Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


3.
Scattering and Structures
3.1 Introduction
The scattering of X-rays from crystals provided information on the locations of
atoms within solids that had been the object of speculation for centuries, and it
rapidly led to a huge scientific enterprise of structure determination, but before the
first careful experiments of 1912 it was far from obvious that anything could come
of it at all.
M. von Laue was struck in 1912 by the intuition that X-rays might scatter
off crystals in the way that ordinary light scatters off a diffraction grating. This
hunch preceded any mathematical attempt to quantify the size and character of the
effect, and if it seems obvious in retrospect, one might keep in mind that neither
the periodic character of crystals nor the wave nature of X-rays was known with
certainty at the time. Laue discussed
his idea with colleagues Sommerfeld, Wien and others with the result of
encountering a strong disbelief in a significant outcome of any diffraction
experiment based upon the regularity of the internal structure of crystals.
It was argued that the inevitable temperature motion of the atoms would
impair the regularity of the grating to such an extent that no pronounced
diffraction maxima could be expected. —Ewald (1962), p. 42
This argument against diffraction effects was in fact quite reasonable. Consider,
for example, the prospect of scattering off
NaCl.
The study of elastic deformations
was well developed in 1912, and salt crystals were known to be anisotropie, but
characterized approximately by Young's modulus Y (defined in Section 12.3.2) of
5-10
11
ergs cm
-3
. The chlorine atom was known to have a mass of 35 g mole
-1
,
and sodium 23 g mole
-1
; from salt's known density of 4.29 g cm~
3
, a charac-
teristic spacing of d = 2.5 · 10
-8
cm between sodium and chlorine could be de-
termined. To account for the observed Young modulus, one would need a spring
constant between atoms on the order of % = Yd =10
4
dyne cm
-1
. Making use
of the equipartition theorem, the characteristic excursion of atoms due to thermal
fluctuations should therefore be expected to be x =
^/2kßT/%
ss 2
·
IO
-9
cm. This
distance is fortunately smaller than the interatomic spacing; it has to be, otherwise
the crystals would melt. However, the best estimate for the wavelength of X-rays
based upon diffraction around small slits was also of order 10~
9
cm. A diffraction
grating whose vibrations are the same size as the incoming waves hardly seemed
to pose a promising experiment, and Sommerfeld was reluctant to devote resources
or personnel. The work was carried out by Friedrich et al. (1912) with equipment
43
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.

44
Chapter 3. Scattering and Structures
pirated from elsewhere; it successfully produced diffraction spots from copper sul-
fate on only the second attempt, the first having failed when photographic plates
were placed in front of the crystal in the mistaken expectation that reflected X-rays
would produce the largest effect. Laue's mathematical theory for the directions of
X-ray maxima followed only a few hours later. At first the theory did not encom-
pass the possibility of a lattice with a basis, with the result that some predicted
spots were mysteriously absent from the experiment.
There was some question, at first, about whether the spots observed on the
photographic plate were truly due to diffraction from the crystal. The doubts were
largely dispelled by a series of experiments which seemed to rule out any other
possibility. First the crystalline sample was removed, and it was checked that the
diffraction pattern disappeared. Second, the crystal was replaced by a powdered
sample of the same material, and again distinct spots disappeared. Finally, the ori-
entation of the crystal was altered slightly, and the spots were observed slightly
displaced on the photographic plate. Several years were to pass before it could cor-
rectly be explained why thermal agitation did not obliterate the X-ray interference,
but by summer of 1912 the phenomenon was well on its way to wide acceptance
throughout Germany.
The discovery of X-ray diffraction contains many elements that have been ab-
solutely characteristic of important developments in condensed matter physics.
The enterprise began with a theoretical notion to which, however, there were com-
pelling theoretical objections. The experiment rapidly encountered a highly regular
set of phenomena with which theory agreed only uneasily in its first incarnation.
A rapid simultaneous development of theory and experiment in concert then pro-
duced a new set of concepts and a powerful new experimental tool.
