Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Symmetries
13
Point group symmetries: Choose any lattice point as the origin. Both rectangular
and centered rectangular lattices can be reflected about the x or y axes, and
each is invariant under a 180° rotation.
Because the rectangular and centered rectangular lattices share the same point
group symmetries, they are said to belong to the same crystal system, but they
are not the same lattice.
One might protest that the rectangular and centered rectangular lattices are ob-
viously different because their primitive vectors are different. However, making
this argument would lead one to conclude that two square lattices of different size
are different as well. The correct question to ask in determining if two lattices
are the same is whether one structure can be deformed continuously into the other
without losing symmetries along the way. For example, if one lattice is twice the
size of another, but otherwise the same, one would want to call them the same. Al-
though centered rectangular and rectangular lattices share point group symmetries,
they are different lattices and have different space groups. They are different be-
cause there exists no way to deform the first continuously into the second without
temporarily destroying some symmetries, as indicated in Figure 1.10.
Mirror plane.
..broken..
...and restored
Rectangular
^
Centered Rectangular
Figure 1.10. In deforming the rectangular lattice into the centered rectangular lattice,
reflection symmetry about the y axis is destroyed.
A more formal expression of this same idea uses the idea of a change of co-
ordinates. Suppose one has a first group of symmetry operations, G =
5i
+
a
and
a second group G'
—
Jl' + a'. Then the two groups are equivalent if there exists a
single matrix S (change of coordinates) such that
S~
l
3lS + S~
l
ä =
3i'
+ a.
(1.8)
In other words, G and G' must be the same up to linear changes of coordinate
systems. This definition of equivalent lattices is the same as the previous one,
because once one has the matrix S, then there is a family of matrices
S,
= (!-/)+»,
(1.9)

14 Chapter 1. The Idea of
Crystals
which varies smoothly between the unit matrix and S as t varies between 0 and
1.
It generates explicitly a smooth deformation of one lattice into the other while
preserving the group operations.
1.3.3 Role of Symmetry
Perhaps symmetry is more important for physicists to understand the world than it
is for the world
itself.
Most of
the
exact statements in physics result from symmetry
arguments, and often symmetry provides the only path to making any substantive
statement about complicated assemblies of matter. Its importance persists in con-
densed matter physics, although the discipline's domain includes disordered and
noisy systems. Helpful books on the formal theory of symmetry in physics include
Heine (1960) and Tinkham (1964).
Problems
1.
Honeycomb lattice:
(a) Verify that the honeycomb lattice described by Eq. (1.5) has properly been
constructed so that the distance between all neighboring points is identical.
(b) From Table 2.1, the lattice spacing of graphene is 2.46Â. Find the distance
between nearest neighbors (Figure 1.3).
(c) Find the density of graphene in gm/cm
2
.
2.
Hexagonal lattice:
(a) The hexagonal lattice may be viewed as a special case of
the
centered rectan-
gular lattice. Referring to Figure 1.4 and to the conventional unit cell depicted
there, find the ratio c/a for which the centered rectangular lattice would be-
come hexagonal.
(b) Enumerate the symmetries of the hexagonal lattice, and compare them with
the symmetries of the centered rectangular lattice.
3.
Nanotube structures: To form a single-walled nanotube, one rolls up a sin-
gle atomic layer of graphite, graphene, as shown in Figure 1.11. Referring
to Figure 1.11 (A) and (B), all nanotubes can be indexed with two integers m
and n where c = ma\ + nai and a\ and
a.2
are primitive vectors; one created
in this way is an (m, n)-nanotube.
(a) Are all structures labeled by distinct pairs of integers m, n
G
(—oo, oo)
dif-
ferent from each other?
(b) What are the indices for the nanotube appearing in (C)?
(c) Suppose that the atoms of a two-dimensional crystal lie at locations R, and
that the crystal is rolled into a cylinder by pulling together two atoms sepa-
rated by vector c as shown in Figure 1.11 (A). Write down an explicit expres-
sion for locations of atoms after the sheet has been rolled into a tube.

Problems 15
Figure 1.11. How to roll a sheet of graphene into a nanotube.
4.
Allowed symmetry axes:
(a) Consider a two-dimensional Bravais lattice that is left invariant after rotation
by angle
Θ
around the origin. Suppose the lattice to have points at (0, 0) and
(a, 0). By requiring the image of
(a,
0) under rotations through ±θ to be in the
Bravais lattice, find
a
simple expression that implicitly specifies all possible
rotation axes.
(b) Prove that the only allowed axes are twofold, threefold, fourfold, and sixfold.
In particular,
it
is impossible for a Bravais lattice to have
a
fivefold rotation
axis.
5.
Two-dimensional ground states: Portions of this problem are most easily
carried out with the aid
of a
computer algebra program or brief compiled
programs.
(a) Consider a collection of particles in two dimensions whose energy is
«w
where
^(r)
= <
Uoexp(-r)(^-l) if r
<
1.5
(U1)
I
0
else,
and r,j is the distance (measured, say, in Â) between particles
/
and
j.
