Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


Example 2.10 Ethylene oxide is produced in a catalytic steady-flow reactor. A
feed consisting of 70% C
2
H
4
and 30% O
2
(mole percent) is fed into the reactor at
a rate of 100 mol/s. The following chemical reactions take place in the reactor:
Reaction 1: 2C
2
H
4
þ O
2
! 2C
2
H
4
O
Reaction 2: C
2
H
4
þ 3O
2
! 2CO
2
þ 2H
2
O
Reaction 3: C
2
H
4
þ 2O
2
! 2CO þ 2H
2
O
An analysis of the exit stream indicates that its composition is 41.17% C
2
H
4
,
37.65% C
2
H
4
O, and 7.06% O
2
(mole percent). Determine:
a. The conversions of ethylene and oxygen
b. The yield of ethylene oxide
c. The selectivity of ethylene oxide
Solution Since each chemical reaction has a species that does not participate in
the other two, the three reactions are independent, and their stoichiometric coef-
ficients are
(s
C
2
H
4
)
1
¼2(s
O
2
)
1
¼1(s
C
2
H
4
O
)
1
¼2(s
H
2
O
)
1
¼0(s
CO
2
)
1
¼0(s
CO
)
1
¼0
(s
C
2
H
4
)
2
¼1(s
O
2
)
2
¼3(s
C
2
H
4
O
)
2
¼0(s
H
2
O
)
2
¼2(s
CO
2
)
2
¼2(s
CO
)
2
¼0
(s
C
2
H
4
)
3
¼1(s
O
2
)
3
¼2(s
C
2
H
4
O
)
3
¼0(s
H
2
O
)
3
¼2(s
CO
2
)
3
¼0(s
CO
)
3
¼2
D
1
¼1 D
2
¼0 D
3
¼1
To calculate the required quantities, first determine the extents of the reactions.
Using Eqs. 2.3.11 and 2.3.12, the molar fraction of species j in the outlet stream is
y
j
¼
F
j
in
þ
P
n
I
m
(s
j
)
m
_
X
m
F
tot
in
þ
P
n
I
m
D
m
_
X
m
(a)
Writing (a) for C
2
H
4
,C
2
H
4
O, and O
2
,
y
C
2
H
4
¼
70 2
_
X
1
_
X
2
_
X
3
100
_
X
1
þ
_
X
3
¼0:4117 (b)
y
C
2
H
4
O
¼
2
_
X
1
100
_
X
1
þ
_
X
3
¼0:3765 (c)
y
O
2
¼
30
_
X
1
3
_
X
2
2
_
X
3
100
_
X
1
þ
_
X
3
¼0:0706 (d)
60 STOICHIOMETRY

Solving (b), (c), and (d),
_
X
1
¼16,
_
X
2
¼2, and
_
X
3
¼1mol=s. Using Eq. 2.3.11,
the molar flow rates of the respective species are
F
C
2
H
4
¼70 2(16) þ(2)1 ¼ 35 mol=s (e)
F
C
2
H
4
O
¼2(16) ¼32 mol=s(f)
F
O
2
¼30 þ(1)16 þ(3)2 þ(2)1 ¼6mol=s (g)
F
CO
2
¼2(2) ¼4 mol=s (h)
a. Using the conversion definition (Eq. 2.6.1b) together with (e) and (g), the
conversions of C
2
H
4
and O
2
are, respectively,
f
C
2
H
4
¼
70 35
70
¼ 0:50 (i)
f
O
2
¼
30 6
30
¼ 0:80 ( j)
b. The desirable reaction is Reaction 1 and the limiting reactant is O
2
. Hence,
using Eq. 2.6.14, the yield of ethylene oxide (on the basis of the oxygen
fed) is
h
C
2
H
4
O
¼
1
2
32
30
¼ 0:533 (k)
c. Using Eq. 2.6.18, the selectivity of ethylene oxide is
s
C
2
H
4
O
¼
1
2
32
30 6
¼ 0:667 (l)
Example 2.11 Dichloromethane is formed by reacting methane and chlorine.
