Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


1.3 Examples 291
Tab. 1.4: Amounts of additives to 1 mL sample.
Sample 1 mL Sample 1 mL
8 mol/L urea 0.75 g 9 mol/L urea 0.94 g
0.5% (w/v) CHAPS 8 mg 2% (w/v) CHAPS 36 mg
0.28% (w/v) DTT 5 mg 1% (w/v) DTT 18 mg
1.25% (v/v) Pharmalytes 3–10*) 20 mL 2% (v/v) Pharmalytes 3–10*) 36 mL
0.002% Bromophenol blue**) 2 mL 0.002% Bromophenol blue**) 2 mL
Total volume 1.6 mL 1.85 mL
*) or IPG buffer of respective pH interval
**) from 1% Bromophenol blue solution
1.3
Examples
The volumes are given for 24 cm IPG strips only. For paper bridge
loading 18 cm strips are recommended. The sample would be diluted
in 450 mL for anodal and 350 mL for cathodal loading.
Example 1:
Escherichia coli
extract (ideal as test sample) Sample:
lyophilized cells of E. coli strain B (ATCC 11303), Sigma EC-11303
Lyophilized E. coli 30 mg Measured protein content
Lysis solution 1 mL ~12 mg protein/mL
Freeze (–20 C) and thaw two times in a microcentrifuge tube.
Centrifuge for 10 min with 13,000 rpm.
24 cm IPG strip Sample supernatant Rehydration solution
Analytical run: (~180 mg protein)
Rehydration loading 15 mL mix with 435 mL
Cup loading 15 mL*) at anodal side 450 mL (>6 h pre-rehydration)
Preparative run: (~1 mg protein)
Rehydration loading 83 mL mix with 367 mL
Cup loading 83 mL at anodal side 450 mL (>6 h pre-rehydration)
*) Mix with 50 mL rehydration solution before you apply it.
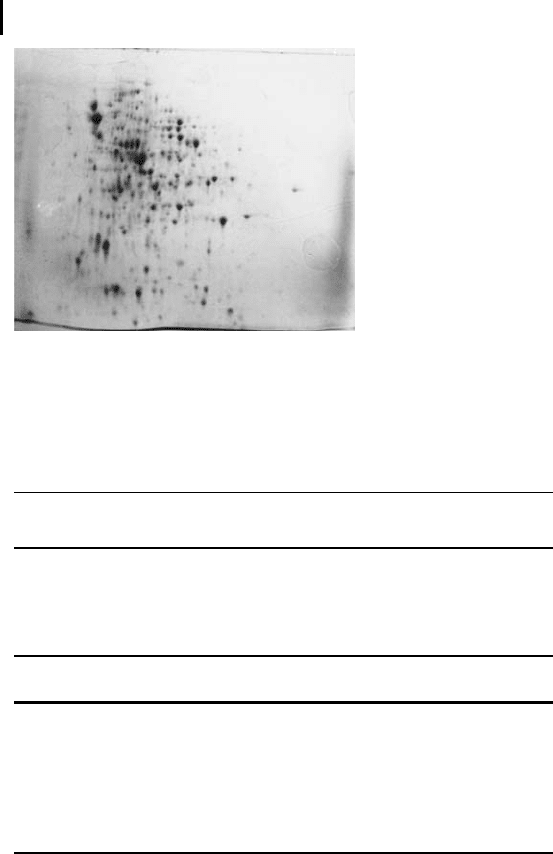
Figure 1.1 shows a typical 2-D electrophoresis pattern of an E. coli
extract applied with rehydration loading.
Step 1: Sample Preparation292
Fig. 1.1: E. coli extract (~600 mg protein), IPG DryStrip,
pH 3–10, Ettan DALT gel, fast Coomassie staining.
Example 2:
Saccharomyces cerevisiae
(yeast cell lysate)
Lyophilized yeast 300 mg
lysis solution 2.5 mL
Sonicate for 10 min at 0 C.
Centrifuge for 10 min at 10 C with 42,000 g.
