Bowker M., Davies P.R. (Eds.) Scanning Tunneling Microscopy in Surface Science, Nanoscience and Catalysis
Подождите немного. Документ загружается.

exposed to 1 atm CO and annealed at 150
C in this environment for 4 h, the
restructured surface is smooth compared to the rough surface formed in H
2
(Figure 7.16a). The surface restructured in CO (Figure 7.16c) has a width of
approximately 1–30 Å and does not s how missing row reconstruction. In fact, the
high-pressure CO lifts the missing row reconstruction and thus forms a flat 1 1
structure separated b y steps.
Another example of the flexibility of the Pt catalyst is the reconstruction of a
stepped Pt(1 1 1) crystal with adsorbed sulfur upon exposure to CO [25]. Single-crystal
Pt(1 1 1) cut at an angle of approximately 5
from the (1 1 1) direction consists of
numerous terraces with a width of 20–60 Å separated by steps with single-atom
height. The adsorption of sulfur atoms restructures the clean stepped Pt(1 1 1)
surface with single-atom steps into a sulfur-adsorbed surface with double-atom
Figure 7.15 STM images of a lifted Pt(1 0 0) surface under 1 Torr CO at 300 K (a) and 420 K (b).
204
j
7 Surface Mobility of Atoms and Molecules Studied with High-Pressure STM
height. The width of the terraces of the double-step surface is twice that of the original
sample: 40–120 Å (Figure 7.17). A similar doubling of step height and terrace width
was also observed on other metal surfaces [26]. This phenomenon is rationalized by a
decrease in the step edge –step edge repulsive interaction that prevents the energet-
ically favorable coalescence of steps. Surprisingly, the exposure of this double-atom
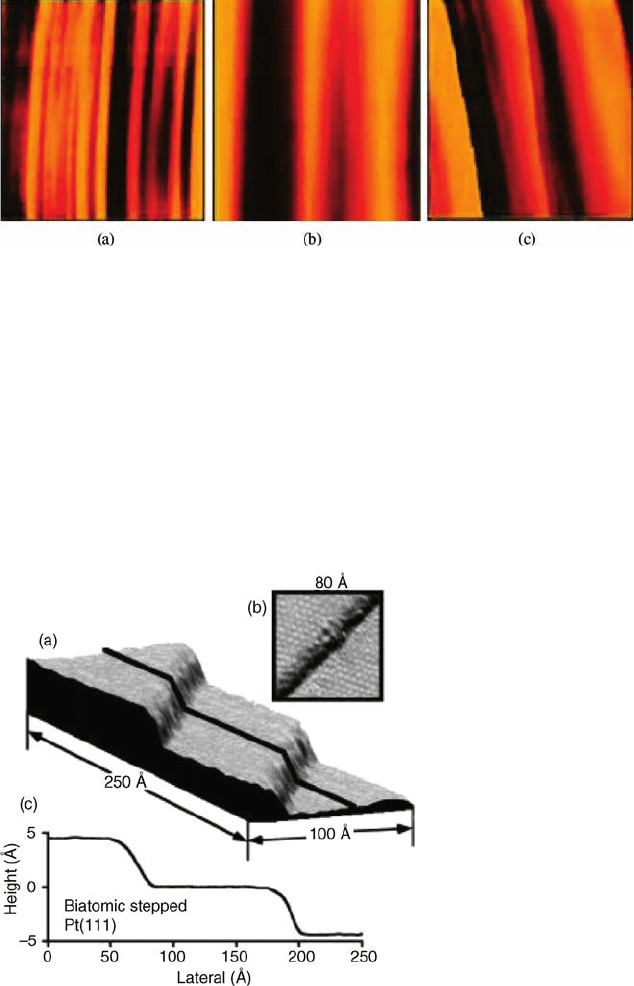
Figure 7.17 (a) A 250 Å 100 Å STM image of
the stepped Pt(1 1 1) surface after depositing
0.25 LM of S and then annealing at 600
C.
(b) Magnified image showing the p(2 2)-S
structure covering the terraces. (c) Line-profile
analysis for the section marked with a line in (a).
This analysis clearly shows the double-step
heights formed by S chemisorption.
