Coker A.K. Fortran Programs for Chemical Process Design, Analysis, and Simulation
Подождите немного. Документ загружается.

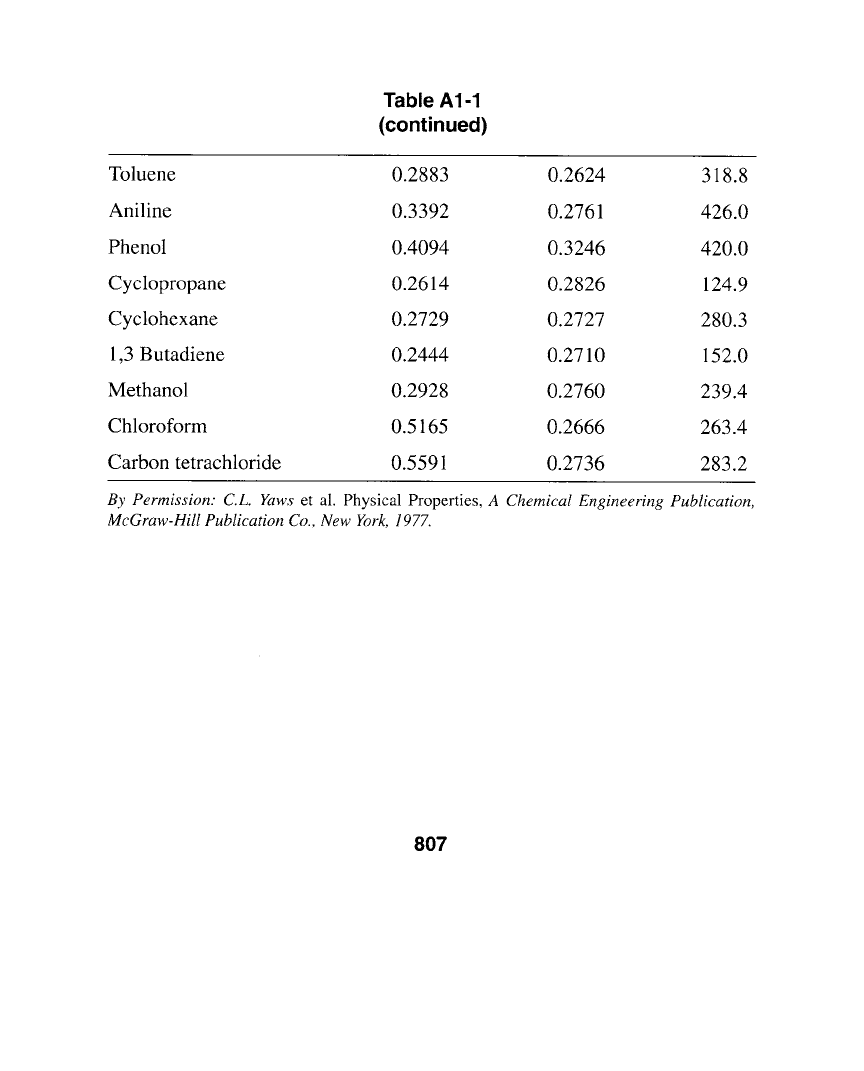
Table A1-1
(continued)
Toluene 0.2883 0.2624 318.8
Aniline 0.3392 0.2761 426.0
Phenol 0.4094 0.3246 420.0
Cyclopropane 0.2614 0.2826 124.9
Cyclohexane 0.2729 0.2727 280.3
1,3 Butadiene 0.2444 0.2710 152.0
Methanol 0.2928 0.2760 239.4
Chloroform 0.5165 0.2666 263.4
Carbon tetrachloride 0.5591 0.2736 283.2
By Permission: C.L. Yaws
et al. Physical Properties,
A Chemical Engineering Publication,
McGraw-Hill Publication Co., New York, 1977.
807
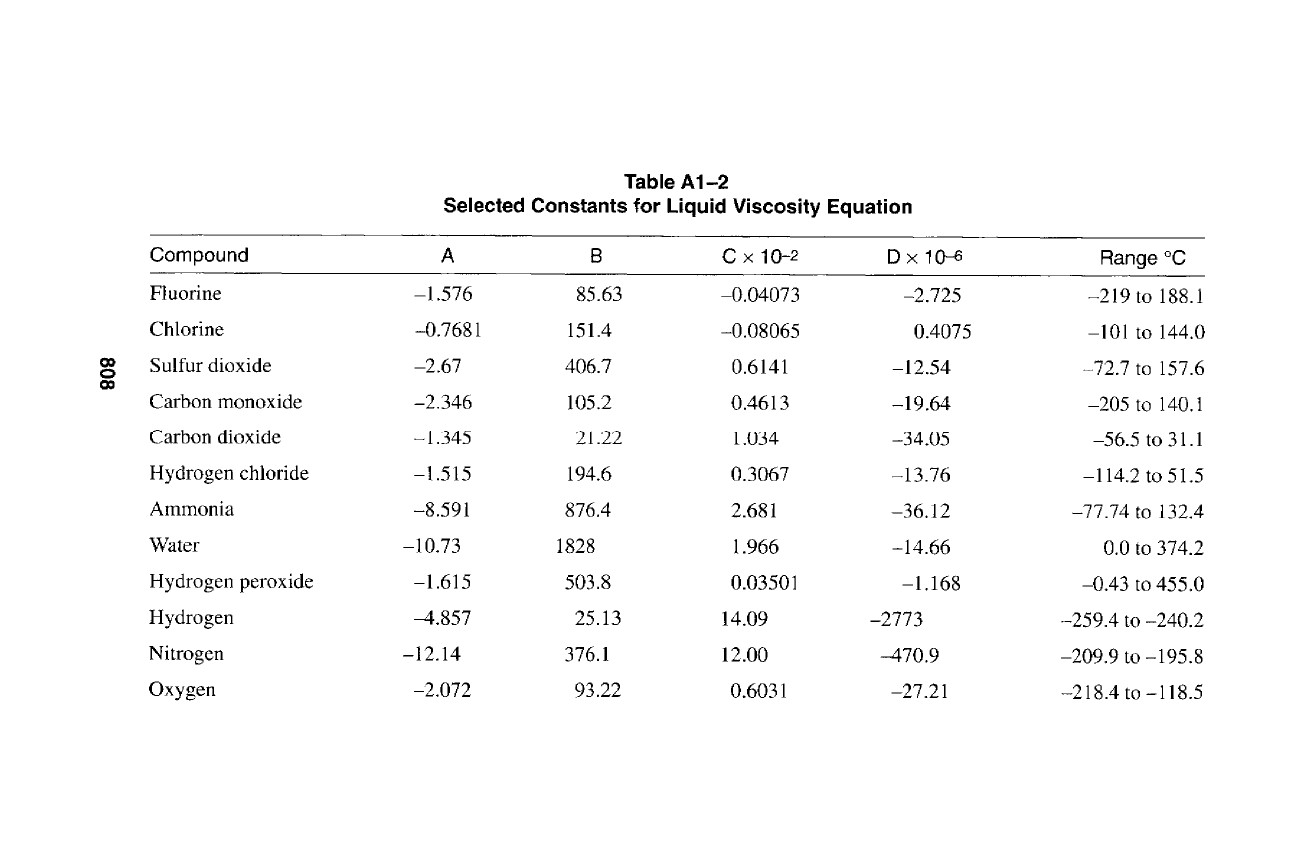
C
0
IJJ
0
(3
._~
~.~
~.~
c
o
{.}
?
r-
rr
0
~=-
X
0
×
0
c-
O
E
0
0
I ~ ~
[ I I
