Bowker M., Davies P.R. (Eds.) Scanning Tunneling Microscopy in Surface Science, Nanoscience and Catalysis
Подождите немного. Документ загружается.


overlayer and the Rh(1 1 1) surface. It has been proposed in this study that the regions
of local (2 2) symmetry are connected in the (9 9) structure via the larger eightfold
rings in order to release the interfacial strain. Support for this conjecture has been
obtained very recently by growth experiments of V-oxide on a Rh(15 15 13) surface,
which is vicinal to the Rh(1 1 1) [14]; the results are summarized in Figure 6.8c. Here,
the (9 9) structure has been found on wide Rh(1 1 1) terraces, whereas a (2 2)
structure was observed on small terraces, where the step edges may provide the strain
relief.
As a particular highlight in the V-oxide–Rh(1 1 1) phase diagram, we mention the
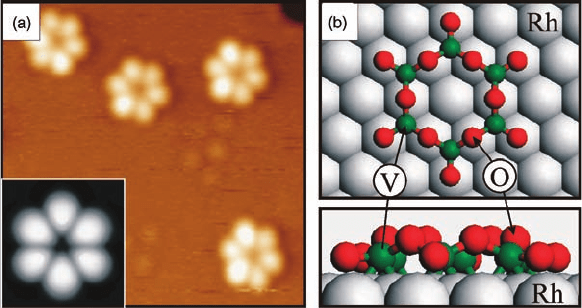
spontaneous formation of quasi-zero-dimensional V-oxide clusters, which are illus-
trated in Figure 6.9a and b [4, 19]. These star-like clusters form at very low Vcoverages
Figure 6.8 The (9 9) V-oxide phase:
(a) large-scale STM image (2000 Å 2000 Å,
þ2.0 V, 0.05 nA). The inset shows a high-
resolution STM image (60 Å 60 Å, þ2.0 V,
0.1nA); (b) DFT model of the (9 9) V
36
O
54
phase. The (9 9) and (2 2) unit cells are
indicated with solid and dashed lines, respec-
tively (V green, O red, Rh gray). The inset shows
a DFT-simulated image. (c) STM images
of the Rh(15 15 13) surface covered with a
V
2
O
3
-type overlayer: (center) large-scale image
(500Å 500 Å, þ2.0 V, 0.1 nA), (left) magnified
view of the (9 9) structure on the highlighted
wide terrace area (45 Å 45Å, þ2.0 V, 0.1nA),
and (right) magnified view of the (2 2) V-oxide
superstructure on the highlighted narrow
terraces (75 Å 75 Å, þ1.0 V, 0.1 nA).
(Reproduced with permission from Refs [4, 14].)
164
j
6 STM Imaging of Oxide Nanolayer Model Systems
(<0.1 ML) at intermediate values of the chemical potential of oxygen in a narrow range
of the surface phase diagram. With the help of DFTcalculations, the stars have been
identified as planar V
6
O
12
cluster molecules, which are unstable in the gas phase but
are stabilized by the interactions at the interface with the Rh substrate [19]. They thus
constitute a novel form of oxide cluster material. The V
6
O
12
clusters become mobile
at elevated temperature, which has allowed us to study their diffusion behavior by
variable-temperature STM measurements. The clusters hop and diffuse across the
surface as entire molecular units, which provides an easy means for oxide mass
transport across a metal surface [19]. The V
6
O
12
clusters self-assemble into two-
dimensional oxide phases of mixed valent character, the (5 5) V
11
O
23
structure and
the (5 3H3)-rect V
13
O
21
structure (see the inset of Figure 6.7a), depending on the
gradient of the chemical potential of oxygen [101]. This self-organization of spon-
taneously formed V
6
O
12
cluster building blocks at a metal surface into different
two-dimensional V-oxide nanostructures, directed by the chemical potential of
oxygen, is a process that might be of interest for the controlled design of ultrathin
oxide nanolayers [101].
