Kenneth E. Gonsalves, Craig R. Halberstadt, Cato T. Laurencin, Lakshmi S. Nair. Biomedical Nanostructures
Подождите немного. Документ загружается.

47. Zimmermann TS, et al. RNAi-mediated gene silencing in non-human primates.
Nature 2006;441:111114.
48. Schiffelers RM, et al. Cancer siRNA therapy by tumor selective delivery with
ligand-targeted sterically stabilized nanoparticle. Nucleic Acids Res 2004;32:e149.
49. Lorenz C, Hadwiger P, John M, Vornlocher HP, Unverzagt C. Steroid and lipid
conjugates of siRNAs to enhance cellular uptake and gene silencing in liver cells.
Bioorg Med Chem Lett 2004;14:49754977.
50. Kunath T, et al. Transgenic RNA interference in ES cell-derived embryos
recapitulates a genetic null phenotype. Nat Biotechnol 2003;21:559561.
51. Xia H, Mao Q, Paulson HL, Davidson BL. siRNA-mediated gene silencing in vitro
and in vivo. Nat Biotechnol 2002;20:10061010.
52. Bridge AJ, Pebernard S, Ducraux A, Nicoulaz AL, Iggo R. Induction of an
interferon response by RNAi vectors in mammalian cells. Nat Genet 2003;34:
263264.
53. Sledz CA, Holko M, deVeer MJ, Silverman RH, Williams BR. Activation of the
interferon system by short-interfering RNAs. Nat Cell Biol 2003;5:834839.
54. Persengiev SP, Zhu X, Green MR. Nonspecific, concentration-dependent stimula-
tion and repression of mammalian gene expression by small interfering RNAs
(siRNAs). RNA 2004;10:1218.
55. Boden D, Pusch O, Lee F, Tucker L, Ramratnam B. Human immunodeficiency
virus type 1 escape from RNA interference. J Virol 2003;77:1153111535.
56. Paroo Z andCorey DR. Challenges for RNAi in vivo. Trends Biotechnol
2004;22:390394.
57. Braasch DA, et al. Biodistribution of phosphodiester and phosphorothioate
siRNA. Bioorg Med Chem Lett 2004;14:11391143.
58. Harborth J, et al. Sequence, chemical, and structural variation of small interfering
RNAs and short hairpin RNAs and the effect on mammalian gene silencing.
Antisense Nucleic Acid Drug Dev 2003;13:83105.
356
BIOMEDICAL NANOSTRUCTURES

CHAPTER 14
Multiscale Coculture Models
for Orthopedic Interface Tissue
Engineering
HELEN H. LU and I-NING E. WANG
14.1 INTRODUCTION
Biological tissues and organs are complex systems whose maintenance and
repair are regulated through cellcell, cellmatrix interactions, as well as
physical and chemical stimuli. These extrinsic and intrinsic signals collectively
contribute to organ homeostasis and physiological functi on. Cellular interac-
tions, in general, consist of one or all of the following: direct cellcell
communications as well as extracellular matrix- and soluble factor-mediated
effects. Cellcell communications imply direct physical contact between cells,
while cellmatrix interactions are initiated by the binding of transmembrane
proteins such as integrins to the extracellular matrix or biomaterial surface. In
addition, secreted soluble factors and cytokines serve as chemical messengers
directing both local and systemic cellular communi cations. These signaling
molecules can be classified as autocrine, paracrine, or endocrine factors. While
cell-to-cell contact may be required to initiate one or all of the above
interactions, the secretion of soluble factors enables interactions beyond the
immediate vici nity of the cell population and facilitates communication
between adjacent tissues and organ systems.
The manipulation of cell microenvironment via modulation of cellcell
communication and cellmatrix interaction is important for the regeneration
of complex tissue systems [1–3]. Currently, the specific mechanisms underlying
the interactions between different types of connective tissue cells are not well
understood. Physiologically relevant in vitro and in vivo models are thus needed
in order to investigate the role of cellular communications in multitissue
BiomedicalNanostructures, Editedby KennethE.Gonsalves,CraigR.Halberstadt,CatoT.Laurencin,
and Lakshmi S. Nair
Copyright # 2008 John Wiley & Sons, Inc.
