Bowker M., Davies P.R. (Eds.) Scanning Tunneling Microscopy in Surface Science, Nanoscience and Catalysis
Подождите немного. Документ загружается.

contrast to the usual Al
2
O
3
stoichiometry. The registry of the substrate and oxide is
provided by the interfacial Al atoms, which are bonded to the Ni rows of the NiAl
part of the interface. This leads to interfacial row matching and to the stress along the
[1 1 0] direction of the oxide, which causes the incorporation of line defects and
the observed antiphase domain boundaries [45]. The latter have also been modeled
in terms of an oxygen-deficient defect structure in the recent work of Kresse and
coworkers [48].
The NiAl(1 1 0) surface is not the only Al-containing alloy surface that can support
a highly ordered oxide overlayer. The formation of a well-ordered alumina film
on Ni
3
Al(1 1 1) by high-temperature oxidation has been reported first by Rovida
et al. [49, 50]. The Wandelt group has taken up the idea subsequently and has refined
the preparation procedure [51]. Rosenhahn et al. recognized from STM images [52]
that two long-range superstructures in the nanometer range can be identified: the
so-called network structure with a periodicity of approximately 2.4 nm and the dot
structure with a periodicity of approximately 4.2 nm, the dot structure being a
(H3 H3)R30
subset of the network structure. SPA-LEED and more extensive
STM investigations of Degen et al. [53] revealed that only the dot structure is a real
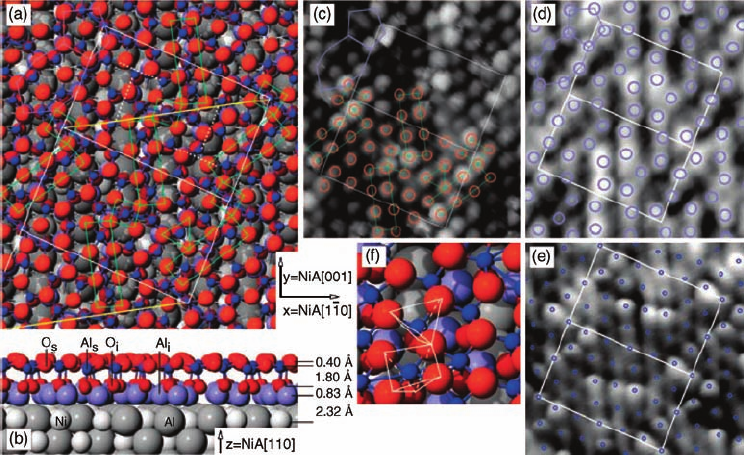
Figure 6.2 (a) Top and (b) side view of the
DFT- and STM-based model for the ultrathin
aluminum oxide film on NiAl(1 1 0).
(c–e) Experimental STM images of the film at
(c) and (d) room temperature and at (e) low
temperature. Sample bias voltage and tunneling
current values are (c) 2.5 mV/1.4 nA, (d) 0.2 V/
0.9 nA, and (e) 0.5 V/0.3 nA. Two oxide unit cells
are marked by white rectangles, the diagonal
along which the oxide is commensurate is
yellow, and the parallelogram enclosed by the
yellow and white lines delimits the simulation
cell. Green rectangles and squares highlight
oxygen atoms in a square arrangement. Circles
indicate the Al and O positions from (a) and
(b) in the corresponding colors. (f) Close-up of
the structure. (Reproduced with permission
from Ref. [47].)
154
j
6 STM Imaging of Oxide Nanolayer Model Systems
superstructure of the alumina film, and they refined the unit cell of the (H67 H67)
R48
superstructure to 4.16 nm. Since atomic resolution of the alumina film could
not be achieved in this latter work, the building blocks of the oxide layer and its
structural relation at the atomic scale to the substrate could not be established. The
full structural complexity of the alumina on Ni
3
Al(1 1 1) has been unraveled by the
noncontact atomic force microscopy (AFM) study by Gritschneder et al. [54], who
found that the main structural element of the oxide film is a lattice of hexagons that is
pinned to the periodicity of the substrate. The surface unit cell is defined by the dot
structure, with the network structure being formed by a honeycomb-like topographic
modulation as a result of the substrate-overlayer pinning. Recently, Kresse and
coworkers [55] have applied their experience with the alumina model on NiAl(1 1 0) to
the determination of the structure of alumina on Ni
3
Al(1 1 1). Combining exper-
imental STM images with extensive DFTmodeling, they found a structural similarity
between the alumina films on the two NiAl alloy substrates, with an AlOAlO
stacking sequence, square and triangular arrangements of atoms at the surface, and a
hole in the unit cell reaching down to the Ni
3
Al substrate. In simulated STM images,
these holes have been identified with the bright contrast of dots in the dot
structure [55]. It is these holes that provide the anchoring centers for the growth
of monodisperse metal clusters [56] and make this alumina overlayer an excellent
nanotemplate for growing regular arrays of nanoparticles.
