Bowker M., Davies P.R. (Eds.) Scanning Tunneling Microscopy in Surface Science, Nanoscience and Catalysis
Подождите немного. Документ загружается.

An A
b
defect-free region (the clean-off area) was created by scanning across the
surface at þ3 V before exposing to water, as shown in Figure 8.4. According to the
assignment of Schaub et al. [15] exposure to water should have little effect in the clean-
off area as there would be no O
b
-vacs with which to react. However, exposing the
surface to water in fact replenishes the A
b
defects in the clean-off area and reduces the
number of A
d
defects (Figure 8.4c). If water dissociates in O
b
-vacs then the number of
O
b
-vacs is expected to decrease and the number of OH
b
species to increase, thus A
b
defects were reassigned as OH
b
and A
d
defects reassigned as O
b
-vacs. This also
means that approximately þ3 V scans and tip pulses remove hydrogen from OH
b
.
While the mechanism for this hydrogen desorption has not yet been elucidated, it
may be related to an empty state identified by Onda et al. [28] approximately 2.4 eV
above E
F
.
Returning to Figure 8.4 because the STM images were recorded from the same
area before and after exposure to water, it can be seen that some of the new OH
b
take
the positions of the reacting O
b
-vacs. Thus, Bikondoa et al. [16, 17] imaged water
dissociating in the O
b
-vacs forming one OH
b
in place of the O
b
-vacs and another OH
b
elsewhere, consistent with the mechanism depicted in Figure 8.1. STM measure-
ments at low temperature show that water dissociates at least down to approximately
187 K [19], a conclusion confirmed by high-resolution electron energy loss
spectroscopy [29].
Surprisingly, the number of OH
b
in positions previously taken by O
b
-vacs always
appears slightly higher than elsewhere. Accordingto the mechanism in Figure 8.1, an
equal number of OH
b
species should appear at the O
b
-vacs and at a distance from
them. Wendt et al. [18, 19] suggest that when the water molecules dissociate, the two
resulting OH
b
lie initially in pairs, adjacent in the [0 0 1] direction. As individual OH
b
from the pair are difficult to resolve with STM, each OH
b
pair therefore appears as a
single feature, which would account for the apparent anomaly. This interpretation is
supported by the observation of three apparent sizes of type-A defects in some STM
images [17– 20, 30]. The largest of these is assigned to OH
b
pairs, the next largest to
isolated OH
b
, and the smallest to O
b
-vacs, an assignment corroborated in recent STM
simulations of O
b
-vacs and OH
b
[30, 31].
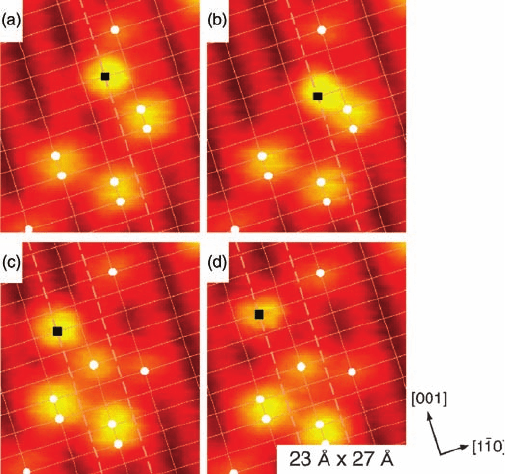
Further evidence for the OH
b
pairs is given in an STM movie recorded at
approximately 187 K. The movie shows the OH
b
pairs separating across the O
b
rows
via proton exchange with water molecules [19]. Key frames from the movie are shown
in Figure 8.5. This water-assisted OH
b
diffusion mechanism is supported by
calculations, which show that the barrier to diffusion is lowered by the exchange
with water. The same diffusion mechanism is also observed for isolated OH
b
and
because of the misassignment of OH
b
and O
b
-vacs by Schaub et al. [15], this water-
mediated diffusion of OH
b
was incorrectly reported as oxygen-mediated diffusion of
O
b
-vacs [32, 33] via a mechanism inconsistent with subsequent isotope studies [34].
