Bowker M., Davies P.R. (Eds.) Scanning Tunneling Microscopy in Surface Science, Nanoscience and Catalysis
Подождите немного. Документ загружается.

also found, in the presence of 2 10
7
Torr H
2
at 65 K, vacancy aggregates with four
or more vacancies are also filled by H atoms from the dissociation of H
2
within the
aggregates and transformed into a single vacancy or totally annihilated. Through a
series of STM movies on the diffusion of surface hydrogen vacancies, the authors
found that only an aggregate with three or more vacancies could be annihilated by H
2
dissociation. The dimers or isolated vacancies are never occupied by H atoms.
Instead, the dimers always dissociate creating isolated vacancies with an average
lifetime of 10 min at 65 K. From these data, the authors concluded that three or more
empty palladium sites are necessary for the dissociation of H
2
molecules. This
finding is rather surprising since it has traditionally been assumed that two
neighboring empty sites are sufficient for the dissociation of a diatomic molecule.
The discovery of the active sites for H
2
dissociation on Pd(1 1 1) illustrates the power
of in situ STM in addressing the elementary steps of surface reactions and in testing
the conventional assumptions in catalysis.
3.3.3
Determining the Sites for Chemisorption on Oxide Surfaces
On reduced oxide surfaces, the diffusion of an adsorbate is often limited by the
localized bonding, either ionic or covalent, between the adsorbate and the oxide
substrate. The relatively slow diffusion of adsorbates allows chemisorption and
diffusion on oxides to be studied by STMat elevated temperatures. For example, TiO
2
is an excellent photocatalyst for dissociation of water and decomposition of organic
molecules, critical to pollution control and the hydrogen economy. Studies of the
adsorption and diffusion of water, oxygen, and organic molecules on TiO
2
are of
primary importance to our understanding of photocatalysis by TiO
2
.
Being the most stable phase of TiO
2
, the rutile TiO
2
(1 1 0) crystal has been studied
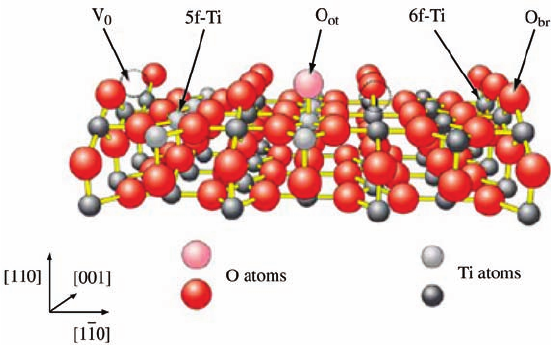
most extensively by STM and other surface science techniques [37]. Figure 3.6 shows
a structural model of the rutile TiO
2
(1 1 0)-(1 1) surface. The surface contains two
types of titanium atoms that form rows along the [0 0 1] direction. Rows of six-
coordinated Ti atoms alternate with five-coordinated terminal Ti atoms, which miss a
single O atom perpendicular to the surface. The surface also contains two kinds of
oxygen atoms, that is, three-coordinated oxygen atoms, sitting in the surface plane,
and bridging oxygen atoms, sitting above the surface plane and bonded to two six-
coordinated Ti atoms. Undersaturated bridging oxygen atoms can be easily removed
from the surface by annealing, electron bombardment, or ion sputtering to form
bridging oxygen vacancies. Bridging oxygen vacancies are the most common and
well-defined defects on the TiO
2
(1 1 0) surface.
STM studies on the adsorption and diffusion of small molecules on the TiO
2
(1 1 0)
surface began in the late 1990s [37–39] and have provided a general understanding of
the important role of bridging oxygen vacancies in the dissociation of water, oxygen,
and small organic molecules. However, due to the difficulties in distinguishing active
surface sites and dissociation products, only recently have the fundamental steps
of adsorption and dissociation processes been understood with the help of
in situ STM.