357
regeneration and organ homeostasis. This review will first discuss the relevance
of cellular interactions for engineering complex tissues for orthopedic repair,
focusing on the regeneration of the multitissue interface between bone and soft
tissues such as ligaments and tendo ns. After a brief overview of the different
types of coculture models, their application in elucidating the mechanisms of
interface regeneration through controlled interactions between connective
tissue cells (e.g., osteoblasts, fibroblast, and chondrocytes) will be reviewed,
highlighting the potential of controlled heterotypic cellular interactions for
interface tissue engineering and biological fixation of soft tissue grafts.
14.2 CELLULAR INTERACTIONS AND THE SOFT
TISSUE-TO-BONE INTERFACE
Soft tissues such as tendons or ligaments insert into bone through a fibrocartilage
interface [4–10], and the reestablishment of this physiologic interface is essential
for the clinical efficacy of soft tissue-based grafts. This complex interface consists
of several continuous yet distinct tissue regions, with each region exhibiting
characteristic cellular phenotype and matrix composition. It is thus likely that
heterotypic cellular interactions are essential for the maintenance and repair of
the distinct matrix zones at the interface between soft tissue and bone. Using the
insertion site of anterior cruciate ligament (ACL) to bone as a model system, this
review highlights the utility of in vitro coculture models for elucidating the role of
heterotypic cellular interactions in interface regeneration.
Anatomically, the ACL-to-bone interface consists of three distinct yet
continuous tissue regions: ligament, fibrocartilage, and bone. The fibrocartilage
region is further divided into nonmineralized and mineralized zones. The ligament
proper contains fibroblasts embedded in collagen-rich matrix. The nonminer-
alized fibrocartilage matrix consists of ovoid chondrocytes, and collagen types I
and II are found within the proteoglycan-rich matrix. Hypertrophic chondrocytes
reside in the mineralized fibrocartilage zone with a pericellular matrix containing
collagen type X [8, 11]. The last region is subchondral bone, within which
osteoblasts, osteocytes, and osteoclasts are embedded in a type I collagen matrix.
The multitissue organization and controlled heterogeneity are believed to be
important for minimizing stress concentrations and facilitating the transfer of
complex loads between soft and hard tissues [5, 12]. At present, it is not well
understood how this complex interface is formed, and the inability to regenerate
this interface following ligament reconstruction surgery is one of the primary
causes of graft failure [13–16].
While the mechanism of interface regeneration is not known, it has been
observed in vivo that tendon-to-bone healing following ACL reconstruction
results in the formation of a fibrovascular tissue layer within the bone tunnel
[17–20]. During the healing process, this layer reorganizes into a fibrocartilage-
like tissue. Although the location of this neo-fibrocartilage is nonanatomical,
these in vivo observations demonstrate that a fibrocartilage-like tissue can be
358 BIOMEDICAL NANOSTRUCTURES
regenerated between the soft tissue and bone. Specifically, the formation of a
fibrocartilage layer only at regions where the tendon graft directly contacts the
bone tissue suggests that the interactions between cells derived from tendon
(e.g., fibroblasts) and bone tissue (e.g., osteoblasts) are important for
fibrocartilage regeneration [2]. In particular, osteoblast fibroblast interactions
may lead to the transdifferentiation of osteoblasts and/or fibroblasts, as well as
the recruitment of progenitor or mesenchymal stem cells to the soft tissue-to-
bone interface. Thes e recruited progenitor or stem cells are likely to be
responsible for fibrocart ilage formation, and their eventual differentiation may
also be regulated by osteoblastfibroblast interactions.
To test the above hypothesis, in vitro coculture models have been reported,
which evaluate the role of heterotypic cellular interactions on the development
of fibrocartilage-specific markers [21–23]. The cell types prevalent at the native
ligament-bone insertion include fibroblasts, fibrochondrocytes, and osteo-
blasts, each residing in their own phenotypic extracellular matrix. Therefore,
the ideal coculture model for orthopedic interface tissue engineering must be
able to recapitulate the complex three-dimensional and multiscale (macro-,
micro-, and nanolevel) interactions between these distinct cell types.
