Coker A.K. Fortran Programs for Chemical Process Design, Analysis, and Simulation
Подождите немного. Документ загружается.

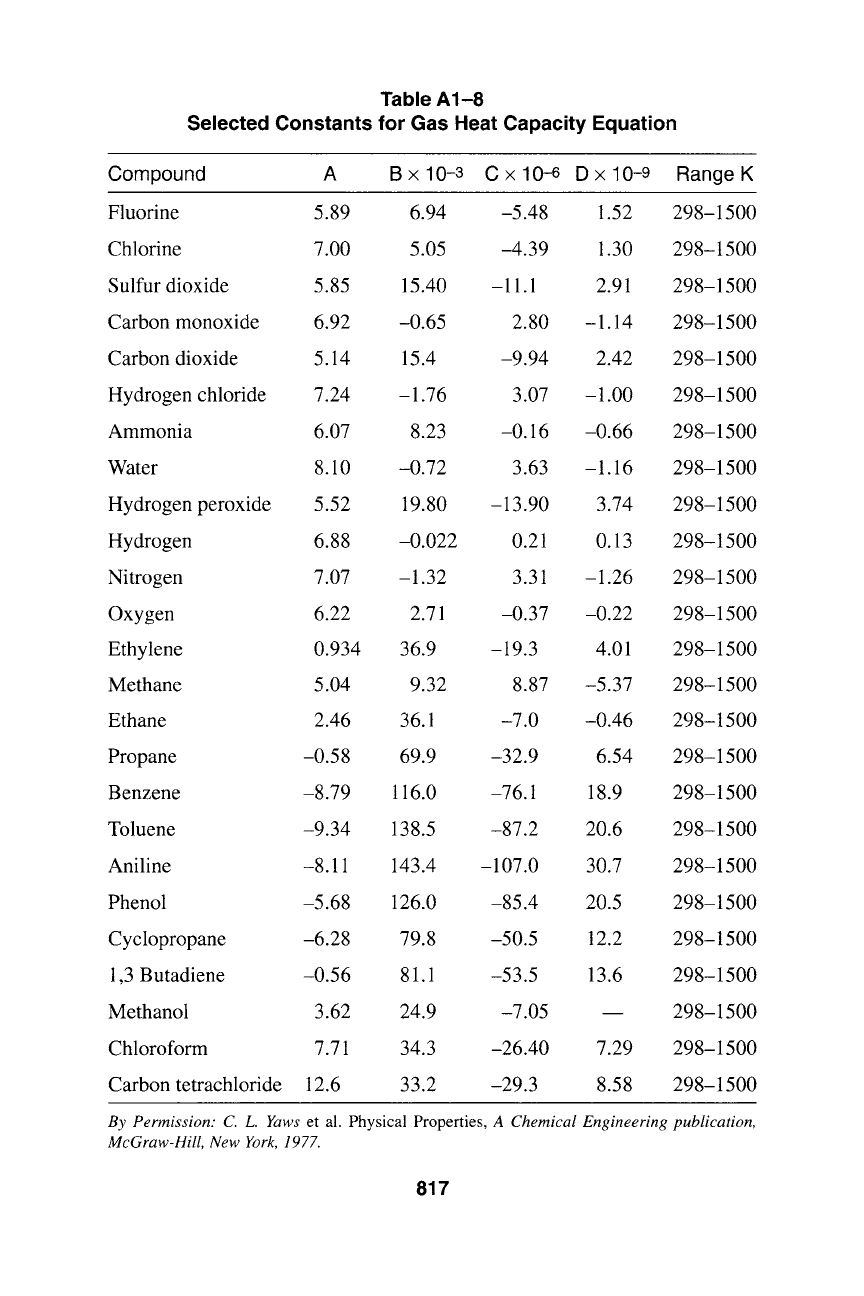
Table A1-8
Selected Constants for Gas Heat Capacity Equation
Compound A B x 10-3 C x 10-6 D x 10-9 Range K
Fluorine 5.89 6.94 -5.48 1.52 298-1500
Chlorine 7.00 5.05 -4.39 1.30 298-1500
Sulfur dioxide 5.85 15.40 -11.1 2.91 298-1500
Carbon monoxide 6.92 -0.65 2.80 -1.14 298-1500
Carbon dioxide 5.14 15.4 -9.94 2.42 298-1500
Hydrogen chloride 7.24 -1.76 3.07 -1.00 298-1500
Ammonia 6.07 8.23 -0.16 -0.66 298-1500
Water 8.10 -0.72 3.63 -1.16 298-1500
Hydrogen peroxide 5.52 19.80 -13.90 3.74 298-1500
Hydrogen 6.88 -0.022 0.21 0.13 298-1500
Nitrogen 7.07 -1.32 3.31 -1.26 298-1500
Oxygen 6.22 2.71 -0.37 -0.22 298-1500
Ethylene 0.934 36.9 -19.3 4.01 298-1500
Methane 5.04 9.32 8.87 -5.37 298-1500
Ethane 2.46 36.1 -7.0 -0.46 298-1500
Propane -0.58 69.9 -32.9 6.54 298-1500
Benzene -8.79 116.0 -76.1 18.9 298-1500
Toluene -9.34 138.5 -87.2 20.6 298-1500
Aniline -8.11 143.4 -107.0 30.7 298-1500