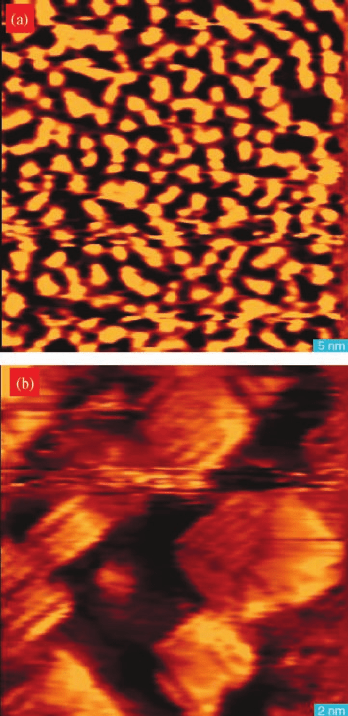
Figure 7.16 (a) Topographic image of Pt(1 1 0)
annealed at 150
C in 1.6 atm H
2
for 5 h,
showing (1 n) missing row reconstruction
randomly nested. Image size: 730 Å 700 Å
vertical range Dz ¼10 Å. (b) Topographic
image of Pt(1 1 0) annealed at 150
C in 1 atm
O
2
for 5 h. Image size: 900 Å 780 Å vertical
range Dz ¼25 Å. (c) Topographic image of Pt
(1 1 0) annealed in 1 atm CO for 4 h. Image size:
770 Å 740 Å vertical range Dz ¼42 Å.
7.4 Mobility and Flexibility of Catalyst Surfaces
j
205
step surface to a background of CO (1 10
6
Torr) causes the step with double-atom
height to split into two monoatomic height steps (Figure 7.18). This suggests the
flexibility of the Pt catalyst surface under a relatively high pressure of reactant gas. In
fact, the restructuring of the adsorbate-covered surface by coadsorption of a second
reactant gas stems from three factors: a repulsive electrostatic interaction upon CO
adsorption at the step edge, the mechanistic interaction, and an effective repulsive
interaction resulting from entropy [25].
7.5
Adsorbate Mobility During Catalytic Reactions
Heterogeneous catalysis is by nature a surface phenomenon. An efficient catalytic
system under reactive conditions demands a delicate energetic balance. For a
catalytic reaction to occur, reactant molecules must adsorb on a surface, react, and
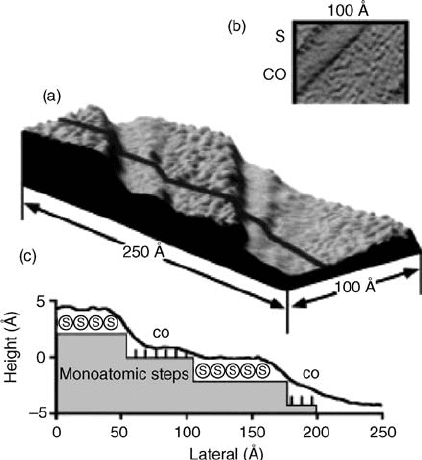
Figure 7.18 (a) A 250 100 Å STM image of
the S-covered Pt(1 1 1) after adsorption of CO
from a background pressure of 1 10
6
Torr
that is maintained during imaging. The double
steps visible in Figure 7.17 before CO
coadsorption are split into monoatomic steps
separated by new terraces that contain
exclusively CO. These new terraces appear as
smooth bands in the image. Sulfur is
compressed in the remaining alternating
terraces and appears as disordered maxima in
the image. (b) A close view of the alternating
CO- and S-covered areas. (c) A cross section of
eight averaged scan lines along the line marked
in (a), showing the single-atom step heights.
The difference in heights of the S- and CO-
covered terraces is due to the difference in
tunneling probability,which is lower throughCO
than through S. The large circles represent S
atoms and the small lines represent CO
molecules.
206
j
7 Surface Mobility of Atoms and Molecules Studied with High-Pressure STM
then desorb in a cyclical process, leaving the active site of the catalyst free for the
next set of reactants. Depending on the specific reaction, this may involve multiple
species with differing adsorption energetics and mechanisms. There are also a wide
variety of reaction mechanisms. For bimolecular reactions, these can be classified
into two types: a reaction between two adsorbed species, which is known as the
Langmuir–Hinshelwood mechanism, and a reaction between an adsorbed species
and a colliding gas-phase species, which is known as the Eley–Rideal mechanism.
