Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.

In the absence of ferrous ions in the bulk solution this becomes:
14 Landolt
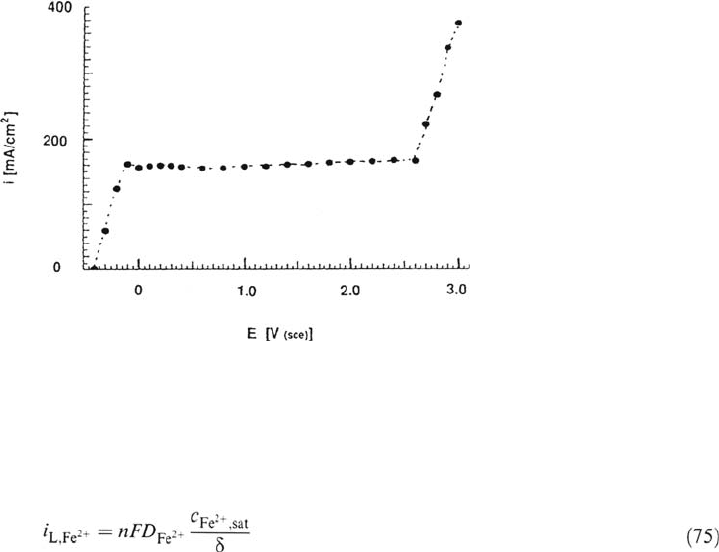
Figure 6 Anodic current-potential curve for iron dissolution in FeCl
2
showing a limit-
ing current plateau due to a salt film. (From Ref. 10.)
Figure 6 shows an anodic current-voltage curve for iron dissolution in 4 M FeCl
2
,
measured with a rotating-disk electrode at 200 rpm. The limiting current plateau
extends over more than 2 V. A thin salt film covers the electrode surface under
these conditions [10]. At very high potentials the salt film is no longer stable, leading
to dissolution of the metal at a high rate. The described transport mechanism
involving salt film formation is of importance in pitting corrosion, where under
certain conditions salt precipitation can occur at the bottom of a pit, leading to
transport-controlled dissolution [5,6].
CORROSION CELLS
In the preceding sections the electrochemical theory of simple electrode reactions
and of mixed electrodes relevant to uniform corrosion was developed. The described
concepts are also applicable to corrosion cells, where anodic and cathodic areas are
spatially separated. However, in the case of corrosion cells the rate of corrosion
depends not only on charge transfer kinetics and on mass transport conditions at the
anode and the cathode but also on the resistivity of the electrolyte and the geometry
of the cell, which determine the internal resistance. This is illustrated by Figure 7,
which shows schematically a corrosion cell formed between an anode and a cathode
of equal surface area facing each other. Also shown is the electrical analogue of the
corrosion cell. The voltage source corresponds to the difference in corrosion
potentials of electrodes 1 and 2 when they are not connected to each other:
ΔE
cor
= E
cor,2
– E
cor,1
. Once they are connected, a current flows whose magnitude
depends on all elements present in the circuit. The nonohmic resistances R
p,1
and
Copyright © 2002 Marcel Dekker, Inc.
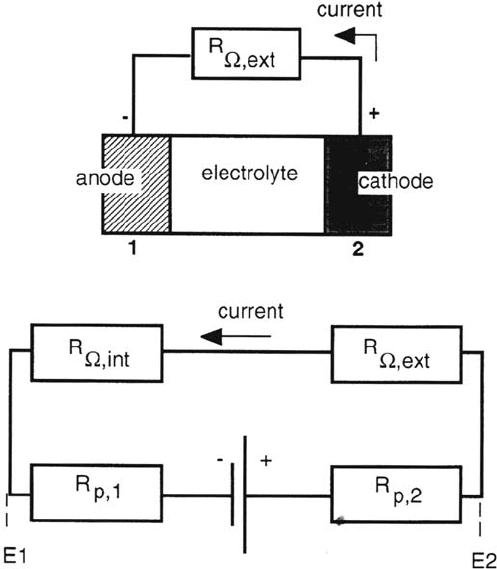
R
p,2
represent the polarization resistance of the anode and the cathode, respectively.
Because the electrode potential usually varies in a nonlinear way with current
density (for example, following Butler-Volmer kinetics) the value of the polarization
resistances R
p,1
and R
p,2
varies as a function of current density. The ohmic
resistances R
Ω,int
and R
Ω,ext
represent the internal cell resistance and the resistance
of the external circuit, respectively. The latter term is usually negligible.