3.2 Theory of Scattering from Crystals
3.2.1 Special Conditions for Scattering
X-rays created a world-wide sensation at the end of the nineteenth century because
they could produce images of the interior of the human body. This means that
they can travel through several centimeters of solid matter, attenuating weakly, and
come out the other side. When X-rays travel through crystals this is usually what
happens.
However, there is an important exception. When the wavelength of the X-
rays is chosen precisely right, and simultaneously the orientation of the crystal is
precisely right, there is constructive interference between the waves scattered by
successive atoms. Intense narrow beams of X-rays emerge from the crystal in a
finite number of special directions. Friedrich, Knipping, and von Laue were not
so lucky as to stumble exactly upon the frequency of radiation needed to create
the effect. Their X-rays contained a broad spectrum of radiation, and happened
to contain the special waves (Section 3.3.1). The same considerations apply to
neutrons and electrons, two other types of waves that can reveal atomic positions
in condensed matter. Their interaction with matter is mainly linear, so it is enough
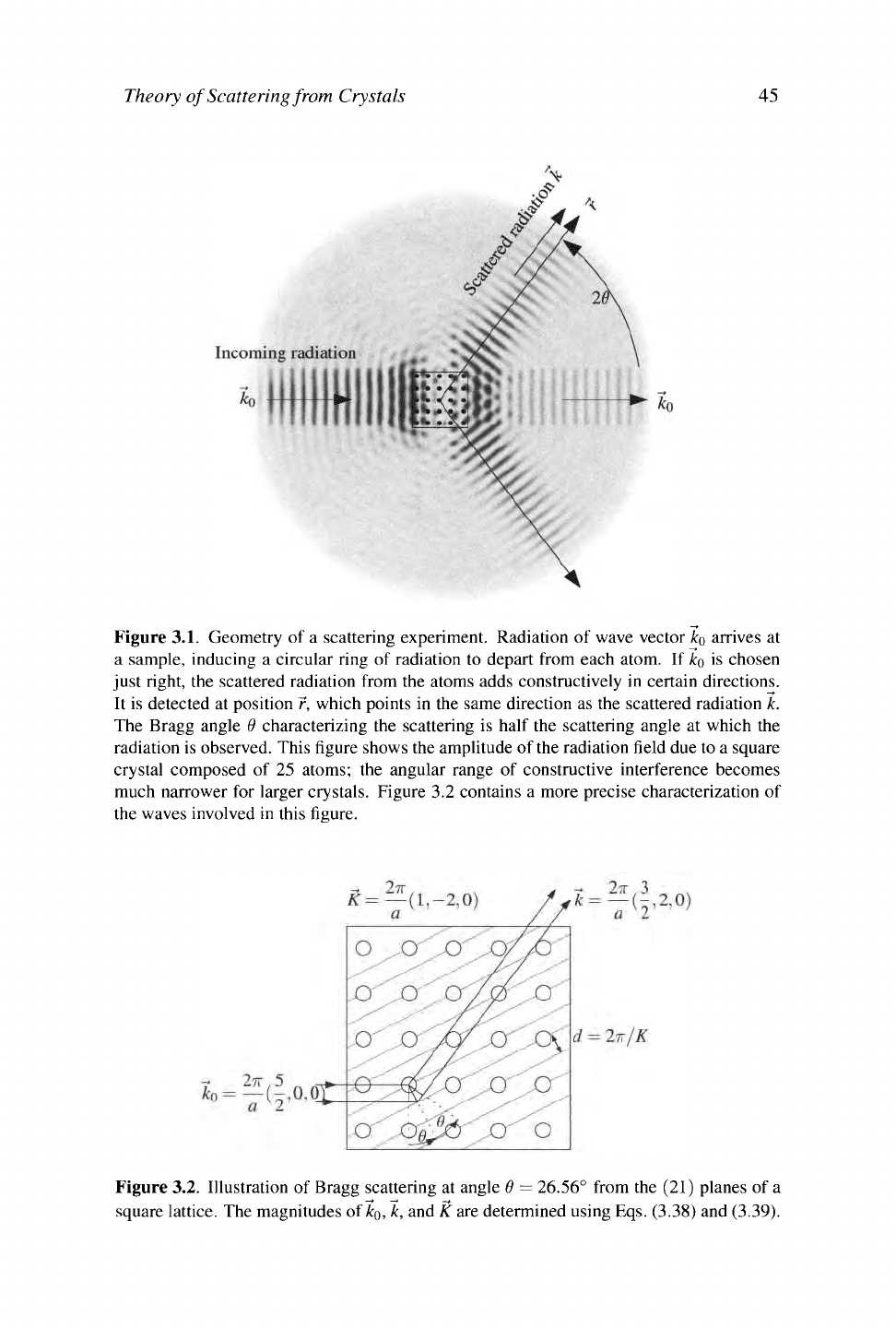
Theory of Scattering from Crystals 45
Figure 3.1. Geometry of a scattering experiment. Radiation of wave vector
ko
arrives at
a sample, inducing a circular ring of radiation to depart from each atom. If
ko
is chosen
just right, the scattered radiation from the atoms adds constructively in certain directions.