The collision frequency, the sticking coefficient, the residence time, the diffusion
rates of the surface species, the concentration of active sites, and the presence of
poisoning species all play an important role in the rate of reaction. For reactions
involving two adsorbed species, the density of surface vacancies and their role in the
diffusion rates of the adsorbed species will have a large effect on the turnover rate.
This complex interplay of energetic factors is the central issue of all catalytic
behavior [13].
There are a wide variety of techniques that can provide molecular-level information
about a surface [13]. The majority of these techniques give ensemble averages of a
large area and many are restricted to a working condition of high vacuum. STM can
provide a valuable complement to surface studies on well-defined extremely flat
systems because it is capable of atomic resolution imaging of the surface structure
and morphology under a wide range of pressures and temperatures. We first applied
STM technique to study catalytic reactions in situ [2]. This technique combines the
ability to prepare well-ordered atomically clean samples with the capability to study
those samples under realistic catalytic conditions. This section will discuss the
application of STM to catalytically active systems and the effect it has had on the
characterization of the adsorbate layer.
Much attention has been given to the pressure gap in heterogeneous catalysis [27],
which refers to the energetic disparity in systems at UHVand at industrially relevant
pressures. Under UHVconditions, a sample might average 10
4
gas-phase collisions
per surface atom per second compared to 10
9
collisions per surface atom per second
at atmospheric pressure. The energy transfer from these gas-phase collisions as well
as from adsorption, desorption, and reaction events could result in a significantly
higher occurrence of high-energy barrier processes compared to the low-pressure
system. These factors underline the advantages of studying catalytic systems in situ
with high-pressure STM.
7.5.1
Ethylene Hydrogenation on Pt(1 1 1)
CO poisoning of the ethylene hydrogenation reaction on Pt(1 1 1) at high pressure
was studied by our group [28]. A clean Pt(1 1 1) surface was prepared with sputtering
and annealing cycles in the sample preparation chamber and then transferred to the
STM chamber. Surface morphology of this single-crystal model catalyst was exam-
ined after the reactant gas was introduced into the STM chamber. At both 20 mTorr
H
2
(Figure 7.19a) and with a mixture of 20 mTorr H
2
and 20 mTorr C
2
H
4
(Figure 7.19b), there was no ordered structure detected in the STM images. When
7.5 Adsorbate Mobility During Catalytic Reactions
j
207

the H
2
/C
2
H
4
mixture was dosed into the chamber, the hydrogenation reaction was
monitored by the production of ethane in the mass spectra. The absence of ordering
was attributed to diffusion of the adsorbate species on timescales much faster than
the scan speed of the STM tip, which was estimated to be 10 mms
1
. This phenom-
enon has been observed by Dunphy et al., who were able to show an ordered structure
through a correlation analysis of an adsorbed layer diffusing slightly faster than the
acquisition time of the STM images [29]. When CO was added to the H
2
/C
2
H
4
mixture, the reaction was poisoned and an ordered structure, a Moir
e interference
pattern, was observed by STM (Figure 7.17c and d). This Moir
e pattern stems from
the interference between the hexagonal surface lattice of the Pt atoms of Pt(1 1 1) and
a different hexagonal lattice for the adsorbed CO molecules.
The adsorption of hydrogen, ethylene, and CO on Pt(1 1 1) was extensively
studied. Molecular hydrogen dissociatively adsorbs on the catalytic Pt(1 1 1) surface
Figure 7.19 (100 Å 100 Å) STM images of
the Pt(1 1 1) surface under different
pressures: (a) 20 mTorr H
2
, (b) 20 mTorr H
2
and 20 mTorr ethylene, and (c) 20 mTorr H
2
,
20 mTorr ethylene, and 2.5 mTorr CO.
The presence of CO induced the
formation of a (H19 H19)R23.4
structure
on the surface. (d) 200 Å 200 Å STM image
showing two rotational domains of
(H19 H19)R23.4
.
208
j
7 Surface Mobility of Atoms and Molecules Studied with High-Pressure STM
and the dissociated hydrogen atoms bind preferentially to threefold hollow sites [30].