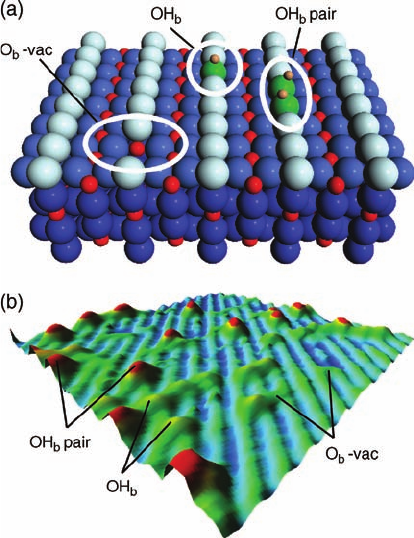
Figure 8.6 summarizes our current knowledge of the appearance of point defects
in STM images. The most prevalent point defects on sputtered/annealed TiO
2
(1 1 0)
1 1 surfaces have been identified as O
b
-vacs, OH
b
, and OH
b
pairs and these are
shown in a ball model together with an STM image decorated with a number of all
three types of defects.
224
j
8 Point Defects on Rutile TiO
2
(1 1 0): Reactivity, Dynamics, and Tunability

Figure 8.4 Sequential (285 Å 250 Å) STM
images of TiO
2
(1 1 0). (a) Sputtered/annealed
TiO
2
(1 1 0) with approximately 3.5 and 5.5% ML
A
b
and A
d
defects, respectively. The blue crosses
denote A
d
defects that are also present in (b),
the red circles denote A
b
defects, and the yellow
circles denote A
b
defects that are removed to
form the image in (b). (b) Following a þ3V
scan. The yellow lines indicate the approximate
boundaries beneath which the þ3 V scan was
applied. Blue crosses and red circles denote A
d
and A
b
defects, respectively. (c) Following
exposure to 0.1 L water. Blue crosses and filled
red circles, respectively, denote A
d
and A
b
defects that were present in (b). Open red circles
indicate positions where A
b
defects were
present in (b) but not in (c). Red crosses denote
new A
b
species that reside where A
d
defects
were positioned in (b) and black crosses denote
new A
b
species appearing elsewhere. The
images are duplicated for clarity in (d), (e), and
(f). (Modified with permission from Ref. [16].)
8.3 Water Dissociation at Oxygen Vacancies and the Identification of Point Defects
j
225
The unambiguous assignment of OH
b
and O
b
-vacs in STM images also paved the
way for the same point defects to be identified in NC-AFM images. Most NC-AFM
images of TiO
2
(1 1 0) show bright rows alternating with dark rows and with dark
depressions appearing on the bright rows. Early interpretations of the NC-AFM
contrast were based on geometrical considerations so that the bright rows were
assigned to O
b
rows and the depressions to O
b
-vacs [35]. The assignment of the bright
rows to O
b
rows was backed up by adsorbing a probe molecule (formic acid) [36] and
by checking the alignment of the bright rows with coexisting strands of the 1 2
reconstruction [37].
Pang et al. exploited information accessible from STM by scanning an area with
NC-AFM then rescanning the same area with STM within minutes [38]. The
experiment was revisited by simultaneously recording STM and NC-AFM images,
an example being shown in Figure 8.7 (Pang et al., unpublished work). In both
experiments, the positions of OH
b
and O
b
-vacs in the STM images correlate
with dark depressions in the NC-AFM images, thus showing that both OH
b
and
O
b
-vacs give rise to dark depressions in NC-AFM. The positions of these OH
b
species
and O
b
-vacs indicate that the bright rows in the NC-AFM image must correspond to
O
b
rows.
Figure 8.5 Sequential images of TiO
2
(1 1 0) showing the splitting
of an OH
b
pair mediated by a water molecule at 187 K.OH
b
groups
are labeled with white circles and water on Ti
5c
sites is labeled with
black squares. Solid grid lines intersect the O
b
sites whereas
dashed gridlines are along Ti
5c
rows. (Modified with permission
from Ref. [19].)
226
j
8 Point Defects on Rutile TiO
2
(1 1 0): Reactivity, Dynamics, and Tunability
In the true surface topography, OH
b
are protrusions and O
b
-vacs are depressions.
As such, the appearance of both OH
b
and O
b
-vacs as depressions in the NC-AFM
image is significant because it means that NC-AFM images do not reproduce the
true surface topography, as is often assumed. In further support of this, Pang et al.
recorded images in the same area with different contrasts, between which there
was presumably some change to the tip (Figure 8.8) [38]. In Figure 8.8a, type-A
defects appear as dark spots in the bright O
b
rows as they do in Figure 8.7b
(Contrast I). However, in Figure 8.8b, the same type-A defects appear as bright
spots between bright rows, which means the bright rows must be assigned to Ti
5c
rows (Contrast II).