The following reactions take place in the reactor:
Reaction 1: CH
4
þ Cl
2
! CH
3
Cl þ HCl
Reaction 2: CH
3
Cl þ Cl
2
! CH
2
Cl
2
þ HCl
Reaction 3: CH
2
Cl
2
þ Cl
2
! CHCl
3
þ HCl
Reaction 4: CHCl
3
þ Cl
2
! CCl
4
þ HCl
A feed consisting of 40% methane and 60% chlorine is fed to a steady-flow reac-
tor at a rate of 100 mol/min. The desirable product is dichloromethane. At the
outlet, the molar flow rate of methane is 10 mol/min and the stream contains
2.6 CHARACTERIZATION OF REACTOR PERFORMANCE 61

three times monochloromethane as dichloromethane, and the content of the latter
is twice that of trichloromethane. Determine:
a. The limiting reactant
b. The conversions of methane and chlorine
c. The yield of dichloromethane
d. The selectivity of dichloromethane.
Solution The stoichiometric relation between the desirable product (dichloro-
methane) and the reactants is obtained by adding Reaction 1 and Reaction 2:
CH
4
þ 2Cl
2
! CH
2
Cl
2
þ 2HCl
This stoichiometric relation represents the desirable reaction, and its stoichio-
metric coefficients are
s
CH
4
¼1 s
Cl
2
¼2 s
CH
3
Cl
¼ 1 s
HCl
¼ 2 D ¼ 0
a. To identify the limiting reactant, use Eq. 2.5.3 for the two reactants:
F
CH
4
in
s
CH
4
¼
40
1
¼ 40 and
F
Cl
2
in
s
Cl
2
¼
60
2
¼ 30
Hence, the limiting reactant is chlorine.
b. To determine the conversions, we have to calculate the extents of the given
four independent reactions. The stoichiometric coefficients of the species in
the four reactions are
(s
CH
4
)
1
¼1(s
CH
4
)
2
¼ 0(s
CH
4
)
3
¼ 0(s
CH
4
)
4
¼ 0
(s
Cl
2
)
1
¼1(s
Cl
2
)
2
¼1(s
Cl
2
)
3
¼1(s
Cl
2
)
4
¼1
(s
CH
3
Cl
)
1
¼ 1(s
CH
3
Cl
)
2
¼1(s
CH
3
Cl
)
3
¼ 0(s
CH
3
Cl
)
4
¼ 0
(s
CH
2
Cl
2
)
1
¼ 0(s
CH
2
Cl
2
)
2
¼ 1(s
CH
2
Cl
2
)
3
¼1(s
CH
2
Cl
2
)
4
¼ 0
(s
CHCl
3
)
1
¼ 0(s
CHCl
3
)
2
¼ 0(s
CHCl
3
)
3
¼ 1(s
CHCl
3
)
4
¼1
(s
CCl
4
)
1
¼ 0(s
CCl
4
)
2
¼ 0(s
CCl
4
)
3
¼ 0(s
CCl
4
)
4
¼ 1
Using Eq. 2.3.11, the species molar flow rates at the reactor outlet are
(F
CH
4
)
out
¼ 40
_
X
1
(F
Cl
2
)
out
¼ 60
_
X
1
_
X
2
_
X
3
_
X
4
62 STOICHIOMETRY

(F
HCl
)
out
¼
_
X
1
þ
_
X
2
þ
_
X
3
þ
_
X
4
(F
CH
3
Cl
)
out
¼
_
X
1
_
X
2
(F
CH
2
Cl
2
)
out
¼ 0 þ
_
X
2
_
X
3
(F
CHCl
3
)
out
¼ 0 þ
_
X
3
_
X
4
(F
CCl
4
)
out
¼ 0 þ
_
X
4
Using the given information,
40
_
X
1
¼ 10
(F
CH
3
Cl
)
out
(F
CH
2
Cl
2
)
out
¼
_
X
1
_
X
2
_
X
2
_
X
3
¼ 0:5
(F
CH
2
Cl
2
)
out
(F
CHCl
3
)
out
¼
_
X
2
_
X
3
_
X
3
_
X
4
¼ 2
(F
CHCl
3
)
out
(F
CCl
4
)
out
¼
_
X
3
_
X
4
_
X
4
¼ 2
The solution of these four equations is
_
X
1
¼ 30
_
X
2
¼ 22
_
X
3
¼ 6
_
X
4
¼ 2 mol=min
The species flow rates at the reactor outlet are
(F
CH
3
Cl
)
out
¼8(F
CH
2
Cl
2
)
out
¼16 (F
CHCl
3
)
out
¼4(F
CCl
4
)
out