Find
the crystal structure in Figure 1.4 which provides a minimum energy state for
this potential, and the equilibrium lattice spacing, assuming no particles are
at a distance less than 1. The potential has been chosen so that only nearest
neighbors interact in the ground state. Do not check all crystal structures
explicitly, only the square and hexagonal lattices.

16 Chapter 1. The Idea of
Crystals
(b) Suppose that φ is replaced by
4>(r)
= \
&>exp(-r)(—-1) ifr< 1.5
(L12
)
I
0
else.
Show that particles would collapse into
a
state of high density. It is not nec-
essary to perform sums numerically: Consider what happens when particles
are so closely spaced that they can be thought of as constituting a continuous
distribution.
(c) Taking for
φ
φ(ή
=
φο^χ
ν
(-ή(^-ή
(1.13)
and assuming that the ground state is a lattice of the same symmetry as in part
(a),
find the equilibrium lattice spacing and energy per particle within 10%.
This sum does need to be performed numerically.
References
J. Emsley (1998), The Elements, 3rd ed., Clarendon Press, Oxford.
H. Goldstein (1980), Classical Mechanics, 2nd ed., Addison-Wesley, Reading, MA.
P.
Groth (1906-1919), Chemische Krystallographie, W. Engelmann, Leipzig.
R. J. Haiiy (1801), Treatise on Minerology, Conseil des Mines, Paris.In French.
V. Heine (1960), Group Theory in Quantum Mechanics, Pergamon, New York.
J. C. Heyraud and J.
J.
Métois (1980), Establishment of the equilibrium shape of metal crystallites
on
a
foreign substrate: Gold on graphite, Journal of Crystal Growth, 50, 571-574.
S. G. Lipson (1987), Helium crystals, Contemporary Physics, 28, 117-142.
C. H. MacGillavry (1976), Fantasy & Symmetry: The Periodic Drawings of
M.
C. Escher, H.
N.
Abrams, New York.
J. B. Marion and S. T. Thornton (1988), Classical Dynamics of Particles and Systems, 3rd ed., Har-
court Brace Jovanovich, San Diego.
J. C. Meyer, A. K. Geim, M.
I.
Katsnelson, K. S. Novoselov, T. J. Booth, and S. Roth (2007), The
structure of suspended graphene sheets, Nature, 446,
60-63.
N.
Steno (1671), The Prodromus to
a
Dissertation Concerning Solids Naturally Contained Within
Solids: Laying
a
Foundation
for the Rendering
a
Rational Accompt Both of
the
Frame and the
Several Changes
of
the Masse
of
the Earth,
as
also
of
the Various Productions
in
the Same,
London, F. Winter. Translation of: De solido intra solidum naturaliter. Readex Microprint, 1973
(Landmarks of Science)
J. J. Thomson (1907), The Corpuscular Theory of Matter, Charles Scribner's Sons, New York.
M. Tinkham (1964), Group Theory and Quantum Mechanics, McGraw-Hill, New York.

2.
Three-Dimensional Lattices
2.1 Introduction
In order to classify the crystalline solids found in nature, one must study three-
dimensional lattices. While the subject involves few concepts not already present in
the two-dimensional case, the number of possible structures is bewilderingly large.
Fortunately, a large number of the elements adopts particularly simple structures.
On the other hand, alloys and compounds explore countless different forms, of
which over 400,000 have now been cataloged; these cannot be summarized in any
neat way despite the aid provided by the theory of symmetry.
There are 32 distinct point groups consistent with crystalline symmetry in
three dimensions, which were first enumerated by Hessel (1830), spurred on by
the desire to classify the shapes of naturally occurring rock crystals. The three-
dimensional Bravais lattices were first correctly enumerated by Bravais (1850);
solution of this problem may seem easier than that of finding all point groups, but
it is more abstract, since crystal surfaces are visible, while lattices are deduced as
an economical explanation for their appearance. Listing of lattices with bases was
begun by Sohncke (1879), who found 65 lattices. The full number is 230, and
these were enumerated by Fedorov (1895) and Schonflies (1891). Fedorov had
priority in most respects, but only following correspondence between the two sci-
entists were the final errors corrected; in one of Schönflies' early papers, 227 space
groups appear. Many other historical details are related by Ewald (1962), Phillips
(1971),
and Hoddeson et al. (1992).
This chapter is designed more for reference than for recreational reading. At
first the listing of crystal structures may appear more like a dull catalog of animals
in some distant land than basic physics. Yet knowledge of crystal structures is
the foundation on which much of the rest of condensed matter physics rests. The
detailed calculation of electronic and mechanical properties of solids depends on
knowing where the atoms lie.
