Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.

TABLE A.3a Continued
Batch Reactor Plug-Flow Reactor CSTR
Definitions Dimensionless operating time:
t ;
t
t
cr
Dimensionless reaction extent:
Z
m
(t) ;
X
m
(t)
(N
tot
)
0
Reference concentration:
C
0
;
(N
tot
)
0
V
R
0
Composition of the reference state:
y
j
0
;
N
j
0
(N
tot
)
0
Dimensionless space time:
t ;
t
sp
t
cr
¼
V
R
v
0
t
cr
Dimensionless reaction extent:
Z
m
;
_
X
m
(F
tot
)
0
Reference concentration:
C
0
;
(F
tot
)
0
v
0
Composition of the reference stream:
y
j
0
;
F
j
0
(F
tot
)
0
a
Initial state is the reference state; inlet stream is the reference stream.
460

TABLE A.3b Design Equations for Ideal Reactors—General Form
a
Batch Reactor Plug-Flow Reactor CSTR
Design equation
for the mth
independent
reaction
dZ
m
dt
¼ r
m
þ
X
n
D
k
a
km
r
k
!
t
cr
C
0
V
R
V
R
0
dZ
m
dt
¼ r
m
þ
X
n
D
k
a
km
r
k
!
t
cr
C
0
Z
m
out
Z
m
in
¼ r
m
þ
X
n
D
k
a
km
r
k
!
t
t
cr
C
0
Auxiliary
relations
Species molar composition:
N
j
¼ (N
tot
)
0
N
tot
(0)
(N
tot
)
0
y
j
(0) þ
X
n
I
m
(s
j
)
m
Z
m
"#
Constant-volume reaction:
V
R
¼ V
R
(0)
Volume of variable-volume gas-phase reactor:
V
R
¼ V
R
(0)
N
tot
(0)
(N
tot
)
0
þ
X
n
I
m
D
m
Z
m
"#
u
u(0)
P(0)
P
Species concentration in constant-volume
reactor:
C
j
¼ C
0
V
R
0
V
R
(0)
N
tot
(0)
(N
tot
)
0
y
j
(0) þ
X
n
I
m
(s
j
)
m
Z
m
"#
Concentration in variable-volume gas-phase
reactor:
C
j
¼ C
0
N
tot
(0)
(N
tot
)
0
y
j
(0) þ
P
n
I
m
(s
j
)
m
Z
m
N
tot
(0)
(N
tot
)
0
þ
P
n
I
m
D
m
Z
m
u(0)
u
P
P(0)
Species molar flow rate:
F
j
¼ (F
tot
)
0
(F
tot
)
in
(F
tot
)
0
y
j
in
þ
X
n
I
m
(s
j
)
m
Z
m
"#
Volumetric flow rate of liquid phase:
v ¼ v
in
¼ v
0
v
in
v
0
Volumetric flow rate of gas phase:
v ¼ v
0
(F
tot
)
in
(F
tot
)
0
þ
X
n
I
m
D
m
Z
m
"#
u
u
in
P
in
P
Species concentration in liquid phase:
C
j
¼
F
j
v
¼ C
0
v
0
v
in
(F
tot
)
in
(F
tot
)
0
y
j
in
þ
X
n
I
m
(s
j
)
m
Z
m
"#
Species concentration in gas phase:
C
j
¼
F
j
v
¼ C
0
(F
tot
)
in
(F
tot
)
0
y
j
in
þ
P
n
I
m
(s
j
)
m
Z
m
(F
tot
)
in
(F
tot
)
0
þ
P
n
I
m
D
m
Z
m
u
in
u
P
P
in
(Continued)
461

TABLE A.3b Continued
Batch Reactor Plug-Flow Reactor CSTR
Definitions Dimensionless operating time:
t ;
t
t
cr
Dimensionless reaction extent:
Z
m
(t) ;
X
m
(t)
(N
tot
)
0
Reference concentration:
C
0
;
(N
tot
)
0
V
R
0
Composition of reference state:
y
j
0
;
N
j
0
(N
tot
)
0
Dimensionless space time:
t ;
t
sp
t
cr
¼
V
R
v
0
t
cr
Dimensionless reaction extent:
Z
m
;
_
X
m
(F
tot
)
0
Reference concentration:
C
0
;
(F
tot
)
0
v
0
Composition of reference stream:
y
j
0
;
F
j
0
(F
tot
)
0
a
Initial state is different of the reference state or inlet stream is different of the reference stream.
462

