Marcus P. Corrosion mechanisms in theory and practice
Подождите немного. Документ загружается.


concentrations. The bulk nitrogen concentration (N
b
) expressed in wt %, in
equilibrium with nitrogen at 1 atm is given by [26]
N
b
= 0.098 exp – (ΔH
s
/RT)
with ΔH
s
= 30.2 kJ mol
–1
(endothermic).
The heat of surface segregation ΔH
seg
is the difference between the heat of
chemisorption of 1/2N
2
and the heat of segregation
ΔH
seg
= –108.5 – 30.2 = –138.7 kJ mol
–1
(exothermic)
From direct segregation experiments with Fe-N alloys the value ΔH
seg
= –110 kJ
mole
–1
has been found [19].
Oxygen
Data in Figure 2 refer to polycrystalline metals. As shown in Table 4, the heat of
oxygen adsorption is approximately the same for the three metals Cr, Mo, and W.
By contrast, the enthalpy of atomization is much lower for Cr than for Mo and
reaches a very high value for W. This indicates that the Cr–Cr bond strength is
much lower than the bond strength for the other metals. It is expected that
phenomena involve disruption of the metal lattice in the presence of oxygen such
that the transition from adsorption to oxidation will be much easier on Cr. This
aspect is considered in more detail in a later section.
Sulfur
Most data on the thermodynamics of sulfur adsorption were derived from adsorption
isotherms achieved in H
2
S-H
2
gas mixtures [28,29]. At high temperature, sulfur is
reversibly adsorbed on a metal surface in a range of H
2
S partial pressures below that
required for the formation of the most stable sulfide. At a fixed temperature the
Adsorption From Gas Phase 25
Table 3 Initial Heat of Nitrogen Adsorption (E
ad,o
), E
bM-M
, and M-N Binding Energy
on Iron [22,23]
Table 4 Thermodynamic Data for Cr, Mo, and W [27]
Copyright © 2002 Marcel Dekker, Inc.

adsorption isotherm reflects the surface coverage at equilibrium with the H
2
S-H
2
gas composition that determines the chemical potential of sulfur in the gas phase.
The isosteric heat (enthalpy) of adsorption referred to H
2
S can be determined
from isotherms measured at different temperatures by means of the Van t’Hoff
equation:
[∂ ln(PH
2
S/PH
2
)/∂T]
θ
= −Δ
Η
θ
/RT
2
where θ is the sulfur coverage.
From ΔH
θ
the heat of adsorption with S
2
(g) as the reference state can be
deduced, as well as the binding energy of adsorbed sulfur. Exchange between the
bulk solid solution and the surface, namely segregation and dissolution, can also
be taken into account. The various equilibria between the surface and either the
bulk metal or the gas phase are interrelated according to:
26 Oudar
The heat of surface segregation is equal to the difference between the heat of
adsorption and the heat of sulfur dissolution from the gas phase. Data obtained on
nickel and iron are shown in Table 5. In this table ΔH
a
corresponds to the reaction
M + ½ S
2
→ MS
ad
ΔH
a
ΔH
f
is the heat of the bulk sulfide formation according to
x/y M + ½ S
2
→ 1/yM
x
S
y
ΔH
f
All values refer to half-saturation coverage by sulfur.
The heat (enthalpy) of formation of chemisorbed sulfur on nickel with respect
to ½ S
2
is –247 kJ mol
–1
, i.e., –158 kJ mol
–1
with H
2
S as the reference state for
sulfur (the heat of formation of gaseous H
2
S is 89 kJ mol
–1
). It is ~ 75 kJ mol
–1
more
exothermic than the enthalpy of formation of Ni
3
S
2
. This means that S adsorbed is
energetically more stable than S in Ni
3
S
2
. A similar trend is observed for Fe. Indeed,
Table 5 Heat of Sulfur Adsorption ΔH
a
(Referred to ½ S
2
), Heat of Sulfide Formation
ΔH
f
(Referred to 1/y M
x
S
y
), Heat of Dissolution from the Gas Phase (
½
S
2
), and Heats of
Segregation
a
In Ref. 29, a higher absolute value was derived by using wrongly for ΔH dissol. the value
corresponding to Fe
γ
instead of Fe
α
.