Similar contrast inversions were reported by Lauritsen et al. [39] who also
reproduced the tip change in simulations. The tip change was modeled as a
switch between a tip with a positive potential and one with a negative potential.
The positive tip gives images with Contrast I while the negative tip produces images
with Contrast II.
Figure 8.6 (a) Schematic models of the three most prevalent
types of point defects on sputtered/annealed TiO
2
(1 1 0) surfaces.
(b) (120 Å)
2
perspective STM image showing the same defects.
The image is color contoured so that Ti
5c
rows appear green,
O
b
rows appear blue, O
b
-vacs appear green, OH
b
yellow, and
OH
b
pairs appear red.
8.3 Water Dissociation at Oxygen Vacancies and the Identification of Point Defects
j
227

A third contrast regime, which appears more rarely, has also been reported in
which bright rows are imaged with bright spots on them [40, 41]. Fukui and
Iwasawa [40] assigned the bright rows to O
b
rows and the bright spots to OH
b
based
on geometrical considerations. This assignment was supported by simulations that
employed a charge-neutral tip [41].
Figure 8.7 Simultaneously recorded
80 Å 150 Å STM and NC-AFM images of
TiO
2
(1 1 0). (a) A current map represents
the STM image. Because the tip is oscillating
during NC-AFM measurements, the time-
averaged current is recorded. The current
map is color contoured so that OH
b
appear
as broad, red spots and O
b
-vacs appear as
narrow, yellow spots. Arrowheads point at
three O
b
-vacs and circles are drawn over
three OH
b
, respectively. (b) The simultaneously
recorded NC-AFM image with the arrow-
heads superimposed in blue and the circles
superimposed in red, both indicate dark
depressions. (From Pang et al., unpublished
work.)
Figure 8.8 115 Å 185 Å NC-AFM images of TiO
2
(1 1 0).
(a) Before the tip change. (b) After the tip change. Purple arrow-
heads indicate coincident type-A defects. (Modified with
permission from Ref. [38].)
228
j
8 Point Defects on Rutile TiO
2
(1 1 0): Reactivity, Dynamics, and Tunability
8.4
O
2
Dissociation at Oxygen Vacancies
Like water, the interaction of O
2
with TiO
2
also has implications for photocatalysis. As
with water again, reactions of O
2
with TiO
2
are also important because O
2
will form
part of the environment in many TiO
2
applications.
It has been known for some time that the spectroscopic signature of O
b
-vacs can be
healed by exposure to O
2
[42–46]. In addition, Epling et al. [47] show that temperature-
programmed desorption (TPD) spectra of water and ammonia are perturbed when
the surface is predosed with O
2
. This implies that oxygen is left on the surface in some
form when O
b
-vacs are healed by O
2
, As such, Epling et al. proposed that one O
b
-vac is
healed per O
2
molecule with the other O atom being adsorbed at a Ti
5c
site (O
ad
), a
dissociation mechanism supported by theoretical calculations [48, 49].
Bikondoa et al. followed the dissociation of O
2
using STM [16, 17]. Any convolution
of the O
2
reaction with O
b
-vacs and that with OH
b
was avoided by removing OH
b
from an area of the surface by scanning at þ3 V. The same area was then
imaged before and after exposure to approximately 0.6 L O
2
at room temperature,
as shown in Figure 8.9. Following O
2
exposure, the four O
b
-vacs highlighted in
Figure 8.9a are healed and four bright features appear nearby on the bright Ti
5c
rows.
This suggests that O
2
dissociates in the O
b
-vacs, one O atom filling the vacancy and
the other forming O
ad
on a nearby Ti
5c
site. Wendt et al. [18] performed similar
experiments at low temperature, showing that O
2
dissociates in O
b
-vacs at least down
to 120 K.
Detailed analysis of the O
ad
positions following dissociation at room temperature
shows that most O
ad
are found one lattice constant (in the [0 0 1] direction) away
from the O
b
-vac that is filled, the rest being immediately adjacent and two lattice
constants away [50]. As there is little thermal diffusion of O
ad
on TiO
2
(1 1 0), the
separation of O
ad
from the closest positions to the reacting O
b
-vacs was attributed
to the energy released during the exothermic dissociation of O
2
(calculated at
3.5 eV [18]) in a similar way to that observed, for example, for Cl
2
dissociation
on TiO
2
(1 1 0) [51].
8.5
Alcohol Dissociation at Oxygen Vacancies
The reactivity of TiO
2
with alcohols is important in a number of technological
applications. For example, alcohols can be used as energy carriers in renewable
sources and they are also employed as model pollutants so that environmental
cleaning strategies can be tested.
