Coker A.K. Fortran Programs for Chemical Process Design, Analysis, and Simulation
Подождите немного. Документ загружается.


i0
18
20
19
21
44
90
95
PROGRAM PROG63
***************************************************************
THIS PROGRAM WILL CALCULATE THE PUMP EFFICIENCY BASED ON
THE FLUID FLOW RATE AND THE HEAD USING THE EQUATIONS
DEVELOPED BY C. BRANAN (The Process Engineer's Pocket Handbook,
Gulf Publishing Company, Houston 1976).
****************************************************************
H = HEAD, ft.
Q = VOLUMETRIC FLOWRATE, gpm.
HBHP = HYDRAULIC HORSEPOWER, bhp.
BHP = BRAKE HORSEPOWER, bhp.
****************************************************************
DIMENSION EFF(20), H(20), Q(20), HBHP(20), BHP(20)
OPEN (UNIT=3, FILE='DATA63.DAT', STATUS='OLD', ERR=I8)
OPEN (UNIT=l, FILE='PRN')
READ (3, *, ERR=I9) N
DO I0 I=l, N
READ ( 3, *, ERR=I9) Q(I), H(I)
CONTINUE
GO TO
44
WRITE (*, 20)
FORMAT (6X, 'FILE DOES NOT EXIST')
GO TO 999
WRITE (*, 21)
FORMAT (6X, 'ERROR MESSAGE IN THE DATA VALUE')
GO TO 999
WRITE (i, 90)
FORMAT (///, 25X, ' EFFICIENCY AND BRAKE HORSEPOWER CALCULATIONS' , /
IH ,78(IH*) ,//)
WRITE(l, 95)
FORMAT (5X,'FLOWRATE gpm', 10X,'HEAD ft.', 6X,'EFFICIENCY
* 6X,'BRAKE HORSEPOWER Bhp',/IH , 78(IH*))
%',
SPGR=I.0
DO 35 I=I,N
IF (Q(1) .EQ. 0.0) THEN
EFF(1) = 0.0
HBHP(1) = 0.0
ELSE
ENDIF
467

35
96
i00
ii0
45
120
C
999
VALI = 0.2855"H(I)
VAL2 = 3.78*H(I)*Q(I)*(10.0**(-4))
VAL3 = 2.38*H(I)*(Q(I)**2)*(10.0**(-7))
VAL4 = 5.39"(H(I)*'2)*(I0.0"*(-4))
VAL5 = 6.39*(H(I)**2)*Q(I)*(10.0**(-7))
VAL6 = 4.0*(H(I)**2)*(Q(I)**2)*(10.0**(-10))
EFF(I)=(80.0-VALI+VAL2-VAL3+VAL4-VAL5+VAL6)
HYDRAULIC BRAKE HORSEPOWER REQUIRED BY THE PUMP
PUMP EFFICIENCY IS ASSUMED = 1.0
HBHP(I)=(Q(I)*H(I)*SPGR)/(3960.0*I.0)
BHP(I)=HBHP(I)/(EFF(I)/100.0)
CONTINUE
DO I =i, N
WRITE (I, 96)Q(I),H(I),EFF(I),BHP(I)
FORMAT (5X, F8.1, 10x, F8.1, 10X, F8.1, 10X, F8.1)
ENDDO
WRITE(*, i00)
FORMAT (5X , "FLOWRATE gpm', 10X, 'HEAD ft.', 6X 'EFFICIENCY
6X,'BRAKE HORSEPOWER Bhp',/iH ,78(IH*))
DO 45 I =i, N
WRITE(*, II0)Q(I), H(I), EFF(I) ,BHP(I)
FORMAT (5X, F8.1, 10X, F8.1, 10X, F8.2, 10X, F8.1)
CONTINUE
WRITE (1,120)
FORMAT (IH ,78(IH-))
FORM FEED THE PRINTING PAPER TO TOP OF THE NEXT PAGE.
WRITE (i, *) CHAR(12)
CLOSE (3, STATUS='KEEP')
CLOSE (i)
STOP
END
%',
468