In summary, the alumina nanolayers formed by the high-temperature oxidation
on NiAl alloy surfaces are structurally and chemically very different from the bulk-
terminated surfaces of the various Al
2
O
3
phases, and they thus provide very
prototypical examples of oxide phases with novel emergent properties because of
interfacial bonding and thickness confinement effects.
6.3.2
Titanium Oxide Nanolayers
There is considerable interest in titanium oxides owing to their importance in
many areas, such as photo-assisted oxidation, heterogeneous catalysis and gas
sensors [57–59]. Titanium dioxide (TiO
2
), in particular, is one of the most prominent
materials in the industrial catalysis used for the selective reduction of NOx [60, 61],
photocatalysis for pollutant elimination [62] or organic synthesis [63], photovoltaic
devices [64], sensors [65], and paints [66]. Additional applications include its use as a
food additive [67], in cosmetics [68], and as a potential tool in cancer treatment [69].
Titaniumoxide is one of the best-characterized model systems in surface science [70].
Most of the studies were performed on single-crystal surfaces, but at present,
ultrathin Ti-oxide films grown on metallic substrates are a subject of intensive
investigation. Self-assembled, low-dimensional Ti-oxide nanostructures could be of
potential interest for applications in electronic devices, nanocatalysts, and gas
sensors. Controlling the oxidation state and the dimensions of these nanostructures
may allow the production of a new class of technologically important materials.
Ultrathin Ti-oxide films have been grown on various metal surfaces, Pt(1 1 1)
[71–75], Pt(1 0 0) [33, 34], Pt(1 1 0) [76], Mo(1 0 0) [77, 78], Mo(1 1 0) [79, 80],
6.3 Case Studies: Selected Oxide–Metal Systems
j
155
Mo(1 1 2) [81], W(1 0 0) [82], Ni(1 1 0) [83–86], and Ru(0 0 0 1) [87]. The group of
Somorjai was first to report the growth of ordered TiOx overlayers on a Pt(1 1 1)
substrate [71]. The as-grown films were disordered but annealing between 500 and
700
C in an oxygen background caused the formation of a hexagonal (H43 H43)
R7.6
superstructure, which was observed in LEED and STM for coverages ranging
between 1 and 5 ML, and was tentatively ascribed to a reconstructed TiO
2
(1 1 1)
surface. Annealing at higher temperatures in UHV produced a second ordered
structure, with a reduced Ti
4
O
7
stoichiometry, according to XPS data [71]. Ten years
after this work, Granozzi and coworkers [72] reexamined this system, focusing their
investigation on the submonolayer to monolayer coverage range and revealed the
complexity of the TiOx-Pt(1 1 1) phase diagram in great detail. These researchers
evaporated Ti reactively onto a clean Pt(1 1 1) surface, kept at room temperature,
in an oxygen pressure of 1 10
6
mbar. By varying the latter in a postoxidation step
between UHV and 1 10
6
mbar, they observed in LEED and STM six different
oxide structures in the coverage range up to 1.2 ML; these are summarized in
Figure 6.3. At low Ti coverage (0.4 ML) and after annealing at 400
Cin10
7
mbar
O
2
, a hexagonal overlayer with a lattice constant of 6 Å has been obtained, which is
incommensurate with the Pt(1 1 1) substrate (Figure 6.3a). For this kagom
e-phase
(k-TiOx), the authors have tentatively proposed a structure model with a formal
Ti
2
O
3
stoichiometry, where the Ti and O ions are arranged in a honeycomb
lattice, identical to that of surface-V
2
O
3
layers on Pd(1 1 1) [88]. At higher Ti
coverages, between 0.8 and 2 ML, and annealing temperature of 700
C and oxygen
pressure of 5 10
6
mbar, a rectangular structure (Figure 6.3b) grows in the form
of rectangular-shaped islands. On the basis of XPS results and because of the
similarity of the STM images to those reported for a rect-VO
2
phase on Pd(1 1 1) by
Surnev et al. [89], this rectangular structure has been ascribed to an interface-
stabilized phase with a TiO
2
stoichiometry. Striking similarities to the structure of
V-oxide overlayers on Pd(1 1 1) [15] and Rh(1 1 1) [18, 90] surfaces have also been
recognized for Ti-oxide phases with a zigzag- (Figure 6.3c and d) and wagon-
wheel-like appearance (Figure 6.3e and f) in the STM images [72]. More recent
work of the same group, combined with DFT calculations [74, 75], has yielded
structural models for the zigzag TiOx phases in terms of layers with Pt–Ti–O
stacking and stoichiometries varying between TiO
1.33
(z-TiOx) and TiO
1.2
(z
0
-TiOx).