64
j
3 In Situ STM Studies of Model Catalysts
Wendt et al. [40] and Bikondoa et al. [41] studied the adsorption and dissociation of
water and O
2
on TiO
2
(1 1 0). In situ STM studies, in parallel with the use of DFT
methods, allow surface features such as bridging oxygen vacancies, adsorbed oxygen
atoms, surface hydroxyls, and adsorbed water to be distinguished. Figure 3.7a
illustrates the difference between a bridging oxygen vacancy, a surface hydroxyl,
and adsorbed water in their appearance in STM images. Due to electronic effects,
STM resolves the five-coordinated terminal Ti rows as bright rows whereas the
bridging oxygen rows are imaged as dark rows. The nuances in the appearance of
oxygen vacancies and hydroxyls in STM images are distinguished by visualizing
oxygen vacancies being transformed into OH species in situ. The authors have also
found that applying voltage pulse (3 V) over the hydroxyls desorbs individual H
atoms from the hydroxyl groups while leaving the oxygen vacancies intact. Using in
situ STM, Wendt et al. [42] demonstrated that water dissociation takes place at
bridging oxygen vacancies of the TiO
2
(1 1 0) surface at 187 K and form paired
hydroxyl groups, with one positioned at the oxygen vacancy site and the other formed
at the nearest bridging oxygen site. The diffusion of these pairs is inhibited at 187 K
but can be initiated in the presence of neighboring water molecules adsorbed in the
five-coordinated Ti trough. Through the interaction with neighboring water mole-
cules, hydrogen atoms from the paired hydroxyl groups are transferred to adjacent
bridging oxygen rows, causing the cross-row diffusion of hydroxyl groups.
Dohnalek and coworkers [43, 44] have further measured the diffusion kinetics of
hydroxyl groups (or H atom) at room temperature and above. In situ STM
images (Figure 3.8) have confirmed that H
2
O dissociates at the bridging oxygen
Figure 3.6 The ball model of the TiO
2
(1 1 0) surface. Large gray
(red) balls represent O atoms, small light gray (gray) balls five-
coordinated surface Ti atoms (5f-Ti), and small black balls six-
coordinated Ti atoms (6f-Ti). The bridging oxygen atoms (O
br
),
single oxygen vacancies (V
O
), and O atoms adsorbed on top of the
5f-Ti row (O
ot
) are indicated. (Reprinted with permission from
Ref. [40]. Copyright 2005, Elsevier.)
3.3 Visualizing the Pathway of Catalytic Reactions
j
65
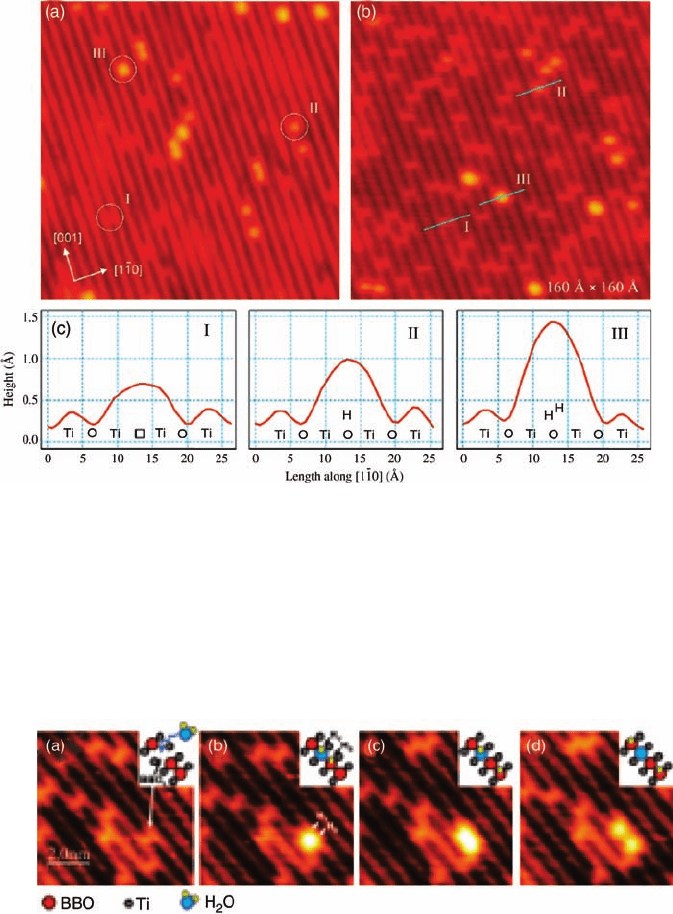
Figure 3.7 STM images (16 nm 16 nm) of clean, reduced
TiO
2
(1 1 0) samples showing the difference between bridging
oxygen vacancies, surface hydroxyls, and adsorbed water. The
sample in (a) was less reduced than the sample in (b). (c) STM
height profiles along the ½1
10 direction of species indicated in
(b). (Reprinted with permission from Ref. [40]. Copyright 2005,
Elsevier.)
