Bowker M., Davies P.R. (Eds.) Scanning Tunneling Microscopy in Surface Science, Nanoscience and Catalysis
Подождите немного. Документ загружается.

3
In Situ STM Studies of Model Catalysts
Fan Yang and D. Wayne Goodman
3.1
Introduction
The surface science approach to studying heterogeneous catalysis dates back to the
pioneering work of Langmuir [1] in the 1910s that addressed the adsorption of gases
on catalyst surfaces. Since then surface science studies of catalytic processes have
played a central role in our understanding of catalysis and have aided in the design
and improvement of catalysts for energy and environmental uses. The goal of surface
science investigations has been to provide structural and spectroscopic information
of catalyst surfaces at the spatial and temporal limit. Scanning tunneling microscopy
(STM), with the capacity to reach the spatial limit at the atomic level, has ignited
considerable interest since its discovery and has become a widely used tool in catalytic
science.
By following a selected area or a molecule at a model catalyst surface, in situ STM
can provide temporal measurements regarding the elementary steps of catalytic
transformations. The capabilities of in situ STM allow one to follow the dynamic
change of surface species and identify what Taylor described as the active site in
catalytic reactions. Such studies also provide kinetic measurements at the atomic
scale, enabling the most precise modeling of macroscopic reactions. With the
development of STM techniques, a large body of in situ STM work has emerged
in the past decade regarding catalytic processes such as adsorption and diffusion,
surface reaction, and catalyst deactivation. These experiments provide invaluable
insights into the fundamental issues of catalysis.
The timescale of catalytically important processes ranges from 10
12
to 10
4
s, with
the chemisorptions and reactions taking place within picoseconds whereas catalyst
deactivation occurs in seconds or minutes. Although the timescale of the funda-
mental process of adsorption and reaction is beyond the time resolution of STM,
information about the pathway and energetics of adsorption/reaction can be acquired
with STM by monitoring the change in the spatial distribution of surface adsorbates.
Indeed, in situ STM can be combined with femtolasers to probe surface processes at
both the spatial and the temporal limits. The feasibility of combining these two
j
55
techniques has been demonstrated in a recent study by Bartels et al. [2]. Such studies
are extremely promising with respect to our understanding of surface chemical
processes. In this chapter, we show how in situ STM has helped to visualize
elementary steps of chemical reaction and to elucidate mechanisms of catalyzed
processes.
Like most surface science techniques, conventional in situ STM studies have been
carried out in UHV on model catalysts consisting of extended planar surfaces. When
extrapolating the information obtained in UHV surface science studies to real-world
catalysis, two issues have generally concerned the catalysis community, namely, the
pressure and material gaps.
The pressure gap refers to the fact that surface science studies are conducted under
UHVconditions (10
10
–10
14
bar), whereasindustrial catalytic reactions typically are
carried out at high pressures (1–1000 bar). Over 10 orders of magnitude difference in
the pressure of reactant gases can drastically change the interaction of reactants on
the catalyst surface, a process essential to a catalytic reaction. The material gap refers
to the gap between the surface structure of metal single crystals often studied in
surface scienceand that of technical catalysts. Real-world catalysts usually consist of
small metal clusters ranging from 1 to 100 nm in size, finely dispersed onto a high-
surface-area oxide support. These metal clusters can have structures and properties
that are quite different from the bulk metal. In catalytic research, it is well
documented that the reactivity and selectivity of catalysts often depend on the size
and shape of supported metal clusters [3]. Furthermore, the presence of the oxide
support can modify the structure and properties of supported metal clusters.
The effect of cluster size and metal support interaction cannot be addressed in
surface science studies on well-defined single crystal metal surfaces. As a local
structural probe, STM has the advantage of addressing these two issues. Although the
operational range of STM extends from UHV to high pressures, there are challenges
in maintaining the stability of STM at catalytically realistic operating temperatures
and pressures. Nevertheless, it is possible to apply STM to study catalytic reactions
under realistic conditions. To bridge the material gap, supported model catalysts,
consisting of small metal clusters supported on planar oxide surfaces, have been
introduced into surface science studies. STM is ideally suited to precisely charac-
terize the structure of these supported model catalysts. In this chapter, we show the
recent progress in bridging the pressure and material gaps by applying in situ STM
to the study of model catalysts under realistic reaction conditions.
3.2
Instrumentation
To visualize the fundamental steps of chemisorptions and reactions that occur at
surfaces, in situ STM investigations typically monitor the diffusion or transformation
of adsorbed molecules. A series of snapshots of preselected surface regions,
compiled into a STM movie, can reveal the evolution of surface phenomena.