The ethylene on Pt(1 1 1) exhibits obvious temperature dependence. Below 50 K,
ethylene physisorbs on the surface. Above 60 K, it forms a di-s-bonded species at two
adjacent hollow sites [31]. At room temperature, this species undergoes proton
transfer to form ethylidyne (CCH
3
), which binds most strongly at hollow sites
with the CC bond perpendicular to the surface. At high pressure and 300 K, another
adsorbate is formed. This adsorbate, a p-bonded ethylene molecule, weakly binds to
an on-top site through p-stacking with the C¼C bond parallel to the surface.
Ethylidyne promotes the formation of the p-bonded species by binding to the
threefold sites and leaving on-top sites open. The weakly p-bonded ethylene molecule
then acts as the reactive surface intermediate for ethylene hydrogenation. CO on
Pt(1 1 1) is one of the most extensively studied model systems in heterogeneous
catalysis. It was studied using several surface techniques such as high-pressure
STM [32], ambient pressure XPS [33], and surface-specific sum frequency generation
vibrational spectroscopy [34]. The structural evolution of adsorbed CO in the entire
pressure range from UHV to one atmosphere was studied with STM [32]. The high-
pressure structure forms a moir
e interference pattern resulting from the hexagonal
symmetry of the adsorbed CO overlaid on the hexagonal packed Pt(1 1 1) surface. The
CO layer is incommensurate with the Pt atoms until the pressure reaches 760 Torr,
when it forms the commensurate (H19 H19)R23.4
-13CO phase. The incommen-
surate phase results from the electrostatic repulsion of the neighboring CO adsorbate
molecules [32].
The absence of a visible ordered structure of hydrogen or ethylene adsorbates
under STM suggests a high mobility of the adsorbed reactant species. The mea-
surement of ethane under the above reaction condition suggests that molecular
mobility is necessary for product formation. The formation of an ordered structure
after CO is introduced into the mixture of high-pressure hydrogen and ethylene
confirmed the mobility of reactant species and the existence of surface vacancies.
More importantly, the comparison of structural information obtained before and
after the introduction of CO demonstrates a method to use high-pressure STM to
study the mobility of molecular adsorbates on catalyst surfaces. This approach can
definitely be applied to studies of the molecular mobility of other catalysis reactions
under realistic conditions. Particularly, the combination of HPSTM with other high-
pressure techniques that identify chemical composition and vibrational signatures
will provide a clear picture of the catalytic mechanism of reactions performed under
realistic conditions.
7.5.2
Hydrogenation of C
6
Cyclic Hydrocarbons on Pt(1 1 1)
The adsorption of cyclohexene, cyclohexane, 1,3-cyclohexadiene, 1,4-cyclohexadiene,
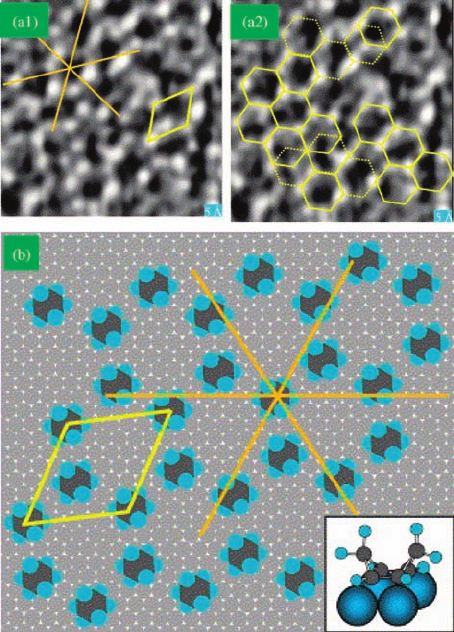
and benzene on Pt(1 1 1) was studied with STM [35, 36]. Figure 7.20a shows an STM
image of 2 10
6
Torr cyclohexene on Pt(1 1 1). The low-pressure structure shows a
hexagonal symmetry with a periodicity of approximately 7 Å that is rotated approx-
imately 18–20
with respect to the [1 1 0] direction of the Pt crystal face. From prior
7.5 Adsorbate Mobility During Catalytic Reactions
j
209
studies of this system using SFG, high-resolution electron energy loss spectroscopy
(HREELS), and thermal desorption spectroscopy (TDS) [37, 38], this structure can be
assigned as a (H7 H7)R19.1
C
6
H
9
p-allyl structure, which preferentially binds in a
chair configuration to threefold hollow sites (inset of Figure 7.20b). The partially
dehydrogenated p-allyl adsorbate structure is similar to that formed from the
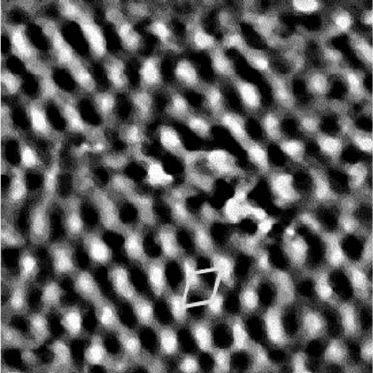
adsorption of low-pressure cyclohexane (Figure 7.21). Molecular cyclohexane is not
the most stable adsorbate species, which leads to a similar dehydrogenation to the p-
allyl covered surface [38]. The adsorbed structure of the further dehydrogenated
species was also imaged.