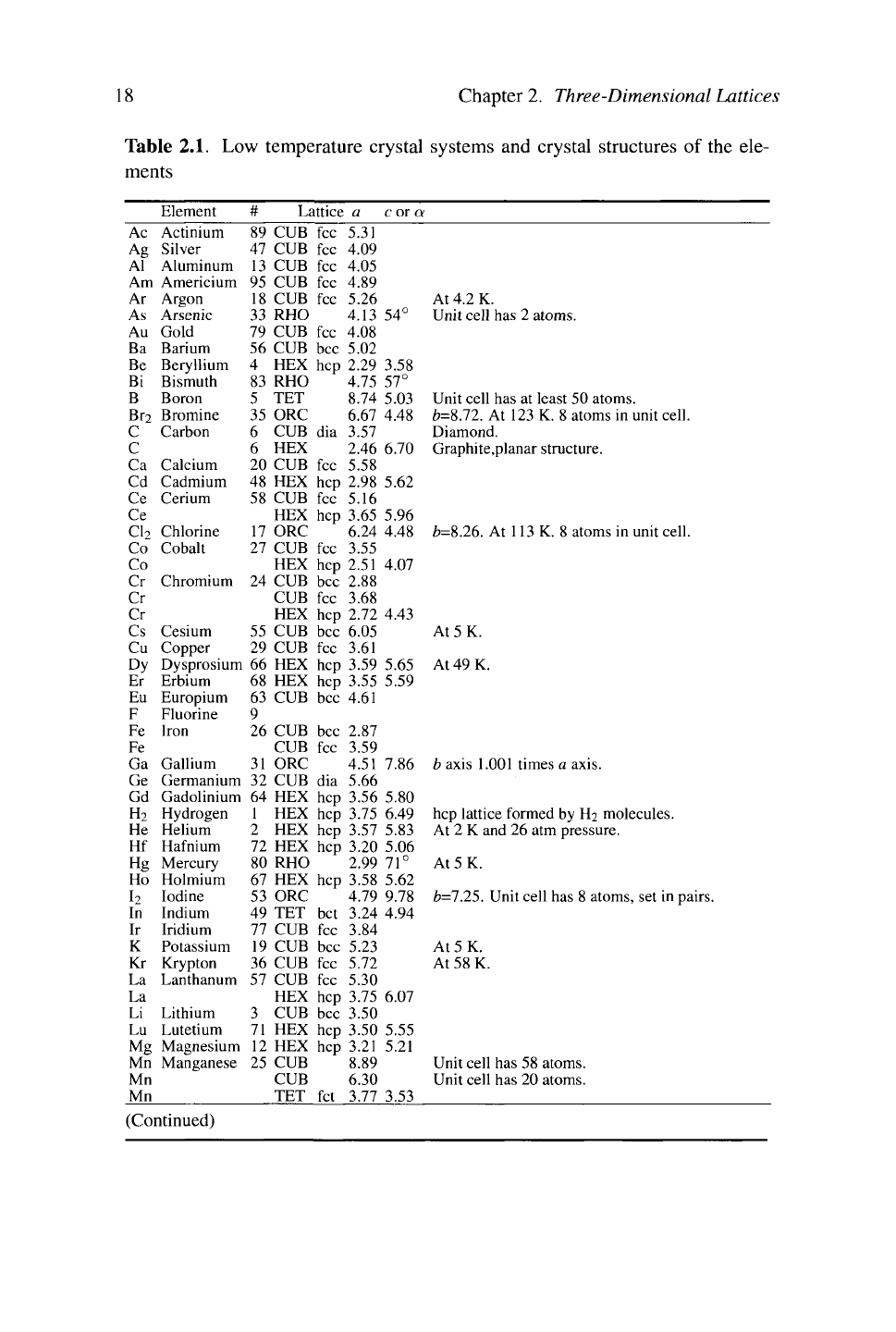
Distribution Among Elements.
A comprehensive account of everything known about crystal structures cannot
be confined to one volume, or ten, let alone a chapter. Still, it is worth giving a
sense of the types of information available, first the elements and then some of
the more common compound structures. The low-temperature crystal structures of
the elements are shown in Table 2.1. Room-temperature crystal structures of the
elements are shown in the periodic table inside the front cover. Not all structures
are known with certainty; boron has a huge unit cell that continues to resist precise
determination. The crystal structures mentioned in the chart and in the periodic
table are defined in the following sections.
17
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.
