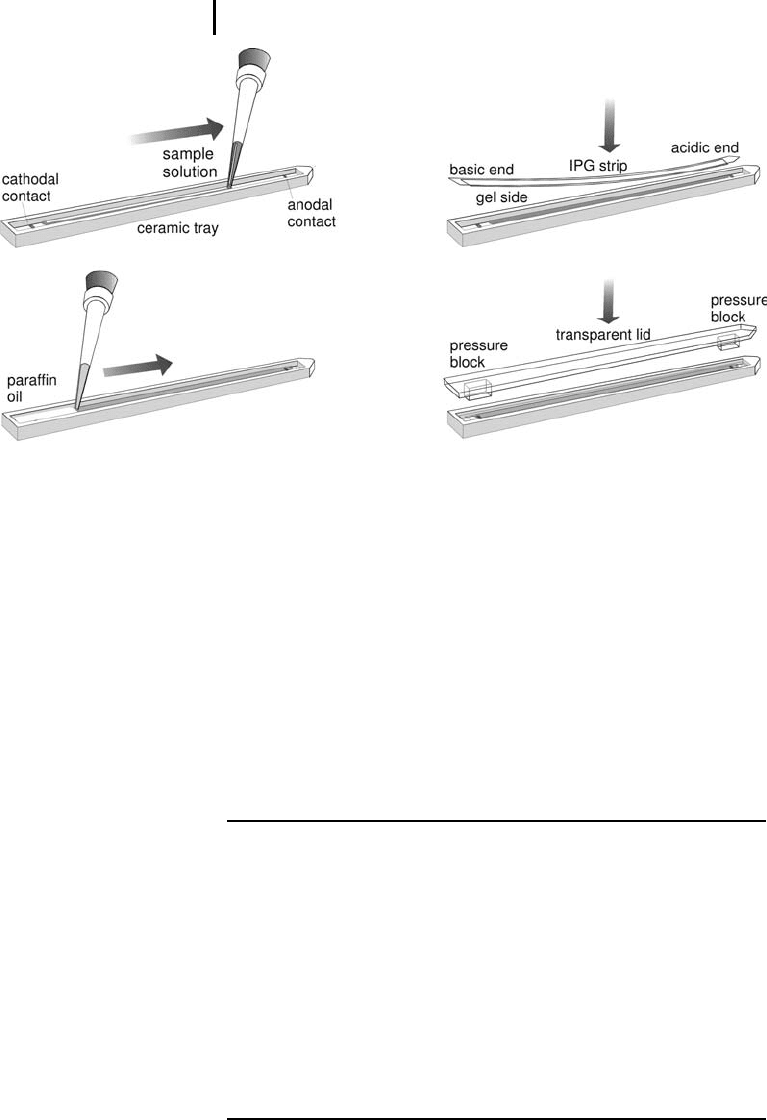
Pipette 2 mL paraffin oil over the strip by start-
ing on both ends of the strip, moving to the cen-
ter.
.
Place the plastic cover on the strip holder.
.
Close the safety lid.
.
Enter the running conditions (see Table 3.7).
Tab. 3.7: “Worst case” conditions for active rehydration loading in strip
holders.
Rehydration time 0 h
20 C
50 mA
Temperature
Current per strip
Strip length 18 cm 24 cm
pH gradient 3–10 L 3–10 NL 3–10 L 3–10 NL
Step 1 60 V 12 h step and hold 12 h step and hold
Step 2 150 V 3 h step and hold 3 h step and hold
Step 3 300 V 3 h step and hold 3 h step and hold
Step 4 1,000 V 6 h gradient 6 h gradient
Step 5,
analytical
10,000 V 20 kVh
2 h 20 min
gradient
20 kVh
2 h 20 min
gradient
40 kVh
4 h gradient
40 kVh
4 h gradient
Total time 16 h 20 min 16 h 20 min 18 h 18 h
The cover fluid prevents urea
crystallization and oxygen
contact.