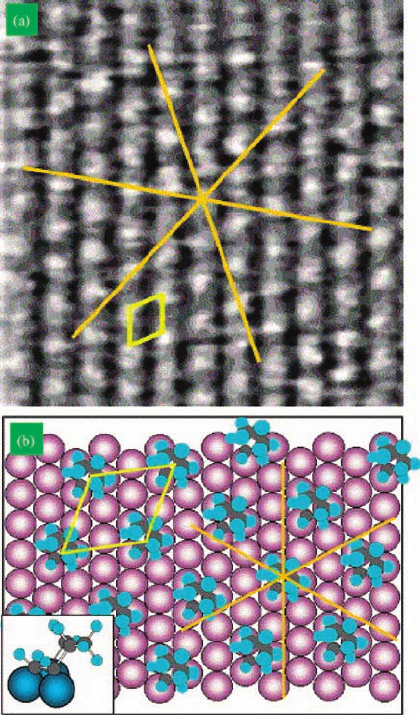
Figure 7.20 (a) 60 Å 60 Å STM image of
Pt(1 1 1) in the presence of 2 10
6
Torr
cyclohexene at 300 K. Periodicity of
approximately 6.5 Å. Spectroscopy studies
have found surface species to be p-allyl.
Lines represent [1 1 0] directions of underlying
platinum. A unit cell of the adsorbate structure is
drawn. (b) Diagram of the proposed (H7 H7)
R19.1
model for the p-allyl structure in
Figure 7.18a. Inset shows bonding structure.
210
j
7 Surface Mobility of Atoms and Molecules Studied with High-Pressure STM

At a pressure of 1 10
5
Torr 1,4-cyclohexadiene, the surface shows ordered
domains of hexagonal rings approximately 18 Å in diameter made up of six adsorbed
molecules with an intermolecular distance of approximately 10 Å (Figure 7.22a).
A(H43 H43)R7.6
structure was proposed as schematically shown in Figure 7.22b.
1,4-Cyclohexadiene adsorbs in a boat configuration on bridge or hollow sites on the Pt
(1 1 1) surface with comparable adsorption energies of 145.6 and 141.6 kJ mol
1
,
respectively [39].
1,3-Cyclohexadiene and benzene form identical structures on Pt(1 1 1) at low
pressures (Figures 7.23 and 7.24). 1,3-Cyclohexadiene dehydrogenates to form
benzene on the surface, while benzene adsorbs molecularly. Figure 7.24b schemat-
ically shows the adsorbed benzene structure at low pressure. The STM images of the
C
6
cyclic hydrocarbons show three different adsorbed structures on Pt(1 1 1).
Cyclohexene and cyclohexane partially dehydrogenate to form p -allyl, 1,4-cyclohex-
adiene adsorbs in a boat configuration, and both 1,3-cylohexadiene and benzene
adsorb as molecular benzene on the surface.