Copyright © 2002 Marcel Dekker, Inc.
it was shown in Figure 3 that there is a correlation, for many metals, between the
heat of chemisorption and the heat of formation of the bulk sulfide that fits
approximately the relation ΔH
ad
= 1.25ΔH
f
(1/y M
ν
S
ν
). The fact that chemisorbed
sulfur is more stable than the three-dimensional sulfide has important consequences
for corrosion processes. Indeed, sulfide on the surface may be dissolved while a
monolayer or a fraction of it remains on the surface and changes, as described later
in sulfur-assisted corrosion mechanisms (Chapter 9). The metal-sulfur bond strength
is easily obtained from the heat of adsorption. The nickel-sulfur bond strength is
–464 kJ mol
–1
, based on –217 kJ mol
–1
as the heat of formation of monoatomic
gaseous sulfur. In the same way, the S–Fe bond strength is found to be –414 kJ mol
–1
.
The heat of segregation of sulfur onto metal surfaces is generally markedly
exothermic. Very low levels of sulfur dissolved in the bulk equilibrate with a very
high surface coverage. Although segregation is an exothermic process, it takes
place only at high enough temperatures to allow the diffusion of sulfur in the solid
state. Usually temperatures above 600°C are required for sulfur to diffuse to the
surface; at 800°C it takes a few minutes to obtain the saturation of the surface by
sulfur adsorbed on a nickel sample containing ~ 10 ppm sulfur. The diffusion
coefficient of sulfur in nickel is [36]
D(1073 – 1498 K) = 1.4 exp(–218.6/RT)
Similarly, sulfur may enrich grain boundaries even when present in the metal at
very low concentration. Values of the heat of grain boundary segregation are
shown in Table 5. One observes that the sulfur binding energy varies in the order
free surface > grain boundaries > solid solution.
Grain boundary segregation of nonmetallic impurities influences many
chemical and mechanical properties [37]. It increases the metal brittleness, favors
hydrogen embrittlement, decreases the intergranular fatigue strength or the creep
rupture life, and favors intergranular corrosion and stress corrosion cracking. In
this respect, sulfur is one of the most detrimental elements. As shown on bicrystals
of nickel, a close correlation exists between the intergranular sulfur segregation
and the intergranular corrosion [38]. At 625°C, the intergranular segregation was
observed for a sample with a sulfur content as low as 0.0005%. Thermodynamic
data on sulfur dissolution in pure metals and alloys and on sulfur diffusion have
been reviewed in Ref. 39.
Halogens
Halogen chemisorption on metals is of special importance in corrosion. This subject
has been reviewed by various authors [40–42]. At room temperature, halogen
molecules are dissociatively adsorbed on transition metals and, depending on the
system and the halogen pressure, the reaction may proceed beyond the adsorption
stage to give halogenide. For a given metal, the halogen binding energy decreases
in the order F > Cl > Br > I. Data at low coverages deduced from desorption kinetics
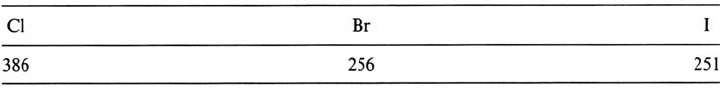
or from the heat of formation of halogenides are shown for Fe in Table 6.
For the refractory metals Nb, Mo, Ta, and W, the heat of adsorption of F and
Cl at low coverage was observed to be nearly identical 450 ± 20 kJ mol
–1
[45]
whatever the metal or the surface orientation. For Br on Nb and Mo the same value
Adsorption From Gas Phase 27
Copyright © 2002 Marcel Dekker, Inc.
of 350 ± 10 kJ mol
–1
was measured [45]. On nickel, chlorine preadsorption totally
inhibits the postadsorption of oxygen, whereas oxygen preadsorption has no
influence on additional adsorption of chlorine [46].