li
Chapter 2. Three-Dimensional Lattices
Table 2.1. Low temperature crystal systems and crystal structures of the ele-
ments
Element
Ac Actinium
Ag Silver
Al Aluminum
Am Americium
Ar Argon
As Arsenic
Au Gold
Ba Barium
Be Beryllium
Bi Bismuth
B Boron
Br2 Bromine
C Carbon
C
Ca Calcium
Cd Cadmium
Ce Cerium
Ce
Cl
2
Chlorine
Co Cobalt
Co
Cr Chromium
Cr
Cr
Cs Cesium
Cu Copper
Dy Dysprosium
Er Erbium
Eu Europium
F Fluorine
Fe Iron
Fe
Ga Gallium
Ge Germanium
Gd Gadolinium
H2 Hydrogen
He Helium
Hf Hafnium
Hg Mercury
Ho Holmium
I2 Iodine
In Indium
Ir Iridium
K Potassium
Kr Krypton
La Lanthanum
La
Li Lithium
Lu Lutetium
Mg Magnesium
Mn Manganese
Mn
Mn
# Lattice a c or a
89 CUB
47 CUB
13 CUB
95 CUB
18 CUB
33 RHO
79 CUB
56 CUB
4 HEX
83 RHO
5 TET
35 ORC
6 CUB
6 HEX
20 CUB
48 HEX
58 CUB
HEX
17 ORC
27 CUB
HEX
24 CUB
CUB
HEX
55 CUB
29 CUB
66 HEX
68 HEX
63 CUB
9
26 CUB
CUB
31 ORC
32 CUB
64 HEX
1 HEX
2 HEX
72 HEX
80 RHO
67 HEX
53 ORC
49 TET
77 CUB
19 CUB
36 CUB
57 CUB
HEX
3 CUB
71 HEX
12 HEX
25 CUB
CUB
TET
fee 5.31
fee 4.09
fee 4.05
fee 4.89
fee 5.26
4.13 54°
fee 4.08
bec 5.02
hep 2.29 3.58
4.75 57°
8.74 5.03
6.67 4.48
dia 3.57
2.46 6.70
fee 5.58
hep 2.98 5.62
fee 5.16
hep 3.65 5.96
6.24 4.48
fee 3.55
hep 2.51 4.07
bee 2.88
fee 3.68
hep 2.72 4.43
bec 6.05
fee 3.61
hep 3.59 5.65
hep 3.55 5.59
bec 4.61
bec 2.87
fee 3.59
4.51 7.86
dia 5.66
hep 3.56 5.80
hep 3.75 6.49
hep 3.57 5.83
hep 3.20 5.06
2.99 71°
hep 3.58 5.62
4.79 9.78
bet 3.24 4.94
fee 3.84
bec 5.23
fee 5.72
fee 5.30
hep 3.75 6.07
bec 3.50
hep 3.50 5.55
hep 3.21 5.21
8.89
6.30
fct 3.77 3.53
At 4.2 K.
Unit cell has 2 atoms.
Unit cell has at least 50 atoms.
6=8.72.
At 123 K. 8 atoms in unit cell.
Diamond.
Graphite.planar structure.
£=8.26.
At 113 K. 8 atoms in unit cell.
At5K.
At 49 K.
b axis 1.001 times a axis.
hep lattice formed by H2 molecules.
At 2 K and 26 atm pressure.
At5K.
b=l.25. Unit cell has 8 atoms, set in pairs.
At5K.
At 58 K.
Unit cell has 58 atoms.
Unit cell has 20 atoms.
(Continued)
Introduction
19
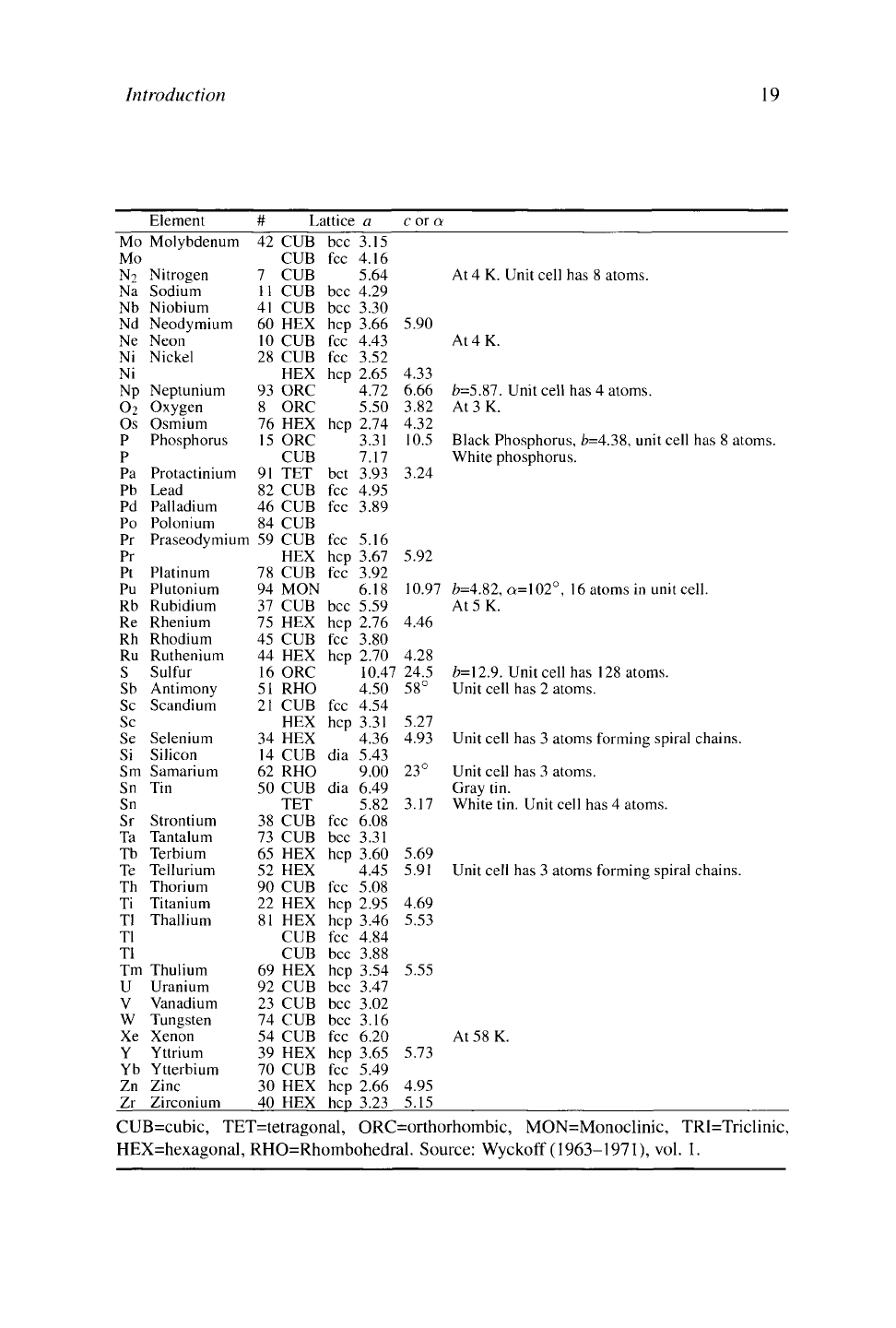
Element
Mo Molybdenum
Mo
N2 Nitrogen
Na Sodium
Nb Niobium
Nd Neodymium