24 cm IPG strip Sample supernatant rehydration solution
Analytical run:
Rehydration loading 30 mL mix with 420 mL
Cup loading 30 mL*) at anodal side 450 mL (>6 h pre-rehydration)
Preparative run:
Rehydration loading 300 mL mix with 150 mL
Cup loading 100 mL at anodal side 450 mL (>6 h pre-rehydration)
*) Mix with 50 mL rehydration solution before you apply it.
Tissue The tissue of interest is sliced with a scalpel to obtain an
appropriately sized piece. It can be frozen with liquid nitrogen and
broken into small fragments in a mortar and pestle.
1.4 Microdialysis 293
Example 3: calf liver Liver acetone powder, calf, Sigma L-7876
Acetone powder 10 mg
Lysis solution 1 mL
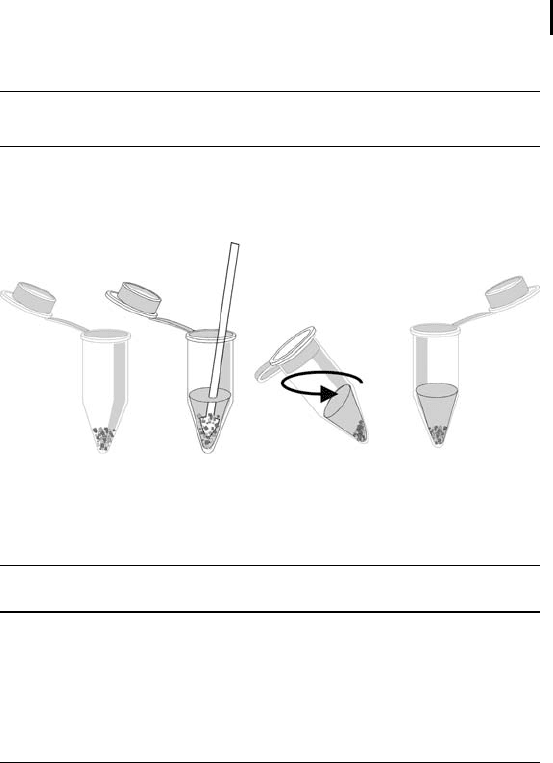
For small tissue amounts a disposable grinding kit is very useful
(see Figure 1.2).
Fig. 1.2: Grinding of tissue material.
Typically 5 mg of the acetone powder or 20–50 mg of animal or
plant tissue are ground in 500 mL of lysis solution.
24 cm IPG strip Sample supernatant Rehydration solution
Analytical run:
Rehydration loading 30 mL mix with 420 mL
Cup loading 30 mL *) at anodal side 450 mL (>6 h pre-rehydration)
Preparative run:
Rehydration loading 300 mL mix with 150 mL
Cup loading 100 mL at anodal side 450 mL (>6 h pre-rehydration)
*) Mix with 50 mL rehydration solution before you apply it.
1.4
Microdialysis
Dialysis of the sample is usually not recommended, because large
membrane areas can cause adsorption of proteins. Less protein gets
lost with specially designed minidialysis tubes. The fastest removal of
small ions is achieved with an 8 kDa cut-off membrane.
Salt and buffer ions can be removed efficiently by dialyzing 2-D
samples against at least 40 sample volume of 8 mol/L urea / 1%
DTT solution for 2 hours to overnight. The 2-D sample should be pre-
pared in lysis buffer. Other, more expensive additives such as CHAPS
In the Tris-glycine buffer system
peptides smaller than 10 kDa
anyhow co-migrate with the
front; they are not displayed in
the gel.

Step 1: Sample Preparation294
and carrier ampholytes do not need to be included in the dialysis
solution. These components are afterwards added back, in the origi-
nal concentrations.
1.5
Precipitation
A crude extract can contain contaminations with phospholipids and
nucleic acids, which are visualized with silver staining as horizontal
streaks in the acidic part of the gel. Protein precipitation is therefore
often employed to remove the contaminating substances. Protein pre-
cipitation is also used to concentrate proteins from samples that are
too dilute for effective 2-D analysis.