WORK FUNCTION VARIATIONS
During adsorption a charge transfer takes place between the metal and the adsorbate
and a dipole moment is established. The effective dipole moment μ of an individual
adsorbate-metal complex can be derived from the measurable change of the work
function Δφ at a coverage n
s
(particles/cm
2
) according to the relation Δφ = 4 Π n
s
μ.
As a consequence of the existence of a dipole layer, pairwise repulsive interactions
may develop.
For hydrogen, dipole-dipole interactions do not operate to a significant
extent and only indirect interactions have to be considered. The indirect interactions,
the forces that operate via the metal substrate, arise from a mutual coupling of the
valence shells of the two adatoms through the substrate valence band. All other
contributions to the interaction energy, essentially short range in nature, are
completely negligible. In some cases such as H/Fe(110) the interactions can be
reasonably treated by the simple lattice gas approximation [47], the calculated
interaction energies being in agreement with the experimental values (4–8 kJ mol
–1
).
As stated before, the order of magnitude of the pairwise repulsive energy is
generally about 10% of the chemisorption energy.
The variations of the work function on the three low-index faces of Fe with
hydrogen adsorption at 140 K are shown in Figure 5. The main characteristics are
(a) the small changes of Δφ, indicating a low adsorbate dipole moment, a minor
net charge transfer between adsorbate and substrate implying some covalent
character of the hydrogen metal bond; (b) the nonlinear variation of φ with coverage,
caused by interactions between adsorbed particles; and (c) the changes of opposite
signs for Δφ on the various crystal faces, namely a decrease of the work function
by 85 mV for Fe(110) in contrast to the increases by 75 and 240 mV for the (100)
and (111) faces, respectively. The negative change of Δφ is somewhat unexpected
because hydrogen is considered to be negatively charged on transition metal surfaces.
A similar result has been reported for H/Pt. A possible explanation is related to the
location of the H atom with respect to the metal surface plane, which affects both
the sign and magnitude of Δφ.
For electronegative species such as oxygen, sulfur, or halogen, the electron
transfer takes place from the metal to the adsorbate, as indicated by the increase of
the work function. Electronic transfer in the opposite direction does occur for less
electronegative adsorbates, Se and Te. This trend is illustrated for Ni(100) covered
28 Oudar
Table 6 Metal-Halogen Binding Energy on Iron (kJ mol
–1
) [43,44]
Copyright © 2002 Marcel Dekker, Inc.

by the same amount of adsorbate (0.5 adsorbed atom per surface metal atom)
(Table 7).
Work function variations induced by sulfur at saturation on single crystals of
copper are shown in Table 8. These variations are larger than on nickel and vary
significantly from one orientation to another.
For S and O, the charge transfer never exceeds one tenth of one electron per
adsorbate atom and the metal-adsorbate bond can be considered mainly covalent.
Results for halogens on Fe (100) (Table 9) show that the charge transfer parallels
the electronegativity of the adsorbate, decreasing in the order Cl > Br > I.
When the adsorption occurs in solution, the resulting charge transfer may
significantly modify the metal-electrolyte potential and affect the electrochemical
processes at the interface. It was shown that on silver the zero charge potential
moves toward higher values with sulfur adsorption [51]. The variation exactly
corresponds to the increase of work function induced by sulfur. The linear correlation
between the zero charge potential and the work function is now well established
both theoretically [52] and experimentally [53].
STRUCTURE OF ADSORBED LAYERS
During the last 20 years, considerable effort has been spent on solving the structure
of adsorbed layers. Achievement in this matter is well illustrated by a handbook
containing a catalog of these structures [54] and a database with a computer graphics
Adsorption From Gas Phase 29
Figure 5 Changes in the work function with hydrogen adsorption on the low-index faces
of iron. (From Ref. 9.)
Table 7 Work Function Variation for Some Elements Adsorbed on Ni(100) [48]
Copyright © 2002 Marcel Dekker, Inc.

program [55]. Only a few examples of structures will be given here. They have
been selected in order to illustrate some of the more general features as well as the
diversity of the overlayer crystallography.
In dilute phases (low coverage), chemisorbed atoms are usually not ordered.