It is detected at position r, which points in the same direction as the scattered radiation k.
The Bragg angle
Θ
characterizing the scattering is half the scattering angle at which the
radiation is observed. This figure shows the amplitude of the radiation field due to a square
crystal composed of 25 atoms; the angular range of constructive interference becomes
much narrower for larger crystals. Figure 3.2 contains a more precise characterization of
the waves involved in this figure.
Figure 3.2. Illustration of Bragg scattering at angle
Θ —
26.56° from the (21) planes of a
square lattice. The magnitudes of
ko,
k, and K are determined using Eqs. (3.38) and (3.39).

46
Chapter 3. Scattering and Structures
to pick
a
single incoming plane wave, study what happens, and build a theory of
more elaborate scattering experiments by superposition.
3.2.2 Elastic Scattering from Single Atom
In a scattering experiment, a plane wave moves into a sample of condensed matter,
travels through it, interacts, comes out, and is measured by detectors far away.
In
the simplest class of scattering experiments, the frequency of the outgoing radiation
is the same as that of the radiation sent in, and the scattering is called elastic. This
phenomenon is most easily explained by invoking wave-particle duality to view
the incoming radiation as photons, neutrons, or electrons that bounce without any
change of energy off atoms in the sample.
Begin by considering a plane wave that collides with a single atom sitting at the
origin and scatters off it. Whether the incoming wave is a neutron or an electron
described by quantum mechanics, or an X-ray described by classical electromag-
netism, the scattered wave ψ takes a particularly simple form far from the atom:
Jkor
ψ « Ae~
,u
"[e'
k
°'
r
+ f(f) 1,
See, for example, Schiff (1968) p. 115 for the
(3.1)
r
'
quantum mechanical version, or Jackson (1999)
Eq. (9.8) for the electromagnetic version.
where the particle enters as a plane wave along ko, the scattering is measured at
a distance
r
that is much larger than the range
of
interaction with the atom,
at
angle
2Θ
relative to the
ko
axis, and
/
is a form factor containing details about the
interaction between the scattering potential and the scattered wave. It is related to
the differential scattering cross-section of the atom by
/.
= =
|f(r)| The intensity of scattered radiation into solid
(3.2)
dÇl atom angle
dii
at distance
r
relative to the incom-
ing beam is dfi
X
da/dQ/r
2
.
Forms for
/
will be given in Section 3.4, but for the moment assume
/
to be known.
Next ask how Eq. (3.1) changes when the same incoming plane wave scatters
off a particle located at R rather than at the origin. In this case, when the radiation
arrives at point R, it has phase exp(i7?
· ko)
relative to what it would have had at the
origin. On the other hand, to reach the observation point, it then has only to travel
a distance
\r —
R\
rather than
r
as previously. Therefore
-
-, - _ -
e
ikj\r—R\
ψ
~
Ae~
iu}>
e
ik(yR
[e
iko
'^~
R
"> +/(r) —1. The leading exponential is
(3.3)
\f
ft\
unchanged from Eq. (3.1).
For sufficiently large r, one may use the approximation
ko\r-R\~k
0
r-ko--R.