On metal surfaces, the surface diffusion of adsorbates is usually so rapid that the
56
j
3 In Situ STM Studies of Model Catalysts
surface temperature must be lowered below room temperature (RT) for successful
STM viewing. Low-temperature (LT) STM, developed in the mid-1990s, not only
potentially allows the determination of reaction intermediates and pathways but also
enables the precise control and measurement of the bond activation processes using
the STM tip.
To measure reaction kinetics, STM should have the capability to resolve adsorbates
at temperatures relevant to catalytic reactions. For this purpose, a variable temper-
ature (VT) STM is required, as well as capabilities for rapid scanning. VT STM with a
typical scan rate of one frame per minute was developed in the mid-1990s.
Considering the scanning probe is a mechanical probe driven by electronics, the
acquisition time of STM images is typically restricted by the mechanical behavior of
the scanning components and the performance of the electronics. In the mid-1990s, a
few STM groups achieved a fast scan rate of approximately 20 frames/s on extended
model catalyst surfaces [4–6]. Working on the compact design of the scanner probe
and using high performance electronics, Frenken and coworkers [7] have recently
pushed the scan rate above the video rate (50 frame/s) on a graphite surface, with
atomic resolution and an image with 256 256 pixels.
Recently, a few groups have taken up the challenge to extend the in situ STM
investigations to high pressures. A major challenge in imaging surfaces with STM
over a wide pressure range is the sensitivity of the tunneling current to extremely
small changes at the tunneling junction resulting from induced instabilities by the
ambient gas. Efforts have emphasized the design of a STM that can work at high
temperatures and pressures with greater stabilities [8–11].
For in situ STM studies at high temperature and pressures, the inability of being
able to track a preselected surface area is often the limiting factor given the tunnel
junction instabilities and sample drifts. To overcome this challenge and to maintain
contact with a specific surface region, it is important to develop experimental
approaches that pattern the surface without influencing the kinetics and dynamics
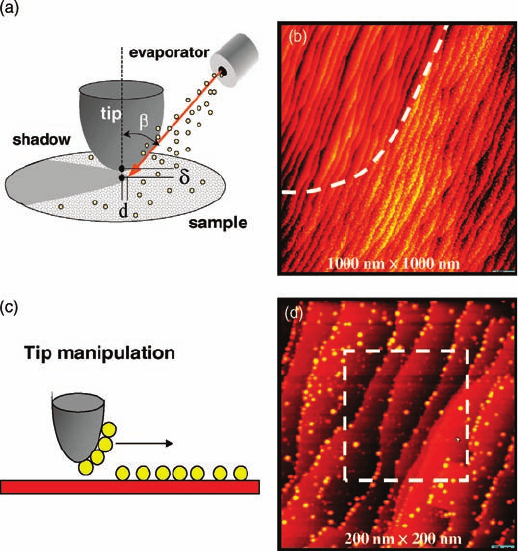
of the particular areas under study. A shadowing technique (Figure 3.1a) has been
developed where metal atoms are dosed with the STM tip in the tunneling position
with the collimated metal flux creating a shadow of the tip on the substrate [12, 13].
For metal clusters supported on an oxide surface, tip manipulation is another method
of choice (Figure 3.1c). This technique removes clusters from a specific area through
aggressive scanning. Using the STM tip to pattern the surface, it is now possible to
monitor a preselected surface area at elevated temperatures while changing the gas
pressure over 12 orders of magnitude.
In addition to the instrumental performance, the STM tip is of primary importance
for in situ STM measurements. Methods for the preparation of STM tips have been
extensively studied with a goal of preparing an atomically sharp tip [14–19]. The STM
tip is important for high-pressure studies with respect to two aspects, tip selection
and in situ tip regeneration. To ensure a continuous DOS near the Fermi level,
transition metals are usually selected to prepare STM tips. In the presence of reactant
gases, especially under high-pressure and high-temperature conditions, the chem-
ical and thermal stability of the STM tip becomes the ultimate limit for reaction
studies and thus the major concern in tip selection. Tungsten tips are very stable in
3.2 Instrumentation
j
57
CO but perform poorly in the presence of O
2
or mixtures of CO and O
2
. Platinum or
platinum alloy tips are stable in O
2
but suffer from adsorption of CO, especially when
the sample surface temperature is above 550 K [20]. Gold is stable in both CO and O
2
but unstable at high temperatures, especially in the presence of water. In addition to
tip selection, in situ tip regeneration or cleaning is also critical for STM studies in the
presence of high-pressure reactant gases because the STM tip is susceptible to
picking up poorly conducting components during extended measurements at
elevated pressures. In situ tip regeneration refers to the method of applying a large
voltage pulse (from a few to hundreds of volts) between tip and sample while the tip is
in tunneling range. This method induces field emission, which cleans and regen-
erates the STM tip. Wintterlin and coworkers [21] recently reported a high-pressure
STM study, in which tungsten tips were used to study ethylene oxidation on Ag(1 1 1);
the tip could be recovered by applying high voltages to the tip (e.g., þ300 V).