Ordering appears at intermediate coverages when interactions between adsorbed
atoms become significant. On dense phases, atoms tend to be localized in the sites
of highest coordination with the highest symmetry: threefold hollow sites on face-
centered cubic (fcc) (111) surfaces, fourfold hollow sites on fee or body-centered
cubic (bcc) (100) surfaces. For electronegative species, the bond distances between
the chemisorbed atom and nearest-neighbor metal substrate atoms are in many cases
nearly the sum of the covalent radius of the metal and the covalent radius of the
adsorbed atom. This is the case for sulfur on nickel, whose crystallographic data are
shown in Figure 6 [56]. These data refer to structures with a small coincidence mesh
between the overlayer and the metal substrate: respectively c(2 × 2) on Ni(100) and
Ni(110) (coverage one half) and (2 × 2) on Ni(111) (coverage one fourth).
Outward surface relaxation induced by adsorption is frequently observed. For
S and O adsorbed on Ni(100) on which the same structures p(2 × 2) and c(2 × 2)
are successively formed at respective coverages one fourth and one half, it was
observed that the first layer distance increases linearly with increasing coverage
and that oxygen is more effective than sulfur [57]. This increase of the metal-metal
bond length certainly provokes a strong decrease of the metal-metal bond strength.
A more complex surface rearrangement occurs when carbon is adsorbed on
Ni(100) [58,59]. Half a monolayer of carbon induces a distortion of the outermost
layer of nickel. On this distorted layer, carbon atoms occupy fourfold hollow sites.
Similar surface rearrangement with possibly very little difference in the bond
strength has been discussed for C/Co(100) [59,60], N/Ni(100) [61], and N/Cr(100)
[62]. Surface stresses generated by carbon, oxygen, and sulfur adsorbed on Ni(100)
have been determined [63]. Results support the concept that surface stress would
be the driving force for the surface reconstruction induced by carbon.
During the surface reconstruction, new adsorption sites are created in order to
optimize the surface chemical bond. In the cases of S on Fe(110) [64] and S on
Ni(111) [65,66], sites with fourfold symmetry seem to prevail. The most drastic
surface rearrangement concerns the S/Ni(111) system, in which the first layer of
nickel reorganizes from a hexagonal plane into a nearly perfect (100) plane in order
30 Oudar
Table 8 Work Function Variations for S Adsorbed on Cu Single Crystals [49]
Table 9 Work Function Variations at Saturation for Halogens on Fe(100) [50]
Copyright © 2002 Marcel Dekker, Inc.

to accommodate the sulfur overlayer (Fig. 7). This overlayer strongly resembles
that formed on Ni(100).
Another well-documented case of surface reconstruction is the H/Ni(110)
system [67]. Above the critical concentration of one monolayer, the topmost
nickel atoms are displaced by approximately 0.4 Å in one direction. This process
may be considered as the prequisite for hydride formation. The importance of
restructuring processes for the chemical reactivity of metals has been discussed
[68]. When the strength of the adsorbate-substrate bond is lower than that of the
substrate-substrate bond, the driving force is not sufficient to induce significant
surface reconstruction. In this case complex structures frequently observed at
high coverages can be described in terms of periodic antiphases or in terms of
close-packed layers of atoms adsorbed on the undisturbed substrate. Halogens on
the dense planes of transition metal seem to belong to this category [42].
Restructuring processes involving surface diffusion of metal atoms over
distances larger than one atomic distance may also be induced by adsorption at
coverages near saturation. In this case, the original surface becomes unstable and
breaks into new crystallographic orientations selectively stabilized by the adsorbate.
This phenomenon, usually called faceting, occurs by step coalescence. The reversible
Adsorption From Gas Phase 31
Figure 6 Schematic configurations of S/Ni(l10)–c(2×2), S/Ni(100)–c(2×2) and
S/Ni(111)–p(2× 2) structures. (From Ref. 56.)
Figure 7 Surface reconstruction induced by adsorption of sulfur on Ni(111). (From Refs.
65 and 66.)