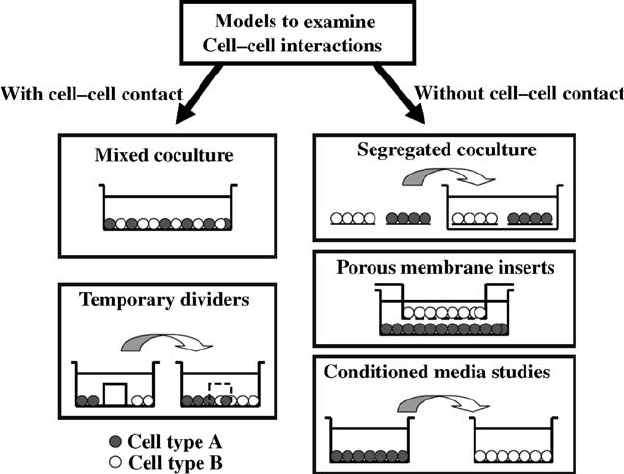
14.3 TYPES OF COCULTURE MODELS
Cellular communications can be bidirectional or multidimensional and may be
exercised at both macro- and microscales. Depending on the hypothesis of
interest, coculture models may be used to discern the individual and collective
effects of physical contact and soluble factors. Specifically, the types of existing
coculture models can be classified as either systems that enable or prevent
cellcell contact (Fig. 14.1). The advantages and disadvantages of each type of
model are briefly discussed below.
14.3.1 Coculture System with Cell–Cell Contact
14.3.1.1 Mixed Coculture The simplest coculture system that permits
physical contact between cells consists of a mixed monolayer culture of the cell
types of interest. This is achieved by combining the different cell suspensions at
the desired coculture ratio, and then seeding the mixture in the well (Fig. 14.1).
Alternatively, the second cell type can be plated directly atop of a preformed
monolayer of the first cell type. The mixed coculture model has been used
successfully to examine interactions between fibroblasts and hepatocytes [24–
26], fibroblasts and bone marrow stromal cells [27], fibroblasts and
chondrocytes [28], as well as osteoblast and chondrocyte interactions [21,29–
31]. This simple and convenient model maximizes the local heterotypic
interactions and can be used to control the level of heterotypic and homotypic
interactions by altering the seeding densities of each cell type. Interpretation of
these coculture results, however, must take into consideration any dilution
MULTISCALE COCULTURE MODELS FOR ORTHOPEDIC INTERFACE TISSUE ENGINEERING 359
effect on cell response due to mixed culture as well as any metabolic differences
between the various cell types.
14.3.1.2 Temporary Dividers Cellcell contact can also be controlled by
establishing physical barriers that are used to organize cell-seeding patterns in
coculture (Fig. 14.1). The barrier may be removed later to permit cell migration
and controlled cellcell physical contact. This model has been used to examine
interactions between fibroblasts and hepatocytes [26], as well as between
osteoblast and fibroblast as shown in Fig. 14.2a [22]. It has the advantage of
been able to excise greater control of the extent of heterotypic and homotypic
interactions, while permitting both physical contact and soluble factor
interactions. This system is, however, experimentally more challenging as a
complete seal between the individual cell compartments is required. Moreover,
cell response and soluble-factor transport in this model is a function of the
physical and chemical properties of the divider material utilized.
14.3.2 Coculture System Without Cell–Cell Contact
14.3.2.1 Segregated Coculture To prevent cellcell contact, a segre-
gated coculture system may be established by first forming individual cultures
FIGURE 14.1 Schematic of models to evaluate heterotypic cellcell interactions.
360
BIOMEDICAL NANOSTRUCTURES
of each cell type, and later cocultivate them in the same environment. For
example, monolayers of each cell type can be preformed on tissue culture
coverslips, and then cocultured together to examine heterotypic cellular
interactions. In contrast to the mixed coculture model, the primary advantage
of this coverslip-based system is that the response of the subpopulation of cells
in coculture can be analyzed. This coculture method has been used to
determine the interactions between chondrocytes and synovial cells [32], as well
as those of fibroblasts and osteoblasts [22]. A disadvantage of the segregated
coculture model is that physical contact cannot be completely prevented in the
long term, as the cells often migrate from the coverslips and form a
heterogeneous culture on the coverslips or in the culture well.