1.5.1
TCA Acetone
Sometimes the TCA and/or the salt from the sample are not comple-
tely removed, which can be observed by horizontal streaking in the
2-D gel. Practice has shown that washing with ice-cold 10% water /
90% acetone instead of pure acetone removes ionic components very
efficiently without any negative effects. Therefore the original proto-
col of Damerval et al. (1986) has been modified here. Furthermore
the modifications according to Gçrg et al. (2000) are implemented
here.
.
Freeze plant material – or other material – in
liquid nitrogen.
.
Grind it with a pestle in a pre-frozen mortar.
.
To 30 mg add 5 mL of 20% TCA in cold acetone
(–20 C) containing 0.2% (w/v) DTT.
.
Precipitate overnight in a freezer.
.
Centrifuge at +20 C with 13,000 rpm for
10 minutes.
.
wash with 90% acetone / 10% water (–20 C)
containing 0.2% (w/v) DTT.
.
Centrifuge again.
.
Resuspend the pellet in lysis buffer.
.
Carefully sonicate.
.
Centrifuge at +20 C with 13,000 rpm for
10 minutes.
For protein precipitation from a liquid the precipitation fluid must be
at least three times larger than the volume of the sample.
For precipitation of proteins
from a cell culture supernatant
often a 100% (w/v) TCA solu-
tion is employed.
Damerval C, DeVienne D, Zivy
M, Thiellement H. Electrophor-
esis 7 (1986) 53–54.
Gçrg A, Obermaier C, Boguth
G, Harder A, Scheibe B, Wild-
gruber R, Weiss W. Electrophor-
esis 21 (2000) 1037–1053.
Avoid heating!

1.5 Precipitation 295
The disadvantage is, however, that some acidic proteins get lost
with this procedure, because they are not precipitated into the pellet.
1.5.2
Ettan 2-D Cleanup Kit
The Ettan 2-D cleanup kit procedure uses a combination of a unique
precipitant and co-precipitant to quantitatively precipitate the sample
proteins. The proteins are pelleted by centrifugation and the precipi-
tate is washed to further remove non-protein contaminants. The mix-
ture is centrifuged again and the resultant pellet is resuspended into
the lysis buffer.
The sample can contain 1 mg to 1 mg protein in a volume of 1 to
100 mL. Protein can be processed from larger volumes by scaling up
the procedure.
1.5.3
Resolubilization
In practice the 2-D cleanup kit and the method according to Wessels
and Flgge hold the highest yield among the precipitation methods.
Nevertheless, there are sometimes difficulties reported to get the pel-
let back into solution. Here are a few important hints how to get the
pellet completely redissolved:
.
Take care that the pellet does not become com-
pletely dry!
.
Pipette repeatedly lysis solution over the pellet,
but do not vortex!
.
Resolubilization can take several hours or over-
night at room temperature. Proteases have been
completely inactivated by precipitation.
.
If this does not work, carefully sonicate with the
sample being placed on ice, with short bursts
and breaks in between.
.
Or use PlusOne Molecular grinding kit to
increase the surface of the pellet
.
Or freeze pellet with lysis solution at –20 C for
15 minutes.
.
If none of the above measures work, use SDS
solution (2% SDS, hot) and then dilute with 9
mol/L urea / 4% CHAPS to an SDS concentra-
tion below 0.1%. Also, as a rule of thumb, the
CHAPS to SDS ratio must be 8:1 in the sample
before application on the IPG strip.
This procedure has the highest
yield of precipitated proteins
and proteins coming back into
solution.
Vortexing can cause oxidation
of some proteins, and then they
will not dissolve anymore.
Do not vortex!
Avoid heating of sample
Particularly useful, when
several mg of proteins have
been precipitated.