(3.4)
r
Using Eq. (3.4) and defining

Theory of Scattering from Crystals
47
- r
k =
KQ —,
k points in the observation direction (3.5)
r r and has the same magnitude as
k(>.
and
q =
k.Q
—
k (3.6)
gives
- _ g'kor+il'R In the denominator of
Eq.
(3.3) one needs only the
ψ ~
Ae~
lu
"
[e °'
r
+ /(f) ]. first term on the right side of Eq. (3.4). However, (3.7)
f within the exponential function, one has to keep
all terms that are large compared to 2π, and re-
quires both terms on the right side of Eq. (3.4).
Note that
n = 2kn sin Θ. Square both sides of
Eq.
(3.6) and use the fact that (3.8)
ko-k =
£Q
cos
2Θ.
The quantity hq describes the momentum
transfer
between the incoming and out-
going waves, and the angle
Θ
is called the Bragg angle.
3.2.3 Wave Scattering from Many Atoms
When one has a large assembly of scatterers, shown in Figures 3.1 and 3.2, the
angular dependence of the scattered radiation is the product of two pieces. The
first results from the fact that each individual scatterer emits radiation with differ-
ent intensities in different directions, described by the form factor f. The second,
which modulates the first, results from interference between the radiation com-
ing from the various objects and therefore contains information about their spatial
correlation.
So consider a large collection of scatterers, placing the origin somewhere in
the middle of them, and observe the scattering from far away. If the scatterers
are dilute, then the total scattered radiation due to the collection will simply be a
sum of the radiation due to each one. Ignoring the contributions that arise when
the light which has scattered off one atom then scatters off of another one before
being observed (multiple
scattering),
as well as ignoring contributions arising from
changes in the state of the scatterer (inelastic
scattering
), one has
ψ-Αβ-^'Ιβ^
+
ΣΜϊ)
e
ik
0
r+iq-R,
Ί
Summing many terms of the sort that arise in (3 9)
Eq.(3.7).
The largest contribution to ψ comes from the incoming beam exp
[iko ·
r],
which also
exits the sample as shown in Figure
3.1.
However this bright spot is restricted to an
outgoing angle of
Θ
= 0, so with the understanding that one will restrict attention
to nonzero scattering angles it is possible to drop the first term on the right-hand
side of
Eq.
(3.9). The intensity per unit solid angle divided by the intensity \A\
2
of
the incoming beam, / =
\ip\
2
r
2
/A
2
is then
Σ
~ (R R )
ReCa
"
that
' Σι
C
'l
2 =
Σ//'
C
'
C
P-
The
functions /,
fitfe'W·-"'''. still depend upon the scattering direction r, but the de- (3.10)
. ., pendence is not made explicit.

48
Chapter 3. Scattering and Structures
This basic equation for weak elastic scattering applies to scattering off matter in all
forms.
The equation is posed in terms of intensity rather than amplitude because
intensity is the quantity measured by experimental instruments.
3.2.4 Lattice Sums
Now ask what happens when all of the scatterers in the sum (3.10) are identical
and arranged in a Bravais lattice. In this case, the scattering intensity is equal to
/ = /
atom
V^
e
'Q(
R
i-
R
i'\ /
alom
is the scattering from a single atom, de- (3.11)
^—' fined in Eq. (3.2).
Inspection of Eq. (3.11) leads to a plausible guess for the wave vectors q that
will produce strong scattering. If one could choose q so that exp(iq-R) =
1
for all
R,
then all terms in Eq. (3.11) would be one, and summing them would produce a
large final result. Otherwise, one might expect terms in Eq. (3.11) to alternate in
sign and to cancel out on average.
This guess is correct, but to justify it one must understand sums of the type
appearing in Eq. (3.11). Such sums determine the behavior of
any
waves interacting
with a periodic lattice; they are important not only for X-ray scattering, but also for
conduction electrons in lattices (Chapter 7). The mathematics of the sum is best
explained in one dimension, and then generalized.
One-Dimensional Sum. In one dimension, lattice points must be of the form
la, where / is an integer and a is the interparticle spacing. So the relevant sum in
Eq. (3.11) becomes
Y>
q
=
Y^e
ilaq
.