Copyright © 2002 Marcel Dekker, Inc.
character of the faceting process associated with the reversibility of adsorption
was demonstrated first in the oxygen-silver system [69]. For iron-chromium alloys
[70] and pure iron [71] the conditions for faceting induced by oxygen adsorption
have been determined. In the case of iron, faceting inhibits the dissociation of
nitrogen on the metal surface [72]. Presumably, the N
2
dissociation step is supp-
ressed by the presence of adsorbed oxygen on the surface. A number of studies
have established the close relationship between faceting and the thermodynamics
of adsorption. Based on the analysis of Herring [73], conditions of faceting have been
evaluated and experimentally demonstrated for oxygen adsorbed on silver [74]. The
influence of carbon on the surface stability of nickel has been extensively studied [75].
TRANSITION FROM ADSORPTION TO OXIDATION
The transformation of the overlayer into a bulk compound is an important area of
study. This aspect is discussed in detail for the oxidation of nickel and refractory
metals Cr, Mo, and W [26]. Chapter 5 of this book is essentially focused on the
oxide growth of thin oxide films. It is well accepted that relaxation or surface recons-
truction plays an important role in this transformation by loosening the metal-metal
bonds. When the reconstructed layer presents strong structural similarities to a
dense plane of the bulk compound, it can be considered as a transition layer between the
metal lattice and the bulk compound lattice. According to a theoretical model [76],
the penetration of the nonmetal atoms into the metal and its conversion into bulk com-
pound would be assisted by the electric field resulting from the charge transfer from
the metal to the adsorbed atom. The magnitude of this field depends on the density
of adsorbed atoms. As a consequence, the onset of the bulk compound formation
occurs only beyond a critical concentration of adsorbed atoms in the overlayer.
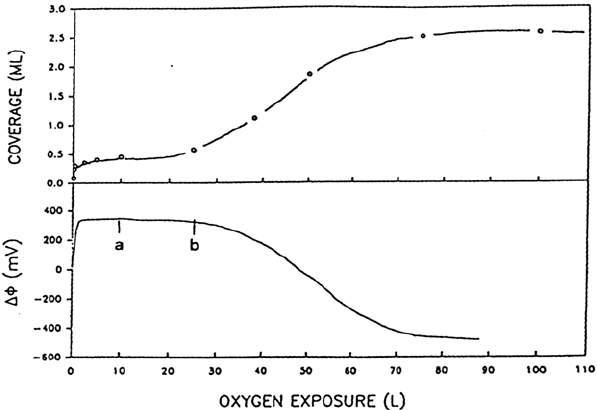
The onset of oxidation has been accurately followed on Ni(100) by measuring
simultaneously the absolute amount of oxygen fixed on the surface by Rutherford
backscattering spectrometry and work function variations as a function of the
exposure time. Results are shown in Figure 8 [77]. At first the work function
increases, indicating a charge transfer from the metal to the adsorbed oxygen
atoms. Well-ordered p(2 × 2) and c(2 × 2) structures successively appear. The onset
of the oxide nuclei is indicated by the decrease of the work function. It occurs
when about a half-monolayer of oxygen is adsorbed in the fourfold symmetry
sites and 0.04 to 0.077 mL of oxygen is incorporated into subsurface sites.
From a comparison of the behavior of Cr, Mo, and W in the presence of
oxygen, some general trends can be drawn [27]. These trends result directly from
the characteristics of the metal and from the energetics of the metal-oxygen
interactions as expressed in Table 4. The propensity for the surface to be
reconstructed under the influence of oxygen and for the metal to be oxidized varies
in the order Cr > Mo > W. It is the reverse of the order of metal-metal bond energies.
For a given metal the ease of reconstructive oxidation varies in reverse order to the
surface density of metal atoms (111) > (100) > (110). This emphasises the necessity
of breaking the metal-metal bonds in order to form the oxide. This breaking is, as one
would expect, favored by a low metal-metal bond strength and a high metal-oxygen
bond strength. The best example is chromium. On this metal the energy dissipated by
32 Oudar
Copyright © 2002 Marcel Dekker, Inc.
the interaction of oxygen is sufficiently large to disrupt the metal-metal bond
directly, without the formation of an oxygen adlayer. Zr and Ta behave similarly
to Cr. By contrast, on W very stable adsorbed overlayers are formed before the
appearance of the oxide. On Mo, which represents an intermediate case, open sur-
faces behave more like Cr and dense surfaces like W.