Urea crystals will develop,
which break up the pellet
See also “SDS procedure”
below
Step 1: Sample Preparation296
1.6
Very Hydrophobic Proteins
1.6.1
Thiourea Procedure
For membrane proteins and other very hydrophobic proteins a combi-
nation urea/thiourea in the solubilization solution can be very helpful
to get more proteins into solution (Rabilloud, 1998). The modified
lysis solution is then composed as follows:
7 mol/L urea 4.2 g
2 mol/L thiourea 1.5 g
With H
2
O
dist
fill up to 9.5 mL
Removal of isocyanate:
.
Warm the tube to get the urea and thiourea com-
pletely in solution: Do not exceed 37 C.
.
Add 100 mg mixed bed ion exchanger Amberlite
IRN-150. Stir for 10 min.
.
Filter through paper.
.
Add the other additives:
4% CHAPS 400 mg
1% (w/v) DTT 100 mg
2% (v/v) Pharmalytes pH 3 to 10*) 500 mL
0.002% Bromophenol blue**) 10 mL
*) or IPG buffer
**) from 1% Bromophenol blue solution
& Important: Prepare the solution freshly, shake to
dissolve the urea, do not warm it higher than
30 C to avoid carbamylation.
This lysis solution can also be produced in larger quantities – without
DTT – and stored frozen in aliquots at a temperature lower than
–60 C.
Always treat thiourea-
containing solutions with
mixed bed ion exchanger.
Repeated freeze-thawing must
be avoided. Add DTT shortly
before use.

1.7 Quantification 297
1.6.2
SDS Procedure
.
Formation of oligomers can be prevented.
.
Organisms with tough cell walls sometimes
require boiling for 5 minutes in 1–2% SDS
before they are diluted with lysis solution.
.
Very hydrophobic proteins might require extrac-
tion with high percentage of SDS.
& Note: The SDS-containing sample solution must
be diluted with lysis solution (9 mol/L urea / 4%
CHAPS) to an SDS concentration below 0.1%.
Also, as a rule of thumb, the CHAPS:SDS ratio
must be 8:1 in the sample before application on
the IPG strip.
Example: human plasma According to Sanchez et al. (1995):
10 mL human plasma Mix with 6.25 mL 10% SDS, 2.3% DTT
Heat for 5 min at 95 C
Dilute with 500 mL lysis solution
& Note: SDS does not always completely separate
from the proteins during IEF, even under high
field strength. SDS can also be removed by
precipitation with the cleanup kit.
1.7
Quantification
The most reliable protein quantification for 2-D IEF samples is
obtained with the Ettan 2-D Quant kit, which is based on precipita-
tion. This procedure is compatible with such common sample pre-
paration reagents as 2% SDS, 1% DTT, 8 mol/L urea, 2 mol/L
thiourea, 4% CHAPS, 2% Pharmalytes and 2% IPG Buffer. The pre-
cipitated proteins are resuspended in a copper-containing solution;
the unbound copper is measured with a colorimetric agent. The color
density is inversely related to the protein concentration. The volume
range of the assay is 1–50 mL and the linear range for quantification
is 0–50 mg. The assay has a sensitivity threshold of 0.5 mg.
Sanchez J-C, Appel RD, Golaz
O, Pasquali C, Ravier F,
Bairoch A, Hochstrass er DF.
Electrophoresis. 16 (1995)
1131–1151.

Step 1: Sample Preparation298
1.8
SDS Samples for 1-D SDS Electrophoresis
SDS sample buffer:
2% (w/v) SDS 2 g
25% Glycerol (v/v) 85% solution 30 mL
50 mmol/L Tris HCl, pH 8.8 1.5 mol/L stock solution 3.5 mL
0.01% Bromophenol blue 10 mg
Water, deionized Make up to 100 mL
Store at room temperature.
.
Dissolve 50 mg of DTT in 100 mL deionized
water.
.
Dissolve 20 mg of iodoacetamide in 100 mL deio-
nized water.
.
Add 10 mL DTT solution to 100 mL SDS sample
buffer.
.
Mix 25 mL sample extract with 25mL SDS sample
buffer.