The most reduced TiOx phases on Pt(1 1 1) comprise the commensurate wagon-
wheel structures, w-TiOx (corresponding to the hexagonal (H43 H43)R7.6
superstructure, reported initially by Boffa et al. [71]) and w
0
-TiOx (with a (7 7)
R21.8
periodicity), which have been interpreted as TiO(1 1 1) bilayers because of
their characteristic contrast in the STM images, in close similarity to bilayers of
FeO(1 1 1) on Pt(1 1 1) [91] and VO(1 1 1) on Rh(1 1 1) [18] surfaces.
Ultrathin Ti-oxide films grown on Pt(1 0 0) by Matsumoto et al. [33, 34] show also
a rich phase diagram of interface-stabilized oxide structures as a function of the Ti
coverage and the oxide preparation conditions. An interesting contrast to this general
growth behavior of oxide nanolayers display the titania bilayers on a (1 2)-Pt(1 1 0)
surface, investigated by Sambi and coworkers [76]. Here, reactive evaporation of
approximately 2 MLTi in an oxygen atmosphere of 1 10
6
mbar was found to result
156
j
6 STM Imaging of Oxide Nanolayer Model Systems

Figure 6.3 STM images of various TiOx structures on Pt(1 1 1):
(a) k-TiOx (30 Å 30 Å, 0.4 V, 1.06 nA); (b) rect-TiO
2
(90 Å 90 Å, þ0.8 V, 1.8 nA); (c) z-TiOx (60 Å 60 Å, þ0.1 V,
1.5 nA); (d) z
0
-TiOx (90 Å 90 Å, þ0.8 V, 1.5 nA); (e) w-TiOx
(75 Å 75 Å, þ1.3 V, 1.9 nA); (f) w
0
-TiOx (126 Å 126 Å, þ0.2 V,
1.0 nA). (Reproduced with permission from Ref. [72].)
6.3 Case Studies: Selected Oxide–Metal Systems
j
157
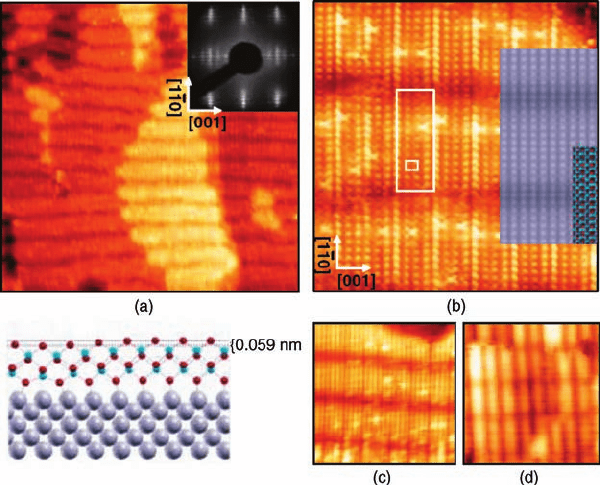
in the formation of a wetting Ti-oxide layer on the Pt(1 1 0) surface, which exhibits a
regular array of dark stripes in STM images, corresponding to a (14 4) superstruc-
ture (Figure 6.4a, c, and d). The high-resolution image (Figure 6.4b) reveals that a
rectangular overlayer unit cell with dimensions 3.0 Å 3.9 Å is at the root of this
coincidence structure, and it is compatible with the unit cell of 2D titania nanosheets
with the lepidocrocite structure, which can be obtained by delamination of layered
titanates (for a model see the inset in Figure 6.4b). DFTcalculations performed in the
same work have revealed that such a lepidocrocite nanosheet is the most stable 2D
titania phase, irrespective of the presence of the substrate, and it can be considered
as a distorted anatase (0 0 1) bilayer obtained by sliding the upper atomic layer
with respect to the lower one by half a unit cell in the [1 0 0] direction. By this
transformation, the resulting lepidocrocite nanosheet is stabilized by 1.24 eV with
respect to the undistorted anatase (0 0 1) bilayer. The authors have concluded that
it is the reduced dimensionality of the oxide overlayer, rather than the interaction
Figure 6.4 (a) Large-area STM image
(620 Å 620 Å, þ0.48 V, 1.4 nA) of a single-
domain lepidocrocite nanosheet on (1 2)-
Pt(1 1 0). The central brighter area is separated
from the lower terrace by a substrate mono-
atomic step. Inset: (14 4) LEED pattern.