Figure 3.8 STM images of the same area on
TiO
2
(1 1 0) at 357 K (V
t
¼1.5 V, I
t
¼0.1 nA)
as a function of time (Dt ¼60 s): (a) clean
TiO
2
(1 1 0) with bridging oxygen (BBO)
vacancies; (b) TiO
2
(1 1 0) with a geminate
hydroxyl pair formed by adsorption and dis-
sociation of a water molecule. H
V
marks the
OH hydrogen and H
B
the hydrogen that split off
from the OH; (c) same area after a single hop
of H
B
; and (d) after subsequent hop of H
V
.
Insets exhibit the ball models illustrating the
corresponding processes. (Reprinted with
permission from Ref. [44]. Copyright 2008,
The American Chemical Society.)
66
j
3 In Situ STM Studies of Model Catalysts
vacancies, producing paired hydroxyl groups. Hydrogen atoms readily diffuse at
room temperature along the bridging oxygen row. However, the two hydrogen atoms
in the paired hydroxyl groups exhibit inequivalent diffusivity. The hydrogen atom
positioned at the healed oxygen vacancy site (H
V
) diffuses much slower than the
one adsorbed at neighbor bridging oxygen sites (H
B
). The different diffusion rates of
H
V
and H
B
were measured between 300 and 410K and the activation barrier of H
B
estimated to be approximately 0.22eV lower than H
V
. The diffusion barrier of
these hydrogen atoms increases with the separation between hydroxyl groups,suggest-
inga repulsive OHOHinteraction.Theauthors speculatedthat a long-livedpolaronic
state is responsible for the inequivalent diffusion rates of H
V
and H
B
.However,the
measured kinetic parameters (prefactors and diffusion barriers) could not be repro-
duced by DFT calculations, suggesting a rather complex diffusion mechanism.
The pathway of dissociation and diffusion discovered in theabove water adsorption
experiment also applies to the adsorption of alcohols on TiO
2
(1 1 0). In situ STM has
been used to study the adsorption of methanol [45] and butanol on TiO
2
(1 1 0) [46].
Figure 3.9 shows a series of STM images obtained on the same area following
exposure to methanol. Figure 3.9a depicts the clean surface before exposure, with
bridging oxygen vacancies marked as yellow circles. The exposure of 0.06 ML
methanol led to the dissociative adsorption of methanol at the bridging oxygen
vacancies of TiO
2
(1 1 0) (Figure 3.9b). The dissociation of methanol forms a methoxy
group at the bridging oxygen vacancy, resolved as bright features in Figure 3.9b, and a
hydroxyl group at the nearest neighbor. The methoxy group has a similar appearance
to the bridging oxygen vacancy in STM images, except that the methoxy group is 0.8 Å
higher than the bridging oxygen vacancy. The hydroxyl group could not be differ-
entiated from the bright features of neighboring methoxy groups in Figure 3.9b.
