Molina-Par?s C., Lythe G. (editors) Mathematical Models and Immune Cell Biology
Подождите немного. Документ загружается.


Chapter 18
Using Mathematical Models to Explore the Role
of Cytotoxic T Lymphocytes in HIV Infection
Helen Fryer and Angela McLean
Abstract The combination of mathematical modelling and data analysis to under-
stand the within-host dynamics of human immunodeficiency virus (HIV) infections
has been one of the most informative uses of mathematical biology in the last
decade. Simple models of viral dynamics together with viral load measurements
provided an early estimate that the turnover of HIV infected cells is very rapid:
most do not survive beyond 1 day. Although this estimate was initially a surprise
to the field, further corroborating evidence has made it widely accepted. More
recently, within-host models have been used to investigate the efficacy of cyto-
toxic T-lymphocytes (CTLs) in controlling HIV infection. Though there is clear
experimental evidence that they play some role, the magnitude of this role remains
contentious. Models have offered three insights on this topic. Firstly, in chronically
infected humans fewer than 20% of HIV-infected cell death is attributed to killing by
CTLs. Secondly, CTLs are more efficient in acute infection than chronic infection,
but not dramatically so, and thirdly, CTLs are markedly more efficient in simian than
human immunodeficiency viral infection. Although based on simple models and re-
peatable data, the main prediction of this work that CTL vaccines might work in
macaques but not in humans is yet to gain recognition. This is despite the fact that
this prediction was borne out by the failure of STEP vaccine. We contend that in
time this assertion too will become more widely accepted.
With an estimated 33 million people affected by HIV/AIDS across the world [1],
a huge research effort is now focussed on understanding the immune response to
Human Immunodeficiency Virus (HIV) in a bid to creating an effective vaccine.
Early HIV vaccines aimed to elicit antibodies, but quickly proved to be completely
ineffective [2]. The reason for this is now clear: even during natural infection, HIV
is able to evade antibody neutralisation, primarily by obstructing antibody binding
sites. Over the last 15 years, several pieces of evidence have demonstrated that cy-
totoxic T lymphocytes contribute to the control of HIV infection in vivo. These
H. Fryer (
)
The Institute for Emerging Infections, The Oxford Martin School,
Department of Zoology, University of Oxford, Oxford OX13PS, UK
e-mail: helen.fryer@zoo.ox.ac.uk
C. Molina-Par´ıs and G. Lythe (eds.), Mathematical Models and Immune Cell Biology,
DOI 10.1007/978-1-4419-7725-0
18,
c
Springer Science+Business Media, LLC 2011
363
364 H. Fryer and A. McLean
include a temporal correlation between the reduction of viremia in acute infection
and the appearance of HIV-specific CTLs [3, 4]; the selection of viral mutants that
evade killing by CTLs [5, 6]; and associations between particular HLA class I alle-
les and life expectancy [7–9]. Though the magnitude of CTLs’ contribution to viral
regulation is contentious, these findings have encouraged the vaccine field to direct
research towards developing a vaccine that elicits CTLs. The aim of such a vaccine
would be to dampen viral replication in hosts who become infected.
In 2004 the first large-scale human efficacy trial (the STEP Study) of a CTL-
based vaccine candidate began. Considerable hope was pinned on this vaccine, but
in 2007 the trial was halted when interim results showed the vaccine to be en-
tirely ineffective both at preventing infection and reducing viral load in those who
become infected [10, 11](Table18.1 and Fig. 18.1). This result is regarded as a
Table 18.1 Results of the STEP study. The number of HIV infections ac-
cording to baseline Ad5 antibody titre. The Merck vaccine trial was halted
early when it became apparent that the infection rate in individuals with
high immunity to the Ad5 vector was higher in those who were vaccinated
than in those who received the placebo. Even in individuals with low Ad5
immunity, the vaccine provided no protection [15]
Number of HIV infections
Ad5 antibody titre prior to innoculation Vaccine Placebo
<18 20/382 20/394
18 < Ad5200 8/140 4/142
200 < Ad5 1000 14/229 7/229
Ad5 > 1000 7/163 2/157
Total 49/914 33/922
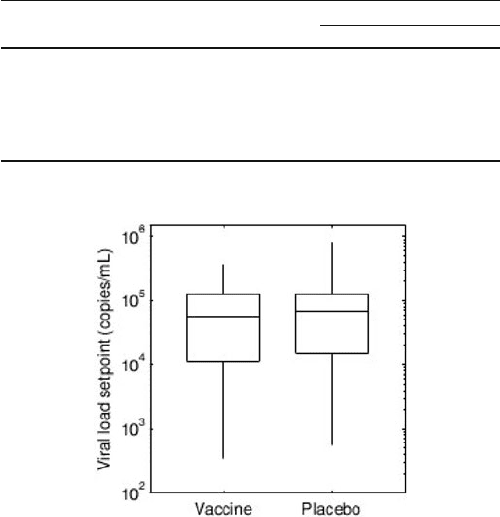
Fig. 18.1 Boxplots of the viral setpoints measured in individuals who became infected in the
STEP study. There was no difference in the viral setpoints measured in the vaccine (N D 46)
and placebo recipients (N D 30). When this analysis was restricted to patients who had low Ad5
immunity, the same result was obtained (data not shown) [15]
18 CTL in HIV Infection 365
major setback for the HIV vaccine field, not least because the candidate (the Merck
HIV DNA/adenovirus complex) had shown the most promise of several proposed
CTL-based vaccines that were tested in monkeys challenged with simian-human
immunodeficiency virus (SHIV)[12]. The failure of this vaccine has reignited doubt
that CTLs play a pivotal role in controlling HIV replication and has thus led the field
to question whether CTLs will ever prove a useful tool for an HIV vaccine. In addi-
tion, the failure has highlighted the inconsistency between CTL efficacy in monkeys
and humans. In light of these issues, this chapter reviews how mathematical mod-
els have been used together with experimental results to address the question: how
effective are CTLs are in controlling HIV-1 infection?