TPD and static secondary ion mass spectrometry (SSIMS) data suggest that
methanol dissociatively adsorbs at O
b
-vacs and molecularly at the Ti
5c
sites [52, 53].
There is also some evidence that methanol also dissociates at other sites apart from
O
b
-vacs, presumably Ti
5c
sites [53–55]. Similar conclusions have been reached for a
series of short-chain (C2–C8) aliphatic alcohols [56–58].
8.5 Alcohol Dissociation at Oxygen Vacancies
j
229

Figure 8.10b shows an STM image of the TiO
2
(1 1 0) surface after a low exposure to
methanol at approximately 300 K [59]. Bright spots are found in place of O
b
-vacs.
These bright spots were attributed to pairs of methoxy/OH
b
, by analogy with the OH
b
pairs formed from water dissociation [18–20]. After dosing more methanol, further
bright spots appear together with a number of darker spots (Figure 8.10c). The darker
spots are assigned to OH
b
that have separated from the methoxy/OH
b
pairs across
the O
b
rows via proton exchange with molecularly adsorbed methanol, through
essentially the same mechanism as described for water in Section 8.3.
Figure 8.9 Reaction of O
2
with O
b
-vacs on
TiO
2
(1 1 0). (a) 100 Å 120 Å STM image of
TiO
2
(1 1 0) following a þ3 V scan to remove
OH
b
. The O
b
-vacs are marked with blue crosses
except for four O
b
-vacs that do not appear in
(b) that are instead marked with purple crosses.
(b) STM image with the same size and scan
parameters as (a), following exposure to 0.6 L
O
2
.O
b
-vacs that coincide with those in (a) are
indicated with blue crosses. Bright spots that
appearonthebrightrows,closetothepositionof
the O
b
-vacs in (a) are indicated with yellow
crosses. The images are duplicated for clarity in
(c) and (d). (e) Schematic representation of the
reaction. Oxygen originating from the exposed
O
2
is colored yellow. The O
ad
is positioned
diagonally adjacent to the O
b
-vac after Du et al.
[50]. (Modified with permission from Ref. [16].)
230
j
8 Point Defects on Rutile TiO
2
(1 1 0): Reactivity, Dynamics, and Tunability

As it is known that high tip bias (approximately þ3 V) can remove OH
b
[16–18],
Zhang et al. performed a high bias scan in order to distinguish the dark and bright
spots. The darker spots were removed but the brighter spots remained, thus
confirming that the darker species are OH
b
and the brighter spots therefore methoxy.
As the methoxy groups take the positions of the O
b
-vacs, this indicates that the
CH
3
OH bond is broken rather than the CH
3
OH bond.
A similar series of STM experiments were performed for 2-butanol with almost
identical results, evidence again being given for ROH cleavage at O
b
-vacs [60]. In
addition to this, for both methanol and 2-butanol, bright spots appeared on the bright
Ti
5c
sites. Zhang et al. did not discuss these features in the case of methanol [59] but it
seems likely that the bright spots arise from either methanol adsorbed at Ti
5c
sites or
methoxy dissociated at Ti
5c
sites. For 2-butanol, one of the bright spots was seen
Figure 8.10 STM images of the same area
before and after exposure of methanol on
TiO
2
(1 1 0) at 300 K. The dosing pressure is
constant in all images so the dose time is
proportional to the exposure. (a) Before
exposure to methanol. (b) After 80 s expo-
sure to methanol. (c) After 110 s exposure to
methanol. (d) The same area as (c) following
a þ3 V scan. Yellow circles show the position
of O
b
-vacs, blue circles show the bright features
on O
b
-vacs, red squares mark the darker spots.
Green arrows point at bright spots on the
bright Ti
5c
rows. (Modified with permission
from Ref. [59].)
8.5 Alcohol Dissociation at Oxygen Vacancies
j
231

filling an O
b
-vac in sequential STM images (Figure 8.11). This sequence was
therefore interpreted as a 2-butanol molecule initially adsorbed at a Ti
5c
site
eventually dissociating in an O
b
-vac.
As the adsorption behavior of ROH on TiO
2
(1 1 0) is similar, where R ¼H, CH
3
,or
2-butyl, Zhang et al. [60] suggest that all alcohols may behave as follows: (i) the ROH
bond breaks at the O
b
-vac sites with RO filling the O
b
-vac and (ii) ROH adsorbs
molecularly at Ti
5c
sites and facilitates diffusion of OH
b
formed from dissociation at
O
b
-vacs.