However, with time, the hydrogen from the hydroxyl group (red dots in Figure 3.9c
and d) diffuses along the bridging oxygen row and across the bridging oxygen rows
through interactions with methanol molecules weakly bounded to the Ti trough. The
diffusing hydrogen atoms were identified by their apparent height in STM images
and by a tip desorption experiment (Figure 3.9e) proposed by Bikondoa et al. [41].
Figure 3.9f gives a graphic illustration of the dissociation and diffusion pathway of
methanol on TiO
2
(1 1 0), which was also observed in the adsorption experiment of
2-butanol (CH
3
CH
2
CH(OH)CH
3
)onTiO
2
(1 1 0) at room temperature [45].
The adsorption and dissociation of O
2
on TiO
2
(1 1 0) have been investigated by
Wendt et al. [40] and Du et al. [47]. Both observed that O
2
molecules adsorb and
dissociate at the bridging oxygen vacancies, with one O adatom healing the vacancy
and the other O adatom bounded to the neighboring five-coordinated Ti site. Du et al.
also analyzed the lateral distribution of the O adatoms upon dissociation and
discovered a transient mobility of O adatoms along the Ti trough in the [0 0 1]
direction. Unlike the dissociative adsorption of O
2
on metal surfaces where both
adatoms have equal diffusivity, the diffusivity of O adatoms on TiO
2
(1 1 0) was found
to be inequivalent. While the O adatoms filling the vacancy are locked in the bridging
oxygen row, O adatoms in the Ti trough are relatively free to move. A majority of O
adatoms on the Ti trough (81%) were found separated from the O adatoms in the
previous vacancy sites by two lattice constants.
3.3 Visualizing the Pathway of Catalytic Reactions
j
67

Figure 3.9 STM images of same area before
and after adsorption of methanol on reduced
TiO
2
(1 1 0) at 300 K (V
t
¼1.0 0.3 V and
I
t
¼<0.1 nA): (a) bare surface; (b) after 80 s
exposure to methanol; (c) after 110 s exposure
to methanol; (d) taken on (c) after spontaneous
tip change; (e) after high bias (3.0 V) sweep of
(c); (f) schematic model of the adsorption
process. Insets show magnified areas marked
by squares. Yellow circles show the position of
bridging oxygen vacancies. Blue circles show the
methoxy groups on oxygen vacancies. Red
squares show H atoms diffusing on bridging
oxygen rows. (Reprinted with permission from
Ref. [45]. Copyright 2006, The American
Chemical Society.)
68
j
3 In Situ STM Studies of Model Catalysts
Zhang et al. [48] have recently measured the stability of bridging oxygen vacancies
on TiO
2
(1 1 0) using in situ STM. Sequences of STM images between 340 and 420 K
suggest that bridging oxygen vacancies migrate along the bridging oxygen row via the
slow diffusion of bridging oxygen atoms with a diffusion barrier of 1.15 eV, in
agreement with DFT calculations. All the above studies suggest that the surface
chemistry of TiO
2
(1 1 0) is dictated by bridging oxygen vacancies, which can account
for approximately10% of the bridging oxygen sites.
However, there are disagreements. Lyubinetsky et al. [49] studied the adsorption
of trimethylacetic a cid ((CH
3
)
3
CCOOH, TMAA), a photoreact ive molecul e, on
TiO
2
(110)atroomtemperature.In situ STM found that the deprotonation of
TMAA to form TMA does not necessarily occur at bridging oxygen vacancies. None
of the hydroxyl groups was found during the adsorption of TMAA. Instead, the
hydrogen a tom was bound to a pair of bridging oxygen atoms and stabilized by
the adjacent TMA groups sitting on the five -coordinated Ti trough. At saturation
coverage, TMAA formed a (2 1) overlayer on the TiO
2
(1 1 0) surface.