3.2 Rehydration Loading and IEF in IPGphor Strip Holders 315
When a new sample type has to be analyzed or the 2-D patterns are
not optimal, it is recommended to apply the following “worst condi-
tions”. Generally, active rehydration provides the best results. For
active rehydration the “rehydration time” in the program is set to “0”,
it is performed as a voltage step.
For preparative sample loads (>1 mg) increase the final focusing
step by 15% of the proposed volt hours.
When an overnight run is finished early in the morning, refocus
the proteins by applying 8,000 V on the strips for 15 minutes before
equilibration, staining, or storage of the strips.
Observations during the run In each IPG strip the Bromophenol
blue tracking dye slowly starts to build a band at the cathodal end,
which migrates towards the anode. When all samples have the same
salt content, they run almost at the same level. There is no reason to
worry, when the Bromophenol blue bands of a few samples migrate
slower, this can always happen.
During the final few hours you will observe a strong black, some-
times also an additional yellow band at the anode: this is Bromophe-
nol blue collecting here, and at the very end it becomes very acidic,
turning the Bromophenol blue to yellow.
.
Before the run is finished: place a tray with
detergent solution next to the instrument for the
strip holders.
Cleaning of the strip holders The strip holders must be carefully
cleaned after each IEF. The solutions must never dry in the strip
holder. Cleaning is very effective, if the strip holders are first soaked a
few hours in a solution of 2–5% of the specially supplied (non-alkali)
detergent in hot water.
The strip holder slot should be vigorously brushed with a tooth-
brush using a few drops of undiluted IPGphor Strip Holder Cleaning
Solution. Then it is rinsed with deionized water.
Sometimes protein deposits are left on the bottom of the strip
holder after IEF. This happens when highly abundant proteins have
been squeezed out of the gel surface at their isoelectric point (see Fig-
ure 1.32 on page 95). It is not always easy to remove these proteins,
particularly when they are sticky like serum albumin. In this case the
strip holders should be boiled in 1% (w/w) SDS with 1% (w/v) DTT
for 30 minutes before the slot is cleaned with the toothbrush.
& Important: Strip holders may be baked, boiled or
autoclaved. But, because of the specially treated
surface they must not be exposed to strong acids
or basis, including alkaline detergents.
Passive rehydration (with no
electric field applied during
rehydration) can be performed
in the reswelling tray.
Clean strip holders should be
handled with gloves to avoid
contamination.
SDS solution in absence of
buffer has a neutral pH.

Step 3: Isoelectric Focusing316
& Note: The strip holder must be completely dry
before use.
3.3
IEF of Rehydration Loaded Strips in the Manifold
The IPGphor chamber must be horizontally leveled on the bench.
IEF separation is carried out at 20 C.
It is highly recommended to connect the IPGphor to an external
computer via the serial port. In this way the electrical conditions can
be monitored. This allows one to judge from the shape of the graphs,
whether the separation will give good or bad 2-D results.
.
Be sure that the manifold is carefully cleaned
and dried.
.
Place manifold on the cooled electrode contact
areas of the power supply.
.