Ne Neon
Ni Nickel
Ni
Np Neptunium
O2 Oxygen
Os Osmium
P Phosphorus
P
Pa Protactinium
Pb Lead
Pd Palladium
Po Polonium
Pr Praseodymiurr
Pr
Pt Platinum
Pu Plutonium
Rb Rubidium
Re Rhenium
Rh Rhodium
Ru Ruthenium
S Sulfur
Sb Antimony
Sc Scandium
Sc
Se Selenium
Si Silicon
Sm Samarium
Sn
Tin
Sn
Sr Strontium
Ta Tantalum
Tb Terbium
Te Tellurium
Th Thorium
Ti Titanium
TI Thallium
Tl
Tl
Tm Thulium
U Uranium
V Vanadium
W Tungsten
Xe Xenon
Y Yttrium
Yb Ytterbium
Zn Zinc
Zr Zirconium
# Lattice
42
CUB
CUB
7
CUB
11
CUB
41
CUB
60
HEX
10
CUB
28
CUB
HEX
93
ORC
8
ORC
76
HEX
15
ORC
CUB
91
TET
82
CUB
46
CUB
84
CUB
1
59 CUB
HEX
78
CUB
94
MON
37
CUB
75
HEX
45
CUB
44
HEX
16
ORC
51
RHO
21
CUB
HEX
34
HEX
14
CUB
62
RHO
50
CUB
TET
38
CUB
73
CUB
65
HEX
52
HEX
90
CUB
22
HEX
81
HEX
CUB
CUB
69
HEX
92
CUB
23
CUB
74
CUB
54
CUB
39
HEX
70
CUB
30
HEX
40
HEX
bcc
fee
bcc
bcc
hep
fee
fee
hep
hep
bet
fee
fee
fee
hep
fee
bcc
hep
fee
hep
fee
hep
dia
dia
fee
bcc
hep
fee
hep
hep
fee
bcc
hep
bcc
bcc
bcc
fee
hep
fee
hep
hep
a
3.15
4.16
5.64
4.29
3.30
3.66
4.43
3.52
2.65
4.72
5.50
2.74
3.31
7.17
3.93
4.95
3.89
5.16
3.67
3.92
6.18
5.59
2.76
3.80
2.70
c or
a
5.90
4.33
6.66
3.82
4.32
10.5
3.24
5.92
10.97
4.46
4.28
10.47
24.5
4.50
4.54
3.31
4.36
5.43
9.00
6.49
5.82
6.08
3.31
3.60
4.45
5.08
2.95
3.46
4.84
3.88
3.54
3.47
3.02
3.16
6.20
3.65
5.49
2.66
3.23
58°
5.27
4.93
23°
3.17
5.69
5.91
4.69
5.53
5.55
5.73
4.95
5.15
At
4
K. Unit cell
has 8
atoms.
At4K.
6=5.87.
Unit cell has
4
atoms.
At3K.
Black Phosphorus, 6=4.38, unit cell
has 8
atoms.
White phosphorus.
6=4.82,
a=102°, 16 atoms
in
unit cell.
At5K.
6=12.9.
Unit cell
has
128 atoms.
Unit cell has
2
atoms.
Unit cell has
3
atoms forming spiral chains.
Unit cell
has 3
atoms.
Gray
tin.
White tin. Unit cell has
4
atoms.
Unit cell
has 3
atoms forming spiral chains.
At 58
K.
CUB=cubic, TET=tetragonal, ORC=orthorhombic, MON=Monoclinic, TRI=Triclinic,
HEX=hexagonal, RHO=Rhombohedral. Source: Wyckoff (1963-1971), vol. 1.
20
Chapter 2. Three-Dimensional Lattices
2.2 Monatomic Lattices
Monatomic lattices are those formed from entirely from atoms of a single element.