Phenol -5.68 126.0 -85.4 20.5 298-1500
Cyclopropane -6.28 79.8 -50.5
1,3 Butadiene -0.56 81.1 -53.5
Methanol 3.62 24.9 -7.05
12.2 298-1500
13.6 298-1500
298-1500
Chloroform 7.71 34.3 -26.40 7.29 298-1500
Carbon tetrachloride 12.6 33.2 -29.3 8.58 298-1500
By Permission: C. L. Yaws
et al. Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
817
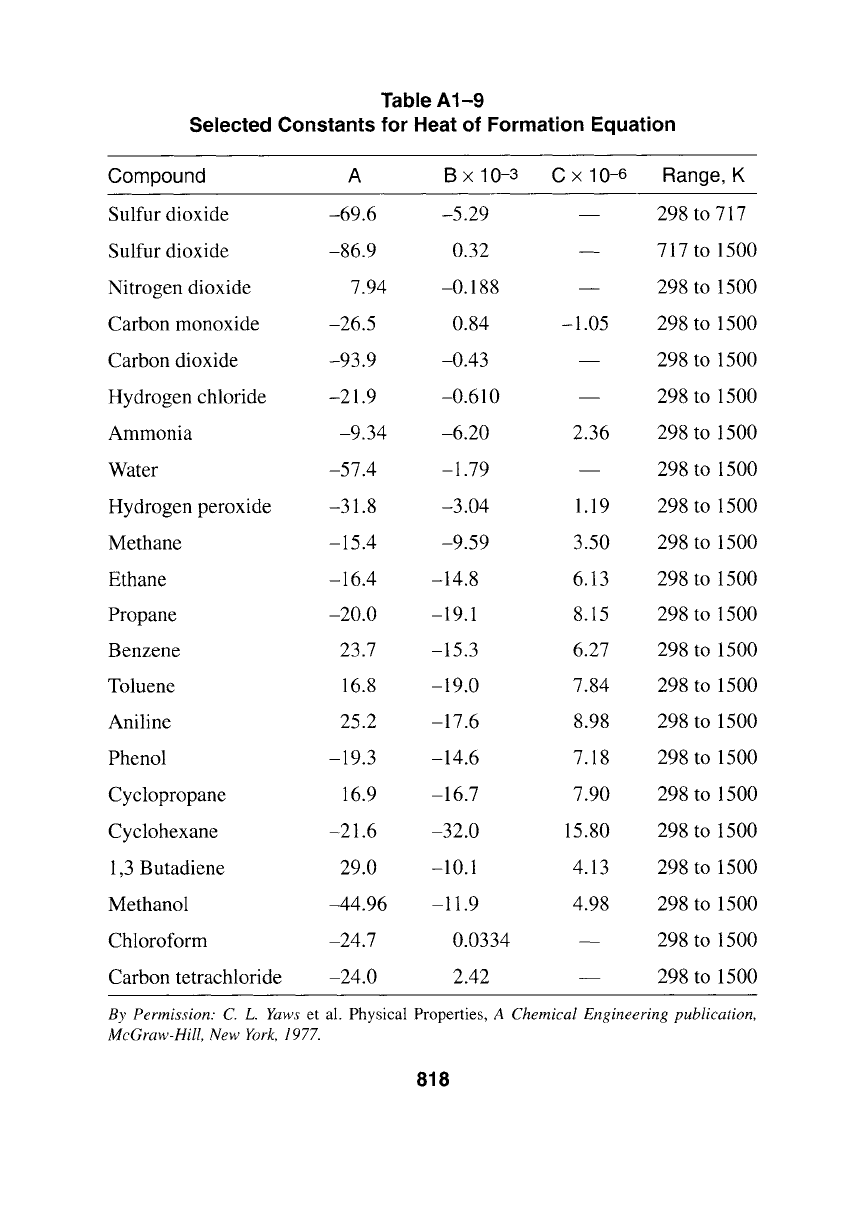
Table A1-9
Selected Constants for Heat of Formation Equation
Compound A B x 10-3 C x 10 -6 Range, K
Sulfur dioxide -69.6 -5.29 298 to 717
Sulfur dioxide -86.9 0.32 717 to 1500
Nitrogen dioxide 7.94 -0.188
Carbon monoxide -26.5 0.84
298 to 1500
- 1.05 298 to 1500
Carbon dioxide -93.9 -0.43 298 to 1500
Hydrogen chloride -21.9 -0.610
Ammonia -9.34 -6.20
298 to 1500
2.36 298 to 1500
Water -57.4 -1.79 298 to 1500
Hydrogen peroxide -31.8 -3.04 1.19 298 to 1500
Methane -15.4 -9.59 3.50 298 to 1500
Ethane
Propane
Benzene
-16.4 -14.8 6.13 298 to 1500
-20.0 -19.1 8.15 298 to 1500
23.7 -15.3 6.27 298 to 1500
Toluene 16.8 -19.0 7.84 298 to 1500