(b) High-resolution STM image (137 Å 137 Å ,
þ0.28 V, 1.65 nA) of the nanosheet. The
overlayer (superstructure) unit cell is indicated
by the small (large) rectangle. Inset:
Tersoff–Hamann simulation of the STM image
with superimposed solid sphere model (oxygen:
dark gray/red; titanium: light gray/blue). Dark
stripes along [0 0 1] are due to overlayer bending
toward the substrate (see the interface side view
in the [0 0 1] plane, lower left corner; Pt: large,
gray). (c),(d) High-resolution STM images
of the nanosheet (136 Å 136 Å, þ0.42 V,
0.9 nA and 180 Å 180 Å, þ1.80 V, 1.0 nA).
(Reproduced with permission from Ref. [76].)
158
j
6 STM Imaging of Oxide Nanolayer Model Systems
with the substrate, that stabilizes the lepidocrocite structure, in contrast to what has
been typically observed for other ultrathin oxide phases on metal substrates, reviewed
in the present work. These findings are also supported by results reported for TiO
2
layers on Pt(1 1 1) [72] and Ni(1 1 0) [85], which exhibit a structure very similar to that
of the lepidocrocite nanosheets.
The group of Thornton has used a Ni(1 1 0) substrate with the aim to grow
epitaxially TiO
2
layers, which can mimic the behavior of rutile TiO
2
(1 1 0) single-
crystal surfaces. Postoxidation in an oxygen pressure of 1 10
7
mbar at 800 K of Ti
films with a coverage exceeding 1 ML was reported to result in 3D Ti-oxide islands
with a structure resembling the surface of the bulk rutile TiO
2
(1 1 0) surface [83–85].
For a Ti coverage between 1 and 2 ML, the unit cell dimensions of the rutile-like
islands were 2.9 Å 6.2 Å, that is, slightly contracted from single-crystal TiO
2
(1 1 0)
(2.96 Å 6.5 Å), but thicker films display the relaxed rutile surface lattice constant.
The question to what extent such titania layers can resemble bulk samples is of
significant relevance for many technological applications and was treated in a recent
study by this group [86]. Here, titania films with a thickness of up to 4 ML have been
subjected to reductive treatments at elevated temperatures and an oxygen pressure
of 1 10
7
mbar. Annealing at temperature between 873 and 1023 K causes a 1 2
reconstruction of the surface of the rutile TiO
2
(1 1 0) islands, in close similarity to
the well-documented reduction behavior of bulk TiO
2
(1 1 0) crystals [92–94]. Higher
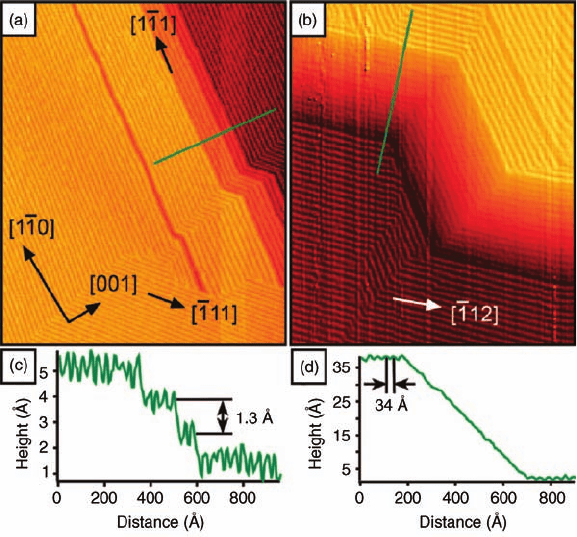
annealing temperatures of up to 1100 K result in a completely changed film
morphology, with the original rutile islands being replaced by a continuous film
displaying step edges and facets in STM images (Figure 6.5). Here, regularly spaced
lines with a periodicity of approximately 34–35 Å can be seen, which correspond to
the intersections of {1 3 2} and {1 2 1} families of crystallographic shear planes with
the (1 1 0) surface and the facets, as previously reported for single-crystal titania
surfaces [95]. As the crystallographic shear planes are bulk defects rather than surface
defects, the authors conclude that ultrathin TiO
2
films on Ni(1 1 0) behave at least
structurally in a bulk-like manner. The question to what extent this also holds for
other physical and chemical properties of TiO
2
nanolayers remains to be answered by
future experiments.