Figure 3.1 Methods of patterning the surface
for in situ STM studies. (a) Schematics of
shadow technique. (Reprinted with
permission from Ref. [12]. Copyright 2002,
Wiley, Inc.) (b) STM image of the surface created
by shadow technique. The shadow area
uncovered by metal clusters is distinguished
from the area covered with metal clusters by the
white dash line. (c) Schematics of the tip
manipulation. (d) STM image of the surface
created by tip manipulation. The dash rectangle
in (d) shows the area where most clusters are
picked up by the STM tip. This area with lower
cluster densities can be distinguished from the
rest of the surface and serves as a nanomarker
for in situ STM studies.
58
j
3 In Situ STM Studies of Model Catalysts
Gas purification is another important issue for high-pressure STM studies. When
the surface is exposed to high pressures, even highly diluted impurities may
completely contaminate the surface. Purification of all reactant gases using liquid
N
2
can greatly improve the operating pressure range for high-pressure reaction
studies and prevent the electrical breakdown often induced by humidity when
backfilling the STM chamber [13]. For a flow-reactor system, where purification of
large volume of gases is required, a heated zeolite filter is effective in removing
carbonyl contaminants from the gas flow [22, 23].
3.3
Visualizing the Pathway of Catalytic Reactions
With the introduction of LT and VT STM, it is now possible to monitor the
fundamental steps of chemical reactions, that is, reactant chemisorption, diffusion,
and catalytic transformation. A detailed review covering this subject was published by
Wintterlin in 2000 [24]. Since then, in situ STM studies have flourished and expanded
to the visualization of the reaction pathway and kinetics of surface processes. In the
following section, we highlight selected examples of recent progress in using in situ
STM for studying fundamental catalytic processes.
3.3.1
Imaging of Adsorbates and Reaction Intermediates
STM can induce adsorption–desorption and dissociation processes nonthermally
and with spatial control. At low temperatures, with limited surface diffusion, the
motion of adsorbates can be controlled and allows the determination of surface
reaction intermediates. The catalytic oxidation of CO on precious metal surfaces is
one of the most important model reactions in heterogeneous catalysis. Hahn and
Ho [25] have recently used in situ STM to visualize the reaction pathway of CO
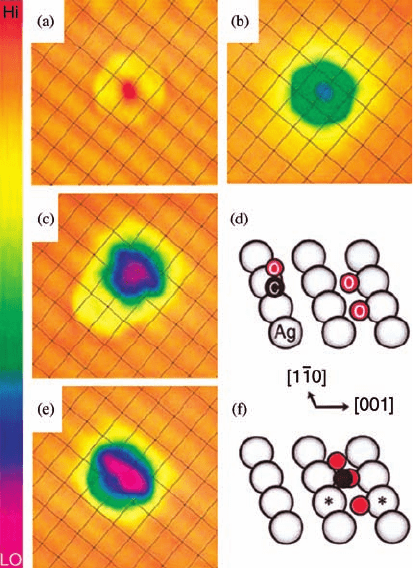
oxidation on Ag(1 1 0) at 4 K. Figure 3.2 depicts the pathway believed to be operative:
the Langmuir–Hinshelwood mechanism. Figure 3.2a and b shows a CO molecule
and a pair of oxygen atoms adsorbed on the Ag(1 1 0) surface, respectively. The oxygen
atom pair was formed by placing the tip over a molecularly adsorbed O
2
and raising
the sample bias to 0.47 V. Subsequently, the bond between two oxygen atoms
is broken and the two oxygen atoms adsorbed at the nearest fourfold sites of the
Ag(1 1 0) surface show slight elongation along the [1
1 0] direction in Figure 3.2b. The
STM tip was then placed over the CO molecule with a þ0.24 V bias applied
repeatedly, causing the molecule to diffuse across the surface. Eventually, the CO
molecule moved close to the pair of O atoms (Figure 3.2c) and then joined them to
form the OCOO complex (Figure 3.2e). With an additional pulse of the sample
bias over the CO molecule, the OCOO complex is decomposed, leaving an oxygen
atom on the surface with the CO
2
desorbing from the Ag surface.