Aniline 25.2 -17.6 8.98 298 to 1500
Phenol -19.3 -14.6 7.18 298 to 1500
Cyclopropane 16.9 -16.7 7.90 298 to 1500
Cyclohexane -21.6 -32.0 15.80 298 to 1500
1,3 Butadiene 29.0 - 10.1 4.13 298 to 1500
Methanol -44.96 -11.9 4.98 298 to 1500
Chloroform -24.7 0.0334 298 to 1500
Carbon tetrachloride -24.0
2.42 298 to 1500
By Permission: C. L. Yaws
et al. Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
818
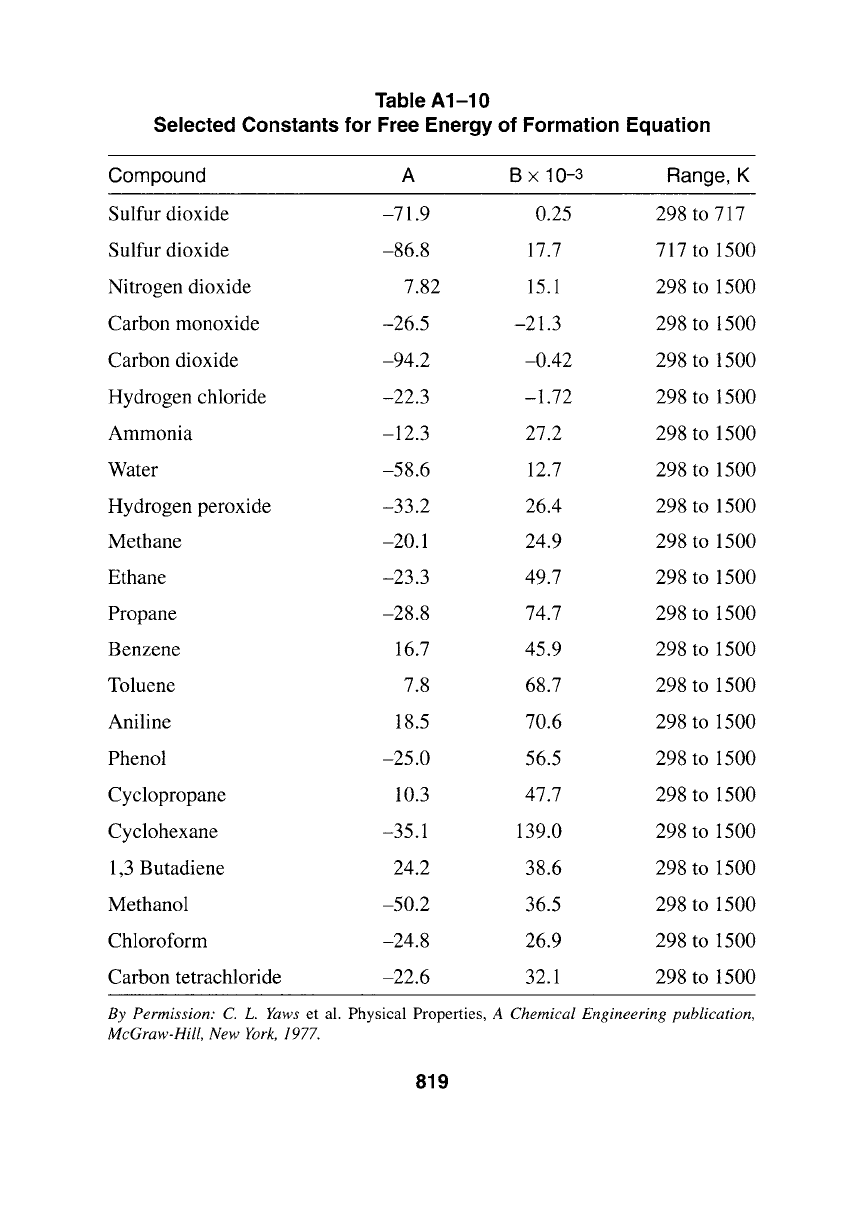
Table A1-10
Selected Constants for Free Energy of Formation Equation
Compound A B x 10-3 Range, K
Sulfur dioxide -71.9 0.25 298 to 717
Sulfur dioxide -86.8 17.7 717 to 1500
Nitrogen dioxide 7.82 15.1 298 to 1500
Carbon monoxide -26.5 -21.3 298 to 1500
Carbon dioxide -94.2 -0.42 298 to 1500
Hydrogen chloride -22.3 -1.72 298 to 1500
Ammonia - 12.3 27.2 298 to 1500
Water -58.6 12.7 298 to 1500
Hydrogen peroxide -33.2 26.4 298 to 1500
Methane -20.1 24.9 298 to 1500
Ethane -23.3 49.7 298 to 1500
Propane -28.8 74.7 298 to 1500
Benzene 16.7 45.9 298 to 1500
Toluene 7.8 68.7 298 to 1500
Aniline 18.5 70.6 298 to 1500