CHAPTER 7
Mass Transfer
INTRODUCTION
Unit operation techniques in the chemical process industries (CPI)
effect the separation of a mixture or mixtures into their components.
These operations are called diffusional or mass transfer operations. Tech-
niques employed in the CPI to effect these separations are: distillation,
absorption, liquid extraction, drying, leaching, crystallization, and gas
adsorption. These processes account for 40% to 70% of both the capital
and operating costs in the CPI. Separation operations significantly
affect energy consumption, product costs, and manufacturing profits.
About 43% of the energy consumed by the U.S. chemical process
industries is used for separation processes [ 1 ]. The method depends on
the state (such as liquid, solid, or gaseous) of the components.
The engineer must adequately size equipment for separating and
recovering products from multicomponent feed streams. In addition, he
or she should consider whether the design is either stagewise or con-
tinuous, tray or packed. The trays act as individual stages and produce
stepwise changes in concentration, while packed towers give gradual
changes in concentration. Designing distillation equipment requires
knowledge of vapor-liquid equilibria in multi-component mixtures.
This chapter considers the vapor-liquid equilibrium of mixtures, con-
ditions for bubble and dew points of gaseous mixtures, isothermal equi-
librium flash calculations, the design of distillation towers with valve
trays, packed tower design, Smoker's equation for estimating the num-
ber of plates in a binary mixture, and finally, the computation of multi-
component recovery and minimum trays in distillation columns.
469

470
Fortran Programs for Chemical Process Design
VAPOR-LIQUID EQUILIBRIUM
A common operation in the CPI for the separation of fluid mixtures
into their components is distillation. The distillation process is a separa-
tion technique that depends on the difference between the compositions
of liquid and vapor phases at equilibrium. This implies that the tem-
peratures and pressures of the phases must be the same, and no compo-
sition change occurs with time. Equilibrium can be achieved after a
long period of thorough mixing and contact between the two phases.
An ideal gas is, by definition, one that follows the ideal gas laws:
PV = nRT (7-1)
where P = absolute pressure
V = total volume
n = number of moles of gas
R = ideal gas constant
T = absolute temperature
The conditions under which a given component or mixture approaches
ideal behavior depend on the critical temperature and critical pressure.
Other principles of gas behavior are Dalton's law of additive pressures
and Amagat's law of additive volumes. These are:
Tc = PA + PB +''" (7-2)
and
V T = VA -4- V B -]--...
(7-3)
where n = total system pressure
p = partial pressure
V a = volume of component A
V B = volume of component B
V t = total volume of system
A,B = components
These laws are correct when conditions are such that each component
and the mixture obey the ideal gas law. The equilibrium between two
phases can be related to the equality of the chemical potential and
defined in terms of the Gibbs free energy as
lai
- - G~ (7-4)
T,P,nj

Mass Transfer 471
m
where Gi is the partial molar Gibbs free energy. From the thermody-
namic relationship between Gibbs free energy, temperature and pres-
sure, and at constant temperature,
dG = VdP (7-5)
where G is the molar Gibbs free energy and V is the molar volume.
Considering a pure fluid of component i and substituting Equation 7-1
into Equation 7-5,
dP
dG = RT
P
= RTd(ln P)
(7-6)
The free energy from Equation 7-6 is restricted to ideal gases, but can
be modified to include real fluids by introducing a new function defined
as the fugacity. Equation 7-6 becomes
dGi = RTd(ln f~) (7-7)
where f~ is the fugacity of pure i with units of pressure. The fugacity
becomes equal to the pressure as the pressure approaches zero.
Thus,
f
lim "~ - 1.0 (7-8)
P
P--90
For a single component in a mixture at constant temperature, the fugac-
ity is defined by
dG i -
RTd(ln f~ ) (7-9)
and
lira
fi -- 1.0
yiP (7-10)
P--90
If a liquid mixture at temperature T and pressure P is in equilibrium
with a vapor mixture at the same temperature and pressure, therefore at
an equilibrium condition, the thermodynamic criterion will be

472 Fortran Programs for Chemical Process Design
fv _ fL (7-11)
i i
where fv_ fugacity of component i in the vapor phase
i
fL_ fugacity of component i in the liquid phase
i
The fugacity of a component in a mixture depends on the temperature,
pressure, and composition of that mixture. The fugacity, f), of any com-
ponent, i, in the liquid phase is related to the composition (mole frac-
tion) of the component in that phase by
fL foc (7-12)
i -- ~/iXi i
where 7~ = the activity coefficient of component i
xi = mole fraction of component i
foL_ the fugacity of component i in the reference (or standard)
i
state
The fugacity of pure liquid i at temperature T and pressure P is given by
s ]
foc _ p
(T)(~i
(T) exp ' dP (7-13)
i vp,i
Lapvp,,
RT
where Pvp,~- the vapor pressure of component i
~- the vapor phase fugacity coefficient of pure saturated
vapor of component i
The exponential term in Equation 7-13 is a correction factor for the
effect of pressure on liquid-phase fugacity and is known as the Poynting
factor. In Equation 7-13, V L
can be replaced by the partial molar vol-
ume of component i in the liquid solution for greater accuracy. For low
L is assumed as the saturated liquid molar vol-
to moderate pressure, V i
ume at the specified temperature. Equation 7-13 is simplified to give
f~ Pvpi
,i ir
exp [ V ~ (e - Pvp'i ) IRT
(7-14)
Equation 7-14 is used to calculate the reference state fugacity of liq-
uids. Any equation of state can be used to evaluate ~. For low to mod-
erate pressures, the virial equation is the simplest to use. The fugacities
of pure gases and gas mixtures are needed for estimating many thermo-
dynamic properties, such as entropy, enthalpy, and fluid phase equilib-
ria. For pure gases, the fugacity is