Recently, we have also employed scanning tunneling spectroscopy measurements
to probe the local electronic structure of the various vanadium oxide surface phases
on Rh(1 1 1) [23]. Figure 6.10 summarizes differential conductance dI/dU spectra
taken at 77 K in constant height (left, blue curves) and constant current (right, black
curves) modes from the (H7 H7)R19.1
-V
3
O
9
(a), (5 3H3)-rect-V
13
O
21
(b),
(9 9)-V
36
O
54
phases (c), and from the star V
6
O
12
clusters (d), discussed above.
The constant current spectra display at higher voltages (>5V) field emission
resonances that were found to be of less diagnostic value for structural details.
Constant height STS spectra from the (H7 H7)R19.1
and (5 3H3)-rect (in the
vanadyl position) V-oxide overlayers show peaks at around þ0.9 and þ2.7 V in the
Figure 6.9 The V
6
O
12
cluster molecules on Rh(1 1 1):
(a) STM image (63 Å 63 Å, þ0.5 V, 0.1 nA). The inset shows a
DFT-simulated image; (b) Relaxed DFT model geometry of the
V
6
O
12
clusters on Rh(1 1 1) in top and side view. (Reproduced with
permission from Ref. [4].)
6.3 Case Studies: Selected Oxide–Metal Systems
j
165
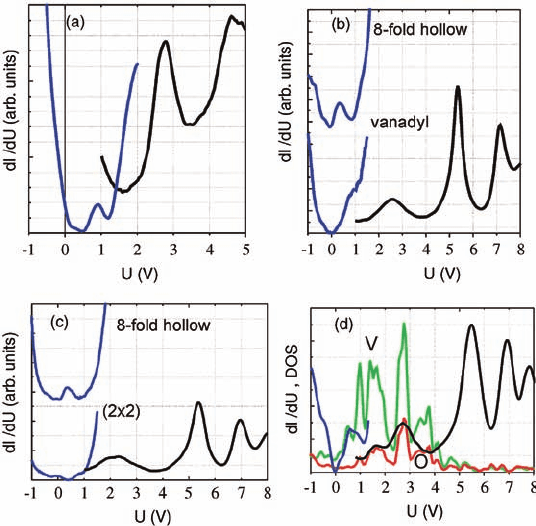
empty states above the Fermi energy (U ¼0 V), which are characteristic of the VO
5
pyramidal building units. Constant height spectra recorded above the eightfold
hollow rings, present in the (5 3H3)-rect and (9 9) phases (see Figures 6.7
and 6.8), also display a similar structure, containing a characteristic peak at þ0.3 V
that has been attributed to vanadium atoms coordinated to bridging oxygen atoms.
STS spectra of the V
6
O
12
clusters (Figure 6.10d) shows peaks at þ0.6, þ1.6, and
þ2.7 V that approximately coincide with peaks of DFT calculated atom-projected
density of states for the V (green) and O (red) atoms. The latter reveal a close
correspondence, as a result of strong hybridization between V and O atoms in the
molecule-type oxide clusters. We emphasize that STS is the only experimental
technique that allows one to measure the electronic states of these surface supported
nanoobjects.
The growth of vanadium oxide overlayers on Rh(1 1 1) converges after a number of
intermediate stages to the formation of a three-dimensional bulk-like epitaxial V
2
O
3
film [90], which is oriented with the (0 0 0 1) plane of its corundum structure parallel
to the Rh(1 1 1) substrate surface. The V
2
O
3
phase is the thermodynamically stable
Figure 6.10 STS differential conductance spectra taken in
constant height (left, blue curves) and constant current (right,
black curves) modes from the (a) (H7 H7)R19.1
, (b) (5 H3)-
rect, (c) (9 9) structures, and (d) from the star cluster. The
DFT-calculated DOS for the oxygen (red) and vanadium (green)
atoms in the star cluster are shown in (d) for comparison.