In the simplest of these, the atoms form a Bravais lattice. This means that atomic
positions are given by sums over three primitive vectors, which must be linearly
independent:
R = n\a\ + «2^2 +
«3«3·
"i- "2·
and
"3
are
inte
g
ers
-
The
subscripts on the (2.1 )
vectors a, do not refer to components of the vec-
tors,
but to three separate three-vectors.
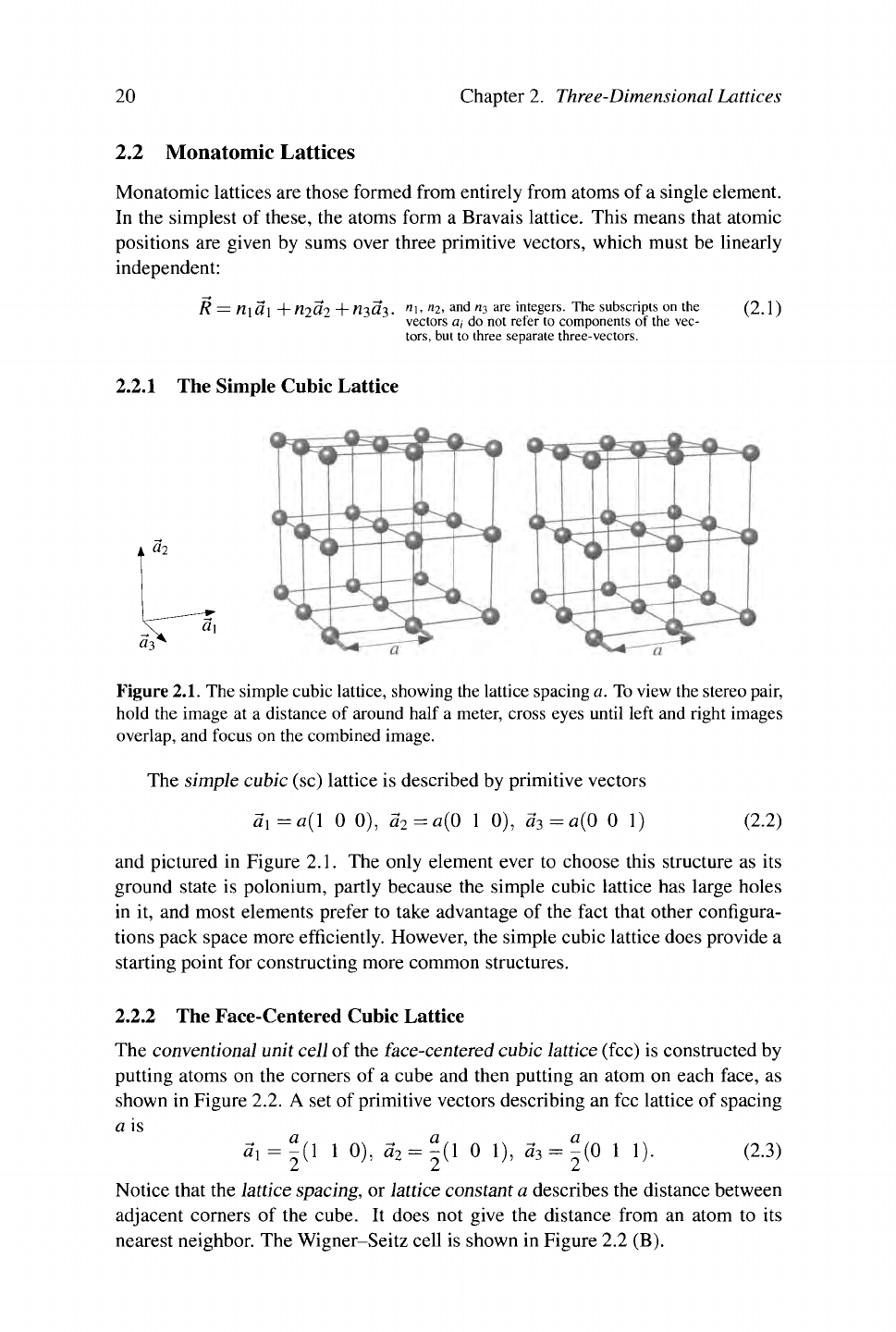
2.2.1 The Simple Cubic Lattice
Figure 2.1. The simple cubic lattice, showing the lattice spacing a. To view the stereo pair,
hold the image at a distance of around half a meter, cross eyes until left and right images
overlap, and focus on the combined image.
The simple cubic (sc) lattice is described by primitive vectors
a\=a{\
0 0), a
2
= a(0 1 0), α
3
= α(0 0 I) (2.2)
and pictured in Figure 2.1. The only element ever to choose this structure as its
ground state is polonium, partly because the simple cubic lattice has large holes
in it, and most elements prefer to take advantage of the fact that other configura-
tions pack space more efficiently. However, the simple cubic lattice does provide a
starting point for constructing more common structures.
2.2.2 The Face-Centered Cubic Lattice
The conventional unit cell of the face-centered cubic lattice (fee) is constructed by
putting atoms on the corners of a cube and then putting an atom on each face, as
shown in Figure 2.2. A set of primitive vectors describing an fee lattice of spacing
a is
2i = |(l 1 0), 3
2
= |(1 0 1), 3
3
= |(0 1 1). (2.3)
Notice that the lattice spacing, or lattice constant a describes the distance between
adjacent corners of the cube. It does not give the distance from an atom to its
nearest neighbor. The Wigner-Seitz cell is shown in Figure 2.2 (B).