14.3.2.2 Porous Membrane Inserts The advent of cell culture membrane
inserts reduced many of the experimental difficulties associated with coculture
and has lead to the development of reliable and reproduci ble coculture systems.
The transwell
1
inserts are designed with pores small enough to prevent cell
migration, yet still large enough to permit transport of interactive fact ors and
other biomolecules. The insert also provides an additional culturing surface,
while effectively preventing heterotypic cellcell contact. This method has been
widely used to investigate the mechanism of endochondral ossification by
examining the interactions between chondrocytes and mesenchymal stem cell
[33, 34], chondrocytes, and osteoblasts [35, 36], and osteoblasts and fibroblasts
[37], in addition to a variety of other cell types [38–40]. This coculture model is
attractive due to its reproducibility and ease of experimentation, although the
effects of soluble factors detected are unidirectional. Moreover, extensive cell
growth can cover the pores of the inserts, limiting cellular interactions to only
the bottom first monolayer of cells, which may be insufficient to elicit a
significant response in coculture.
14.3.2.3 Conditioned Media Studies Another widely utilized method for
determining soluble factor effects is through conditioned media studies, during
which the culture media from one cell type is introduced into the culture of the
second cell type. This method has been used in conjunction with direct physical
contact models to investigate the mechanism of interaction between fibroblasts
and stem cells [27, 41], fibroblasts and chond rocytes [28], as well as between
osteoblasts and fibro blasts [42]. The advantages of this model include its
simplicity and the ability to immediately discern any soluble factor-related
effects, plus the potential for subsequent identification of relevant soluble
factors using quantitative assays. An inherent limitation of conditioned media
studies, however, is the issue of nutrient deficiency, as the experimental group is
exposed to the conditioned media and only supplemented partially with fresh
media. Additional challenges including the reproduction of both the temporal
distribution and optimal concentrations of the secreted factors in conditioned
media studies.
MULTISCALE COCULTURE MODELS FOR ORTHOPEDIC INTERFACE TISSUE ENGINEERING 361

14.4 COCULTURE MODELS FOR ORTHOPEDIC INTERFACE
TISSUE ENGINEERING
As discussed in Section 14.2, the ideal coculture model for evaluating the role
of cell ular communications in interface tissue engineering must be able to
recapitulate the complex interactions inherent at the native soft tissue-to-bone
interface. To this end, existing coculture models can be combined and
optimized in conjunction with other systems in order to evaluate interactions
between interface-relevant cells such as osteoblasts, fibroblasts, chondrocytes,
and stem cells. The application of these models to determine the effects of
cellcell contact and soluble factors for interface tissue engineering are
reviewed below.
14.4.1 Coculture Models of Osteoblasts and Fibroblasts
To determine the role of fibroblast and osteoblast interactions in fibrocartilage
formation, a novel coculture model permitting both cell contact and paracrine
interaction was reported by Wang et al. [22]. This model was designed to emulate
the in vivo condition where the tendon is in direct contact with bone tissue during
graft healing following ACL reconstruction. Briefly, as shown in Fig. 14.2a,
osteoblasts and fibroblasts were first seeded on opposing sides of a tissue culture
well. The cells were separated by a hydrogel divider preformed in the center of
the well. Once the cells reached confluence on each side, the divider was removed,
allowing the osteoblasts and fibroblasts to migrate and interact within the
interface region. The two cell types communicated through paracrine and
autocrine factors, as well as eventual physical contact in the interface region. It
was found that osteoblastfibroblast interaction led to a decrease in cell
FIGURE 14.2 Examples of cellcell interaction models used in interface tissue
engineering: (a) Coculture of fibroblasts and osteoblasts with temporary agarose gel
divider; (b) triculture of fibroblasts and osteoblasts with chondrocytes encapsulated in
3D hydrogel; (c) microscale coculture of fibroblasts and osteoblasts using microfluidic
patterning techniques. FB: fibroblasts, OB: osteoblasts, CH: chondrocytes.