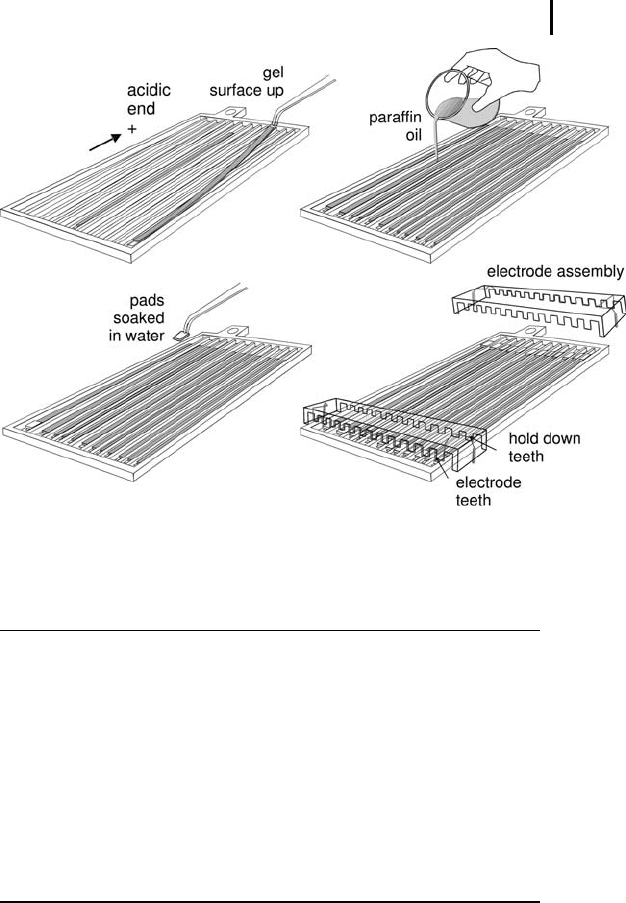
Starting at the basic side, place the IPG strip –
gel side facing up – into the manifold with the
acidic end towards the anode side (see Figure
3.3).
.
Soak electrode pads with deionized water. Blot
them on filter paper and place them on top of
the ends of the strip. The pads should sit com-
pletely on the gel surface. If longer strips are
required for removal of salt, there must be an
overlapping of at least 5 mm.
.
Pour 100 mL Drystrip cover fluid (paraffin oil)
over the strips and electrode pads.
.
The electrode assembly has electrode teeth on
one side and hold-down teeth (for paper bridge-
loading) on the other side. It is important to
choose the correct orientation, to get contact
with the electrode pads.
.
Place the electrode assemblies on the pads.
Secure them in place with the cams.
.
Close the safety lid.
.
Enter the running conditions (see Table 3.8).
See Section 1.5.3.4 on page
91 f for further explanations.
Aligner protrusions along the
grooves inside the manifold
align the rehydrated IPG strips,
keeping them straight and
centered when placed inside the
manifold.
I there are no ready-cut elec-
trode pads available, cut 5 mm
long pads from IEF electrode
strips. The pads must be damp,
not wet.
The cover fluid prevents urea
crystallization and oxygen
contact.
3.3 IEF of Rehydration Loaded Strips in the Manifold 317
Fig. 3.3: Using the manifold for running rehydration-loaded IPG strips.
Tab. 3.8: “Worst case” conditions for the manifold (cup, paper bridge,
rehydration loading).
Rehydration time 0 h
20 C
up to 100 mA
Temperature
Current per strip
Strip length 18 cm 24 cm
pH gradient 3–10 L 3–10 NL 3–10 L 3–10 NL
Step 1 Step and hold 150 V 3 h 3 h
Step 2 Step and hold 300 V 3 h 3 h
Step 3 Gradient 1,000 V 6 h 6 h
Step 4 Gradient 10,000 V 1 h 1 h
Step 5 Step and hold 10,000 V 16 kVh
for 2 h
16 kVh
for 2 h
24 kVh
for 3 h
24 kVh
for 3 h
Total time 14 h 14 h 15 h 15 h
For preparative sample loads (>1 mg) increase the final focusing
step by 15% of the proposed volt hours.
When an overnight run is finished early in the morning, refocus
the proteins by applying 10,000 V on the strips for 15 minutes before
equilibration, staining, or storage of the strips.

Step 3: Isoelectric Focusing318
3.4
Cup Loading IEF
It is highly recommended to connect the IPGphor to an external com-
puter via the serial port. In this way the electrical conditions can be
monitored. This allows you to judge from the shape of the graphs as
to whether the separation will give good or bad 2-D results.
The IPGphor chamber must be horizontally leveled on the bench.