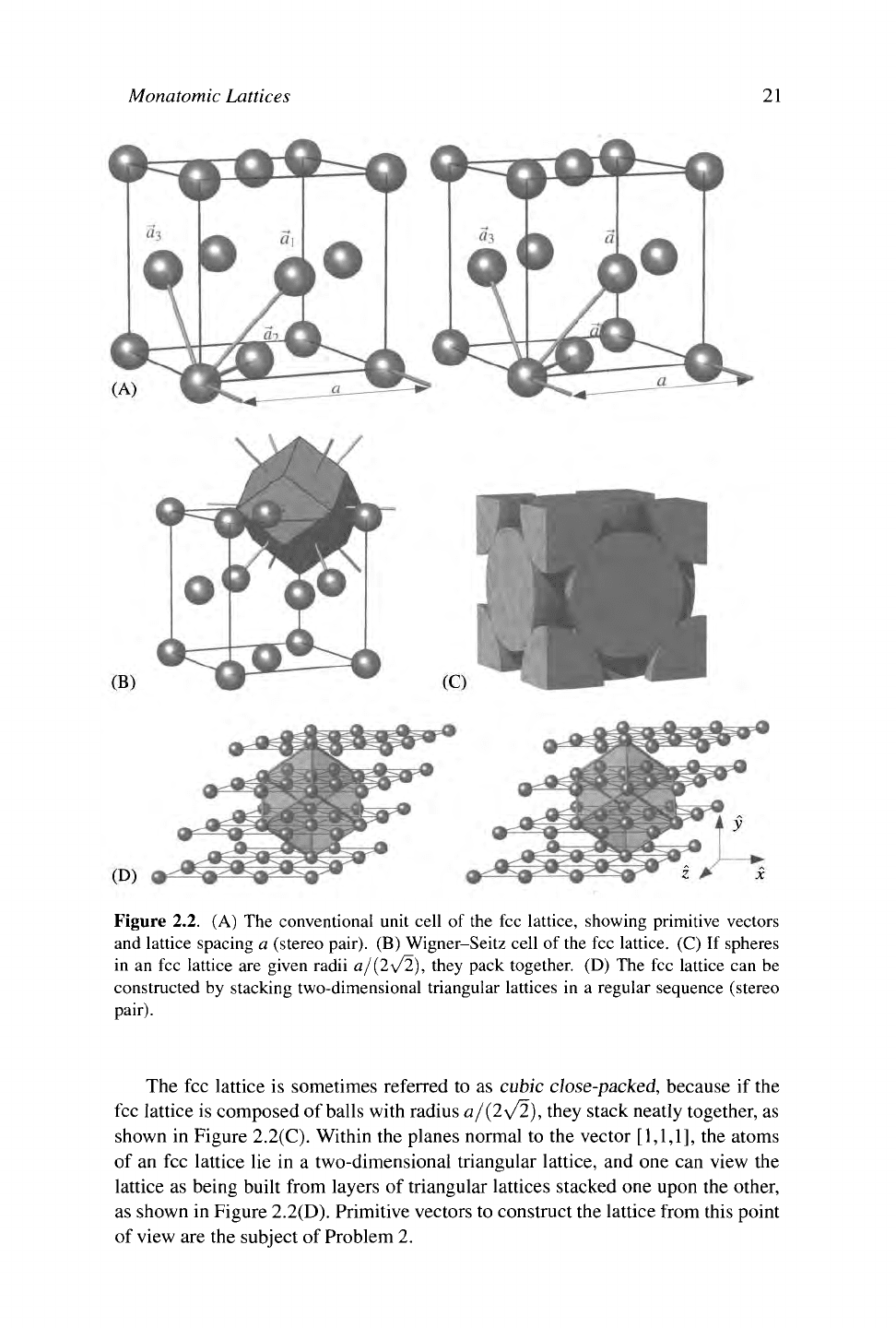
Monatomic Lattices
21
Figure 2.2. (A) The conventional unit cell of the fee lattice, showing primitive vectors
and lattice spacing a (stereo pair). (B) Wigner-Seitz cell of the fee lattice. (C) If spheres
in an fee lattice are given radii a/(2\/2), they pack together. (D) The fee lattice can be
constructed by stacking two-dimensional triangular lattices in a regular sequence (stereo
pair).
The fee lattice is sometimes referred to as cubic close-packed, because if the
fee lattice is composed of balls with radius a/(2\/2), they stack neatly together, as
shown in Figure 2.2(C). Within the planes normal to the vector
[1,1,1],
the atoms
of an fee lattice lie in a two-dimensional triangular lattice, and one can view the
lattice as being built from layers of triangular lattices stacked one upon the other,
as shown in Figure 2.2(D). Primitive vectors to construct the lattice from this point
of view are the subject of Problem 2.