(3.12)
/=o
This sum is calculated in Appendix A, which yields the results
JNaq _ i
Σ
«
=
7^τ
(3
·
13)
2
ÙÎNaqjl
sin aq/2
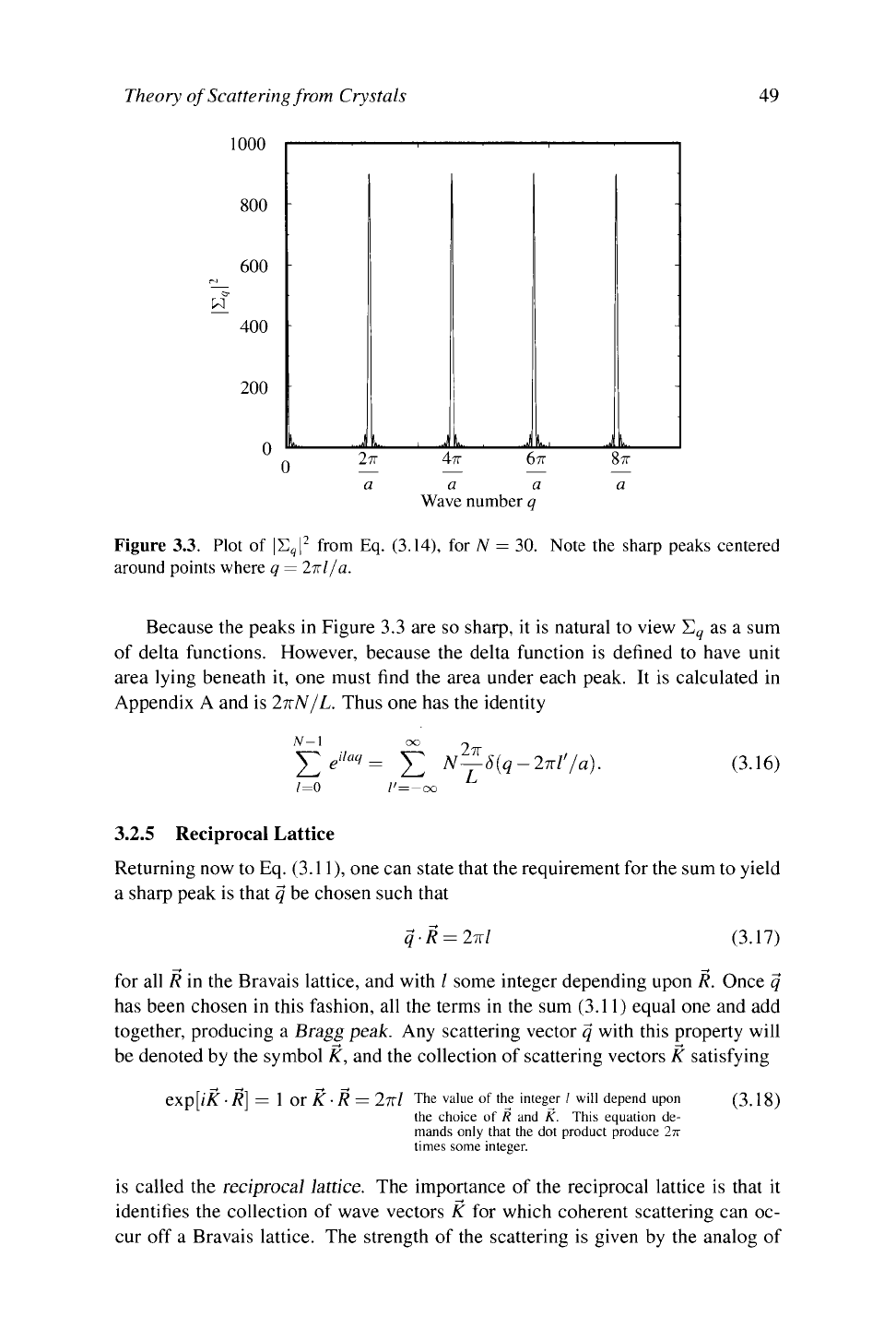
A plot of Eq. (3.14) appears in Figure 3.3. The graph contains a number of very
sharp identical peaks, separated by regions where the scattering intensity is nearly
zero.