IEF separation is carried out at 20 C.
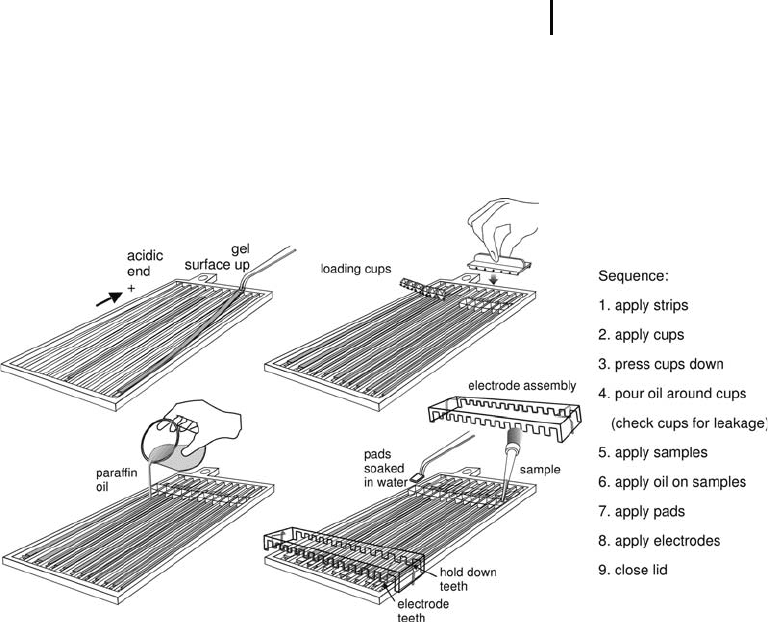
Figure 3.4 shows all the parts needed and the proper placements of
strips, pads, sample cups, and electrodes.
.
Be sure that the manifold is carefully cleaned
and dried.
.
Place the manifold on the cooled electrode con-
tact areas of the power supply.
.
Starting at the basic side, place the IPG strip –
gel side facing up – into the strip holder with the
acidic end towards the anode side. Be sure that
the protruding film at the basic end touches the
end of the groove.
.
Apply the loading cups at correct side of the
strip. Press them down to prevent leakage.
& Note: The cup can straddle on the alignment
protrusions, if necessary.
.
Soak electrode pads with deionized water. Blot
them on filter paper and place them on top of
the ends of the strip. The pads should sit com-
pletely on the gel surface. If longer pads are
required for removal of salt, there must be an
overlapping of at least 5 mm.
.
Pour 100 mL Drystrip cover fluid (paraffin oil)
over the strips, around the cups. With this mea-
sure leakages are detected, because oil would
flow into a cup.
.
Dilute samples with rehydration solution to
50–100 m L for optimum protein entry.
.
Pipette samples into the cups.
.
Pipette 20 mL paraffin oil on each sample.
.
The electrode assembly has electrode teeth on
one side and hold-down teeth (for paper bridge-
loading) on the other side. It is important to
See Section 1.5.3.4 on page
91 f for further explanations.
For the course: use analytical
protein load here.
Never use new manifold
without cleaning them before
the first run.
Aligner protrusions along the
grooves inside the manifold
align the rehydrated IPG strips,
keeping them straight and
centered when placed inside the
manifold.
Mostly anodal sample applica-
tion is employed.
If there are no ready-cut elec-
trode pads available, cut 5 mm
long pads from IEF electrode
strips. The pads must be damp,
not wet.
The cover fluid prevents urea
crystallization and oxygen
contact.
In higher concentrated samples
more proteins would tend to
aggregate and precipitate.
3.5 Paper Bridge Loading 319
choose the correct orientation, to get contact
with the electrode pads.
.
Place the electrode assemblies on the pads.
Secure them on place with the cams.
.
Close the safety lid.
.
Enter the running conditions (see Table 3.8).
Fig. 3.4: Using the manifold for cup loading.