Phenol -25.0 56.5 298 to 1500
Cyclopropane 10.3 47.7 298 to 1500
Cyclohexane -35.1 139.0 298 to 1500
1,3 Butadiene 24.2 38.6 298 to 1500
Methanol -50.2 36.5 298 to 1500
Chloroform -24.8 26.9 298 to 1500
Carbon tetrachloride -22.6 32.1 298 to 1500
By Permission: C. L. Yaws
et al. Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
819
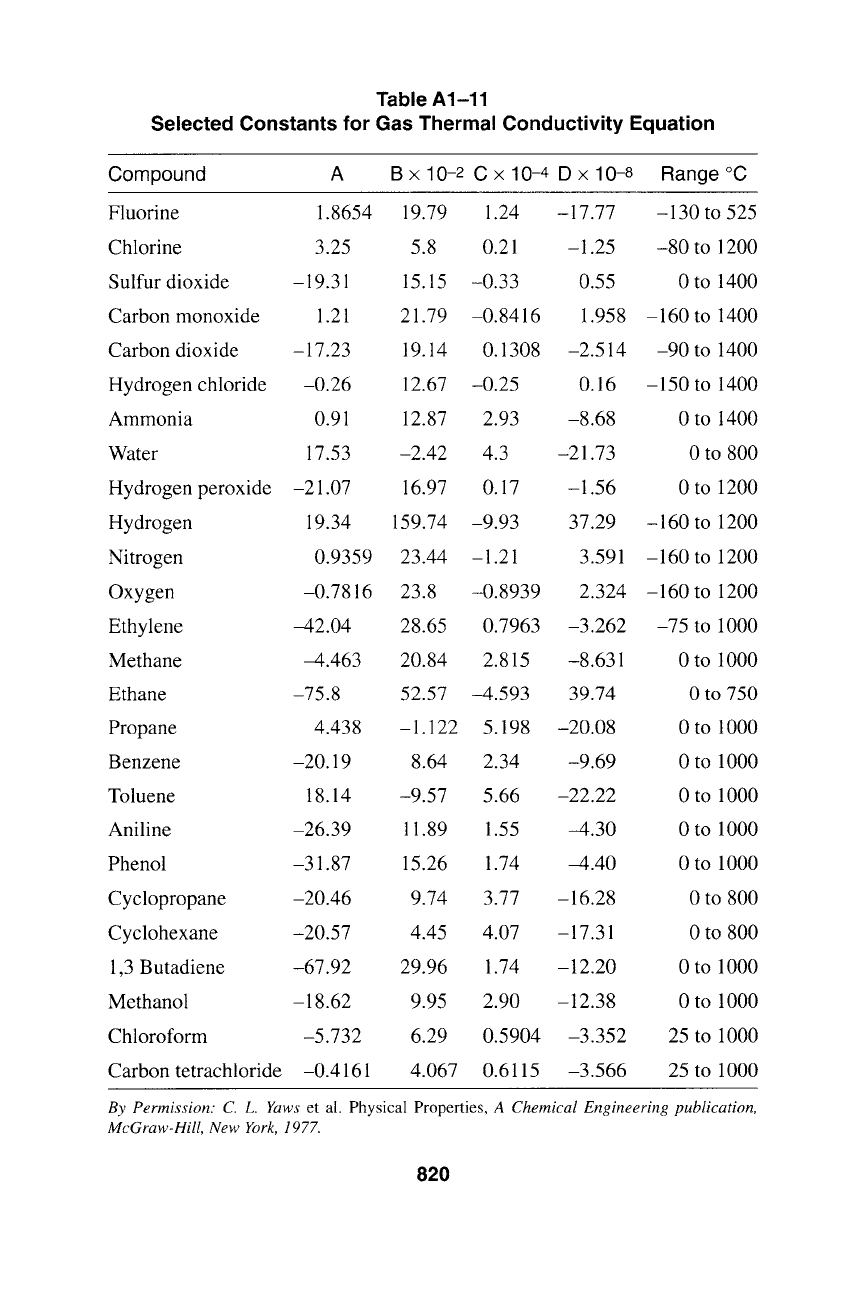
Table A1-11
Selected Constants for Gas Thermal Conductivity Equation
Compound A
B x 10 -2 C x 10 -4 D x 10-8 Range ~
Fluorine 1.8654 19.79 1.24 -17.77 -130 to 525
Chlorine 3.25 5.8 0.21 -1.25 -80 to 1200
Sulfur dioxide -19.31 15.15 -0.33 0.55 0 to 1400
Carbon monoxide 1.21 21.79 -0.8416 1.958 -160 to 1400
Carbon dioxide -17.23 19.14 0.1308 -2.514 -90 to 1400
Hydrogen chloride -0.26 12.67 -0.25 0.16 -150 to 1400
Ammonia 0.91 12.87 2.93 -8.68 0 to 1400
Water 17.53 -2.42 4.3 -21.73 0 to 800
Hydrogen peroxide -21.07 16.97 0.17 -1.56 0 to 1200
Hydrogen 19.34 159.74 -9.93 37.29 -160 to 1200
Nitrogen 0.9359 23.44 -1.21 3.591 -160 to 1200