It is easily seen from the figure that for a given potential difference ΔE
cor
the
current in the corrosion cell, and hence the corrosion rate at the anode, is the higher
the smaller the value of the different resistances present. For example, if one
increases the distances between the electrodes or the resistivity of the solution, the
current between anode and cathode will become smaller. In practice, the anode and
the cathode of a corrosion cell are usually not parallel and the current density
therefore varies locally with distance. This is schematically illustrated by Figure 8,
which shows an anode and a cathode in contact with each other in such a way that
their surfaces exposed to an electrolyte are in the same plane. Such a situation could
arise when two different metals are joined together. The figure shows schematically
Electrochemical Basis of Corrosion 15
Figure 7 Schematic presentation of a corrosion cell consisting of an anode and a cathode
immersed in the same electrolyte and in electrical contact through an external resistance.
Also shown is the electrical analogue of the corrosion cell including the voltage source
ΔE
corr
, the polarization resistances R
p,1
and R
p,2
of the anode and the cathode, respectively,
the internal resistance R
Ω,int
due to the electrolyte, and the external resistance R
Ω,ext
.
Copyright © 2002 Marcel Dekker, Inc.
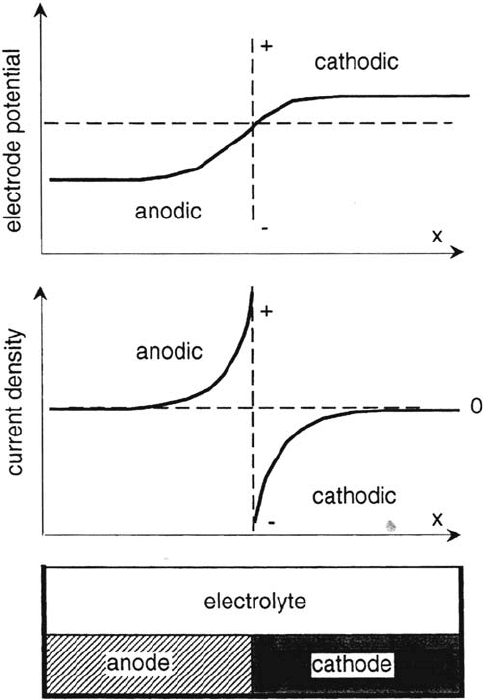
how the potential and the current density vary as a function of the distance from
the electrical contact. At the contact, the potentials of the cathode and the anode
are the same and where the local current density in principle should be infinitely
high. In reality, the current density at the contact will always be finite because of
limitations due to mass transport and reaction kinetics. Far from the contact the
value of the potential approaches that corresponding to the corrosion potential of
the metals in the absence of galvanic coupling. The current density in the metal
far from the contact becomes zero. The distance where this occurs depends on
geometric and electrochemical parameters and on the electrolyte conductivity.
The variation of current density and of potential as a function of distance can
in principle be calculated using the theory of current and potential distribution in
electrochemical systems [5,6,11]. In practical corrosion systems this is rarely done,
16 Landolt
Figure 8 Schematic presentation of the variation of the current density and of the
electrode potential as a function of the distance from the contact of two metals immersed
in an electrolyte.
Copyright © 2002 Marcel Dekker, Inc.
however, because numerical simulations taking into account all meaningful
parameters are rather onerous.
Corrosion cells not only are formed when two different metals are in contact
with each other but also can be the result of differences in the corrosive environment.
A typical example is differential aeration cells, which are due to differences in
accessibility of oxygen to the surface of a metal. Because the value of the corrosion
potential depends on the kinetics of the anodic as well as the cathodic partial
reactions, a different accessibility of oxygen results in a difference in corrosion
potential and hence in the establishment of a corrosion cell.
REFERENCES
1. A. J. Bard, R. Parsons, and J. Jordan, Standard Potentials in Aqueous Solutions,
Marcel Dekker, New York, 1985.
2. M. Pourbaix, Atlas of Electrochemical Equilibria in Aqueous Solutions, 2nd ed,
NACE, Houston, 1974.
3. K. J. Vetter, Electrochemical Kinetics, Academic Press, New York, 1967.
4. J. O’M. Bockris and A. K. N. Reddy, Modern Electrochemistry, Plenum, New York,
1970.