Mass Transfer 473
lnf- 1 ioVd P (7-15)
RT
The fugacity can be calculated from Equation 7-15 once the P-V-T
behavior of the fluid is known from an equation of state. The fugacity
coefficient is the ratio of the fugacity of a substance to its pressure. For
a pure substance,
f
= -- (7-16)
P
For the vapor phase, the composition is nearly always expressed by the
mole fraction, yi
fv
~i = i
(7-17)
Yi P
For a mixture of ideal gases, 0i = 1. The fugacity coefficient, ~)i, depends
on temperature and pressure. The fugacity coefficient is normalized,
such that as P --+ 0, r --+ 1 for all components. At a low pressure, ~)i is
assumed to be unity.
When the volume is known as a function of the pressure, either from
direct measurement or from an empirical equation of state, changes in
fugacity may be found by integration. Thus
lnf2 1 I]~
= VdP (7-18)
fl RT ,
Since ~ = 1 when P = 0, absolute values of the fugacity coefficient can
be determined. Therefore,
ln, ln f
- --- V-
dP
P RT P
P dP
-So
(Z- 1)
(7-19)
Methods for estimating fugacities of liquid and vapor phases are given
by Reid,
et al.
[2], and by Walas [3].
Raoult's law states that the partial pressure in the liquid phase can be
expressed as
o
PA -- XAPA
(7-20)