3.5
Paper Bridge Loading
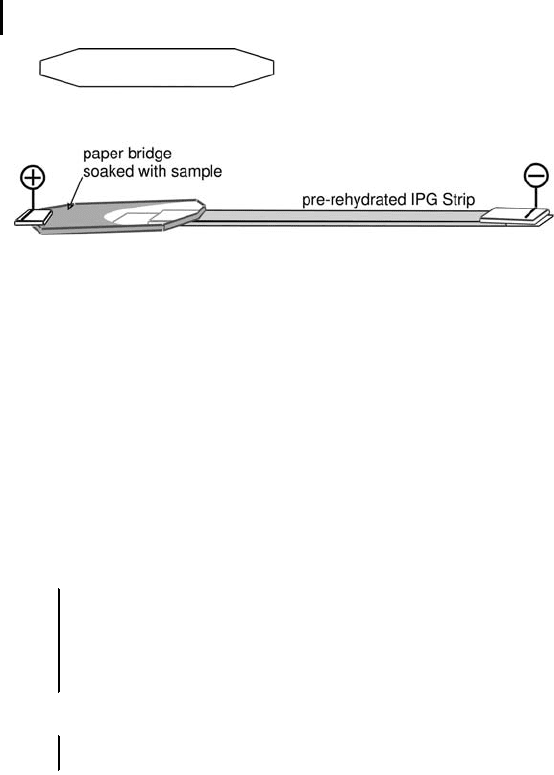
There are pre-cut paper bridges on the market. If there are no ready-
cut bridges available, cut 5 cm long and 0.8 mm wide pieces from
very clean filter card board material. They should have pointed 3 mm
wide ends as shown in Figure 3.5. The electrode assembly has elec-
trode teeth on one side and hold-down teeth for paper bridge loading
on the other side. Therefore the length of the paper bridge is impor-
tant.
The procedure works in principle as described for rehydration
loaded IPG strips. The only difference: the paper bridges, soaked in
500 mL sample solution are placed between IPG strip end and the
electrode pad, as shown in Figure 3.5. If this would give a better
separation, the paper bridge can also be placed at the cathodal side.
As described in the paper by Kane et al. (2006) the paper bridge can
be sequentially applied on different narrow pH gradients.
Kane LA, Yung CK, Agnetti G,
Neverova I, Van Eyk JE. Proteo-
mics 6 (2006) 5683–5687.
Step 3: Isoelectric Focusing320
Fig. 3.5: Schematic drawing of the shape of a paper bridge and
paper bridge loading for the example of anodal sample application.
Note: The paper bridge is not shown to scale! Use the dimensions
given in the text.
For running conditions choose the settings in Table 3.5.
Cleaning of the manifold The manifold must be carefully cleaned
after each IEF. The solutions must never dry in the manifold. Clean-
ing is very effective, if the manifold is first soaked for a few hours in a
solution of 2–5% of the specially supplied detergent in hot water.
The manifold grooves should be vigorously brushed with a tooth-
brush using a few drops of undiluted IPGphor Strip Holder Cleaning
Solution. Then it is rinsed with deionized water.
& Important: The manifold may be baked, boiled
or autoclaved. But, because of the specially
treated surface they must not be exposed to
strong acids or basis, including alkaline
detergents.
& Note: The manifold must be completely dry
before use.
The IPG strips are directly transferred to equilibration in SDS buffer
for the second dimension run or stored in a deep freezer at –60 C to
–80 C.
When the IPGphor is connected to an external computer via the
serial port, the electrical conditions are monitored. The shapes of the
graphs indicate whether the separation will give good or bad 2-D
results.
Alternatively some of the strips can be stained for inspection as to
whether the IEF step was successful.
Cleaning of the manifold is
easier than the regular strip
holder.
The clean manifold should be
handled with gloves to avoid
contamination.
See Section 1.5.3.4 on page
91 f for further explanations.