Wendt et al. [50] recently studied the interaction between O
2
and TiO
2
(1 1 0) surface
in detail and suggested that bridging oxygen vacancies are only the minor sites that
account for O
2
dissociation. Even though bridging oxygen vacancies account only for
approximately 10% of surface bridging oxygen sites, exposing the clean TiO
2
(1 1 0) to
a few Langmuirs ofO
2
could not fully remove all bridging oxygen vacancies. To isolate
the influence of bridging oxygen vacancies in O
2
dissociation, the authors created a
perfect TiO
2
(1 1 0) surface by exposing the TiO
2
(1 1 0) surface to water at room
temperature. Hydroxyl groups, formed via water dissociation, covered all bridging
oxygen vacancies, yielding a vacancy-free TiO
2
(1 1 0) surface (Figure 3.10a).
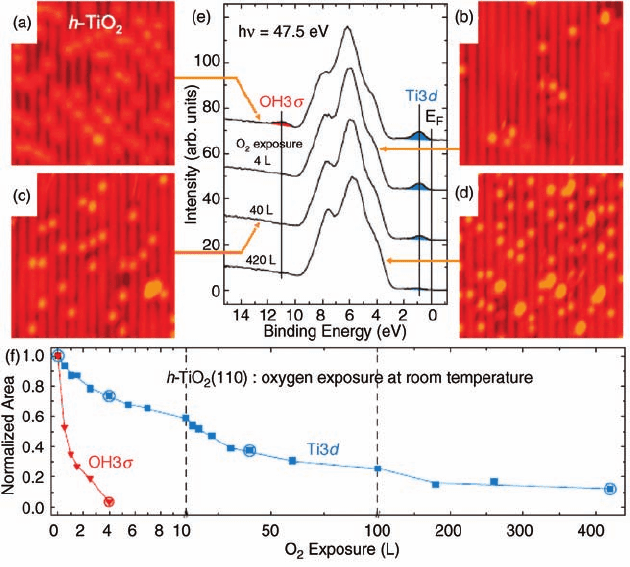
Figure 3.10c and d illustrates that O
2
exposure can fully remove surface hydroxyl
groups and create a TiO
2
(1 1 0) surface with perfect bridging oxygen rows, as
previously suggested in TPD studies [51]. With the titration of hydroxyl groups,
oxygen adatoms on the five-coordinated Ti row (O
ot
) also increase (Figure 3.10b).
However, the increase in oxygen adatoms does not seem to stop even after all the
hydroxyl groups have been replaced with oxygen (Figure 3.10c and d). Paired O
ot
atoms start to appear on the five-coordinated Tirow during extended O
2
exposure. On
the basis of these observations, the authors showed that a second and primary O
2
dissociation channel is operative on the five-coordinated Ti row.
STM results combined with photoelectron spectroscopy (PES) on the valence
state of TiO
2
(1 1 0) further show that the removal of all hydroxyl groups by oxygen,
leading to a perfect TiO
2
(1 1 0) surface, only slightly attenuates the Ti 3d defect
state (Figure3.10e). The full attenuation of Ti3d state requires 420 L of O
2
. Figure 3.9f
plots the evolutions of the Ti 3d defect state and the OH 3s state over O
2
exposure
and suggests the Ti 3d defect state is not mainly caused by bridging oxygen vacancies.
The authors suggest that other types of defects, Ti
3 þ
interstitials that form during
the reduction of TiO
2
(1 1 0) and are hidden beneath the surface, are primarily
responsible for the formation of the Ti 3d defect state and the dissociative adsorption
of O
2
.