The locations of
the
peaks are determined by searching for the points at which
the denominator of Eq. (3.14) vanishes. They occur whenever
aq/2 = Ιπ
=>■
q = 2πΙ/α.
/ is an
integer.
(3.15)
Glancing back at Eq. (3.12), one sees that peaks in the sum Σ
9
correspond exactly
to the choices for q such that all terms in the sum (3.12) equal 1 and thus add
coherently. For any other choice of q, the terms in the sum add with different
phases and signs, giving a much smaller result.
Theory of Scattering from Crystals
49
JUL
, il , IL· il
2π 4π 6π 8π
a a a a
Wave number q
Figure 3.3. Plot of |Σ
9
|
2
from Eq. (3.14), for N = 30. Note the sharp peaks centered
around points where q = 2πΙ/α.
Because the peaks in Figure 3.3 are so sharp, it is natural to view Y,
q
as a sum
of delta functions. However, because the delta function is defined to have unit
area lying beneath it, one must find the area under each peak. It is calculated in
Appendix A and is 2-KN/L. Thus one has the identity
N—Ì
OO n.
Y^
e
n
aq=
J2 Ν^δ(ς-2πΙ'/α).
(3.16)
/=0 /' = -oo
3.2.5 Reciprocal Lattice
Returning now to Eq. (3.11), one can state that the requirement for the sum to yield
a sharp peak is that q be chosen such that
q-R = 2nl (3.17)
for all R in the Bravais lattice, and with / some integer depending upon R. Once q
has been chosen in this fashion, all the terms in the sum (3.11) equal one and add
together, producing a Bragg peak. Any scattering vector q with this property will
be denoted by the symbol K, and the collection of scattering vectors K satisfying
C\p[iK R] =
\OTKR
= 2πΙ The value of the integer / will depend upon (3.18)
the
choice of R and K. This equation de-
mands
only that the dot product produce 2π
times
some integer.
is called the reciprocal lattice. The importance of the reciprocal lattice is that it
identifies the collection of wave vectors K for which coherent scattering can oc-
cur off a Bravais lattice. The strength of the scattering is given by the analog of
w
1UUU
800
600
400
200

50 Chapter 3. Scattering and Structures
Eq. (3.16), which according to Eq. (A.25) is
R K
where V is the volume of the system.
Bragg Planes. Why should there be any K that have property (3.18), and why do
they form a lattice? First of all, notice that if one finds any two K that have this
property, then their sum has it. Second of all, suppose that one has found a vector
K so that
KR = 0 (3.20)
for at least three noncollinear Bravais lattice points R. The set of all r that satisfy
K
■
r = 0 is a plane passing through the origin, so the R that satisfy Eq. (3.20) must
lie in such a plane
1Ό
(Figure 3.4), and K must be the normal to that plane. Thus the
direction of every K corresponds to a plane containing points in the Bravais lattice.
However, it is not enough to guarantee that K
■
R = 0 for R lying on the plane
7Ό;
one needs Eq. (3.18) to hold for all R. To achieve this goal, it is essential to note
that the whole Bravais lattice can always be constructed by taking the points that
lie in the plane
CPo,
all of which are of the form
ΙχΟ,-ι
-\-ljfl2 Having chosen a plane, one can then find two (3.21)
primitive vectors that span it.
and stacking multiple copies of this plane next to one other, separated by a third
primitive vector
a-},
as shown in Figure 3.4. There is an infinite number of ways to
decompose a crystal in this fashion into Bragg planes.
Figure 3.4. Any Bravais lattice can be regarded as composed of parallel planes of points,
the Bragg planes. In this case, a simple cubic lattice is built out of parallel planes normal
to (110) (stereo pair).
Choose the magnitude of K so that
α
3
-Κ =
2ττ.
(3.22)

Theory of Scattering from Crystals 51
Once Eq. (3.22) is satisfied, one has for any point in the Bravais lattice
K-R
= K-(l
l
ai+l
2
â
2
+
l
3
â
3
)
=2π/
3
. (3.23)
The integer
IT,
simply counts by how many lattice planes the one containing R is
distant from the origin.