¼2 mol=min
(F
Cl
2
)
out
¼0(F
HCl
)
out
¼60 mol=min
Using Eq. 2.6.1b, the conversions of the two reactants are
f
CH
4
;
(F
CH
4
)
in
(F
CH
4
)
out
(F
CH
4
)
in
¼
40 10
40
¼0:75
f
Cl
2
;
(F
Cl
2
)
in
(F
Cl
2
)
out
(F
Cl
2
)
in
¼
60 0
60
¼1:0
c. Using Eq. 2.6.15, the yield of the dichloromethane (with respect to the
methane) is
h
CH
2
Cl
2
¼
1
1
16 0
40
¼ 0:4
2.6 CHARACTERIZATION OF REACTOR PERFORMANCE 63

d. Using Eq. 2.6.18, the selectivity of the dichloromethane (with respect to the
methane) is
h
CH
2
Cl
2
¼
1
1
16 0
40 30
¼ 0:533
2.7 DIMENSIONLESS EXTENTS
The stoichiometric relations derived so far provide a glimpse at the key role the
reaction extents play in the analysis of chemical reactors. Whenever the extents
of the independent reactions are known, the reactor composition and all other
stated variables (temperature, enthalpy, etc.) can be determined. Unfortunately,
the extent has two deficiencies:
†
It is not a measurable quantity and, consequently, must be related to other
measurable quantities (concentrations, pressure, etc.).
†
It is an extensive quantity depending on the amount of reactants initially in the
reactor or on the inlet flow rate into the reactor.
While the use of calculated quantities may seem, at first, cumbersome and even
counterproductive, it actually simplifies the analysis of chemical reactors with mul-
tiple reactions. In fact, calculated quantities such as enthalpy and free energy are
commonly used in thermodynamics resulting in simplified expressions. Here too,
by using the extents of independent reactions, we formulate the design of chemical
reactors by the smallest number of design equations.
To characterize the generic behavior of chemical reactors, it is preferred to
describe their operations in terms of intensive dimensionless quantities. To convert
the reaction extents to intensive quantities, dimensionless extents are defined. For
batch reactors, the dimensionless extent, Z
m
, of the mth independent reaction is
defined by
Z
m
;
Extent of the mth independent reaction
Total number of moles of reference state
¼
X
m
(N
tot
)
0
(2:7:1)
where (N
tot
)
0
is the total number of moles of a conveniently selected reference state.
The selection of the reference state will be discussed below, but in most appli-
cations the initial state of the reactor is taken as the reference state.
For flow reactors, the dimensionless extent, Z
m
, is defined by
Z
m
;
Extent per time of the mth independent reaction
Total molar flow rate of reference stream
¼
_
X
m
(F
tot
)
0
(2:7:2)
64 STOICHIOMETRY
where (F
tot
)
0
is the total molar flow rate of a conveniently selected reference stream.