~ ~o
~ ~ ~ ~ .~ ~ ~ ~ ,T ~ o
I I I l I I t"--
I
, , , , T , i 7 ' " T '
~ ~ = = ~F.
0
~~.~
~ ~ Z 0
808
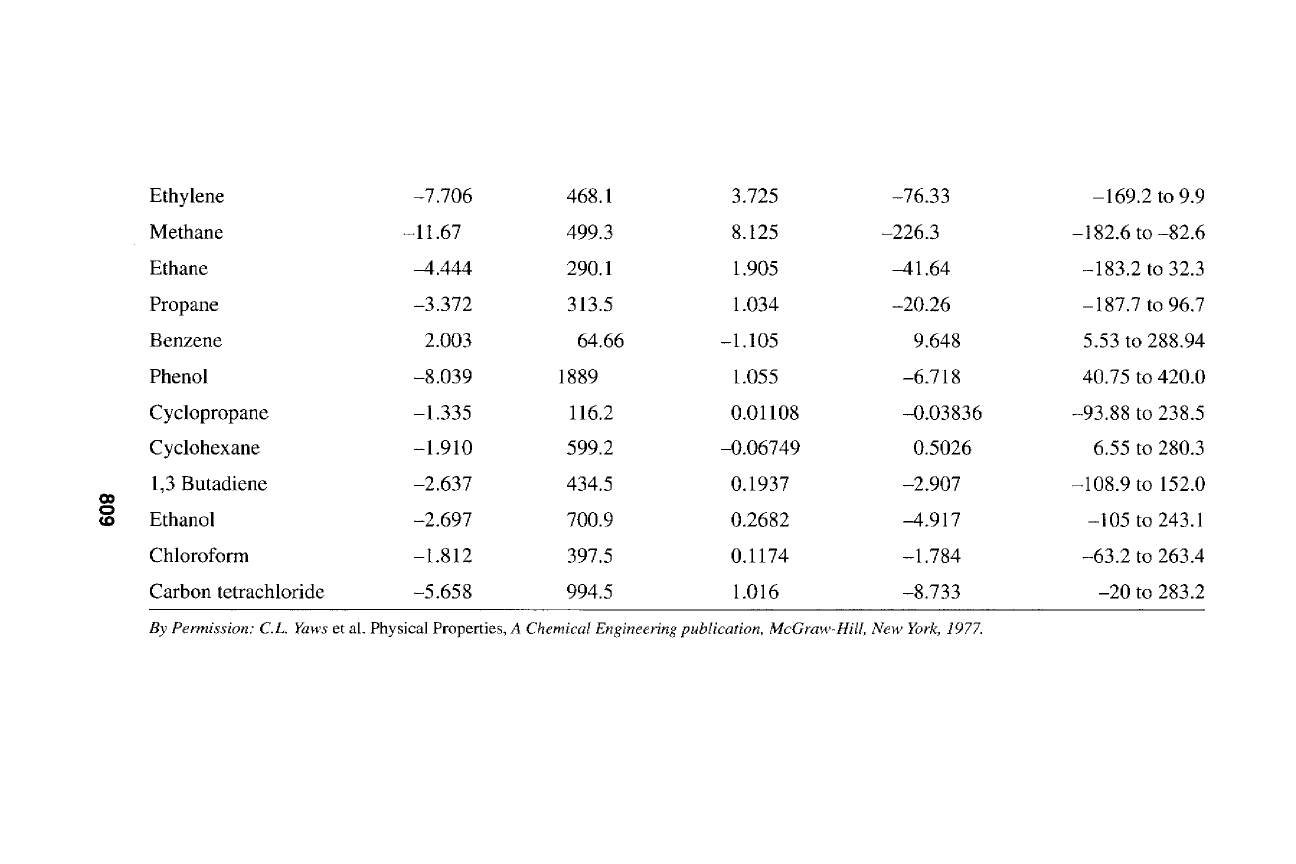
~ ~ ~ $ 2 2 2 2 2 2
- ~ ~ ~ ~ ~- ~ ~ ~ _~ ~ ~
I I ]
I I
~ ~ ~ ~ ~- ~ ~ ~ ~
~ N ~ ~ m m ~ u - m ~ u
c~
809
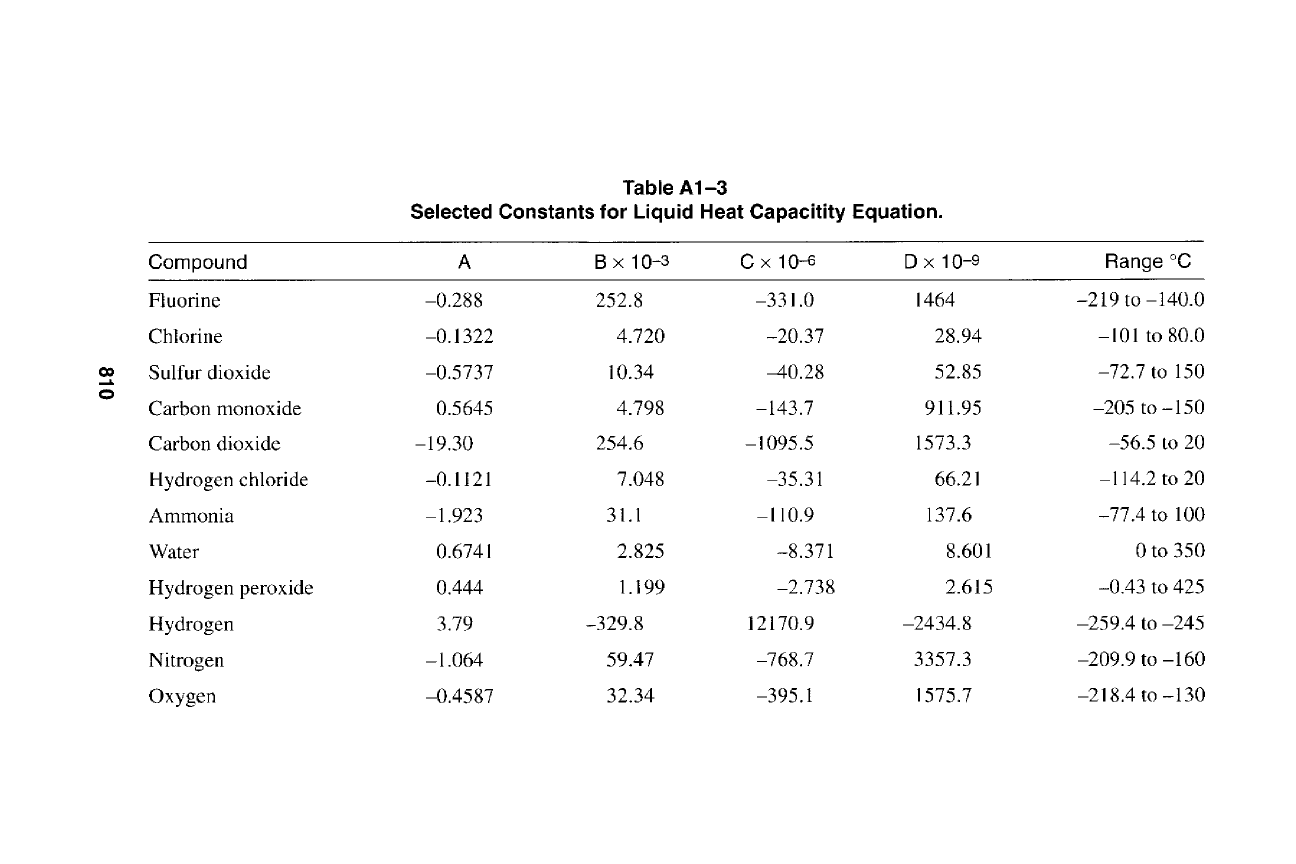
0
ILl
e"
e,,
0
o
f-
m
I
o
x
I
o
x
o
X
m
<
¢-
--,s
o
c-~
E
o
o
~- 0o
I o o o o o I I I
o ~ ~ ~ ~ _ ~ ~ ,~- c~ ,~-
eq cq
I I I I
u ~ C'q e~
I
I
I
I
"~ o o ~ ~'~
° °
~ ~ "~ < ~ ~ • Z 0
810
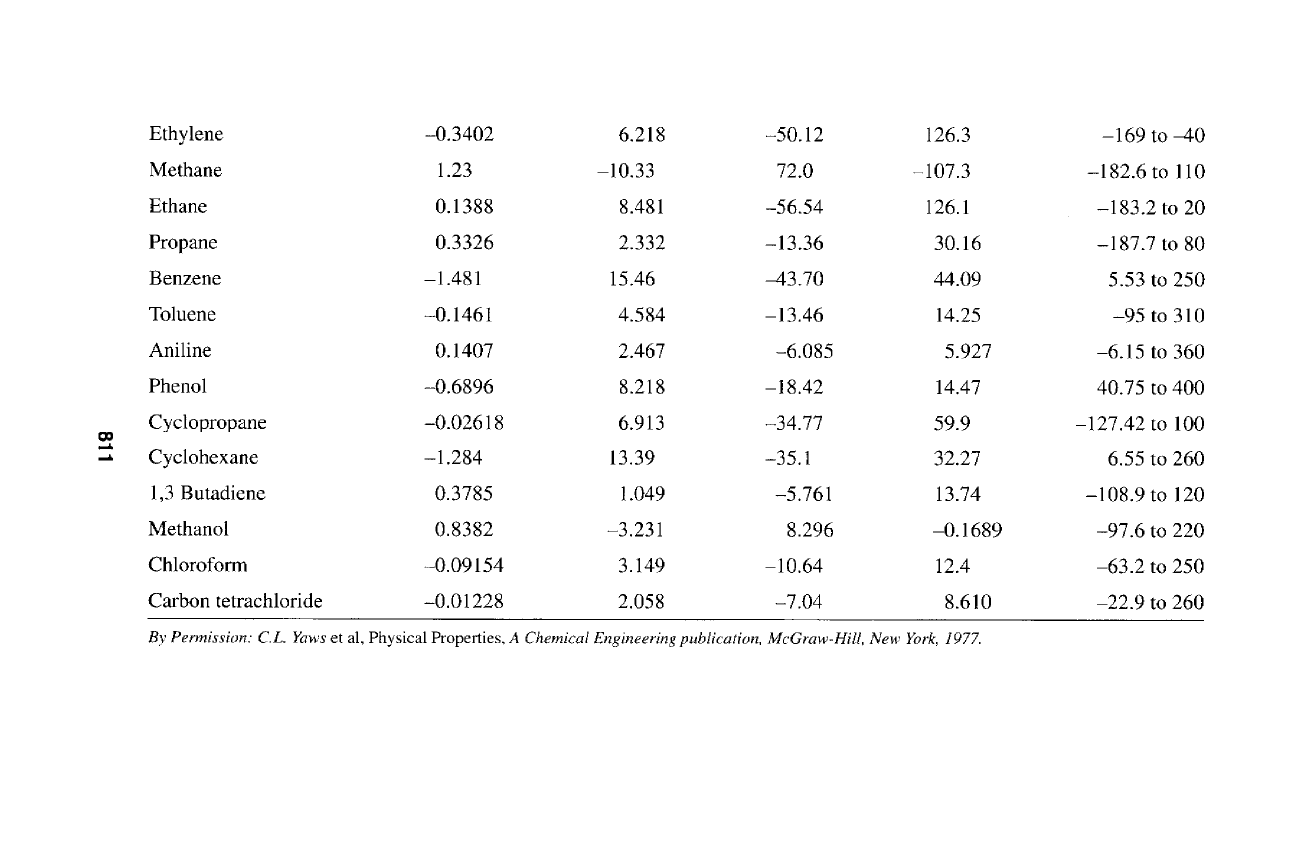
0 0 0 0 0 ~ 0 0 0 0 0 ~ ~ C~
Cq
I ~ I I I ~- eq ~ I I I
I ~ I
I
O0
~D C~ ~'~ ~ I
TM
I'~ ~" ~ ~-,
1
C~. ~ ~ ~ ~- 0
~" ~- --- I ~ cq
~
,-~ ~-q ~ I ~ o~ ~ O~ ~ ~ oo
I
c~
811
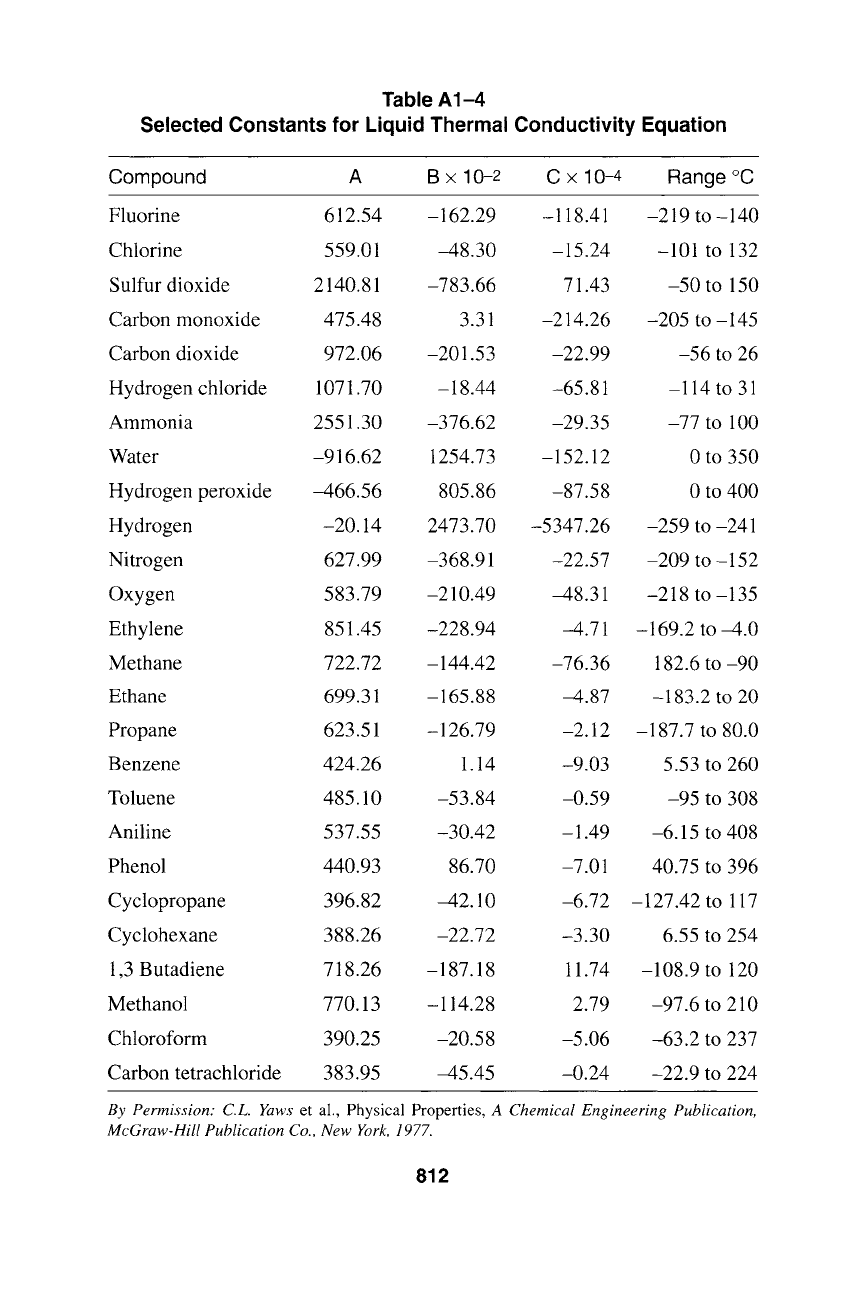