6.3.3
Vanadium Oxide Nanolayers
In the bulk form, vanadium oxides display different oxidation states and VO
coordination spheres and exhibit a broad variety of electronic, magnetic, and
structural properties [96, 97], which make these materials attractive for many
industrial applications. Prominent examples range from the area of catalysis, where
V-oxides are used as components of important industrial catalysts for oxidation
reactions [98] and environment pollution control [99], to optoelectronics, for the
construction of light-induced electrical switching devices [100] and smart thermo-
chromic windows. In view of the importance of vanadium oxides in different
technological applications, the fabrication of this material in nanostructured form
is a particularly attractive goal.
6.3 Case Studies: Selected Oxide–Metal Systems
j
159
We refrain here from giving an extensive overview of studies on the surface
structure of vanadium oxide nanolayers, as this has already been done for up to year
2003 in our recent review [97]. Instead, we would like to focus on prototypical
examples, selected from the V-oxide–Rh(1 1 1) phase diagram, which demonstrate
the power of STM measurements, when combined with state-of-the-art DFT calcula-
tions, to resolve complex oxide nanostructures. Other examples will highlight the
usefulness of combining STMand STS data on a local scale, as well as data from STM
measurements, and sample area-averaging spectroscopic techniques, such as XPS
and NEXAFS, to derive as complete a picture as possible of the investigated system.
The surface phase diagram of vanadium oxides on Rh(1 1 1) has been investigated
in a series of papers of our group [4, 18, 19, 90, 101–103]. It is characterized by
pronounced polymorphism and many different oxide structures have been detected
as a function of coverage and growth temperature. The vanadium oxide structures
for coverages up to the completion of the first monolayer formed on Rh(1 1 1) under
the different preparation conditions may be subdivided into highly oxidized phases
Figure 6.5 STM images of the crystallo-
graphically sheared film: (a) 1700 Å 2100 Å,
þ0.30 V, 0.90 nA; (b) 1500 Å 1800 Å,
þ0.15 V, 0.80 nA. The arrows indicate both the
principal azimuths of the TiO
2
(1 1 0) surface and
the directions of the CS plane intersections at
the (1 1 0) surface. (c) Line profile along the line
indicated in (a). (d) Line profile along the line
indicated in (b). The image in (b) has a shadow
effect applied, and the line profile in (d) is taken
from an image without the shadow applied.
(Reproduced with permission from Ref. [86].)
160
j
6 STM Imaging of Oxide Nanolayer Model Systems
containing vanadium atoms in formal oxidation states of around þ5, into more
reduced oxides with V
2 þ
or V
3 þ
species, and into mixed valency vanadium
oxides [18, 90]. The latter are a peculiarity of the vanadium oxide – Rh(1 1 1) phase
diagram – and occur only in a narrow range of the chemical potential of oxygen. The
various vanadium oxide structures have been investigated with a combination of
experimental techniques, including STM, LEED, high-resolution photoelectron
spectroscopy (HR-XPS, UPS) with the use of synchrotron radiation, and HREELS
(vibrational spectroscopy of phonon modes). The results have been combined with
ab initio DFT calculations to derive the structural models for the vanadium oxide
surface phases. We emphasize that the synergy of the different experimental
techniques in combination with a high-level theory was absolutely necessary to
achieve the success obtained in the analysis of these very complex oxide structures.
In the following section, we illustrate the complexity of the structures observed with
just a few examples.