362
BIOMEDICAL NANOSTRUCTURES
proliferation, a reduction in osteoblast-mediated mineralization, accompanied
by an increase in the mineralization potential of fibroblasts [23].
This osteoblastfibroblast model is limited in its ability to distill the relative
contributions of soluble factor and cellcell contact to these observed changes,
and the specific response of each cell type in coculture is not known. Further
refinement of the model using the coverslip-based segregated coculture system
[22] revealed that the observed changes in cell phenotype are indeed cell type-
specific. For example, doubling the number of osteoblasts in coculture resulted
in greater suppression of cell proliferation, suggesting that the decreased
mitotic activity is affected by osteoblasts instead of fibroblasts. In addition,
conditioned media studies have been performed to investigate the relative
contribution of cell cell contact and soluble factors. Shan et al. introduced
three types of conditioned media (osteoblasts, fibroblasts, and osteobl ast-
fibroblast coculture) to individual culture of fibroblasts or osteoblasts, and
examined the proli feration and differentiation of each cell type over time [42].
It was found that both autocrine and paracrine effects were responsible for the
changes in phenotype observed due to osteoblastfibroblast coculture. The
magnitude of response of the cultures treated with cond itioned media was,
however, signifi cantly lower than those seen in coculture [42], suggesting that
cellcell contact also plays a role in cell transdifferentiation. Interestingly, the
osteoblastfibroblas t cocultured media elicited distinct responses when
compared to media from single cultures of either osteoblasts or fibroblasts.
These observations confirm that osteoblast and fibroblast respond differently
when they are in a cocultured or single-cultured environment.
Findings from these coculture studies collectively demonstrate that
osteoblastfibroblas t interactions modulate cell phenotypes and may lead to
transdifferentiation. While it is not known which or if any of these cells are
directly responsible for interface regeneration, their interactions most likely
have a downstream effect, either in terms of directing osteoblast or fibroblast
transdifferentiation, or in the recruitment and induction of progenitor or stem
cells for fibrocartilage formation.
14.4.2 Coculture Models of Osteoblasts and Chondrocytes
As the postnatal ligament-to-bone interface comprised chondrocyte-like cells [43],
the effects of osteoblast and chondrocyte coculture also needs to be examined.
Using a layered mixed coculture model, Jiang et al. seeded an osteoblast
monolayer atop a condensed chondrocyte micromass [21]. This model permitted
direct physical contact between these two cell types, while maintaining the required
3D culture for chondrocytes. It was found that while the chondrocytes continued
to synthesize collagen type II, proteoglycan deposition was significantly lower in
coculture. Alkaline phosphatase activity remained unchanged in the osteoblasts,
while their mineralization potential was significantly reduced due to coculture.
These results suggest that osteoblastfibroblast and osteoblastchondrocyte
interactions are key modulators of cell phenotypes.
MULTISCALE COCULTURE MODELS FOR ORTHOPEDIC INTERFACE TISSUE ENGINEERING 363
14.4.3 Coculture and Triculture Models of Osteoblasts,
Chondrocytes, and Fibroblasts
Recently, Wang et al. reported on a triculture model of fibroblasts,
chondrocytes, and osteoblasts; the three cell types dominant in their respective
region of the interface, namely ligament, fibrocartilage, and bone (Fig. 14.2b)
[44]. To ensure their phenotypic spherical morphology, chondrocytes were
encapsulated within the hydrogel divider used in the previous osteo-
blastfibroblast coculture model (Fig. 14.2a) [22]. Once again, a reduction in
the proliferation of both osteoblasts and fibroblasts due to heterotypic cell
interactions was found, while the number of cho ndrocytes remained relatively
constant over time in the hydrogel. Triculture led to reduced osteoblast-
mediated mineralization, accompanied by increa sed fibroblast mineralization .
The chondrocytes continued to produce proteoglycans and the expression of
both collage n types I and II were detected in the interfacial region.
This model was subsequently used to compare the effects of
fibroblastosteoblast interaction on the response of interface-relevant cells
such as bone marrow stromal cells and ligament fibroblasts [45]. In this study,
the stromal cells or fibroblasts were encapsulated in the hydrogel insert instead
of chondrocytes. While minimal response was observed with fibroblasts, the
stromal cells measured significantly higher ALP activity during triculture.