Oxygen -0.7816 23.8 -0.8939 2.324 -160 to 1200
Ethylene -42.04 28.65 0.7963 -3.262 -75 to 1000
Methane -4.463 20.84 2.815 -8.631 0 to 1000
Ethane -75.8 52.57 -4.593 39.74 0 to 750
Propane 4.438 -1.122 5.198 -20.08 0 to 1000
Benzene -20.19 8.64 2.34 -9.69 0 to 1000
Toluene 18.14 -9.57 5.66 -22.22 0 to 1000
Aniline -26.39 11.89 1.55 -4.30 0 to 1000
Phenol -31.87 15.26 1.74 -4.40 0 to 1000
Cyclopropane -20.46 9.74 3.77 -16.28 0 to 800
Cyclohexane -20.57 4.45 4.07 -17.31 0 to 800
1,3 Butadiene -67.92 29.96 1.74 -12.20 0 to 1000
Methanol -18.62 9.95 2.90 -12.38 0 to 1000
Chloroform -5.732 6.29 0.5904 -3.352 25 to 1000
Carbon tetrachloride -0.4161 4.067 0.6115 -3.566 25 to 1000
By Permission." C. L. Yaws
et al. Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
820
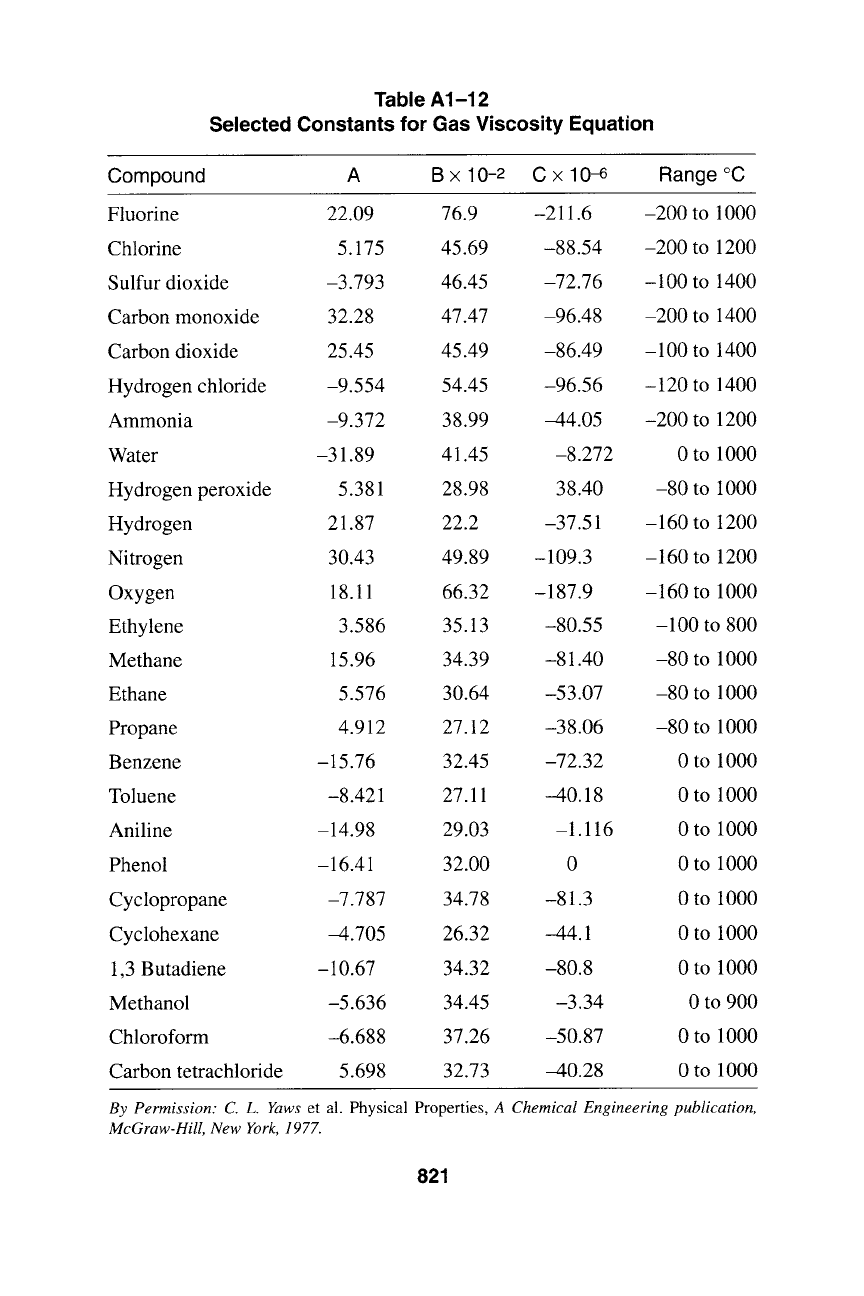
Table A1-12
Selected Constants for Gas Viscosity Equation
Compound A B x 10-2 C x 10-6 Range ~
Fluorine 22.09 76.9 -211.6 -200 to 1000
Chlorine 5.175 45.69 -88.54 -200 to 1200
Sulfur dioxide -3.793 46.45 -72.76 -100 to 1400