Indeed, the importance of Ti
3 þ
interstitials has also been realized in previous in
situ STM studies of the reoxidation of TiO
2
(1 1 0) [52–54]. It is noted that Ti
3 þ
3.3 Visualizing the Pathway of Catalytic Reactions
j
69
interstitials diffuse to the surface in the presence of O
2
and form TiO
x
species, which
serve as the building blocks for the regrowth of TiO
2
(1 1 0) plane. Using in situ STM,
Bowker and coworkers [52] measured the reoxidation kinetics of reduced TiO
2
(1 1 0)
surface at temperatures between 573 and 1000 K and oxygen pressures of 5 10
8
to
2 10
6
mbar. By monitoring the TiO
x
species that diffused and coalesced on the
TiO
2
(1 1 0) surface, the surface growth rate could be measured through the change of
island morphology. This growth rate was found to be linear with respect to the oxygen
partial pressure. The regrowth of TiO
2
(1 1 0) has a low activation energy of
approximately 25 kJ/mol, suggesting a high mobility of Ti
3 þ
interstitials in the
presence of oxygen. Previous studies on the reduction of TiO
2
(1 1 0) [37] have shown
the rutile TiO
2
(1 1 0) bulk serves as a huge reservoir for Ti
3 þ
interstitials during
reduction, so that the surface is maintained near-stoichiometry and is thermody-
namically stable.
Figure 3.10 (a–d) STM images (105 Å 105 Å)
of the TiO
2
(1 1 0) surface covered with
hydroxyls [h-TiO
2
(1 1 0)] and then exposed to
increasing amounts of O
2
at room temperature.
(e) Selected PES valence-band spectra recorded
on an h-TiO
2
(1 1 0) surface that was exposed to
O
2
at RT. Arrows indicate the representative
STM images. (f) Normalized integrated
intensities of the OH 3s (red) and Ti 3d (blue)
features for O
2
exposures up to 420 L from PES
spectra; circles indicate intensity values that
were obtained from the spectra shown in (e).
(Reprinted with permission from Ref. [50].
Copyright 2008, The American Association for
the Advancement of Science.)
70
j
3 In Situ STM Studies of Model Catalysts
Ti
3 þ
interstitials are essentially oxygen vacancies within the bulk whereas bridging
oxygen vacancies are basically undercoordinated Ti ions at the surface. It is not
surprising to expect that Ti
3 þ
interstitials in the subsurface play a role in the
dissociation of adsorbed molecules. In the above STM study by Wendt et al.,itis
worth noting that subsurface Ti
3 þ
interstitials were neither visualized nor seen to
diffuse to the TiO
2
(1 1 0) surface during O
2
adsorption at room temperature.
Considering PES usually probes the top few layers at the surface, the complete
attenuation of the Ti 3d defect state suggests that Ti
3 þ
interstitials within those layers
have been oxidized and therefore quenched. Details of how the excess charge of Ti
3 þ
interstitials involves in the bond breaking of O
2
molecules remain to be elucidated.
Nevertheless, the above studies demonstrate the power of in situ STM in tracing the
active sites, hidden or unhidden. The finding of mobile defects in a rigid TiO
2
(1 1 0)
will stimulate more investigations on other reducible oxides and encourage revisiting
their surface chemistry.
3.3.4
Visualizing Reaction Intermediates and the Mechanism of Hydrogen Oxidation
In situ STM has also been used to study the pathway of surface reactions and to
measure their kinetics. The hydrogen oxidation reaction, catalyzed by Pt group
metals to produce water, was the first catalytic reaction discovered in 1823. This
reaction is still a core catalytic reaction at the heart of fuel cell technologies. Although
the oldest of catalytic reactions, the mechanism of catalytic hydrogen oxidation is still
unclear, especially for its surprising reactivity at or below the water desorption
temperature (170 K). It has been proposed that the reaction proceeds via the
combination of dissociatively adsorbed O atoms (O
ad
) and H atoms (H
ad
), forming
hydroxyl groups that subsequently bind with another H
ad
to form water. The
formation of hydroxyl groups has been postulated as the rate-limiting step. However,
the hydroxyl group, as a reaction intermediate, could not be confirmed in early
surface spectroscopic studies. It has not been until the past decade, with the help of in
situ STM, that the reaction mechanism has become apparent for the hydrogen
oxidation reaction on metal surfaces.