5. H. Kaesche, Die Korrosion der Metalle 3, Springer-Verlag, Berlin, 1990.
6. D. Landolt, Corrosion et chimie des métaux, Presses polytechniqes et universitaires
romandes, Lausanne, 1993.
7. M. Stern and A. L. Geary, J. Electrochem. Soc. 104:56 (1957).
8. C. Wagner and W. Traud, Z. Electrochem. 44:391 (1938).
9. V. Levich, Physicochemical Hydrodynamics, Prentice Hall, Englewood Cliffs, NJ,
1962.
10. R. D. Grimm, A. C. West, and D. Landolt, J. Electrochem. Soc. 139:1622 (1992).
11. J. Newman, Electrochemical Systems, 2nd ed., Prentice Hall, Englewood Cliffs, NJ,
1991.
Electrochemical Basis of Corrosion 17
Copyright © 2002 Marcel Dekker, Inc.
2
Introduction to Surface Reactions:
Adsorption from Gas Phase
Jacques Oudar
Laboratorie de Physico-Chimie des Surfaces, Université Pierre et Marie Curie, Ecole
Nationale Supérieure de Chimie de Paris, Paris, France
INTRODUCTION
Atoms at a metal surface exhibit unsaturated bonds that are available for fixing
reactive species, atoms or molecules, present in the gas or liquid surrounding the
surface. Such a reaction, when limited to one monolayer or a fraction of a mono-
layer, is known as an adsorption phenomenon or chemisorption. Similarly, atoms
present in the bulk metal may diffuse toward and enrich the surface by so-called
thermal segregation. Segregation may also occur by selective evaporation of the
metal in vacuum or in an inert gas or by selective dissolution of the metal in a liquid
phase (anodic segregation). Whatever the mechanism of surface enrichment, there
is strong experimental evidence that the same structural and chemical states can
be achieved by adsorption or segregation.
The presence of adsorbed species at the metal-liquid interface may greatly
influence electrochemical processes involved in corrosion, such as anodic
dissolution, cathodic reaction, or passive film formation. Most of our understanding
of adsorption has been acquired by studying gas-metal interactions at low pressure
by means of ultrahigh-vacuum techniques. Low-pressure data cannot be directly
related to electrochemical interfaces without some caution. On a polarized
interface, the adsorption of charged species such as ions is potential dependent
and requires the replacement of solvent molecules. Adsorbed ions may also retain
part of their charge and interact with the solvent molecules. There is good evidence
that small cations retain part of their solvated shell in the adsorbed state. It is
expected that similarities between both kinds of adsorption will be more significant
in systems characterized by strong interactions and nearly complete ion discharge.
In many systems, the reaction may proceed beyond the adsorption stage to
the formation of a bulk compound; this transition may occur at gas-metal as well
at metal-liquid interfaces.
In the following we will especially refer to adsorbates that play a direct role
in electrochemical processes (hydrogen, oxygen, water) or that may influence the
19
Copyright © 2002 Marcel Dekker, Inc.
formation of the passive layer (carbon, nitrogen, sulfur, halogens). We will focus
mainly on transition metals as substrates.
Finally, a short introduction to gas adsorption on oxides will be given.
We will examine especially water adsorption, which is of special interest for
understanding the behavior of passive films in aqueous solution.
MOLECULAR VERSUS DISSOCIATIVE ADSORPTION
Diatomic gases such as CO, NO, N
2
, O
2
, and H
2
can be molecularly or dissociatively
adsorbed on metals. In the case of CO and NO, the borderline between both types
of adsorption has been determined [1]. At room temperature N
2
is dissociatively
adsorbed on W, Mo, and Fe, but not on Ni and Cu; O
2
on all metals but Au; and
H
2
on transition metals but not on Al, Ag, and Au.