(Reproduced with permission from Ref. [23].)
166
j
6 STM Imaging of Oxide Nanolayer Model Systems
phase of V-oxide under the given experimental conditions of ultrahigh vacuum
experiments. A pertinent question of interest in relation to the physical and chemical
properties of the V
2
O
3
(0 0 0 1) surface is its surface termination. We have addressed
this question and have found, by a combined experiment–theory effort [102, 103],
that the most stable surface termination is a vanadyl V¼O terminated (1 1) surface.
This is remarkable, since V¼O groups are not VO coordination units contained in
the bulk V
2
O
3
corundum structure; they do occur, however, in the layered structure of
the V
2
O
5
bulk phase. The occurrence of vanadyl units as the most stable surface
termination of V
2
O
3
is clearly unexpected and of interest to the large field of V-oxide
catalysis. Apart from the vanadyl-terminated (1 1) surface, two other surface
terminations could be produced, a (H3 H3)R30
structure, containing V¼O
vacancies, and a V-terminated (1/H3 1/H3) surface, which requires highly
reducing preparation conditions [102, 103]. The lack of bulk-type V-oxide phases
on Rh(1 1 1) with an oxidation state higher than þ3 can be ascribed to kinetic effects,
that is, the limited speed of oxidation under the employed oxidation conditions
(oxygen pressures of max 10
6
mbar).
This limitation has recently been circumvented by Kuhlenbeck and coworkers [31,
32], who reported that highly oxidized V-oxide layers with V
6
O
13
and V
2
O
5
stoichio-
metries can be prepared on a Au(1 1 1) substrate, using a high-pressure cell, where
oxygen pressures of up to 50 mbar can be applied for in situ oxidation of V atoms.
At submonolayer to monolayer coverages, two well-ordered interface-stabilized
V-oxide structures have been observed in STM and LEED, with a V oxidation state
estimated to be close to þ5, according to NEXAFS spectra taken in the V L-edge and
O K-edge region [31]. By comparing the polarization dependence and the shape of
NEXAFS spectra from these monolayer films and from bulk V
2
O
5
(0 0 1) single
crystals, the authors concluded that the monolayer V
2
O
5x
structures should contain
V¼O groups and suggested that these are part of VO
5
building units, in close
similarity to the highly oxidized V
3
O
9
structures on Rh(1 1 1) [18]. After the
completion of the first layer, a two-layer V
6
O
13
(0 0 1)-like wetting film was observed
in STM [31], which is the precursor for the bulk-type V
2
O
5
(0 0 1) growth. The
V
2
O
5
(0 0 1) phase appears at a V coverage of approximately 1.5 ML in the form of
elongated 3D islands, rotationally aligned with the V
6
O
13
(0 0 1) wetting layer [31],
which eventually merge to form a continuous film [32]. Kuhlenbeck and co-
workers [32] have established that atomically flat and nearly defect-free V
2
O
5
(0 0 1)
overlayers can be prepared by cycles of successive Vdepositi on and pos toxidation, as
illustrated in the STM images in Figure 6.11a and b. The corresponding LEED
image (Figure 6.11c) shows a ring patte rn, indicating that the V
2
O
5
(0 0 1) domains
have a rand om azimuthal orientation, suggesting a weak film–substrate interaction.
The V
2
O
5
stoichiometry has been confirmed by XPS and angle-dependent NEXAFS.
The application of these techni ques was found, however, to lead to the for mation of
X-ray-induced O vacancies in the V
2
O
5
film, as revealed by S TM. Accordingly, O
vacancies were not randomly distributed on the surface, but as pairs or rows, as a
result of a concerted reduction process. The V
2
O
5
film was found to be thermally
stable upon annealing in UHV up to 500
C, starting to sublime above this
temperature, as revealed by temperature-prog rammed desorption (TPD) spectra.