Wintterlin and coworkers [55, 56] studied the hydrogen oxidation reaction on
Pt(1 1 1) using in situ STM. The study was conducted as a titration experiment, in
which the surface was precovered with O
ad
and then the O-terminated surface
subsequently exposed to H
2
molecules. The O-terminated Pt(1 1 1) surface was
prepared by exposing the clean Pt(1 1 1) surface to 10 L of O
2
, followed by annealing at
225 K to dissociate O
2
. The surface was then exposed to 8 10
9
mbar H
2
at 131 K
and monitored by STM, as shown in Figure3.11. The Pt(1 1 1) surface was precovered
by a (2 2)-O
ad
layer, where O
ad
atoms were imaged as dark dots (Figure 3.11a). Upon
exposure to H
2
, a few bright islands form on top of the (2 2)-O
ad
layer
(Figure 3.11b). These islands continue to grow and eventually develop into an
ordered layer with hexagonal and honeycomb phases (Figure 3.11c). These two
phases are characterized as a surface hydroxyl (OH
ad
) overlayer, formed by hydrogen
bonding. Additional diffusing islands seen in Figure 3.11c are attributed to adsorbed
3.3 Visualizing the Pathway of Catalytic Reactions
j
71
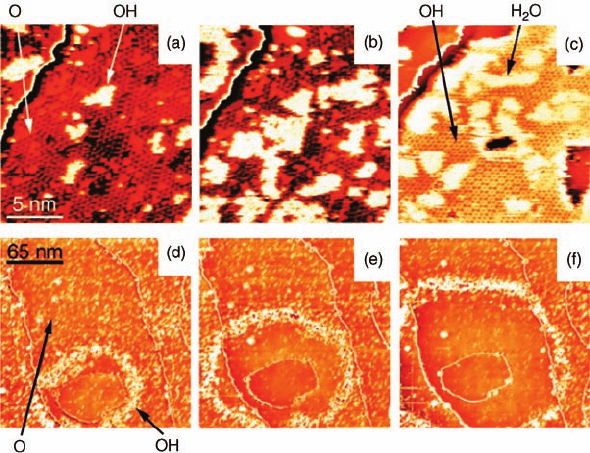
H
2
O islands (H
2
O
ad
). Figure 3.11a–c shows the formation of hydroxyl group as the
reaction intermediate and reveal the atomic details of hydrogen oxidation catalyzed by
Pt(1 1 1).
Figure 3.11d–f presents snapshots of STM images of the O
ad
-terminated Pt(1 1 1)
surface exposed to 2 10
8
mbar H
2
at 112 K. The imaged area includes several
surface terraces covered with numerous small bright dots and a bright ring that
expands with time. These small bright dots have been assigned to small OH
ad
islands
that appear after H
2
exposure. The white ring, termed as the reaction front, is
concentrated with small OH
ad
islands and continues to grow as the reaction
progresses, suggesting a fast reaction at the boundaries between O
ad
atoms and
the diffusing H
2
O
ad
. The fast reaction between O
ad
and H
2
O
ad
produces two OH
ad
,
which in turn forms H
2
O
ad
through a rapid reaction with H
ad
atom. Since the
combination of O
ad
and H
ad
to form OH
ad
is the rate-limiting step, the presence of
H
2
O
ad
removes this kinetic limit and promotes an autocatalytic cycle until depletion
of O
ad
atoms. STM images thus illustrate an autocatalytic reaction mechanism that
accounts for the low activation barrier and high reactivity of hydrogen oxidation at or
Figure 3.11 Series of successive STM images,
recorded during dosing of the O-covered
Pt(1 1 1) surface with H
2
.(a–c) Frames
(17 nm 17 nm) from an experiment at 131 K
[P(H
2
) ¼8 10
9
mbar]. The hexagonal pattern
in (a) is the (2 2)-O structure; O atoms appear
as dark dots and bright features are the initial
OH islands. In (c), the area is mostly covered by
OH, which forms ordered structures. The white,
fuzzy features are H
2
O-covered areas.