The hydrogenation of unsaturated C
6
cyclic hydrocarbon molecules on Pt(1 1 1)
was studied with STM at high pressure with the same approach as the hydrogenation
of ethylene in Section 7.5.1. At 300 K, with 200 mTorr hydrogen and 20 mTorr
cyclohexene, the hydrogenation reaction forms cyclohexane. The absence of an
ordered structural feature of the reactant species in the STM images indicates a
mobile adsorbed layer under high pressure. When 5 mTorr CO was then added to the
chamber in addition to the 200 mTorr hydrogen and 20 mTorr cyclohexene already
present, the reaction was poisoned and the Moir
e interference pattern indicative
of high-pressure CO structure was observed. The experiment on Pt(1 1 1) with
Figure 7.21 75 Å 75 Å STM image of Pt(1 1 1) in the presence
of 2 10
6
Torr cyclohexane at 300 K. Periodicity of approximately
7 Å. Spectroscopic studies suggest that the surface species is
p-allyl.
7.5 Adsorbate Mobility During Catalytic Reactions
j
211
200 mTorr hydrogen and 20 mTorr cyclohexene was performed at 353 K.The increase
in temperature favors the dehydrogenation reaction pathway.Thus, both cyclohexane
and benzene products were measured and the rate of cyclohexane formation was
found to be twice that of the benzene. When 5 mTorr CO was introduced into the
chamber, the reaction was poisoned but no ordered structure was observed in STM
images. Since CO desorbs at 410 K in UHV [40] and the mobility of reactant species
increases at elevated temperature, it is not surprising that the more weakly bound
high-pressure structure becomes disordered at 353 K. Despite the mobile adsorbate
layer, the reaction was poisoned, which indicates that the rate of vacancy generation
was still not sufficient to activate the reaction. When the sample was subsequently
cooled, the STM images showed an ordered CO structure on the surface. This study
Figure 7.22 (a) 50 Å 50 Å STM image
showing the structure formed by 1,4-cyclo-
hexadiene on Pt(1 1 1) at 1 10
5
Torr and
300 K. Lines in the [1 1 0]-type directions of
the underlying platinum lattice are drawn.
The surface species form hexagonal units in
domains containing a few unit cells and in
antiphase relationship with each other, as
shown in the image on the right (dashed
hexagons are extrapolated cells). The periodicity
and rotation suggest a (H43 H43)R7.6
structure. (b) Diagram of the proposed
(H43 H43)R7.6
1,4-cyclohexadiene
structure. Inset shows bonding orientation.
212
j
7 Surface Mobility of Atoms and Molecules Studied with High-Pressure STM
was able to identify the low-pressure adsorbate structure of all of the C
6
hydrocarbon
species and correlate the mobility of the adsorbed layer with the reactivity of the
catalytic versus the poisoned surface.
These systematic studies suggest that an intrinsic connection between the adsor-
bate structure, mobility, and the formation of product can be established with the aid
of structural information obtained from high-pressure STM. It further demonstrated
the importance of STM in studies of heterogeneous catalysis at high pressure.
7.5.3
CO/NO Coadsorption on Rh(1 1 1)
The coadsorption of CO and NO on Rh(1 1 1) was studied by high-pressure STM [41].
CO and NO conversion on rhodium catalysis is part of the catalytic converter reaction
for automobile emissions and has been extensively studied. After cleaning the Rh(1 1
1) crystal with sputtering and annealing cycles, the sample was isolated in the high-
pressure cell, which was then filled with 500 mTorr CO. CO was imaged as a (2 2)-3
structure binding on Rh top sites. When 150 mTorr NO was introduced into the
chamber, individual bright spots began to accumulate within the (2 2)-3 structure.
The bright spots were observed with an apparent height 0.3 Å more than CO and were
assigned to molecularly adsorbed NO. When the NO pressure was increased to
700 mTorr, adsorbed NO accounted for roughly one-quarter of the surface layer on
the CO precovered surface at room temperature. By counting the mole fraction of top
site NOas a function of the molar fraction of gas-phase NO, an Arrhenius relationwas
plotted yielding the relative binding energies of CO and NO on top sites [41]. CO was
determined to bind 66 5 meV more strongly than NO at top sites. Figure7.25 shows
Figure 7.23 100 Å 100 Å image of Pt in the presence of
1 10
5
Torr 1,3-cyclohexadiene at 300 K. The surface structure,
(2H3 2H3)R30.0
, is similar to that formed by benzene.
7.5 Adsorbate Mobility During Catalytic Reactions
j
213