Moreover, expression s of interface-relevant markers such as collagen type II
and proteoglycans were detected in triculture with the bone marrow stroma l
cells. These results provide new evidence that osteoblastfibroblast interac-
tions may initiate the differentiation of stem cells or progenitor cells for
fibrocartilage regeneration.
14.5 MACRO- AND MICROSCALE COCULTURE
The coculture models described above have evaluated cellular interactions
only at the macroscopic level, while it is well known that physiologically
relevant interactions are conducted at microscopic and nanolevels. Modern
methods for spatial control over cell attachment and spreading have
facilitated an unprecedented level of sophistication in the investigation of
cellcell interactions. Micropatterning and microfluidic models have been
utilized for the microscale coculture of a variety of cells types [1, 23, 4651].
Bioinspired micropatterning of cells have been reported using photolitho-
graphy, microcontact printing, micromolding, inkjet printing, and dip-pen
spotting [4650, 52]. A distinct advantage of these microscale systems is that
the diffusion distances between the different cell types can be controlled and
designed to match the biological scenario, leading to physiologic and
relatively rapid interaction times. Biomimetic pattern of cells can also be
achieved with high fidelity. Moreover, the smaller solution volume and lower
concentration of effector molecules required at the microscale level, increase
364 BIOMEDICAL NANOSTRUCTURES
the overall sensitivity of the system in its ability to detect the local effects of
cellcell contact and secreted factors when compared to the macroscale
models.
Although promising, micropatterning is limited by the fact that chemical or
biochemical modification of the surface is usually required. Consequently, the
selection of adhesion proteins, as well as the underlying chemistry and physical
properties of the surface, have significant effects on the stability of the cell
pattern and the eventual cellcell interactions. Moreover, long term evaluation
of cellular interactions is difficult as cell migration often disrupts the
micropattern over time. Microfluidic systems have therefore been developed
to circumvent these limitations associated with micropatterning. The primary
advantage of the microfluidic model resides in its ability to provide high
resolution spatial control of fluid flow and factor concen tration, as well as the
potential for long term microscale coculture.
For interface tissue engineering, a microscale cocultu re model is also
physiologically more relevant, as the native human ligament-to-bone interface
spans merely 200300 mm [8]. Tsai et al. recently investigated the effects of
osteoblast and fibroblast micropatterning on cellular function and organiza-
tion [23]. This microscale coculture model (Fig. 14.2c) was fabricated using soft
lithography and replica molding, and utilized microfluidics to exert spatial
control and cell patterning. Not surprisingly, differenc es in cell growth and
differentiation were detected at the microscale when compared to the
macroscale model [22]. These observations emphasize the importance of
considering interaction scale in coculture models.
14.6 TWO-DIMENSIONAL (2D ) AND THREE-DIMENSIONAL
(3D) COCULTURES
In addition to the interaction scale, the effects of 2D and 3D substrates on
cellular communications during coculture must be considered. The majority of
published coculture studies of connective tissue cells have been performed on
2D surfaces, while it is well known that cell response is distinctly different in
the 3D environment. Cukierman et al., evaluated human foreskin fibroblast
adhesion to various 2D and 3D matrices [53], and observed that a cell-derived
3D matrix was significa ntly more effective in promoting cell adhesion and
migration. Similarly, Spalazzi et al. evaluated osteoblast and cho ndrocyte
coculture as a function of scaffold architecture [3], and found temporal
morphological changes in chondrocytes seeded on extracellular matrix
preformed by osteoblasts on either a 2D (flat disc) or 3D (microsphere-based
scaffold) substrate. Specifically, the tim e required for nonphe notypic chon-
drocyte spreading was prolonged on the microsphere scaffolds when compared
to the 2D discs. The expression of adhesion complexes or the characteristics of
the preformed extracellular matrix likely differ between 2D and 3D substrates
[3, 53].
MULTISCALE COCULTURE MODELS FOR ORTHOPEDIC INTERFACE TISSUE ENGINEERING 365