Carbon monoxide 32.28 47.47 -96.48 -200 to 1400
Carbon dioxide 25.45 45.49 -86.49 -100 to 1400
Hydrogen chloride -9.554 54.45 -96.56 -120 to 1400
Ammonia -9.372 38.99 -44.05 -200 to 1200
Water -31.89 41.45 -8.272 0 to 1000
Hydrogen peroxide 5.381 28.98 38.40 -80 to 1000
Hydrogen 21.87 22.2 -37.51 -160 to 1200
Nitrogen 30.43 49.89 -109.3 -160 to 1200
Oxygen 18.11 66.32 -187.9 -160 to 1000
Ethylene 3.586 35.13 -80.55 -100 to 800
Methane 15.96 34.39 -81.40 -80 to 1000
Ethane 5.576 30.64 -53.07 -80 to 1000
Propane 4.912 27.12 -38.06 -80 to 1000
Benzene -15.76 32.45 -72.32 0 to 1000
Toluene -8.421 27.11 -40.18 0 to 1000
Aniline -14.98 29.03 -1.116 0 to 1000
Phenol -16.41 32.00 0 0 to 1000
Cyclopropane -7.787 34.78 -81.3 0 to 1000
Cyclohexane -4.705 26.32 -44.1 0 to 1000
1,3 Butadiene -10.67 34.32 -80.8 0 to 1000
Methanol -5.636 34.45 -3.34 0 to 900
Chloroform -6.688 37.26 -50.87 0 to 1000
Carbon tetrachloride 5.698 32.73 -40.28 0 to 1000
By Permission: C. L. Yaws
et al. Physical Properties,
A Chemical Engineering publication,
McGraw-Hill, New York, 1977.