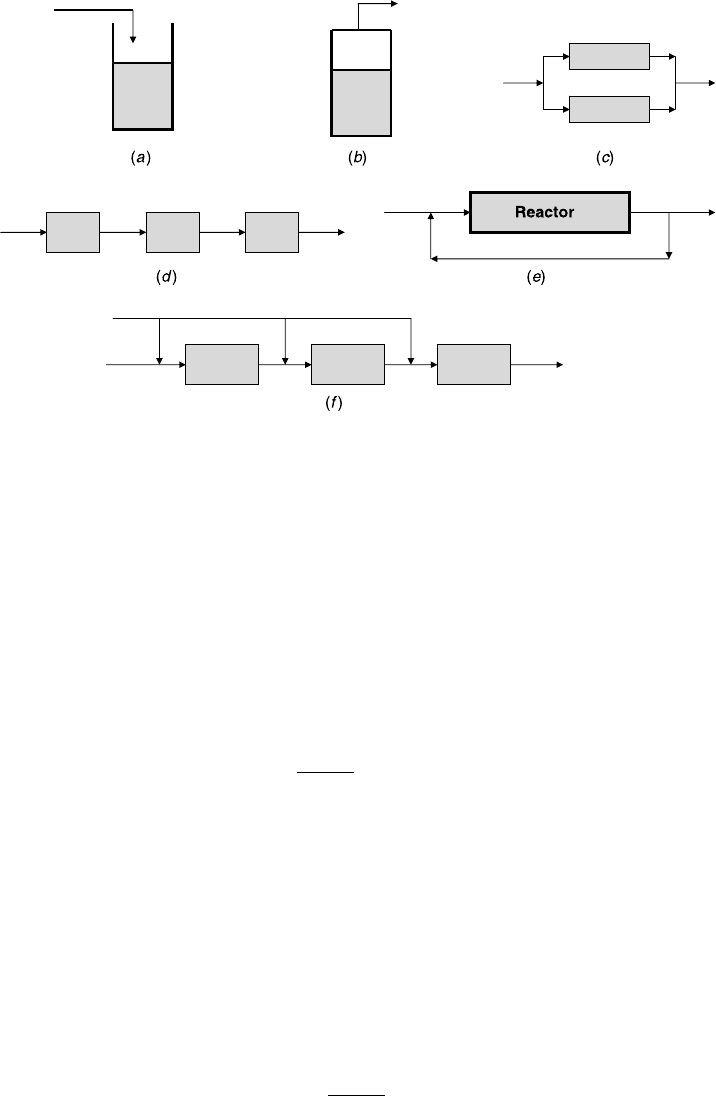
These definitions should apply for all reactor configurations; some are shown in
Figure 2.2, The selection of the reference stream is discussed below. Note that
the numerical values of dimensionless extents depend on the reference state or
reference stream as well as on the chemical formulas used to represent the chemical
transformations.
For batch reactors, using Eqs. 2.7.1 and 2.3.3, the species composition reduces to
N
j
(t) ¼ (N
tot
)
0
N
tot
(0)
(N
tot
)
0
y
j
(0) þ
X
n
I
m
(s
j
)
m
Z
m
(t)
()
(2:7:3)
where y
j
(0) ¼ N
j
(0)=N
tot
(0) is the initial molar fraction of species j in the reactor.
When the initial state is selected as the reference state, Eq. 2.7.3 reduces to
N
j
(t) ¼ (N
tot
)
0
y
j
0
þ
X
n
I
m
(s
j
)
m
Z
m
(t)
"#
(2:7:4)
Similarly, using Eq. 2.3.4, the total molar content in the reactor
N
tot
(t) ¼ (N
tot
)
0
N
tot
(0)
(N
tot
)
0
þ
X
n
I
m
D
m
Z
m
(t)
()
(2:7:5)
Figure 2.2 Various reactor Configurations: (a) semibatch, (b) distillation reactor, (c) split
feed, (d ) cascade, (e) recycle reactor, and ( f ) side injection.