22
Chapter 2. Three-Dimensional Lattices
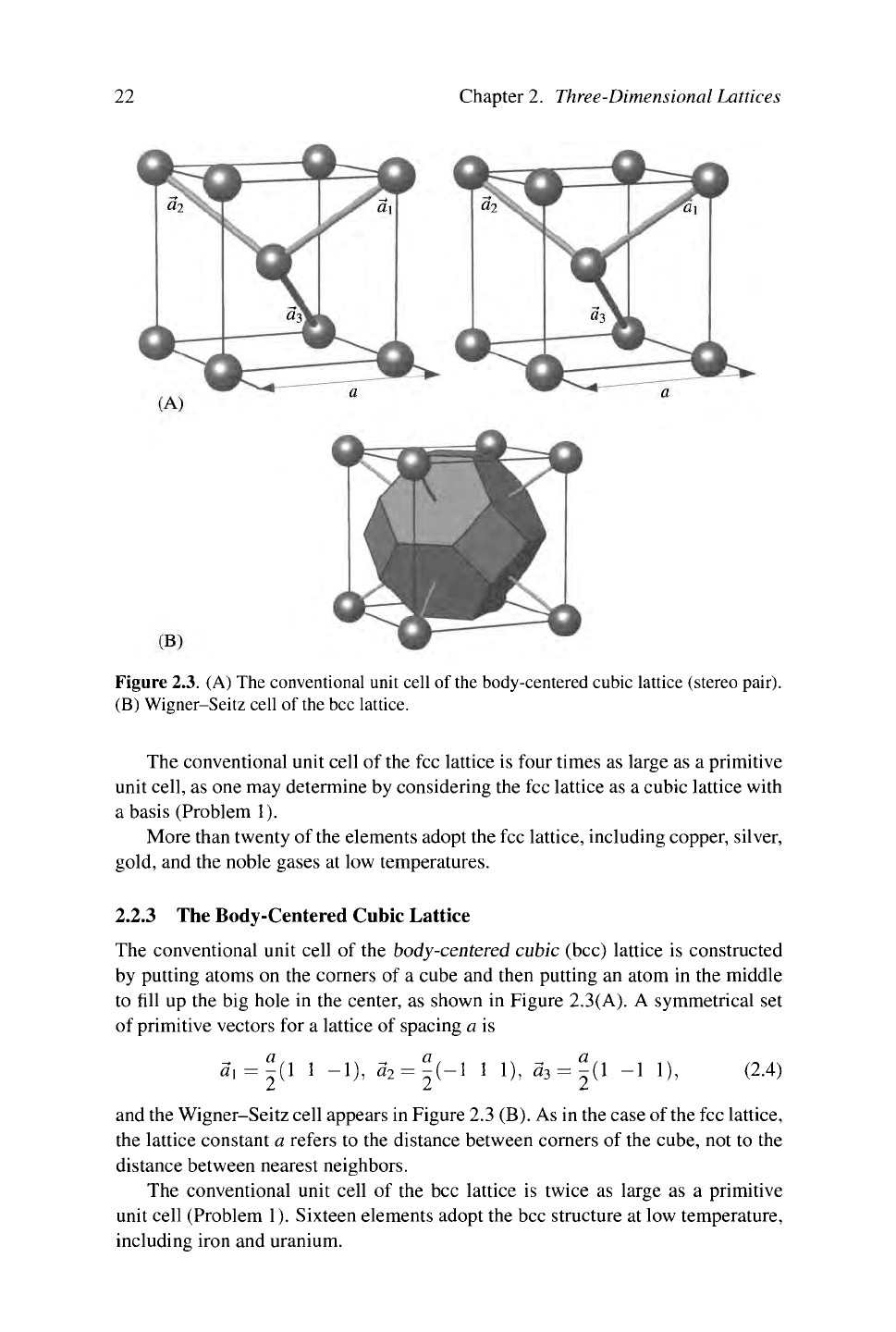
Figure 2.3. (A) The conventional unit cell of the body-centered cubic lattice (stereo pair).
(B) Wigner-Seitz cell of the bcc lattice.
The conventional unit cell of the fee lattice is four times as large as a primitive
unit cell, as one may determine by considering the fee lattice as a cubic lattice with
a basis (Problem 1).
More than twenty of the elements adopt the fee lattice, including copper, silver,
gold, and the noble gases at low temperatures.
2.2.3 The Body-Centered Cubic Lattice
The conventional unit cell of the body-centered cubic (bcc) lattice is constructed
by putting atoms on the corners of a cube and then putting an atom in the middle
to fill up the big hole in the center, as shown in Figure 2.3(A). A symmetrical set
of primitive vectors for a lattice of spacing a is
«i = |(1 1 -1), «2 = |(-1 1 1), «3 = |(1 "I 1), (2-4)
and the Wigner-Seitz cell appears in Figure 2.3 (B). As in the case of the fee lattice,
the lattice constant a refers to the distance between corners of the cube, not to the
distance between nearest neighbors.
The conventional unit cell of the bcc lattice is twice as large as a primitive
unit cell (Problem 1). Sixteen elements adopt the bcc structure at low temperature,
including iron and uranium.