The chapter begins with a discussion of experimental and observational results
that provide insight into the role of CTLs in controlling HIV infection. This is fol-
lowed by a review of models of viral dynamics that have been used to interpret
patient viral load data in relation to this question. Finally, a model of the infection of
CD4 T cells with either the wildtype viral strain or a CTL escape mutant strain [13]
is discussed. We show how this model has been used to analyse CTL escape mutant
outgrowth data to quantify the magnitude of the CTL response. Further, we show
how it has been used to compare the strength of the HIV-specific CTL response in
humans to that of the SIV/SHIV-specific response in macaques [14].
Experimental and Observational Results that Provide Insight
into the Role of CTLs in Controlling HIV Infection
Today there is considerable evidence to suggest that CTLs contribute to the control
of HIV infection in vivo. What is not clear is exactly how important their contribu-
tion is at each stage of infection. Measuring their contribution directly is not possible
and there is some evidence against a significant role for CTLs; notably certain corre-
lations predicted on the basis of a strong CTL response have not been observed. Data
indicating an important role for CTLs in controlling HIV in infected individuals are
summarised in Fig. 18.2.
One of the early observations in favour of a role for CTLs came in 1994 when
a temporal correlation was established between the reduction in viremia in acute
infection and the appearance of HIV-specific CTLs [3, 4]. The appearance of the
CTLs preceded the appearance of antibodies, indicating that antibodies were not
responsible for the observed effect (Fig. 18.2(a)). Other evidence is provided by
the selection of CTL escape mutations in vivo [5, 6]; by in vitro experiments that
have revealed CTLs capable of exerting potent antiviral effects towards HIV [16–19]
(Fig. 18.2(b)); and by the high levels of anti-HIV-1 CTLs seen in established HIV-1
infection. These can often be detected directly from the peripheral blood without
in vitro stimulation. This confirms that the CTLs are ‘at work’, rather than simply
waiting to be stimulated into action.
Probably the best evidence in favour of a significant contribution by CTLs, how-
ever, comes from clear associations between progression to AIDS and different
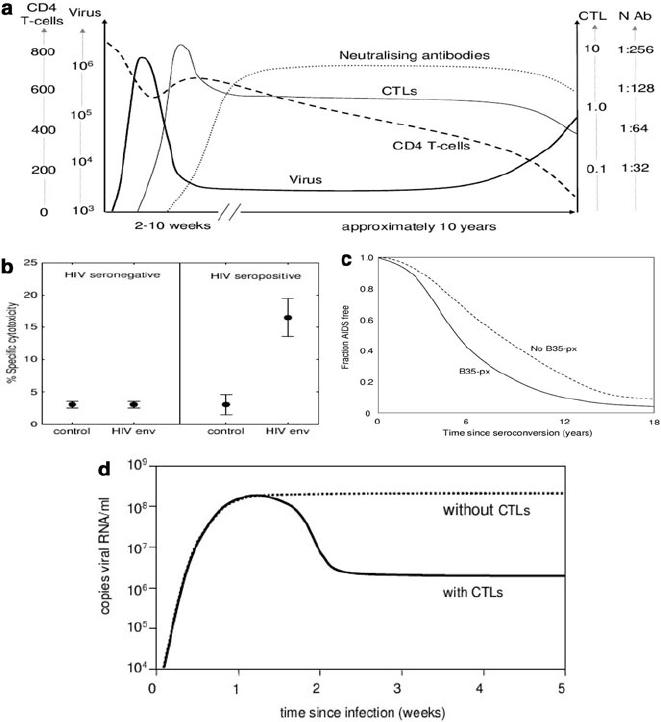
366 H. Fryer and A. McLean
Fig. 18.2 (a) The time course of natural HIV infection in a typical individual. A temporal cor-
relation exists between the containment of viremia during acute infection and the appearance of
HIV-specific CTLs. The measurements shown are the CD4
C
T cell count (cells/l), the plasma
viral load (RNA copies/ml), the CTL response (% effector CTL in PBMC) and the neutralising
antibody titre [31]. (b) Evidence that CTLs target HIV-infected cells in vitro. The mean percent
specific CTL cytotoxicity and corresponding standard error is presented for four seropositive and
five seronegative individuals. The percent specific cytotoxicity is shown for the effector:target ra-
tio 25:1. Cytotoxicity is measured in vitro against two vaccinia recombinants: one containing the
env region of the HIV genome and one containing the bacterial lacZ gene used as a control. The
HIV-specific response to the env/Vac complex was significantly elevated in seropositive individu-
als compared to the response to the lacZ/Vac (control) complex in the same hosts and compared to
the env/Vac complex in seronegative individuals. Note that some background cytotoxicity directed
against the Vaccinia virus is expected in all hosts [16]. (c) The association between the HLA B35Px
allele and progression to AIDS. Infected individuals who carry at least one copy of the HLA B35Px
allele progress to AIDS more rapidly than those who contain no copies of HLA B35px [20, 22].