2.7 DIMENSIONLESS EXTENTS 65

When the initial state is selected as the reference state, Eq. 2.7.5 reduces to
N
tot
(t) ¼ (N
tot
)
0
1 þ
X
n
I
m
D
m
Z
m
(t)
"#
(2:7:6)
For steady-flow reactors, using Eq. 2.7.2, Eq. 2.3.11 reduces to
F
j
¼ (F
tot
)
0
(F
tot
)
in
(F
tot
)
0
y
j
in
þ
X
n
I
m
(s
j
)
m
Z
m
()
(2:7:7)
where (F
tot
)
in
is the total molar flow rate of the inlet stream and y
j
in
¼ F
j
in
=(F
tot
)
in
is
the molar fraction of species j in the inlet stream. When the inlet stream is selected
as the reference stream, Eq. 2.7.7 reduces to
F
j
¼ (F
tot
)
0
y
j
0
þ
X
n
I
m
(s
j
)
m
Z
m
"#
(2:7:8)
Similarly, using Eq. 2.3.12, the total molar flow rate in the reactor
F
tot
¼ (F
tot
)
0
(F
tot
)
in
(F
tot
)
0
þ
X
n
I
m
D
m
Z
m
()
(2:7:9)
When the inlet stream is selected as the reference stream, Eq. 2.7.9 reduces to
F
tot
¼ (F
tot
)
0
1 þ
X
n
I
m
D
m
Z
m
"#
(2:7:10)
Example 2.12 The gas-phase catalytic oxidation of ammonia is investigated in
an isothermal batch reactor. The following reactions take place in the reactor:
Reaction 1: 4NH
3
þ 5O
2
! 4NO þ 6H
2
O
Reaction 2: 4NH
3
þ 3O
2
! 2N
2
þ 6H
2
O
Reaction 3: 2NO þ O
2
! 2NO
2
Reaction 4: 4NH
3
þ 6NO ! 5N
2
þ 6H
2
O
Initially, the reactor contains 4 mol of NH
3
and 6 mol of O
2
, and its pressure is
2 atm. At time t, the reactor pressure is 2.12 atm, and an analysis of the reactor
content indicates that mole fraction of NH
3
is 0.07547, and of N
2
is 0.1132.
Using the dimensionless extents, determine the reactor composition.
66 STOICHIOMETRY

Solution First we have to determine the number of independent reactions, and
to select a set of independent reactions. We construct a matrix of stoichiometric
coefficients for the given reactions:
NH
3
O
2
NO H
2
ON
2
NO
2
4 5 4600
4 3 0620
0 1 2002
406650
2
6
6
6
4
3
7
7
7
5
(a)
We conduct a Gaussian elimination procedure and reduce Matrix (a) to
4 5 4600
012010
004012
0 0 0000
2
6
6
4
3
7
7
5
(b)
Matrix (b) is a reduced matrix, and, since it has three nonzero rows, there are
three independent chemical reactions. The nonzero rows in Matrix (b) provide
the following set of independent reactions (stoichiometric relations):
Reaction 1: 4NH
3
þ 5O
2
! 4NO þ 6H
2
O
Reaction 5: 2NO ! O
2
þ N
2
Reaction 6: 4NO ! N
2
þ 2NO
2
Note that the second and third rows in Matrix (b) represent two chemical reac-
tions that are not among the original reactions, and we denote them as Reactions
5 and 6. The stoichiometric coefficients of these three independent chemical
reactions are
(s
NH
3
)
1
¼4(s
O
2
)
1
¼5(s
NO
)
1
¼ 4(s
H
2
O
)
1
¼ 6(s
N
2
)
1
¼ 0
(s
NO
2
)
1
¼ 0 D
1
¼ 1
(s
NH
3
)
5
¼ 0(s
O
2
)
5
¼ 1(s
NO
)
5
¼2(s
H
2
O
)
5
¼ 0(s
N
2
)
5
¼ 1
(s
NO
2
)
5
¼ 0 D
5
¼ 0
(s
NH
3
)
6
¼ 0(s
O
2
)
6
¼ 0(s
NO
)
6
¼4(s
H
2
O
)
6
¼ 0(s
N
2
)
6
¼ 1
(s
NO
2
)
6
¼ 2 D
6
¼1
To determine the species compositions, we express them in terms of the extents
of these three independent reactions, Z
1
, Z
5
, and Z
6
. Selecting the initial state as
2.7 DIMENSIONLESS EXTENTS 67

the reference state, and using Eq. 2.7.4,
N
NH
3
(t) ¼ N
tot
(0)by
NH
3
(0) þ (s
NH
3
)
1
Z
1
þ (s
NH
3
)
5
Z
5
þ(s
NH
3
)
6
Z
6
c
¼ 10(0:4 4Z
1
) (c)
N
N
2
(t) ¼ N
tot
(0)by
N
2
(0) þ (s
N
2
)
1
Z
1
þ (s
N
2
)
5
Z
5
þ (s
N
2
)
6
Z
6
c¼10(5Z
1
þ Z
6
) (d)
Using, Eq. 2.7.6,
N
tot
(t) ¼ N
tot
(0)[1 þ D
1
Z
1
þ D
5
Z
5
þ D
6
Z
6
] ¼ 10(1 þZ
1
Z
6
) (e)