474
Fortran Programs for Chemical Process Design
where x A is the mole fraction of component A, and PA is the vapor
pressure of component A.
For the vapor phase, Dalton's law can be expressed as:
PA = gYA
(7-21)
where YA
is the mole fraction of component A. Combining Equation 7-
20 and Equation 7-21
XAP A -- /1;y A
K_YA _ _
PA
x A
(7-22)
where K is the equilibrium constant.
Few multicomponent systems exist for which completely general-
ized equilibrium data are available. The most widely available data are
those for vapor-liquid systems, and these are frequently referred to as
vapor-liquid equilibrium distribution coefficients or K value. The K
values vary with temperature and pressure, and a selectivity that is equal
to the ratio of the K values is used. For vapor-liquid systems, this is
referred to as the relative volatility and is expressed for a binary system as
K A
o: - (7-23)
KB
where
K A = yA/XA
and
K B = yB/XB
YA" XB
~ZA_ B = (7-24)
Xa" YB
Equation 7-24 can be expressed in terms of the more volatile compo-
nent A, as
~X A
YA =
(7-25)
1 + (0~-
1)X A
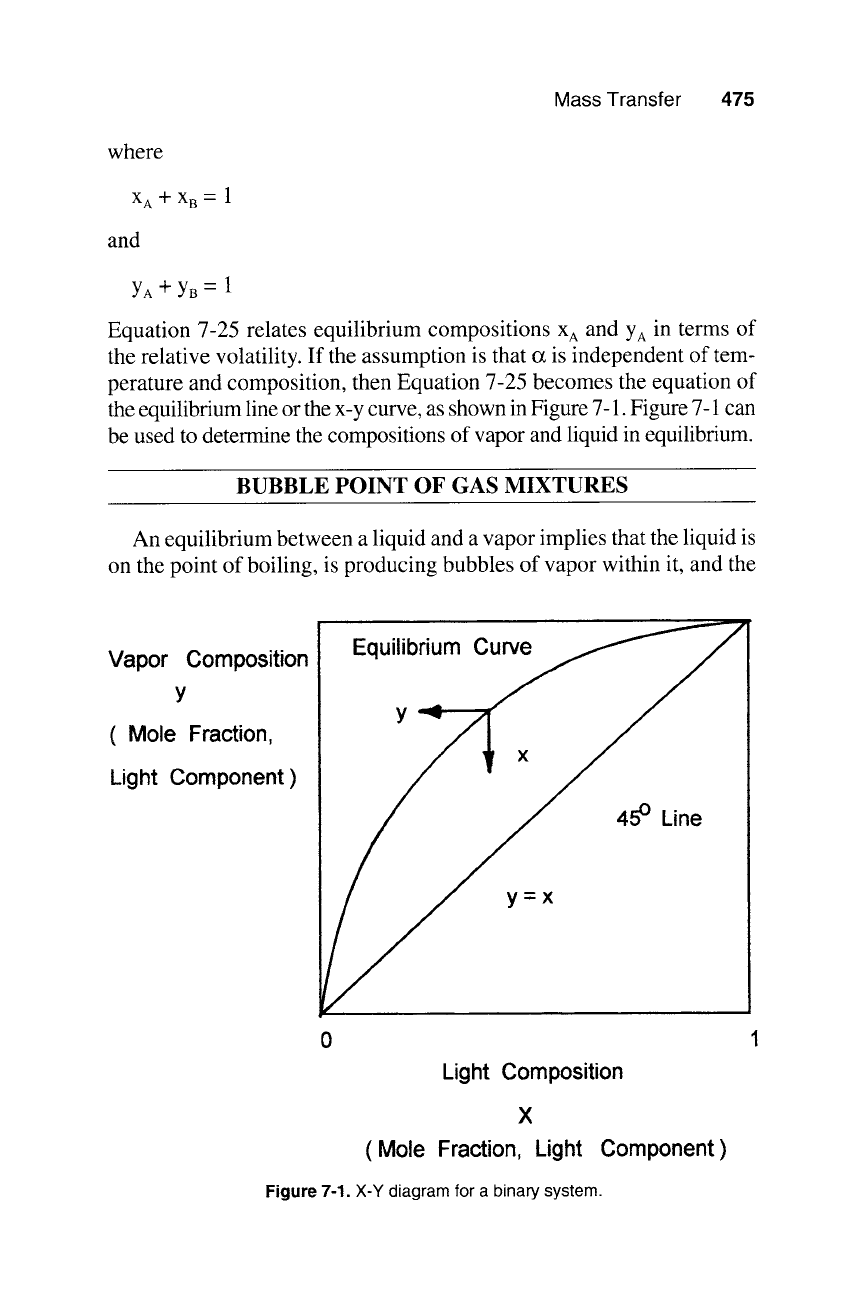
Mass Transfer 475
where
XA+XB= 1
and
yA+YB= 1
Equation 7-25 relates equilibrium compositions
X A
and YA in terms of
the relative volatility. If the assumption is that o~ is independent of tem-
perature and composition, then Equation 7-25 becomes the equation of
the equilibrium line or the x-y curve, as shown in Figure 7-1. Figure 7-1 can
be used to determine the compositions of vapor and liquid in equilibrium.
BUBBLE POINT OF GAS MIXTURES
An equilibrium between a liquid and a vapor implies that the liquid is
on the point of boiling, is producing bubbles of vapor within it, and the
Vapor Composition
Y
( Mole Fraction,
Light Component )
Eq
0
Light Composition
X
( Mole Fraction, Light Component)
Figure
7-1. X-Y diagram for a binary system.

476
Fortran Programs for Chemical Process Design
vapor is on the point of condensing or forming a dew. These conditions
are referred to as saturated liquid and saturated vapor. For the particular
total pressure, the liquid is at its bubble point temperature and the
vapor at its dew point temperature. The amount of vapor formed is
assumed to be so small as to have no effect on the liquid composition.
For a binary system,
1-K 2
x, = (7-26)
K I - K 2
and
x 2 = 1.0- x 1 (7-27)
In many cases where there are more than two components in the liquid
mixture, no direct solution as Equation 7-26 exists. The basic definition
of the equilibrium coefficient is
Yi-" Kixi
(7-28)
For a stable system, the total mole fraction is unity, which is
y~ - 1.0 - ~ K~x~ (7-29)
Equation 7-29 is used to determine the bubble point temperature and
pressure. In using Equation 7-29, the temperature or pressure is fixed,
while the other parameter is varied until the criterion for a stable system
is satisfied. A combination of temperature and pressure is altered, if the
summation of the calculated vapor composition is different from unity.
There is no known direct method that will allow a reasonable estima-
tion of the amount of change required. However, Dodge [4], Hines and
Maddox [5] have provided techniques for reducing the number of trials
that is required.
DEW POINT CALCULATIONS
The dew point of a vapor is that combination of temperature and
pressure at which the first drop of liquid condenses. The dew point cri-
terion can be expressed as:
Yi (7-30)
Zxi-1.0-~K ~