Table A1-4
Selected Constants for Liquid Thermal Conductivity Equation
Compound A B x 10-2 C x 10-4 Range ~
Fluorine 612.54 -162.29 -118.41 -219 to -140
Chlorine 559.01 -48.30 -15.24 -101 to 132
Sulfur dioxide 2140.81 -783.66 71.43 -50 to 150
Carbon monoxide 475.48 3.31 -214.26 -205 to -145
Carbon dioxide 972.06 -201.53 -22.99 -56 to 26
Hydrogen chloride 1071.70 -18.44 -65.81 -114 to 31
Ammonia 2551.30 -376.62 -29.35 -77 to 100
Water -916.62 1254.73 -152.12 0 to 350
Hydrogen peroxide -466.56 805.86 -87.58 0 to 400
Hydrogen -20.14 2473.70 -5347.26 -259 to-241
Nitrogen 627.99 -368.91 -22.57 -209 to -152
Oxygen 583.79 -210.49 -48.31 -218 to -135
Ethylene 851.45 -228.94 -4.71 -169.2 to -4.0
Methane 722.72 -144.42 -76.36 182.6 to -90
Ethane 699.31 -165.88 -4.87 -183.2 to 20
Propane 623.51 -126.79 -2.12 -187.7 to 80.0
Benzene 424.26 1.14 -9.03 5.53 to 260
Toluene 485.10 -53.84 -0.59 -95 to 308
Aniline 537.55 -30.42 -1.49 -6.15 to 408
Phenol 440.93 86.70 -7.01 40.75 to 396
Cyclopropane 396.82 -42.10 -6.72 -127.42 to 117
Cyclohexane 388.26 -22.72 -3.30 6.55 to 254
1,3 Butadiene 718.26 -187.18 11.74 -108.9 to 120
Methanol 770.13 -114.28 2.79 -97.6 to 210
Chloroform 390.25 -20.58 -5.06 -63.2 to 237
Carbon tetrachloride 383.95 -45.45 -0.24 -22.9 to 224
By Permission: C.L. Yaws
et al., Physical Properties,
A Chemical Engineering Publication,
McGraw-Hill Publication Co., New York, 1977.
812
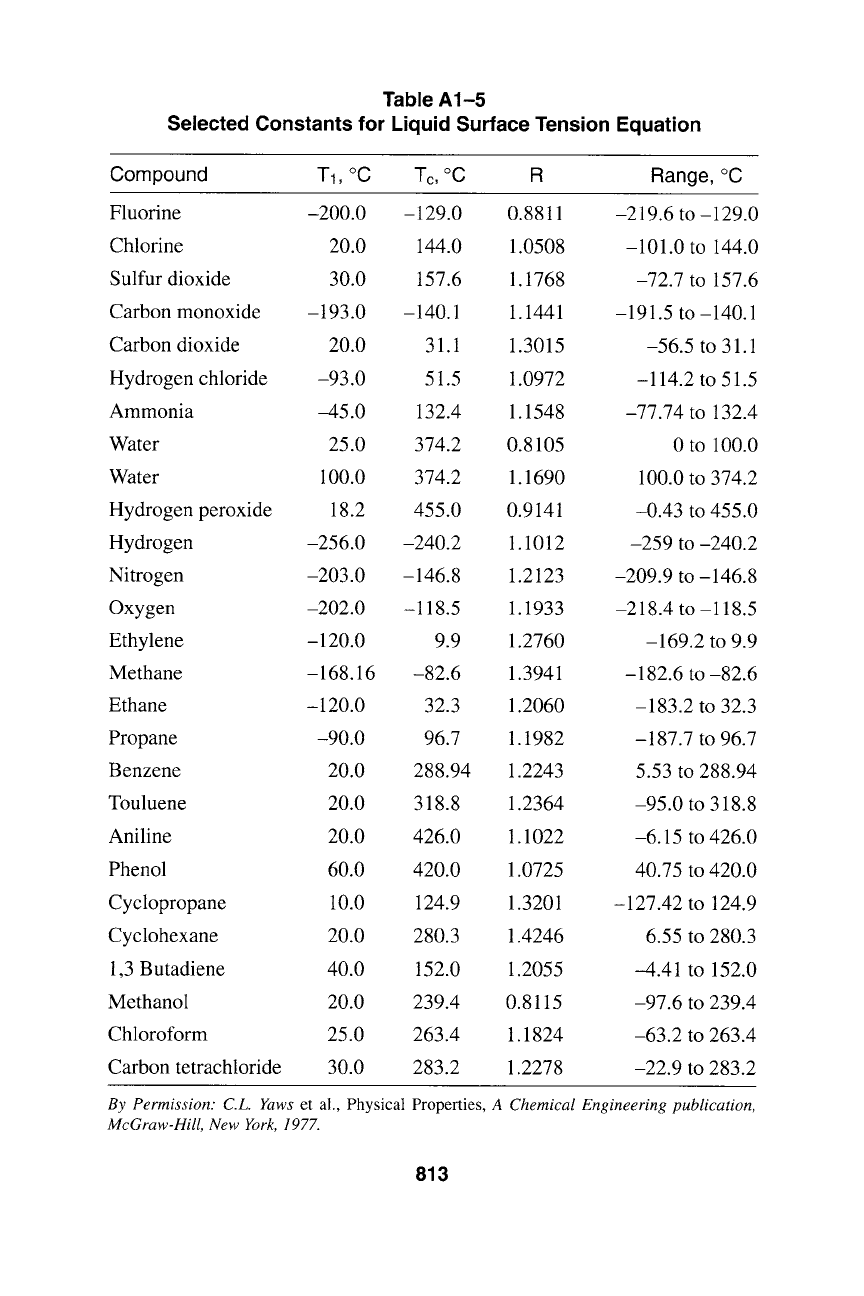