Using the given data, and assuming ideal gas behavior,
P(t)
P(0)
¼
N
tot
(t)
N
tot
(0)
¼ 1 þZ
1
Z
6
¼ 1:06 (f)
y
NH
3
(t) ¼
0:4 4Z
1
1 þ Z
1
Z
6
¼ 0:07547 (g)
y
N
2
(t) ¼
5Z
5
þZ
6
1 þ Z
1
Z
6
¼ 0:1132 (h)
Solving Eqs. (f), (g), and (h), Z
1
¼ 0.08; Z
5
¼ 0.10; Z
6
¼ 0.02. Using Eqs. 2.7.4
and Eq. 2.7.6, the compositions of the other species are
y
NO
(t) ¼
4Z
1
2Z
5
4Z
6
1 þ Z
1
Z
6
¼ 0:0377
y
H
2
O
(t) ¼
6Z
1
1 þ Z
1
Z
6
¼ 0:4528
y
O
2
(t) ¼
0:6 5Z
1
þ Z
5
1 þ Z
1
Z
6
¼ 0:283
y
NO
2
(t) ¼
2Z
6
1 þ Z
1
Z
6
¼ 0:0377
2.8 INDEPENDENT SPECIES COMPOSITION SPECIFICATIONS
In the preceding section, we discussed how to determine the number of indepen-
dent chemical reactions and how to select a set of independent reactions. The
number of independent reactions indicates the number of equations that we
should solve to determine the composition of the reactor. To solve these equations,
68 STOICHIOMETRY
the specified species conditions should provide independent information. Below,
we describe a method to determine what sets of species compositions provide inde-
pendent information.
Let us examine more closely the nature of the problem. Consider first a chemical
reactor where the following two reactions take place:
N
2
þ 3H
2
! 2NH
3
2NO þ O
2
! 2NO
2
Since each chemical reaction has at least one species that does not participate in the
other, both reactions are independent. Hence, we need to specify two species com-
positions to determine the extents of the two reactions. At first glance, it seems that
we can specify the composition of any two species. However, a closer examination
of the reactions reveals that none of the species participates in both reactions. By
specifying the amount of two species from the same chemical reaction, we
cannot determine the extents of the second reaction and the reactor composition.
Hence, we have to specify two independent compositions such that each relates
to a distinct independent chemical reaction. Similar situations may arise when
we deal with more complex sets of chemical reactions, where the identification
of independent compositions is not so obvious. Consider the hydrogenation of
toluene where the following two reactions take place:
C
6
H
5
CH
3
þ H
2
! C
6
H
6
þ CH
4
(2:8:1)
2C
6
H
5
CH
3
þ H
2
! (C
6
H
5
)
2
þ 2CH
4
(2:8:2)
These two chemical reactions are independent, yet if we specify the amounts of
toluene and methane, we cannot determine the composition of the other species.
The reason is that, while the two reactions are independent, we cannot select any
two species compositions to provide information on all other species. In this
case, compositions of the toluene and methane do not provide us with a relationship
on the amount of diphenyl and hydrogen. This becomes evident if we multiply
Reaction 2.8.1 by 2 and subtract Reaction 2.8.2:
(C
6
H
5
)
2
þ H
2
! 2C
6
H
6
(2:8:3)
It is clear that specifications of the amounts of toluene and methane do not provide
any information on the amounts of diphenyl and hydrogen.
Independent composition specifications depend on the relationships among indi-
vidual species and the chemical reactions taking place in the reactor, but they are
invariant of the specific set of independent reactions selected. Since the set of inde-
pendent reactions generated by the reduced matrix of the Gaussian elimination
2.8 INDEPENDENT SPECIES COMPOSITION SPECIFICATIONS 69