8.6
Diffusion of Oxygen Vacancies and Surface Hydroxy
The surface diffusion of defects and adsorbates is of obvious importance in
heterogeneous catalysis, as this process brings the reactants together. Understanding
the dynamics of molecules on oxide surfaces is also a key step toward the realization
of working molecular electronics. We note here that diffusion of O
b
-vacs really means
diffusion of O
b
into the vacancy, which leaves another O
b
-vac in the position vacated
by the O
b
. Similarly, diffusion of OH
b
occurs by diffusion of the H atom.
Zhang et al. [61] recorded STM images between 350 and 423 K to investigate the
diffusion of O
b
-vacs. Figure 8.12 shows two sequential STM images taken at 400 K,
with a difference image being shown in Figure 8.12c. The difference image clearly
shows that the O
b
-vacs diffuse along the O
b
rows. Diffusion was never observed
across the rows in the temperature range investigated. Hopping rates for O
b
-vacs
along the rows were determined at seven temperatures so that an Arrhenius plot
could be performed. This yielded a diffusion barrier of E
b
¼1.15 eV that closely
matches the value given by their DFT calculations (E
b
¼1.03 eV) [61]. Calculations
also show that it is energetically unfavorable for O
b
-vacs to be positioned adjacent to
or close to another O
b
-vac along the O
b
row [48, 61, 62]. This is reflected in the spatial
distribution of O
b
-vacs in STM images. If O
b
-vacs are positioned randomly over the
surface, statistically a 10% density of vacancy sites should lead to 10% of the O
b
-vacs
existing as pairs, yet O
b
-vac pairs have not been reported [61].
Figure 8.11 Sequential STM images showing
a 2-butanol molecule initially adsorbed on a
Ti
5c
site dissociating at an O
b
-vac. (a) Before
dissociation, the 2-butanol molecule is
indicated with a green arrowhead. (b) After
the reaction, the 2-butoxy takes the position
of the O
b
-vac (red arrowhead) and the OH
b
sits adjacent (red cross). (c) Schematic
representation of the dissociation process.
(Reprinted with permission from Ref. [60].
Copyright 2007, American Chemical Society.)
232
j
8 Point Defects on Rutile TiO
2
(1 1 0): Reactivity, Dynamics, and Tunability

As for OH
b
diffusion, Zhang et al. [20, 63] also show that diffusion occurs along the
[0 0 1] direction, distinct from the [1
1 0] direction water-assisted diffusion reported by
Wendt et al. [19]. This is demonstrated in Figure 8.13. Furthermore, by carefully
analyzing the positions of the OH
b
it was shown that the initial hop away from OH
b
pairs is usually taken by the OH
b
formed from the hydrogen, which splits off from the
OH fragment that fills the O
b
-vac, a preference that holds at least from 300 to 372 K.
Therefore, in contrast to previous expectations, the two OH
b
that form from
dissociation of a water molecule appear to be inequivalent. Zhang et al. speculate
that this is due to a different charge distribution around each OH
b
; the O
b
-vac
involves two nominally Ti
3 þ
ions so that when water dissociates, the OH fragment
Figure 8.12 64 Å 69 Å STM images of a
TiO
2
(1 1 0) surface recorded at 400 K with O
b
-
vacs present. (a) and (b) are sequential images
recorded 2 min apart. The Ti
5c
rows appear red
and the O
b
rows appear blue. A schematic
model of the surface is shown to scale above
parts of the image in (a) and (b). Ti atoms are
shown red, and oxygen blue with the O
b
rows
shown a lighter blue. (c) Difference image,
the image in (b) being subtracted from that
in (a). Yellow protrusions indicate the original
positions of O
b
-vacs whereas blue depressions
indicate their positions in (b). (Reprinted with
permission from Ref. [61]. Copyright 2007,
American Physical Society.)
Figure 8.13 STM images of a TiO
2
(1 1 0)
surface recorded at 381 K with OH
b
present. (a)
and (b) are sequential images recorded 3 min
apart. The white arrows in (a) markthe positions
of two remaining O
b
-vacs. (c) Difference image,
in which (a) is subtracted from (b). The dark
spots represent the initial hydrogen positions in
(a) whereas the bright spots show their final
positions in (b). The black arrows show the
hopping directions. (Reprinted with permission
from Ref. [63]. Copyright 2008, American
Chemical Society.)
8.6 Diffusion of Oxygen Vacancies and Surface Hydroxy
j
233