Table A1-5
Selected Constants for Liquid Surface Tension Equation
Compound T1, ~ Tc, ~ R Range, ~
Fluorine -200.0 -129.0 0.8811 -219.6 to -129.0
Chlorine 20.0 144.0 1.0508 -101.0 to 144.0
Sulfur dioxide 30.0 157.6 1.1768 -72.7 to 157.6
Carbon monoxide -193.0 -140.1 1.1441 -191.5 to-140.1
Carbon dioxide 20.0 31.1 1.3015 -56.5 to 31.1
Hydrogen chloride -93.0 51.5 1.0972 -114.2 to 51.5
Ammonia -45.0 132.4 1.1548 -77.74 to 132.4
Water 25.0 374.2 0.8105 0 to 100.0
Water 100.0 374.2 1.1690 100.0 to 374.2
Hydrogen peroxide 18.2 455.0 0.9141 -0.43 to 455.0
Hydrogen -256.0 -240.2 1.1012 -259 to -240.2
Nitrogen -203.0 -146.8 1.2123 -209.9 to-146.8
Oxygen -202.0 -118.5 1.1933 -218.4 to-118.5
Ethylene -120.0 9.9 1.2760 -169.2 to 9.9
Methane -168.16 -82.6 1.3941 -182.6 to -82.6
Ethane -120.0 32.3 1.2060 -183.2 to 32.3
Propane -90.0 96.7 1.1982 -187.7 to 96.7
Benzene 20.0 288.94 1.2243 5.53 to 288.94
Touluene 20.0 318.8 1.2364 -95.0 to 318.8
Aniline 20.0 426.0 1.1022 -6.15 to 426.0
Phenol 60.0 420.0 1.0725 40.75 to 420.0
Cyclopropane 10.0 124.9 1.3201 -127.42 to 124.9
Cyclohexane 20.0 280.3 1.4246 6.55 to 280.3
1,3 Butadiene 40.0 152.0 1.2055 -4.41 to 152.0
Methanol 20.0 239.4 0.8115 -97.6 to 239.4
Chloroform 25.0 263.4 1.1824 -63.2 to 263.4
Carbon tetrachloride 30.0 283.2 1.2278 -22.9 to 283.2
By Permission: C.L. Yaws
et al., Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
813
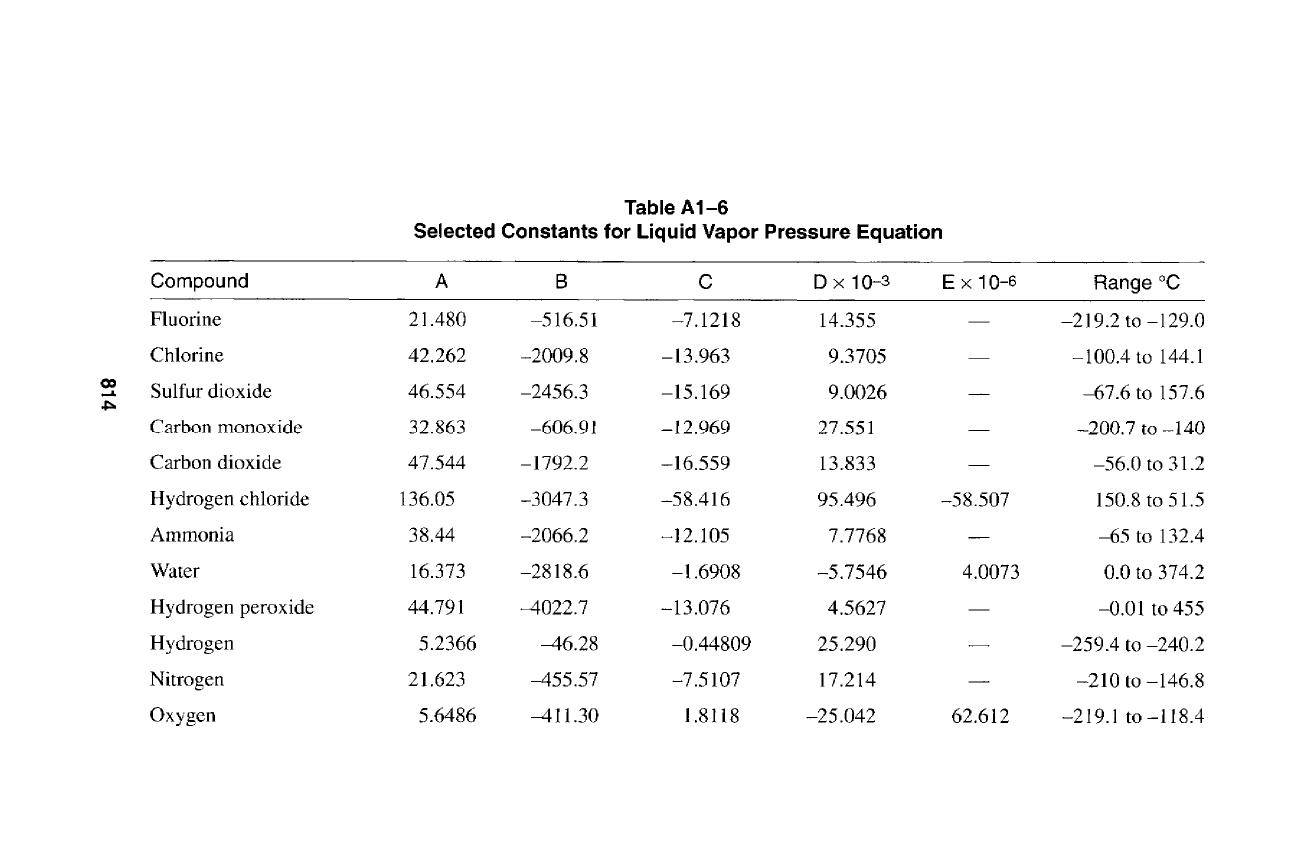
c-
O
k~
-I
O"
ill
13.