.
Heat at 60 C for 5 min.
.
Add 10 mL iodoacetamide solution to the 50 mL
SDS sample solution. Leave it at room tempera-
ture for 30 min.
.
Apply 20 mL of this solution on the SDS gel.
The glycerol is added to
increase the density to prevent
mixing with upper buffer.
Do not heat it, when the
sample contains urea!

299
Step 2: Fluorescence Difference Gel Electrophoresis
2.1
Experimental Design
The experiment setup should be properly designed: Biological repli-
cates ensure that induced biological changes are not mixed up with
inherent biological differences. Gel replicates are only necessary
when an experiment consists of a low number of samples, like two to
six samples. In this case one-time gel replicates are run, which con-
tain inverse labeled samples. Experiments with higher numbers of
samples do not require gel replicates.
Optimally all samples of an experiment should be run together in
the first and second dimension to minimize methodical variation.
This means that an experiment should not consist of more than 12
gels, if possible.
.
Prepare a chart for planned randomization of
the samples and the internal standard among
the gels, like the example in Table 2.1.
Tab. 2.1: Example for “randomized” sample labeling and application
for 12 samples.
Gel Cy2 standard Cy3 Cy5
1 All 12 samples Control 1 Treated 3
2 All 12 samples Control 2 Treated 1
3 All 12 samples Control 3 Treated 2
4 All 12 samples Treated 4 Control 5
5 All 12 samples Treated 5 Control 6
6 All 12 samples Treated 6 Control 4
Disposable gloves must be worn during each step to avoid contam-
ination of the sample.
Proteomics in Practice. A Guide to Successful Experimental Design 2
nd
Ed.
Reiner Westermeier, Tom Naven, and Hans-Rudolf Hçpker
Copyright 2008 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
ISBN: 978-3-527-31941-1
The pooled internal standard is
always labeled with one and
the same CyDye: Cy2 for 3-
color experiments and Cy3 for
2-color experiments.
Running gels of one experiment
in different chambers makes
spot matching more difficult.

Step 2: Fluorescence Difference Gel Electrophoresis300
2.2
Sample Preparation
In the standard labeling protocol, proteins are first denatured in a
lysis buffer; that will eliminate all secondary and tertiary structures of
proteins. The protein concentration should then be determined using
the Ettan 2-D Quant kit procedure. The fluorescent properties of Cy2,
Cy3 and Cy5 can be adversely affected by exposure to light: keep the
exposure of protein labeled with CyDye to all light sources to a mini-
mum.
& During labeling the samples must not contain
primary amines (IPG buffers, carrier ampholytes)
and reductants.
Appropriate sample preparation is crucial for good results. Practice
has shown that labeling efficiency is much higher after sample
cleanup based on precipitation of the proteins. This measure will not
only remove disturbing contaminants like lipids, polysaccharides, salt
ions, and nucleic acids, but also endogeneous peptides from the sam-
ple. Peptides contain also lysines and cysteines, which would be
labeled with a CyDye fluor as well. In a standard buffer system the
small peptides migrate in the front of the second dimension, and
would not be resolved.
2.2.1
Stock Solutions
Sample lysis solutions for DIGE:
Lysis solution 1: 9 mol/L urea, 4% (w/v) CHAPS, 30 mmol/L Tris.
Lysis solution 2: 2 mol/L thiourea, 7 mol/L urea, 4% (w/v) CHAPS,
30 mmol/L Tris.
For pH adjustment: 50 mmol/L NaOH solution
.
Solubilize the protein samples either in sample
lysis solution 1 or 2.
.
Check the protein concentration in the samples:
optimally it would be between 5 mg/mL and 10
mg/mL. But 1 mg/mL to 20 mg/mL are tolerable
for successfully labeling.
.
Check the pH carefully: pipette 2 mL sample on a
pH indicator paper. Read out the pH value
immediately, because the color will shift with
time.
These compounds are added
after labeling had been
completed by adding a 2
sample solution.