6.3 Case Studies: Selected Oxide–Metal Systems
j
167

Figure 6.11 STM images (a) 3000 Å 3000 Å, 3.5 V, 0.2 nA and
(b) 1000 Å 758 Å, 3 V, 0.2 nA (differentiated), and LEED pattern
(c) obtained for a film formed by three successive expositions of
2.6 MLE V and oxidation cycles. (Reproduced with permission
from Ref. [32].)
168
j
6 STM Imaging of Oxide Nanolayer Model Systems
6.3.4
Iron Oxides on Pt(1 1 1)
Iron oxides, which are active materials in the catalysis of dehydrogenation reac-
tions [106, 107], can be grown with good epitaxial order on Pt(1 1 1) surfaces. This was
demonstrated by the work of the Berkeley group, Weiss et al. [108] and Barbieri
et al. [109], who performed a careful LEED analysis of iron oxide films grown on
Pt(1 1 1) up to a thickness of 10 ML. The 10 ML oxide was identified by a dynamical
LEED analysis to be magnetite Fe
3
O
4
, terminated by an unreconstructed polar (1 1 1)
surface that exposes 1/4 monolayer of Fe atoms over a distorted hexagonal close-
packed oxygen layer. It was also noted in this work that at 1 ML coverage the Fe oxide is
of a different kind and forms a hexagonal coincidence structure with respect to the
Pt substrate.
Virtually all subsequent surface science-related studies on Fe oxide films have
been performed using the Pt(1 1 1) surface as a metallic substrate. The established
preparation procedure for well-ordered Fe oxide films on Pt(1 1 1) involves PVD of Fe
in 1–2 ML quantities onto clean Pt(1 1 1), followed by annealing in oxygen at elevated
temperature; this process can be repeated until oxide layers of the desired thickness
have been formed. The preparation of Fe oxides on Pt(1 1 1) and the morphology of
the resulting films as a function of the preparation parameters as well as the
properties of Fe oxides in relation to catalysis have been comprehensively reviewed
by Weiss and Ranke [106].
Iron oxides grow with a Stranski–Krastanov mode on Pt(1 1 1) surfaces. The first
phase that forms, up to coverages of approximately 2.5 ML, is a FeO(1 1 1)-type
structure that grows in a layer-by-layer fashion. Subsequent layers of Fe oxide rapidly
converge to Fe
3
O
4
, which nucleates in the form of 3D islands. Thick films of Fe
3
O
4
are formedby coalescence of the 3D islands [106]. The Fe
3
O
4
(1 1 1) surface is madeup
by Fe atoms in a (2 2) arrangement with respect to the hexagonal oxygen layer of the
subsurface plane [108, 109]. At higher oxygen pressures, Fe
3
O
4
is transformed into
Fe
2
O
3
(0 0 0 1) with a (H3 H3)R30
structural arrangement. This sequence of
phases is roughly in accordance with the one found in the bulk-phase diagram of
Fe oxides, but differences occur in the detailed temperatures and pressures, pointing
toward the importance of kinetic effects in the stabilization of the thin film phases.
All Fe oxide films on Pt have strongly relaxed, unreconstructed bulk-terminated
surfaces, but while the Fe
3
O
4
and Fe
2
O
3
oxide layers are similar to their respective
bulk compounds, the ultrathin FeO layers are true 2D oxide phases that are different
from the FeO bulk and stabilized by the metal–oxide interface.
Galloway et al. [110] were first to apply STM to investigate the growth of Fe oxides
on Pt(1 1 1). For coverages 1ML, atomically resolved STM images have been
obtained and interpreted in terms of a FeO(1 1 1) bilayer with an approximate (9 9)
periodicity. The overlayer can be described with a coincidence structure, where
approximately eight lattice spacings of the FeO fit approximately nine of the Pt(1 1 1)
surface; this gives rise to a Moir
e pattern in STM as shown in Figure 6.12. The
structure contains an atomic spacing of approximately 3.1 Å, a longer range Moir
e
periodicity of approximately 26 Å and is slightly rotated against the Pt substrate.