821

Appendix B
Table B-1
Compressibility Factor of Natural Gas
*****************************************************************************
TEMPERATURE, oF SPECIFIC GRAVITY
6O. .550
PRESSURE psia. COMPRESSIBILITY FACTOR Z
i00. .98160
200. .97934
300. .97263
400. .96270
500. .95042
600. .93651
700. .92156
800. .90610
900. .89053
i000. .87520
ii00. .86041
1200. .84636
1300. .83322
1400. .82110
1500. .81008
1600. .80021
1700. .79151
1800. .78399
1900. .77767
2000. .77253
2100. .76860
2200. .76586
2300. .76432
2400. .76395
2500. .76473
822

Table B-1
(continued)
*****************************************************************************
TEMPERATURE, oF SPECIFIC GRAVITY
60. .550
PRESSURE psia. COMPRESSIBILITY FACTOR Z
2600. .76663
2700. .76960
2800. .77354
2900. .77838
3000. .78401
3100. .79032
3200. .79718
3300. .80447
3400. .81208
3500. .81989
3600. .82780
3700. .83573
3800. .84363
3900. .85144
4000. .85916
4100. .86678
4200. .87436
4300. .88195
4400. .88966
4500. .89761
4600. .90597
4700. .91495
4800. .92478
4900. .93575
5000. .94817
823

Table B-2
Compressibility Factor of Natural Gas
*****************************************************************************
TEMPERATURE, oF SPECIFIC GRAVITY
70. .550
PRESSURE psia. COMPRESSIBILITY FACTOR Z
i00. .98208
200. .98046
300. .97453
400. .96546
500. .95411
600. .94117
700. .92721
800. .91272
900. .89810
i000. .88369
ii00. .86975
1200. .85649
1300. .84406
1400. .83257
1500. .82209
1600. .81267
1700. .80432
1800. .79706
1900. .79089
2000. .78583
2100. .78187
2200. .77903
2300. .77729
2400. .77665
2500. .77710
2600. .77859
2700. .78109
2800. .78452
2900. .78880
3000. .79383
3100. .79951
3200. .80572
3300. .81234
3400. .81926
3500. .82638
3600. .83360
3700. .84085
3800. .84806
3900. .85519
4000. .86223
4100. .86919
4200. .87610
4300. .88303
4400. .89006