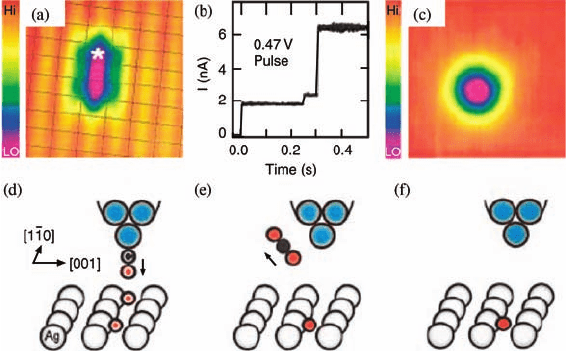
A second reactionpathway wasalso illustratedby moving aCO molecule toward the
absorbedoxygenatoms. In Figure3.3a,anSTMtipwith a COmolecule adsorbedatthe
3.3 Visualizing the Pathway of Catalytic Reactions
j
59
tip end was positioned over anoxygen atom, which lined up with another oxygen atom
fromthe O
2
dissociation.Thetwooxygenatomswereseparatedfromeachotherbytwo
lattice spacings. With a pulse of þ0.47 V sample bias, the CO molecule is detached
from the STM tip and reacted with the oxygen below the tip to form CO
2
. Figure 3.3b
shows the two pulses of tunneling current, corresponding to the CO molecule,
impinging on the surface and reacting with the adsorbed oxygen, and to CO
2
desorption, respectively. Figure 3.3c shows the oxygen atom left on the Ag(1 1 0)
surface. The combined imaging, manipulation, and spectroscopic capabilities of the
STM provide direct visualization of reaction pathways at the single molecule level.
At low temperatures, tip manipulation has been regularly used to promote surface
diffusion, to activate bonds, and to synthesize molecules. Recent progress along these
lines is described by Hla and Rieder [26, 27].
Figure 3.2 STM images obtained with a
CO-terminated tip, V
t
¼70 mV and I
t
¼1 nA.
(a) Isolated CO molecule, (b) two O atoms
(adsorbed on the nearest fourfold hollow sites
along the [1
1 0] direction), (c) CO and two
O atoms separated by 6.1 Å along the [0 0 1]
direction, and (e) OCOO complex. Grid
lines are drawn through the silver surface
atoms. Scan area of (a–c) and (e) is 25 Å 25 Å.
Schematic diagrams for adsorption geometries
of (c) and (e) are shown in (d) and (f ),
respectively; a linear atop and a tilted off-site CO
are implicated. The black (red) circles represent
carbon (oxygen) atoms and the large gray circles
are silver atoms. The sizes of the circles are
scaled to the atomic covalent radii. (Reprinted
with permission from Ref. [25]. Copyright 2001,
The American Physical Society.)
60
j
3 In Situ STM Studies of Model Catalysts
3.3.2
Imaging Chemisorption on Metals
Under catalytic reaction conditions, adsorbates are usually mobile on the surface. The
diffusion of adsorbates has been studied both on metal surfaces and on oxide surfaces
by in situ STM. On metal surfaces, it has been shown that at low surface coverage,
adsorbates are mobile and are distributed randomly on the surface. As the surface
coverage is increased, the interaction between adsorbates also changes such that an
attractive interaction begins to appear, leading to the formation of adsorbate islands.
These adsorbate islands are in equilibrium with the diffusing adsorbates (2D gas) at
the surface. Eventually, with an increase in the adsorbate coverage, the adsorbate
islands grow into an adsorbate overlayer. In situ STM studies by Wintterlin et al. [4]
and by Wong et al. [28] both illustrate that the diffusion rate of adsorbates on metal
surfaces depend on their coverage and/or nearest neighbors. The surface diffusion
coefficient measured for adsorbates on metal surfaces is meaningful only at very low
surface coverages where the adsorbate–adsorbate interaction has a negligible influ-
ence on adsorbate diffusion.
The structure of the adsorbate layer formed at high surface coverage is more
relevant to catalytic reactions at high pressures. Besenbacher and coworkers [29–33]
found for CO on Pt(1 1 0) and Pt(1 1 1) and NO on Pd(1 1 1) that the structure of high
Figure 3.3 Reaction of a CO molecule released
from a CO-terminated tip with an O atom
adsorbed on the surface. (a) STM image, taken
with a CO-terminated tip, of two O atoms
separated by two lattice spacings (2 2.89 Å)
along the ½1
10 direction. Grid lines are drawn
through the silver surface atoms. (b) Tunneling
current during a 1470 mV sample bias pulse
with the CO-terminated tip over one of the two
O atoms (denoted by
). Two current rises
(at 250 and 310 ms) indicate the moments of
desorption and reaction of CO from the tip and
the moment of desorption of CO
2
into vacuum.