6.3 Case Studies: Selected Oxide–Metal Systems
j
169

At higher coverages, (2 2) and (H3 H3)R30
structures have been observed,
which have been ascribed as before [108, 109] to Fe-terminated Fe
3
O
4
(1 1 1) and
a-Fe
2
O
3
(0 0 0 1) phases, respectively.
As mentioned several times in this review, the interpretation of STM images of
oxide surfaces is a nontrivial problem because the relationship between images and
crystallography of the surface is not straightforward. Sautet et al. [111] have suc-
cessfully attempted to determine the origin of the STM image contrast and its
relationship with the atomic positions of the FeO monolayer on Pt using electron
scattering quantum chemistry (ESQC) theory. In their analysis, the Moir
e pattern
has been decomposed into simple high-symmetry FeO/Pt confi gurations containing
Fe in on-top, bridge, and hollow sites. In the real image, there is a continuous
transition between them, but major aspects of the pattern can be understood by
considering the high-symmetry configurations shown in Figure 6.12, bottom part. As
an interesting result of the analysis, it turned out that the highest topographic points
in the images occurred in the bridge and hcp hollow regions, depending on the tip
conditions, but never in the on-top positions, which support the largest corrugation,
Figure 6.12 Current mode, (60 Å 60 Å) STM
image of a monolayer of FeO on Pt(1 1 1). The
Moir
e structure is due to the mismatch between
the 3.1 Å periodicity of the FeO small maxima in
the image and the Pt lattice dimensions
(2.77 Å). The small circular images at the
bottom are calculated images using the ESQC
method for the various high-symmetry epitaxial
configurations associated with the circled
regions in the image. (Reproduced with
permission from Ref. [110].)
170
j
6 STM Imaging of Oxide Nanolayer Model Systems
since there the O atoms are furthest out. This indicates that the electronic contribu-
tions to the contrast dominate over the geometric contributions. The ESQC method
also allowed the analysis of the contributions to the tunneling current of the various
electronic states; both O and Fe orbitals contribute to the tunnel current, thus the
contrast in STM images of FeO layers is due to the interplay of various electronic
effects. Moreover, it was found that the terminating atom of the tip is important – Pt
and O atoms have been used in the simulations – and that simpler treatments of the
STM contrast neglecting tip –surface interactions are insufficient to correctly model
the STM images.
The structural parameters of the FeO(1 1 1) monolayer on Pt(1 1 1) have been
examined by X-ray photoelectron diffraction by Kim et al. [112]; the previously
proposed structure model of Galloway et al. [110] could be confirmed and specified
in terms of its oxygen surface termination. The FeO interlayer distance was found
to be highly compressed by about 50% compared to bulk FeO, 0.68 Å versus 1.25 Å.
This vertical lattice compression is balanced by a lateral lattice expansion as revealed
from the STM images [110, 111], namely, 3.09 Å for the FeO monolayer versus 3.04 Å
in the bulk; the former is necessary to maintain the FeO bond distances at a
reasonable length. As discussed below, this structural rearrangement is driven by the
tendency to minimize the electrostatic surface energy. Kim et al. [112] found only
one of the two possible rotational domains of the oxide monolayer on their surface,
which they ascribed to an OFePt interlayer interaction. This manifestation of
interlayer effects in the growth mode is an interesting aspect in epitaxial growth of
oxide nanolayers. However, Ritter et al. [113] seem to have observed both rotational
domains on a formally identical FeO monolayer on Pt(1 1 1), and the presence of only
one domain in the experiments of Kim et al. may be the effect of some particular
kinetic conditions during surface preparation.