The transition from molecular adsorption to dissociative adsorption for a
homopolar diatomic molecule is well represented by the classical Lennard Jones
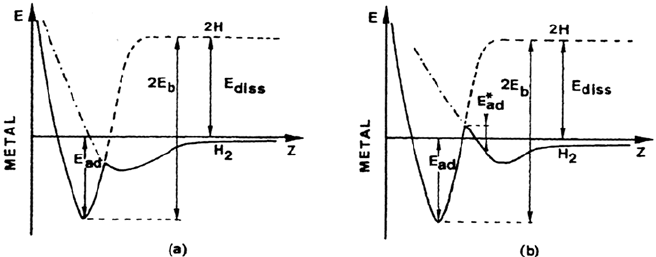
diagram. In this diagram, shown in Figure 1 for hydrogen, the potential energy of
the system is plotted as a function of the distance of the molecule to the metal surface
defined by the first plane of metal atoms. One curve corresponds to molecular
hydrogen, the other to hydrogen atoms. In the first shallow minimum, hydrogen is
molecularly adsorbed. As the metal-hydrogen distance decreases, dissociation
may occur. The much deeper potential well close to the metal surface corresponds
to chemisorbed hydrogen atoms. The energy path is represented in Figure 1 by the
solid line. The dissociation takes place at the intersection between the two potential
curves according to
H
2
(g) + 2M → 2M – H
ad
As shown in Figure 1, the intersection point may be located at positive or at negative
potentials. When it is at a positive potential, the adsorption process is activated and
the height of the activation barrier or activation energy is denoted by E
*
ad
. When the
intersection point is below E = 0, the dissociative adsorption of hydrogen is
non-activated. This is the case for the chemisorption of hydrogen on most transition
metals. The energy balance gives E
ad
+ E
diss
= 2E
b
where E
ad
is the adsorption
20 Oudar
Figure 1 Schematic potential energy diagram for (a) nonactivated and (b) activated
chemisorption of hydrogen on a metal surface.
Copyright © 2002 Marcel Dekker, Inc.
energy, E
diss
is the energy required for the dissociation of the H
2
molecule
(432 kJ /mol), and E
b
is the hydrogen-metal bond energy.
ENERGETICS OF ADSORPTION
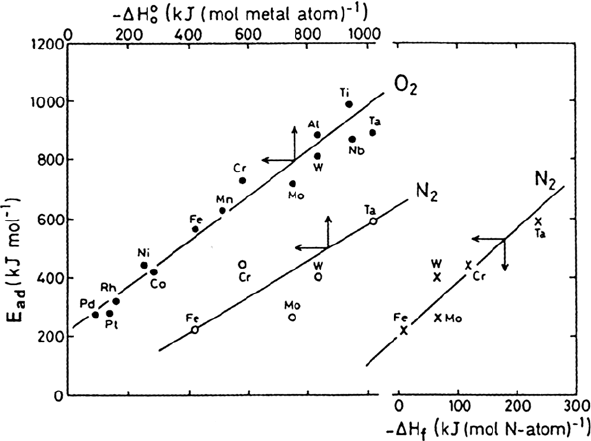
General Trends
Based on analyzing data for many gas-metal systems, some general remarks can
be made:
1. The heat of adsorption is of the same order of magnitude as the heat of
formation of the most stable bulk compound. A linear correlation between
these two thermodynamic quantities has been experimentally established, as
exemplified for O
2
and N
2
in Figure 2 [2,3] and for S
2
in Figure 3 [4].
2. The strength of the adsorbate bond follows the order C > N > O > S > H.
3. The heat of adsorption is not very sensitive to the surface structure. Variation
from one plane to another does not exceed 10% of the total heat of adsorption.
Stronger bonding usually occurs on rougher planes on an atomic scale.
4. The heat of adsorption and consequently the strength of the chemical
adsorption bond usually decrease with increasing surface coverage due to
adsorbate-adsorbate repulsive interactions.
Data mostly derived from studies performed on single crystals are summarized
next for some selected systems.
Adsorption From Gas Phase 21
Figure 2 Correlation between the initial heat of chemisorption (E
ad,0
) and the corresponding
heat of oxide formation for O
2
(•) and N
2
(o) or nitride formation (for N
2
(x)). (From Refs.
2 and 3.)
Copyright © 2002 Marcel Dekker, Inc.
Hydrogen
Hydrogen adsorption on pure metals has been reviewed [5–7]. Fundamentals of
hydrogen-metal interactions with special reference to the hydrogen degradation of
materials in aqueous solution can be found in Ref. 8.