TABLE A.4 Energy Balance Equations for Ideal Reactors
a
Batch Reactor
(Constant Volume)
Plug-Flow
Reactor CSTR
Energy balance
equation
du
dt
¼
1
CF(Z
m
, u)
HTN(u
F
u)
X
n
I
m
DHR
m
dZ
m
dt
d
dt
W
sh
(N
tot
)
0
^
c
p
0
T
0
"#
(For plug-flow reactor, omit the mechanical work term.)
HTN t(u
F
u)
_
W
sh
(F
tot
)
0
^
c
p
0
T
0
¼
X
n
I
m
DHR
m
(Z
m
out
Z
m
in
) þ CF
out
(u
out
1)
CF
in
(u
in
1)
Definitions and
auxiliary
relations
Dimensionless temperature:
u ;
T
T
0
Specific molar heat capacity of the reference state:
Gas Phase Liquid Phase
^
c
p
0
;
X
J
j
y
j
0
c
p
j
(T
0
)
^
c
p
0
;
M
0
c
p
(N
tot
)
0
Dimensionless heat of reaction:
DHR
m
;
DH
R
m
(T
0
)
^
c
p
0
T
0
Dimensionless heat-transfer number:
HTN ;
Ut
cr
C
0
^
c
p
0
S
V
Dimensionless heat-transfer rate:
d
dt
_
Q
F
tot
0
^
c
p
0
T
0
¼ HTN (u
F
u)
Dimensionless temperature:
u ;
T
T
0
Specific molar heat capacity of the reference stream:
Gas Phase Liquid Phase
^
c
p
0
;
X
J
j
y
j
0
c
p
j
(T
0
)
^
c
p
0
;
_
m
(F
tot
)
0
c
p
Dimensionless heat of reaction:
DHR
m
;
DH
R
m
(T
0
)
T
0
^
c
p
0
Dimensionless heat-transfer number:
HTN ;
Ut
cr
C
0
^
c
p
0
S
V
Dimensionless heat-transfer rate for plug-flow reactor:
d
dt
_
Q
F
tot
0
^
c
p
0
T
0
¼ HTN (u
F
u)
(Continued)
463

TABLE A.4 Continued
Batch Reactor
(Constant Volume)
Plug-Flow
Reactor CSTR
Dimensionless heat-transfer rate for CSTR:
_
Q
F
tot
0
^
c
p
0
T
0
¼ HTN t(u
F
u)
Correction factor of heat capacity for liquid phase:
CF(Z
m
, u) ;
M
c
p
M
0
c
p
0
Correction factor of heat capacity for gas phase:
CF(Z
m
, u) ¼
1
^
c
p
0
X
J
j
y
j
0
^
c
p
j
(u) þ
X
J
j
^
c
p
j
(u)
X
n
I
m
(s
j
)
m
: Z
m
"#
Correction factor of heat capacity for liquid phase:
CF(Z
m
, u) ;
_
m
c
p
_
m
0
c
p
0
Correction factor of heat capacity for gas phase:
CF(Z
m
, u) ¼
1
^
c
p
0
X
J
j
y
j
0
^
c
p
j
(u) þ
X
J
j
^
c
p
j
(u)
X
n
I
m
(s
j
)
m
: Z
m
"#
a
Initial state is reference state; inlet stream is the reference stream.
464