(d–f) Frames (220 nm 220 nm) from an
experiment at 112 K [P(H
2
) ¼2 10
8
mbar].
In (d), the surface is mostly O-covered
(not resolved). The bright dots are small OH
islands, most of which are concentrated in the
expanding ring. H
2
O in the interior of the ring is
not resolved here. Thin, mostly vertical lines are
atomic steps. (Reprinted with permission from
Ref. [55]. Copyright 2001, The American
Association for the Advancement of Science.)
72
j
3 In Situ STM Studies of Model Catalysts
below the water desorption temperature. At temperatures higher than 170 K, where
H
2
O starts to desorb, the shortened lifetime of H
2
O
ad
breaks down the autocatalytic
cycle by stopping the fast reaction between H
2
O
ad
and O
ad
to form OH
ad
.
The autocatalytic reaction mechanism apparent at low temperatures is expected to
apply to catalytic hydrogen oxidation at high pressures. In addition, the above study is
the first to use STM to observe the formation of dynamic surface patterns at the
mesoscopic level, which had previously been observed by other imaging techniques
in surface reactions with nonlinear kinetics [57]. This study illustrates the ability of in
situ STM to visualize reaction intermediates and to reveal the reaction pathway with
atomic resolution.
3.3.5
Measuring the Reaction Kinetics of CO Oxidation
Another important catalytic reaction that has been most extensively studied is CO
oxidation catalyzed by noble metals. In situ STM studies of CO oxidation have focused
on measuring the kinetic parameters of this surface reaction. Similar to the above
study of hydrogen oxidation, in situ STM studies of CO oxidation are often conducted
as a titration experiment. Metal surfaces are precovered with oxygen atoms that are
then removed by exposure to a constant CO pressure. In the titration experiment, the
kinetics of surface reaction can be simplified and the reaction rate directly measured
from STM images.
Wintterlin et al. [58] investigated the catalytic oxidation of carbon monoxide on
Pt(1 1 1) using in situ STM. Oxygen atoms were preadsorbed on the Pt(1 1 1) surface
by exposing the surface to 3 L O
2
at 96 K, followed by a short anneal at 293 K to
dissociate O
2
. The oxygen-covered Pt(1 1 1) surface was then cooled to 247 K and
exposed to 5 10
8
Torr CO. STM was used to follow the change of surface adsorbate
structures as a function of the CO exposure time (Figure 3.12). At 247 K, O
ad
atoms
form an ordered (2 2) overlayer, imaged as dark dots. At t ¼0, the Pt(1 1 1) surface is
mainly covered by the (2 2)-O layer together with empty Pt sites, imaged as bright
islands, scattered on the surface. The addition of CO molecules lowers the mobility of
surface oxygen atoms and slowly compresses the (2 2)-O layer into large islands.
The adsorbed CO molecules form ordered c(4 2) domains on the Pt(1 1 1) surface.
As time progresses, the areas of c(4 2) CO domains continue to grow at the expense
of the (2 2)-O islands. From the series of in situ STM images, the rate of CO
oxidation can be estimated based on the reduction rate of the surface areas of the
(2 2)-O islands.
Figure 3.13 plots the dependence of the reaction rate on the surface area or
perimeter of oxygen domains, as a function of time. Approximately, the reaction rate
is linear with respect to the perimeter of the surface oxygen domains, suggesting CO
oxidation mainly occurs along the boundary between the oxygen and the CO domains
on the Pt(1 1 1) surface. The titration experiments were repeated at various
temperatures between 237 and 274 K. An Arrhenius plot gives an activation energy
of 0.49 eV and a prefactor of 3 10
21
cm
2
s
1
, in good agreement with the kinetic
parameters obtained from macroscopic measurements.
3.3 Visualizing the Pathway of Catalytic Reactions
j
73