Experimental data obtained on single crystals (Table 1) show that the initial
heat of adsorption does not depend much on the surface orientation. As seen on Ni,
22 Oudar
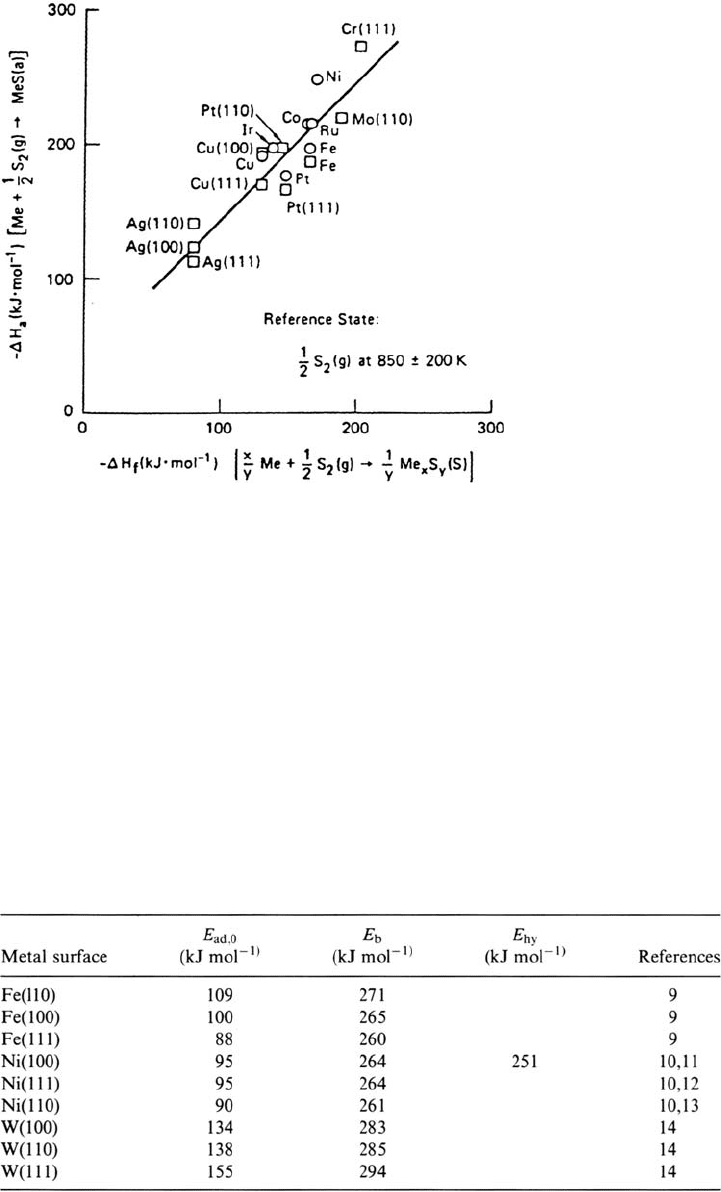
Figure 3 Correlation between the heat (enthalpy) of sulfur adsorption (ΔH
a
)at
half-saturation coverage and the corresponding heat (enthalpy) of sulflde formation (ΔH
f
).
(From Ref. 4.)
Table 1 Some Selected Values for Initial Heats of Adsorption (E
ad,0
), M–H Binding
Energy (E
b
), and Bond Strength, of Diatomic Hydride Molecules (E
hy
)
Copyright © 2002 Marcel Dekker, Inc.
the hydrogen-metal binding energy has the same order of magnitude as the bond
energy in the corresponding hydride. Hydrogen adsorption is sensitive to surface
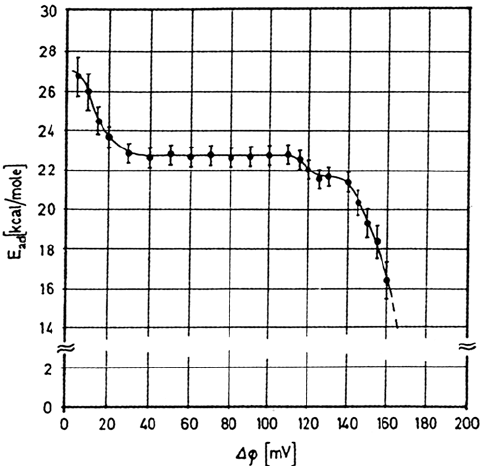
defects. The preferential adsorption on monoatomic steps is reflected by the
increase of the value of E
ad
observed on Ni(100) at small coverages (Fig. 4). When
the hydrogen coverage increases, the isosteric heat of adsorption decreases as far
as repulsive lateral interactions between adsorbed atoms become significant. Most
data in Table 1 concern H species adsorbed at low H
2
pressures (<10
–4
torr). The
nature and binding energy of H adsorbed species formed at higher pressures can
be different. Their possible role in heterogeneous catalysis as deduced from
experiments made on dispersed metals is discussed in detail in Ref. 5.