APPENDIX B
MICROSCOPIC SPECIES
BALANCES—SPECIES
CONTINUITY EQUATIONS
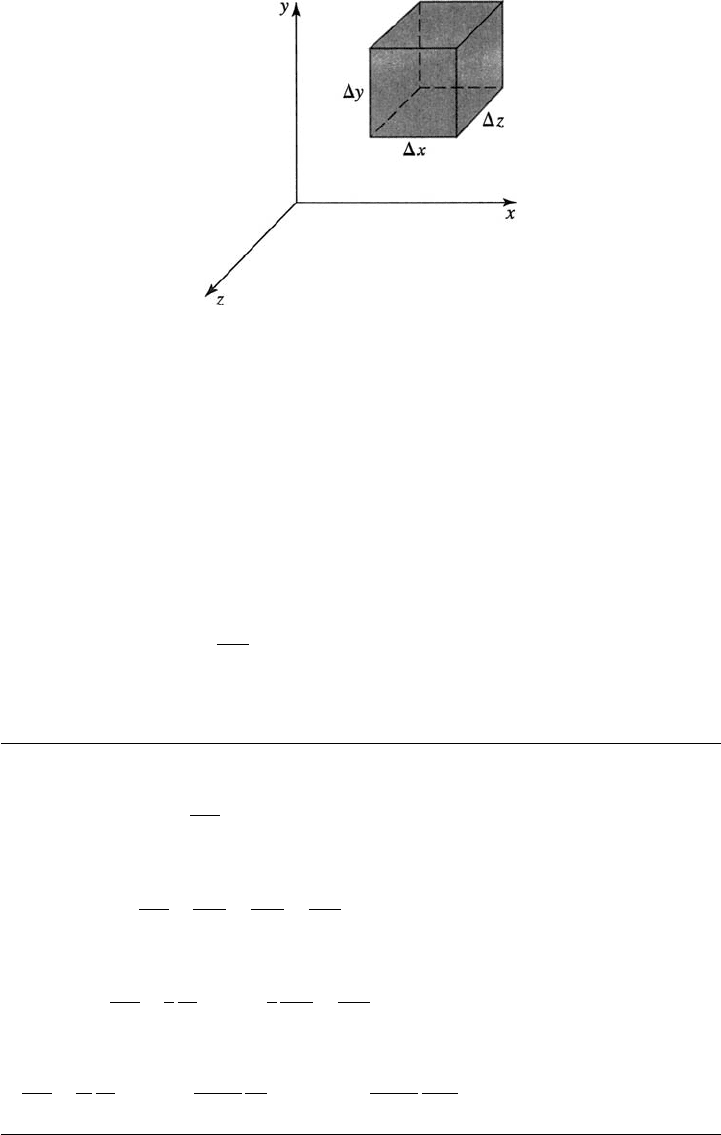
Consider a stationary volume element, Dx Dy Dz shown in Figure B.1, through
which species j flows and in which chemical reactions take place. Let J
jx
, J
jy
,
and J
jz
be the components of the local molar flux of species j, C
j
the local molar
concentration of species j, and (r
j
) the volume-based formation rate of species j
defined by Eq. 3.1.1a. We write a species balance over the element in terms of
the molar flux of species j through the six surfaces of the element; each bracket
corresponds to a term in Eq. 4.0.1:
(J
jx
)
x
Dy Dz þ (J
jy
)
y
Dx Dz þ (J
jz
)
z
Dx Dy
þ (r
j
)Dx Dy Dz
¼
(J
jx
)
xþDx
Dy Dz þ (J
jy
)
yþDy
Dx Dz þ (J
jz
)
zþDz
Dx Dy
hi
þ
d
dt
C
j
Dx Dy Dz
(B:1)
Dividing both sides by Dx Dy Dz and taking the limit, Dx ! 0, Dy ! 0, Dz ! 0,
we obtain
@C
j
@t
þ
@J
jx
@x
þ
@J
jy
@y
þ
@J
jz
@z
¼ (r
j
)(B:2)
In general, we can write Eq. B.2 in vector notation:
@C
j
@t
þrrrrrJ
j
¼ (r
j
)(B:3)
Principles of Chemical Reactor Analysis and Design, Second Edition. By Uzi Mann
Copyright # 2009 John Wiley & Sons, Inc.
465
where rrrrr is the divergence operator of the molar flux of species j. Equation B.3 is
commonly called the species continuity equation. It provides a relation between
the time variations in the species concentration at a fixed point, the local motion of
the species, and the rate the species is formed by chemical reactions. The species
continuity equations for cylindrical and spherical coordinates are given in Table B.1.
To describe the operation of a chemical reactor, we integrate the species
continuity equation over the reactor volume. Multiplying each term in Eq. B.3
by dV and integrating,
ð
V
R
@C
j
@t
dV þ
ð
V
R
(rrrrrJ
j
) dV ¼
ð
V
R
(r
j
) dV (B:4)
Figure B.1 Diagram of the molar flux components in a Cartesian element.
TABLE B.1 Species Continuity Equations
In general vector notation:
@C
j
@t
þrrrrrJ
j
¼ (r
j
)
(A)
For rectangular coordinates:
@C
j
@t
þ
@J
jx
@x
þ
@J
jy
@y
þ
@J
jz
@z
¼ (r
j
)
(B)
For cylindrical coordinates:
@C
j
@t
þ
1
r
@
@r
(rJ
j
r
) þ
1
r
@J
j
u
@u
þ
@J
j
z
@z
¼ (r
j
)
(C)
For spherical coordinates:
@C
j
@t
þ
1
r
2
@
@r
(r
2
J
jr
) þ
1
r sin u
@
@u
(J
ju
sin u) þ
1
r sin u
@J
jf
@f
¼ (r
j
)
(D)
466 APPENDIX B

The first term on the left-hand side reduces to
ð
V
R
@C
j
@t
dV ¼
dN
j
dt
where N
j
is the total number of moles of species j in the reactor. Applying Gauss’s
divergence theorem, the second term on the left-hand side reduces to
ð
V
R
(rrrrrJ
j
) dV ¼
ð
S
R
(J
j
n) dS ¼ F
j
out
F
j
in
where n is the outward unit vector on the boundaries of the reactor. This term
provides the net molar flow rate of species j through the boundaries of the reactor.
Thus, Eq. B.4 reduces to
dN
j
dt
þ F
j
out
F
j
in
¼
ð
V
R
(r
j
) dV (B:5)
which is the integral form of the general species-based design equation of a chemi-
cal reactor, written for species j, and is identical to Eq. 4.1.3.
APPENDIX B 467

APPENDIX C
SUMMARY OF NUMERICAL
DIFFERENTIATION AND
INTEGRATION
C.1 NUMERICAL DIFFERENTIATION
For equally spaced points, the first derivative of function f(x) is approximated (to
error order of Dx
2
) as follows:
Forward differentiation:
df
dx
i
¼
3f (x
i
) þ 4f (x
iþ1
) f (x
iþ2
)
2 Dx
(C:1)
Central differentiation:
df
dx
i
¼
f (x
iþ1
) f (x
i1
)
2 Dx
(C:2)
Backward differentiation:
df
dx
i
¼
3f (x
i
) 4f (x
i1
) þ f (x
i2
)
2 Dx
(C:3)
Principles of Chemical Reactor Analysis and Design, Second Edition. By Uzi Mann
Copyright # 2009 John Wiley & Sons, Inc.
469