i=..
o
¢-
C
0
"0
O0
?
O~
c-
0~
n-
~o
I
O
"r-
X
ill
0
0
rn
<
¢.-
"-1
0
El.
E
0
o
• ~ o~ ~
I
eq
I
~; ~~-
~ ~ N ~ ~ N ~ ~ o G •
I ~ ~ ~ ~ ~ ~ I ~ I I
I I I I I I I
814
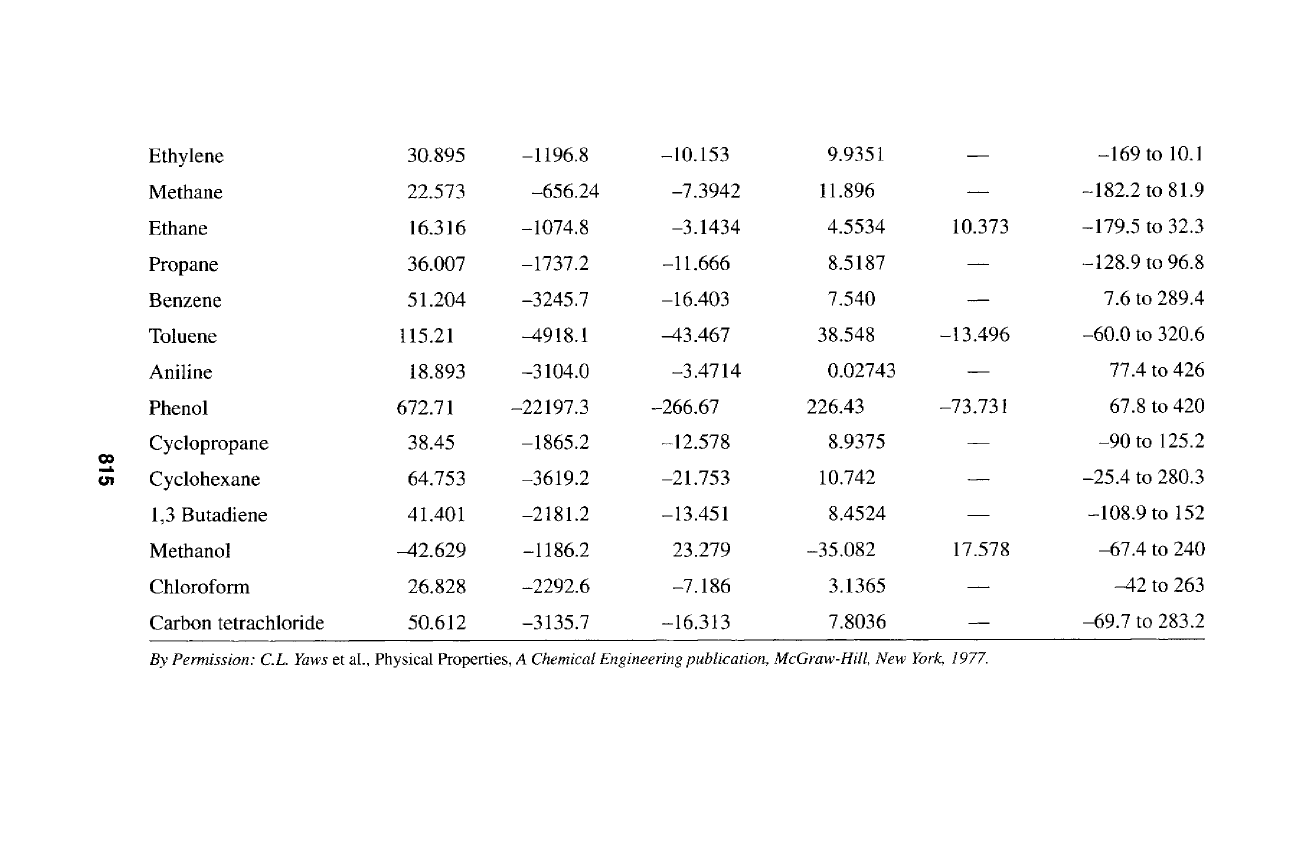
I
c~
I [
~ ~- ~ ~ ~ ~- ~ '43
L
~'~ ~ I ~ ~ "~- ~ ~ <:~ ~C~ ,~ ~ c~ C~
I I I I I C~ I [ I I I
/
~ ~ - ~ ~ -~ - ~'- ~ ~ ~ "~ ~ ~_
@
C~
",~
"~.