4500. .89733
4600. .90499
4700. .91323
4800. .92228
4900. .93241
5000. .94392
824

Table B-3
Compressibility Factor of Natural Gas
*****************************************************************************
TEMPERATURE, oF SPECIFIC GRAVITY
80. .550
PRESSURE psia. COMPRESSIBILITY FACTOR Z
i00. .98250
200. .98147
300. .97625
400. .96798
500. .95750
600. .94547
700. .93243
800. .91886
900. .90513
I000. .89158
ii00. .87845
1200. .86594
1300. .85419
1400. .84330
1500. .83334
1600. .82434
1700. .81634
1800. .80933
1900. .80333
2000. .79834
2100. .79437
2200. .79143
2300. .78952
2400. .78864
2500. .78878
2600. .78990
2700. .79197
2800. .79492
2900. .79868
3000. .80317
3100. .80826
3200. .81387
3300. .81987
3400. .82616
3500. .83264
3600. .83922
3700. .84582
3800. .85239
3900. .85889
4000. .86531
4100. .87165
4200. .87794
4300. .88425
4400. .89066
4500. .89729
4600. .90429
4700. .91185
4800. .92017
4900. .92952
5000. .94017
825
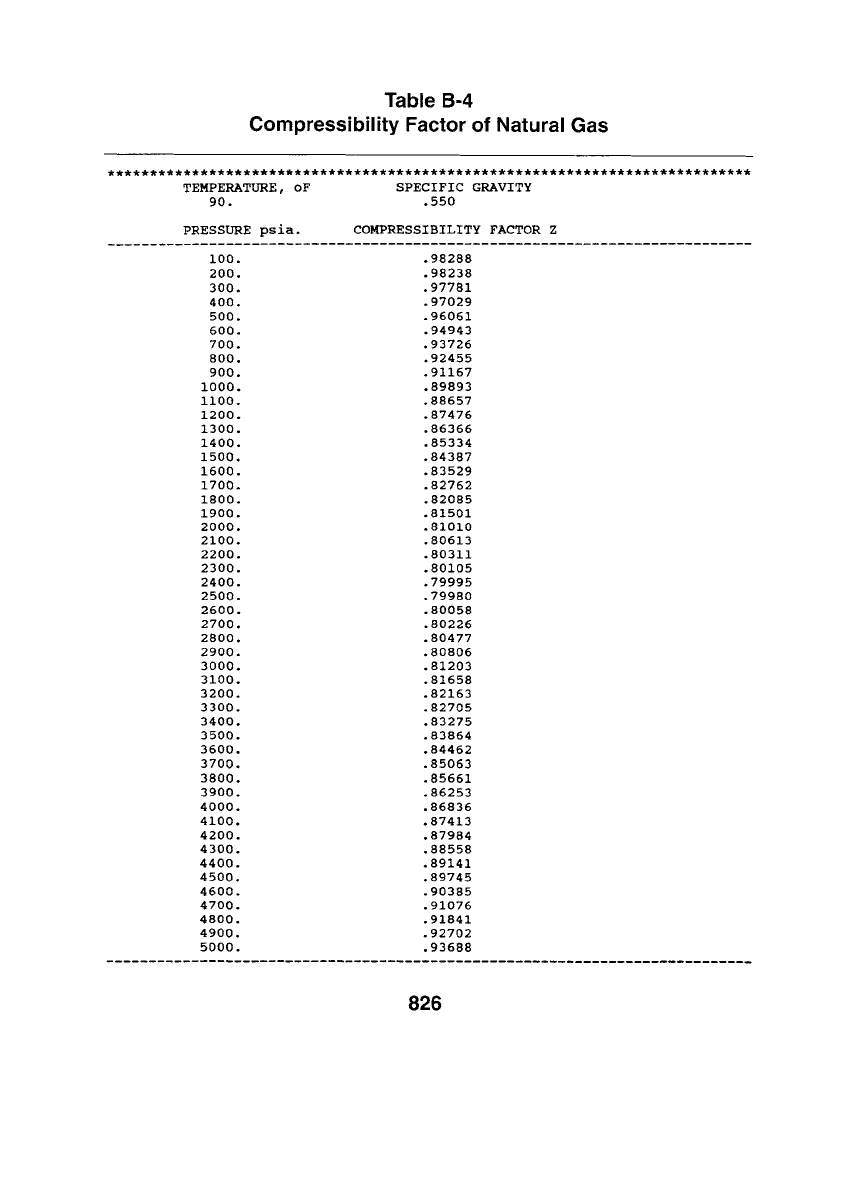
Table B-4
Compressibility Factor of Natural Gas
****************************************************************************
TEMPERATURE, oF SPECIFIC GRAVITY
90. .550
PRESSURE psia. COMPRESSIBILITY FACTOR Z
i00. .98288
200. .98238
300. .97781
400. .97029
500. .96061
600. .94943
700. .93726
800. .92455
900. .91167
i000. .89893
ii00. .88657
1200. .87476
1300. .86366
1400. .85334
1500. .84387
1600. .83529
1700. .82762
1800. .82085
1900. .81501
2000. .81010
2100. .80613
2200. .80311
2300. .80105
2400. .79995
2500. .79980
2600. .80058
2700. .80226
2800. .80477
2900. .80806
3000. .81203
3100. .81658
3200. .82163
3300. .82705
3400. .83275
3500. .83864
3600. .84462
3700. .85063
3800. .85661
3900. .86253
4000. .86836
4100. .87413
4200. .87984
4300. .88558
4400. .89141
4500. .89745
4600. .90385
4700. .91076
4800. .91841
4900. .92702
5000. .93688
826