ADSORPTION AND SEGREGATION ON ALLOYS
The surface composition of an alloy at equilibrium is rarely the same as the bulk
composition. In most cases, the surface tends to be enriched with the metal
component having the lower surface energy. In addition, several mechanisms can
affect the surface composition with respect to its equilibrium composition:
1. In vacuum or neural atmospheres, one component may be selectively
evaporated.
2 In reactive gases, one component may be selectively combined with the
gas to form two- or three-dimensional compounds.
3. In electrolyte, one component may be selectively dissolved and the surface or
the region near the surface selectively enriched with the other component.
These surface modifications may significantly affect the resistance of the
alloy to dry or wet corrosion. Alternatively, corrosion tests can be used as a probe
for determining the surface composition of the alloy. Some of these aspects can be
Adsorption From Gas Phase 33
Figure 8 Adsorption of oxygen on Ni(100). (Top) Absolute coverage of oxygen in
mono-layer (ML) as a function of oxygen exposure in Langmuir (L) at 329 K. (Bottom)
Work function change corresponding to the data in the upper panel. (a) Onset of the filling
of subsurface sites; (b) onset of the oxide nucleation. (From Ref. 77.)
Copyright © 2002 Marcel Dekker, Inc.
illustrated with reference to Fe-Cr alloys. When these alloys are treated at high
temperature under pure flowing hydrogen or in vaccum, chromium is selectively
evaporated. This phenomenon occurs at temperatures of ~ 1000°C, at which the
chromium partial pressure becomes appreciable (~10
6
atm). Alloys superficially
depleted in chromium have lower resistance to dry oxidation or corrosion in acid
solutions. After treatment in pure hydrogen at 1200°C, the chromium content on
the surface of Fe-18Cr or Fe-23Cr alloys can reach a value well below 13%. As a
result, an alloy that is initially passive in acid solution becomes active and is
dissolved until the layer depleted to below the critical value of 13% Cr is consumed.
Chromium evaporation at high temperature can be suppressed by introducing traces
of H
2
S or H
2
O into the hydrogen atmosphere [78]. At a critical partial pressure of H
2
S
or H
2
O well below that corresponding to the formation of bulk compounds (sulfide
or oxide), chromium combines selectively to give a two-dimensional sulfide or
oxide. These very stable two-dimensional compounds, identified by AES [79], act as
a barrier to chromium evaporation. The critical H
2
S or H
2
O partial pressure at
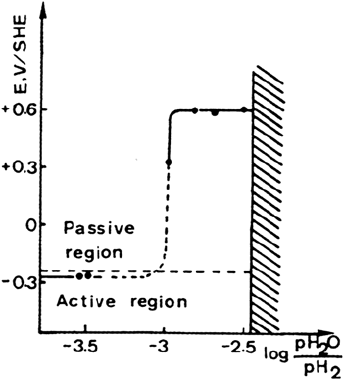
which the chromium evaporation is suppressed can be accurately determined by
measuring the corrosion potential of the alloy after the thermal treatment. This
potential, which depends on the H
2
S or H
2
O content, reflects the surface coverage by
sulfur or oxygen. Its variation with H
2
S or H
2
O partial pressure in the gas phase
shown in Figure 9 is analogous to the respective adsorption isotherms for sulfur or
oxygen at the temperature of the thermal treatment. In the case of sulfur, direct
determination of surface concentration has confirmed that the transition from an
active to a passive corrosion potential occurs at the transition from low to high sulfur
coverage [80].
Surface segregation induced by adsorption is a general phenomenon. The metal
element with the highest affinity to the reacting gas tends to be enrich surface
provided that the reaction temperature is high enough to permit bulk diffusion [81,82].
34 Oudar
Figure 9 Variation of potential of Fe-23Cr alloy as a function of prior thermal treatment
under different atmosphere compositions. (From Ref. 78.)
Copyright © 2002 Marcel Dekker, Inc.