F
815
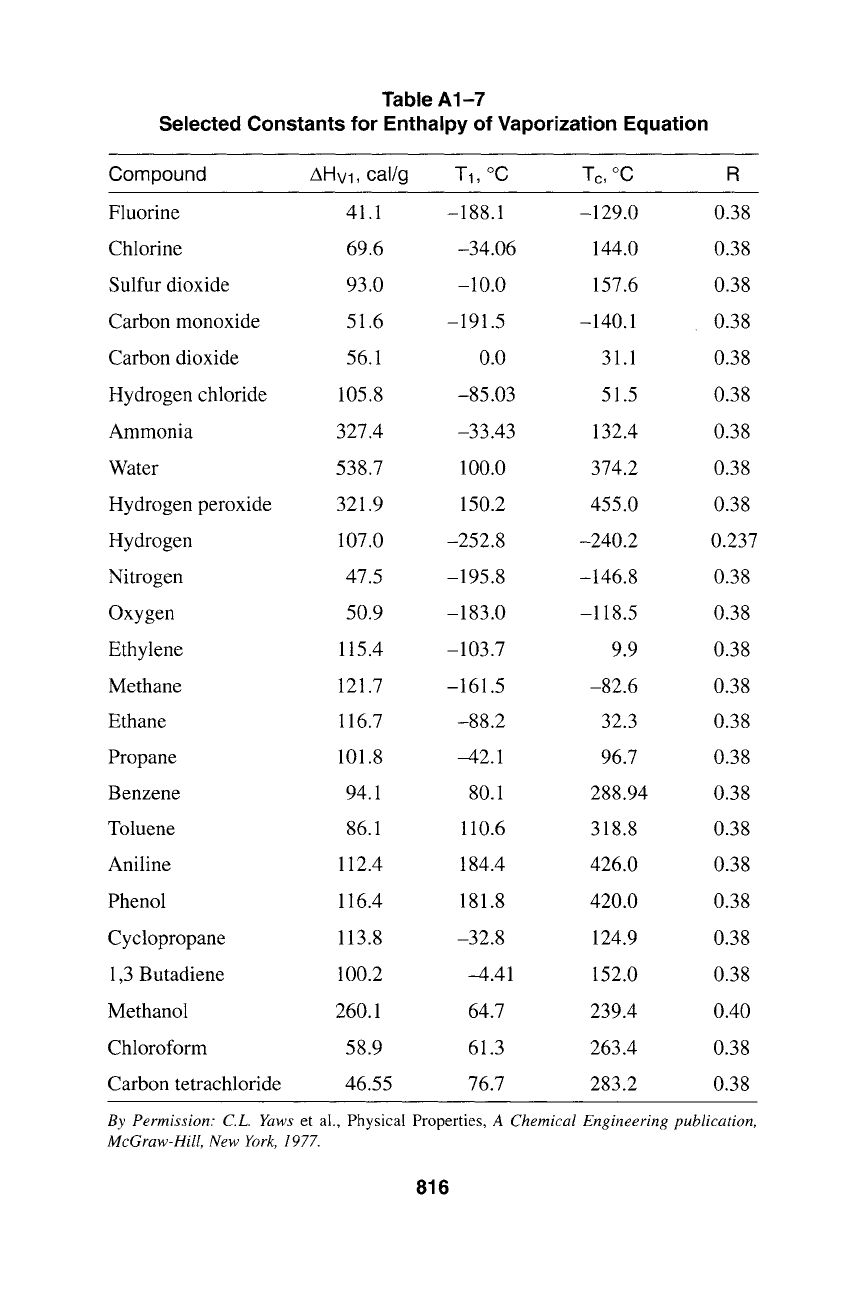
Table A1-7
Selected Constants for Enthalpy of Vaporization Equation
Compound AHvl, cal/g T1, ~ Tc, ~ R
Fluorine 41.1 -188.1 -129.0 0.38
Chlorine 69.6 -34.06 144.0 0.38
Sulfur dioxide 93.0 -10.0 157.6 0.38
Carbon monoxide 51.6 -191.5 -140.1 0.38
Carbon dioxide 56.1 0.0 31.1 0.38
Hydrogen chloride 105.8 -85.03 51.5 0.38
Ammonia 327.4 -33.43 132.4 0.38
Water 538.7 100.0 374.2 0.38
Hydrogen peroxide 321.9 150.2 455.0 0.38
Hydrogen 107.0 -252.8 -240.2 0.237
Nitrogen 47.5 -195.8 -146.8 0.38
Oxygen 50.9 -183.0 -118.5 0.38
Ethylene 115.4 -103.7 9.9 0.38
Methane 121.7 -161.5 -82.6 0.38
Ethane 116.7 -88.2 32.3 0.38
Propane 101.8 -42.1 96.7 0.38
Benzene 94.1 80.1 288.94 0.38
Toluene 86.1 110.6 318.8 0.38
Aniline 112.4 184.4 426.0 0.38
Phenol 116.4 181.8 420.0 0.38
Cyclopropane 113.8 -32.8 124.9 0.38
1,3 Butadiene 100.2 -4.41 152.0 0.38
Methanol 260.1 64.7 239.4 0.40
Chloroform 58.9 61.3 263.4 0.38
Carbon tetrachloride 46.55 76.7 283.2 0.38
By Permission: C.L. Yaws
et al., Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
816