(c) STM image of the same area rescanned after
the pulse, showing CO on the tip has reacted
away. Scan area of (a) and (c) is 25 Å 25Å.
(d–f) are the schematic diagrams for (a–c),
respectively. (Reprinted with permission from
Ref. [25]. Copyright 2001, The American Physical
Society.)
3.3 Visualizing the Pathway of Catalytic Reactions
j
61

adsorbate coverages, formed at low-temperature and low-pressure conditions, is
identical to the structure formed at room temperature and high pressures. This
finding suggests that on metal surfaces, reaction studies at high surface coverage
conducted at low temperature likely connect with real catalytic processes at high
pressures.
By studying the diffusion of surface adsorbates (or adsorbate vacancies), in situ
STM can be used to determine the active site for chemisorptions. Mitsui et al. [34–36]
studied the process of hydrogen dissociation on Pd(1 1 1) using in situ STM. Pd is a
catalyst widely used in hydrogenation and dehydrogenation reactions. Exposure of Pd
to H
2
leads to dissociative adsorption. At approximately 65 K and in the presence of
2 10
7
Torr of H
2
, Pd(1 1 1) is nearly saturated with H atoms leaving only a few
vacancies as sites for the dissociation of adsorbed H
2
molecules (Figure 3.4). Due to
the inversion of the image contrast caused by the adsorption of H atoms on the STM
tip, the empty surface sites are imaged as protrusions. This surface can be used to
model the Pd(1 1 1) surface under high-pressure H
2
at room temperature.
Figure 3.5 shows a sequence of STM images acquired for the same surface region.
Figure 3.5a depicts a number of isolated vacancies (bright spots) separated by more
than one Pd lattice, as well as two aggregates of vacancies (marked by dashed circles).
Each vacancy aggregate consists of a pair of vacancies (dimer) occupying neighboring
fcc sites. The dimers in Figure 3.5 are always imaged as a three-lobed object because
of the fast diffusion of a neighbor H atom, which can hop over bridging sites to
occupy the vacancy pairs without getting close to other H atoms. The vacancy pairs or
dimers are most frequently encountered in the STM study. Isolated vacancies can hop
Figure 3.4 The 6.5 nm 6.5 nm STM image of Pd(1 1 1) with a H
coverage near one monolayer. Numerous H vacancies, visible as
bright protrusions, are present. V
t
¼45 mV and I
t
¼2.7 nA.
Streaks and fractional protrusions are due to vacancies moving
while the tip is scanning over them. (Reprinted with permission
from Ref. [35]. Copyright 2005, Springer.)
62
j
3 In Situ STM Studies of Model Catalysts

randomly on the Pd surface and occasionally coalesce to form aggregates, as shown in
Figure 3.5b. Vacancies A and B aggregate to form a dimer while vacancies C, D, and E
coalesce to form a three-vacancy aggregate. The vacancy dimer remains together for
several minutes and eventually disintegrates back to isolated vacancies. However, the
three-vacancy aggregate disappears and leaves only one vacancy on the surface
(Figure 3.5c). The authors concluded then that two vacancies in the three-vacancy
aggregate are occupied by H atoms from the dissociation of adsorbed H
2
. The authors
Figure 3.5 STM images from a movie showing
the formation, separation, and annihilation of
H-vacancy clusters. The images on the left
(3 nm 2.5 nm) are repeated on the right with
annotations. (a) Five vacancies near the center
are labeled (A–E) and two triangular vacancy
pairs (2V) are marked with dashed triangles for
reference. (b) Vacancies A and B have formed a
2V cluster indicated by the triangle containing
the number 2. Vacancies C–E have formed a
three-vacancy (3V) cluster, indicated by the
larger triangle containing the number 3.
(c) The 2V pair has separated into isolated
vacancies A and B, while the 3V cluster has been
annihilated by dissociative adsorption of a H
2
molecule, leaving a single remaining vacancy C.
The other 2V clusters separated a few frames
later. (Reprinted with permission from Ref. [34].
Copyright 2003, Nature Publishing Group.)
3.3 Visualizing the Pathway of Catalytic Reactions
j
63