Under highly oxidative conditions, that is, p(O
2
) 2 10
7
mbar, T
substrate
¼
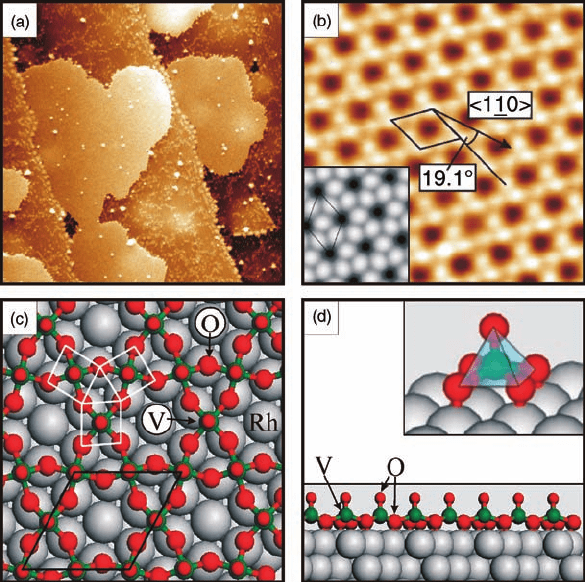
400
C, a V-oxide layer with a (H7 H7)R19.1
structure, here called simply H7,
grows at low submonolayer coverages (Q<0.3 MLE) as two-dimensional islands,
as seen in the STM image in Figure 6.6a [4, 18]. The high-resolution STM image in
Figure 6.6b reveals the structural details of the H7 phase at the atomic scale, with
a hexagonal lattice that consists of three bright protrusions per unit cell. The DFT
calculations [18] have established a model of the (H7 H7)R19.1
structure
(Figure 6.6c and d): it corresponds to a V
3
O
9
oxide phase that contains identical
pyramidal O
4
V¼O building blocks (marked squares in Figure 6.6c). Each pyramid
(inset of Figure 6.6d) has the V atom (in green) in the center, four bridging O atoms
(in red) in the basal plane, and a vanadyl-type O atom at the apex. The presence of the
latter O species in the H7 overlayer has been inferred from HREELS spectra [18],
which contain a characteristic loss at approximately 130 meV because of the stretch-
ing vibrations of vanadyl (V¼O) groups. The pyramids are linked together via the four
bridging O atoms at the interface, which results in three VO
3
units per H7 unit cell,
or in an overall V
3
O
9
stoichiometry. Owing to the fact that the basal O atoms are
shared with the Rh substrate, the formal V
3
O
9
stoichiometry is not incompatible with
the maximum oxidation state of þ5 of the V atoms in the bulk V
2
O
5
structure. The
structure model of the H7-V
3
O
9
phase is confirmed by the good agreement between
the experimental and simulated STM image (inset of Figure 6.6b) and the calculated
phonon spectrum, which reproduces the vanadyl stretching vibrations observed in
the experiment [18].
Within the inverse model catalyst approach, the H7-V
3
O
9
–Rh(1 1 1) nanostruc-
tures have been used to visualize surface processes in the STM with atomic-level
precision [104]. The promoting effect of the V-oxide boundary regions on the
oxidation of CO on Rh(1 1 1) has been established by STM and XPS by comparing
the reaction on two differently prepared H7-V
3
O
9
–Rh(1 1 1) inverse catalyst surfaces,
which consist of large and small two-dimensional oxide islands and bare Rh areas in
between [105]. A reduction of the V-oxide islands at their perimeter by CO has been
observed, which has been suggested to be the reason for the promotion of the CO
oxidation near the metal-oxide phase boundary.
6.3 Case Studies: Selected Oxide–Metal Systems
j
161
Exposing the H7-phase to a reducing environment, for example, by annealing in
UHV or in a hydrogen atmosphere, results in the formation of various reduced 2D
V-oxide phases [18, 101]. In particular, annealing at 650–750 K in vacuum produces
an oxide structure with a rectangular (5 3H3) unit cell, presented in Figure 6.7.