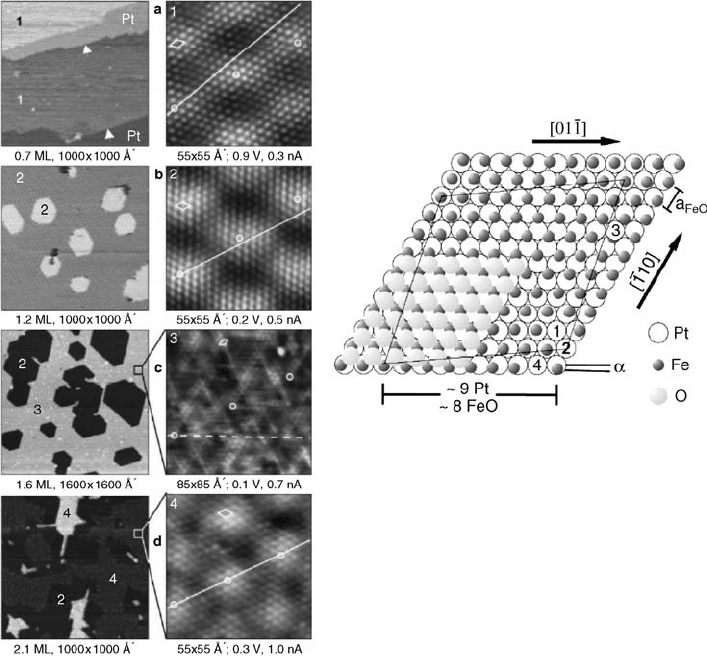
Ritter et al. [113] performed a very detailed study of the initial stages of iron oxide
growth on Pt(1 1 1) and found four different coincidence structures of FeO(1 1 1)-type
phases up to approximately 2.5 ML film thickness. With increasing coverage several
structural changes occur in the FeO layers resulting in coincidence structures with
slightly different lateral lattice constants and rotation misfit angles; all of them
give rise to Moir
e patterns with periodicities between 22 and 38 Å. Large-scale and
atomic resolution STM images of the four coincidence structures found by Ritter
et al. [91, 113] are shown in Figure 6.13 gives a rigid sphere model of the structures
illustrating the different coincidence sites. The FeO(1 1 1)-type overlayer on Pt(1 1 1)
thus exhibits a remarkable variety in terms of lattice parameter and interfacial
registry.
The physical origin of this structural flexibility of the FeO overlayer is still unclear,
the more so since no clear trend is observable in the sequence of lattice parameters of
the coincidence structures. The FeO(1 1 1) phase forming up to coverages of 2–3ML
is clearly stabilized by the interactions with the Pt substrate since FeO is thermo-
dynamically metastable with respect to the higher iron oxides [106, 114]. FeO has the
rock salt structure and the (1 1 1) plane yields a polar surface with a high surface
energy [115], which requires stabilization by internal reconstruction or external
compensation. The structural relaxation observed in the form of the reduced FeO
6.3 Case Studies: Selected Oxide–Metal Systems
j
171
interlayer spacing and the expanded lateral lattice constant is the result of reducing
the distance between the positively charged Fe and the negatively charged O layers,
thereby reducing the surface dipole. Moreover, external compensation of the
FeO(1 1 1) surface dipoles by the image dipoles in the underlying Pt metal substrate
add a further contribution to the stabilization of the FeO overlayer. However, instead
of converging to the FeO bulk lattice with increasing film thickness, the lateral
expansion increases from 3.09 Å at 1 ML to 3.15 Å at >2 ML oxide coverage [91, 113].
Ranke et al. [91] have attempted a qualitative interpretation of the different coinci-
dence structures based on an ionic model of FeO and the decrease of the electrostatic
Figure 6.13 Left panel: Large-scale (left) and
atomic resolution (right) STM images of
epitaxial FeO(1 1 1) films on Pt(1 1 1). Four
different coincidence structures 1–4 are formed
sequentially as the coverage increases. They
exhibit different contrasts in the large-scale
images as indicated by the numbers. Right
panel: Rigid model of the four FeO(1 1 1)
structures formed on Pt(1 1 1). FeO(1 1 1)
bilayers with different lattice constants a
FeO
and
rotated against the platinum surface lattice by
different angles a lead to coincidence sites 1–4.