Because the magnitudes of the vectors K are not arbitrarily small according to
Eq. (3.22) and because the sum of any two is another such vector, the set of K's
must themselves form a lattice. One can find primitive vectors for the reciprocal
lattice, by choosing any three primitive vectors a\, a
2
, and a
3
for the Bravais lattice
and then building three primitive vectors b\, b
2
, and b
3
for the reciprocal lattice
from
- a? x «3
£ι=2π^-^—— (3.24a)
a\ -a
2
x
ατ,
b
2
= 2vr^^—— (3.24b)
a
2
-«3 xa\
a\ x a
2
a
3
-a\ x a
2
-* at x a
2
b
3
= 2π ^ ^ \, (3.24c)
3
and in general K — ^ m\Î>i.
Th
e m are arbitrary integers. (3.24d)
1=1
This construction works because if one takes the dot product of b\ with any prim-
itive vector αιι, the result is either zero or
27Γ.
Therefore, any vector in the direct
lattice dotted into linear combinations of the è/'s gives an integer times 2π as re-
quired.
Table 3.1 records the reciprocal lattices of four common Bravais lattices. The
reciprocal lattice of a fee lattice of spacing a is, a bcc lattice of spacing 4π/α, the
reciprocal lattice of a bcc lattice of spacing a is an fee lattice of spacing 4π/α, and
the reciprocal lattice of an hexagonal lattice is another hexagonal lattice twisted by
30°.
3.2.6 Miller Indices
A traditional notation describing reciprocal lattice vectors, lattice planes, and lat-
tice points is the Miller index. The notation is most commonly used for lattices of
cubic and hexagonal symmetry.
In cubic crystals, one begins by defining three perpendicular coordinate axes x,
y,
and z pointing along the edges of the conventional unit cell.
•
[ijk]
refers to a direction
ix
+ jy + kz (3.25)
in the lattice specified by the three integers i,
j,
and k.

52
Chapter 3. Scattering and Structures
Table 3.1. Conventional primitive vectors of four common Bravais
lattices and their reciprocal lattices.
Lattice
sc
fee
bec
hex
Lattice
Spacing
a
a
a
a, c
Primitive
Vectors
(10 0)
(0 10)
(0 0 1)
(22°)
( 2 0 2)
(0 \ \)
fi i _i)
I 2 2
2>
f_i i i)
V 2 2
2>
f i _i Ì)
\ 2
2 2^
( 1 f o)
H f
o)
(001)
Reciprocal
Lattice
Spacing
2π
a
4π
a
4π
a
4π 2π
\βα e
Reciprocal
Lattice
Primitive
Vectors
(100)
(0 10)
(0 0 1)
fi i _\\
l 2 2
2>
f_i i ì)
v 2 2 2/
f i _i i)
\ 2 2
21
(Ho)
( io Ì)
(o
è £)
( f 20)
(f-^o)
(0 0 1)
• (ijk) refers to a lattice plane perpendicular to the normal vector [ijk]. (ijk)
may also refer to the reciprocal lattice vector of smallest magnitude perpen-
dicular to the plane
(ijk).
• {ijk} refers to the complete collection of lattice planes perpendicular to [ijk].
• ijk refers to the X-ray diffraction peak resulting from scattering off the lattice
planes
{ijk}.
• Negative integers are denoted by / rather than — i.
• (ijk) refers to the set of all lattice planes or reciprocal lattice vectors related
to (ijk) by rotational symmetry.
Examples. For a simple cubic lattice, the reciprocal lattice vector pointing along
(2π/α, 0, 0) is called (100), while the one along (2π/α, 2π/α, 2π/α) is called
(111) and the one along (2π/α, —2π/α, 2π/α) is called (111), and so forth. For
an fee lattice, the smallest reciprocal lattice vector is (111), and it has magnitude
2ν
/
3π/α. The collection of reciprocal lattice points referred to by (100) is (100),
(010),
(001), (TOO), (0Ϊ0), and (00Ϊ).
Alternate Definition. An alternate prescription used by crystallographers to find
the Miller index of a plane is to find a lattice point as near as possible to the plane