The large-scale STM image of Figure 6.7a shows well-ordered oxide islands with
rectangular shapes, elongated along the principal h110i azimuthal directions of the
Rh(1 1 1) substrate. The inset of Figure 6.7a displays the (5 3H3)-rect structure at
a higher magnification, also revealing details on the island boundary; the latter will be
discussed in the next paragraph. The synergy of various experimental and theoretical
methods has allowed us to derive the structure model for the (5 3H3)-rect
phase [18], represented in Figure 6.7b. Accordingly, the structure consists of two
types of building units, the O
4
V¼O tetragonal pyramid and a planar V
6
O
12
hexagon
(indicated on the figure), which are linked together to give the observed rectangular
Figure 6.6 The (H7 H7)R19.1
oxide phase:
(a) large-scale STM image (1000 Å 1000 Å,
þ2.0 V, 0.1 nA) of 2D oxide islands on the
Rh(1 1 1) surface; (b) high-resolution STM
image (5 Å 5Å, þ0.75 V, 0.2 nA). The
(H7 H7)R19.1
unit cell and the Rh(1 1 1)
substrate direction are indicated. The inset
shows a DFT-simulated STM image; (c) DFT
model of the H7-V
3
O
9
phase. Unit cell and
structural units are indicated (V green, O red,
Rh gray); (d) Side view of the V
3
O
9
model. The
inset shows a detailed view of the pyramidal
O
4
V¼O unit. (Reproduced with permission
from Ref. [4].)
162
j
6 STM Imaging of Oxide Nanolayer Model Systems
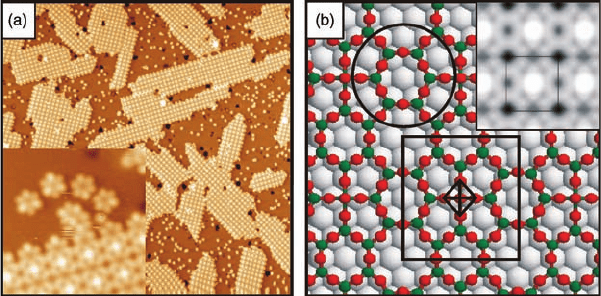
structure. The stoichiometry per unit cell is V
13
O
21
, and thus the (5 3H3)-rect
structure corresponds to a novel mixed-valence vanadium oxide phase, which occurs
only in ultrathin layer form. The simulated STM image (inset of Figure 6.7b) is in
excellent agreement with the experimental one and thus supports the structure
model.
Further annealing the (5 3H3)-rect structure at 750 –800 K leads to the transfor-
mation into a (9 9) phase [4, 18], which grows in the form of compact branched 2D
islands with rounded boundary shapes, as shown in the large-scale STM image in
Figure 6.8a. The high-resolution STM image (inset of Figure 6.8a) reveals a complex
contrast pattern of this phase, with the (9 9) unit cell indicated. In the center of the
unit cell, seven hexagonally arranged depressions are visible, showing a local (2 2)
periodicity. The structure model for the (9 9) phase, derived from the DFT
calculations is presented in Figure 6.8b and involves only one type of building unit,
the planar V
6
O
12
hexagons (encircled). The latter are connected in a complex way,
with eightfold and fivefold rings in between, to give the observed (9 9) periodicity.
Each unit cell contains 36 vanadium atoms and 54 oxygen atoms, yielding basically
aV
2
O
3
stoichiometry; the structure thus contains only V atoms in a formal þ3
oxidation state. The DFT model is strongly supported by the simulated STM image,
shown in the inset of Figure 6.8b; note that every single detail is reproduced in
the simulation. Interestingly, the presence of locally ordered (2 2) areas within the
(9 9) unit cell and the overall V
2
O
3
stoichiometry bear a strong similarity to the
(2 2) surface-V
2
O
3
phase observed on the Pd(1 1 1) surface [88]. We recently
suggested that the absence of a long-range ordered (2 2) structure on Rh(1 1 1) is
due to interfacial strain [23]; while the (2 2)-V
2
O
3
superstructure fits perfectly on
the Pd(1 1 1) lattice, there isa 2.3% lattice mismatch between the relaxed (2 2)-V
2
O
3
Figure 6.7 (a) STM image of (5 H3)-rect vanadium oxide islands
on Rh(1 1 1) (1000 Å 1000 Å, þ1.5 V, 0.1 nA). Inset: enlarged
section of an (5 Ö3)-rect island (70 Å 70 Å, þ0.5 V, 0.1 nA);
(b) DFT-derived model of the (5 H3)-rect structure, unit cell
and structural units are indicated (V green, O red, Rh gray).
Inset: simulated STM image. (Reproduced with permission
from Refs [18, 101].)
6.3 Case Studies: Selected Oxide–Metal Systems
j
163