Here, coincidence structure 2 with its
superstructure cell is shown. (Reproduced with
permission from Ref. [91].)
172
j
6 STM Imaging of Oxide Nanolayer Model Systems

energy of the polar FeO stack by reducing interlayer distances and increasing lateral
lattice constants. While catching the essence of the overall lattice relaxations in the
FeO(1 1 1) nanolayers, a purely ionic description of FeO is questionable, and the
model is too simplistic to explain the sequence of coincidence structures observed
as a function of coverage. High-level ab initio calculations of the various interfacial
configurations will be necessary to shed light on the origin of the observed
coincidence structures of FeO on Pt(1 1 1); this will be no easy task in view of the
large unit cells involved.
6.3.5
Nickel Oxide Nanolayers
Thin nickel oxide (NiO) films are used in many oxidation catalysts and industrial
chemical processes including steam reforming of methane to syngas [116], the
oxidation of methane [117], hydrocarbons [118], methanol [119], CO [120], and other
organic compounds [121, 122]. NiO is also employed in alkaline electrochemical
systems, such as alkaline fuel cells [123, 124]. Owing to its defect structure,
nonstoichiometric nickel oxide is a good p-type semiconductor and is therefore
used as resistive sensors for reducing gases, such as H
2
[125], CO [126], NH
3
[127],
and also NO
2
[128], formaldehyde [129], and methanol [130]. Epitaxial growth of
nickel oxide nanolayers and the characterization of their structure at the atomic level
is thus of considerable fundamental and applied interest.
Historically, ultrathin Ni-oxide layers have been first grown by oxidation of single-
crystal Ni(1 0 0) and Ni(1 1 1) surfaces (see Ref. [131] and references therein). The
lattice parameter of NiO (4.176 Å) is by nearly 20% larger than that of metallic
Ni (3.524 Å), which is not a favorable prerequisite for epitaxial NiO growth. B
€
aumer
et al. [132] have reported a (1 0 0)-oriented NiO film growth by exposing a Ni(1 0 0)
surface to 10
4
lO
2
at 570 K. According to their STM and SPA-LEED data, the film
consists of NiO(1 0 0) crystallites, which are tilted by approximately 8% with respect to
Ni(1 0 0) surface normal. The authors concluded that by means of this tilt the surface
strain, caused by the large mis fit between Ni and NiO lattices, may be partially
relieved [132]. On the other hand, a much more favorable epitaxial relationship for
a NiO(1 0 0) layer growth has been established on the Ni(1 1 1) substrate, that is,
NiO(1 0 0)||Ni(1 1 1) and NiO[0 1 0]||Ni[1
10] with a mismatch of only 1.4%, where
locally ordered (2 2), (2 3), (3 2), and (3 3) superstructure cells of NiO(1 0 0)
have been detected in LEED and STM [133, 134]. In particular, Hildebrandt et al. [134]
used a variable-temperature STM to investigate in situ and in real time the oxidation
of Ni(1 1 1) at elevated temperature. They found that the oxidation starts at the Ni step
edges, which is followed by the transformation of the Ni(1 1 1) terraces into a three-
domain NiO(0 0 1)-like layer, containing a mixture of (2 2), (2 3), and (3 3) NiO
(0 0 1) unit cells, as shown in Figure 6.14. At higher oxygen exposures (>25 l), the
formation of triangular bulk NiO(1 1 1) islands has been observed.
More favorable lattice matching conditions have been achieved by growing
epitaxial NiO layers on dissimilar metal surfaces, such as Au(1 1 1) [135, 136],
Ag(1 0 0) [137–142], Cu(1 0 0) [143], Cu(1 1 1) [144, 145], and more recently
6.3 Case Studies: Selected Oxide–Metal Systems
j
173