A survey of the elements forming bulk hydrides is given in Ref. 6.
Carbon
Despite the fact that carbon is one of the most common elements in many
metals, there are no precise data on its possible role in wet corrosion. The importance
of carbon in metallurgy and heterogeneous catalysis (methanation) has motivated
precise studies of metal single crystals. During exposure of Ni to CO, two kinds of
surface carbon deposit have been identified by Auger electron spectroscopy (AES)
[15]. The first one, limited to a fraction of a monolayer, is the “carbidic form,” so
named because of its similarity to the Auger spectrum of bulk nickel carbide. The
second one is graphite. The carbidic form is an intermediary in the CO hydro-
genation, whereas the graphitic carbon deactives the surface. The transformation of
Adsorption From Gas Phase 23
Figure 4 Isosteric heat of adsorption (kcal/mol) for hydrogen on Ni(100) surface as a
function of hydrogen-induced work function change (which is a direct measure of the surface
concentration of hydrogen). (From Ref. 10.)
Copyright © 2002 Marcel Dekker, Inc.
the carbidic form into graphite occurs beyond a critical carbon coverage. The two
carbon states can be produced by exposure of nickel or iron to gases other than CO
(C
2
H
4
or C
2
H
6
) or by surface segregation.
Most data for determining the binding energy of adsorbed carbon in its
carbidic form have been obtained by segregation studies of single crystals following
a procedure given in Ref. 16. The thermodynamic properties of the different forms
of surface carbon that can be present on Ni and Fe are described in Refs. 17 and 4.
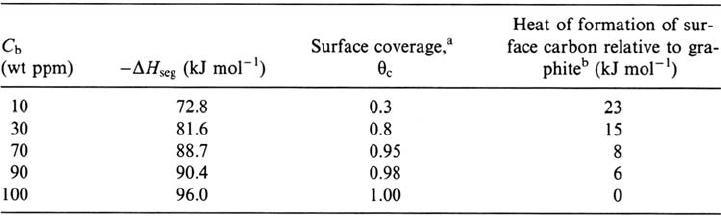
As an example, results obtained for Fe(100) are shown in Table 2. On this surface,
carbon, irrespective of the coverage, is less strongly bonded in the adsorbed layer
than in graphite. The average enthalpy of carbon segregation on Fe(100) (–85 kJ
mol
–1
) is significantly lower than that to grain boundaries (–57 kJ mol
–1
). Surface
site competition between Si and C has been reported on Fe-Si alloys [18,19].
As evidenced by its high reactivity in gaseous hydrogen [15], a “carbidic”
adlayer is probably highly reactive in the presence of hydrogen cathodically
formed in acid solution. The evolution of CH
4
during the cathodic hydrogen
decarburation of steel at room temperature presumably originates from such a
carbon deposit [20]. One may expect the formation of the passive film to be
partially or totally inhibited on a surface partially or totally covered by graphite
that is inert toward oxygen or H
2
O at room temperature.
Nitrogen
Nitrogen adsorption has been especially studied on iron, which is the basis of the
industrial catalyst for ammonia synthesis. This catalytic reaction is now relative-
ly well understood [21]. The main function of iron is to activate the very stable
N
2
molecule by dissociative adsorption. Results in Table 3 show that the Fe–N
binding energy is approximately the same on the three main low-index planes.
However, the rate of N
2
dissociation decreases by many orders of magnitude
when going from Fe(111), the most active for ammonia synthesis, to the less
active Fe(110).
On W the metal-nitrogen bond energy is estimated to be 640 kJ mol
–1
irrespective of the surface orientation [24,25], about 60 kJ mol
–1
higher than
for the Fe–N. Nitrogen, like carbon, dissolves interstitially into cc iron in small
24 Oudar
Table 2 Thermodynamic Properties of Carbon on Fe(100) [4]
a
At 923 K .
b
Data from Ref. 8 based on heat of solution of 95 kJ mol
–1
.
Copyright © 2002 Marcel Dekker, Inc.