(d) The effect of depletion of CTLs during primary SIV infection in macaques. The early control
of virus fails when CTLs are depleted [20,22]
18 CTL in HIV Infection 367
HLA alleles. These alleles encode for the HLA molecules involved in the process
that results in CTL targeting. Notably, HLA types B57, B51 and B27 are associ-
ated with slow progression and HLA B35Px is associated with rapid progression
(Fig. 18.2(c)). Patients who are homozygous at any of the three HLA class I loci
also progress substantially more rapidly to disease [7–9]. More recently, Kiepiela
and colleagues have also shown that the part of the genome targeted by CTLs
is an important determinant of disease progression. They found that an increased
breadth of gag-specific responses is associated with lower levels of viremia, whereas
an increased breadth of env-specific and accessory/regulatory-specific responses is
associated with increased levels of viremia. The reason for these protein-specific
differences is unclear.
HIV research that cannot be conducted in humans is instead tested in non-human
primates infected either with simian immunodeficiency virus (SIV) or the hybrid
virus simian-human immunodeficiency virus (SHIV). The primates used for these
tests are typically rhesus macaques or pigtail macaques, chosen because upon in-
fection they reproduce many of the key elements of HIV infection in humans. Not
only do they experience CD4
C
T cell depletion leading eventually to AIDS-like
symptoms, but they also have similar immune systems to humans. Like humans,
macaques have been shown to elicit CTL responses against immunodeficiency virus
infection. Furthermore, when antibodies specific for CTLs were used to experimen-
tally deplete CTLs during acute SIV/SHIV infection, the early control of virus failed
[20–22](Fig.18.2(d)). When the antibodies were instead given in the chronic phase,
the virus level rose until the effects of the antibody wore off. Depletion of B-cells
had no effect. These results favour a role for CTLs in controlling the set point in
natural HIV infection, though should be viewed in light of the unknown extent to
which results from macaques can be extrapolated to humans.
Evidence of CTL involvement in controlling HIV infection is therefore clear.
Exactly how important they are in this process, however, is put into question by
the absence of certain results. If CTLs were crucial in containing HIV, one might
expect that counts of HIV-specific CTLs would correlate with viral load. Despite
early reports to this effect [23–25], it is now accepted that no such simple correla-
tion exists [26,27]. In an examination of CTL responses directed against the entire
HIV genome in 57 patients, no correlation was found between either the breadth or
the magnitude of the CTL responses and viral load [27]. A second study into the
effects of drug therapy interruptions also showed no correlation between CTL count
and either viral rebound rates at the start of the interruption, or viral clearance rates
at the restart of therapy [26]. Furthermore, neither measures of absolute numbers
nor changes in numbers of CTLs were able to predict viral load in this experiment.
At the very least, these results indicate that different CTLs have different efficacies,
a finding supported by the study by Kiepiela et al. [28] and by two recent stud-
ies that suggest that a correlation may exist between the number of polyfunctional
CTL (as measured by five different immunological markers) and plasma viral load
[29,30].
368 H. Fryer and A. McLean
Modelling HIV-1 Viral Dynamics to Understand What Patient
Viral Load Data Tells Us About the Importance of CTLs
in Controlling HIV
One way in which the importance of CTLs in controlling HIV infection can begin to
be addressed is by investigating how well observed viral load data can be explained
by mathematical models of viral dynamics. Viral dynamics during HIV infection
have been widely analysed and it is now accepted that there are three main phases
to the dynamics in natural infection [31](Fig.18.2(a)). During the first few weeks
(acute infection), viral load rises to a peak before declining and settling to an equi-
librium called the viral set point. The start of this decline is typically associated with
the appearance of CTLs specific for HIV. Once at the set point, viral load remains at
that level for a period of several years (the chronic phase), during which there is
both rapid viral replication and rapid viral loss. The set point differs between hosts
and is predictive of life expectancy – higher set points correlate with shorter life ex-
pectancies. Eventually this balance is disrupted and viral load gradually increases
in concordance with a decline in CD4 count that eventually results in AIDS. Infor-
mation about viral dynamics can also be attained by disrupting the natural course of
infection. This can be achieved when antiretroviral therapy is started. These drugs
disrupt the viral life cycle and when given during chronic infection, lead to viral
load decaying in an exponential manner.
A Simple Model of Virus Infection
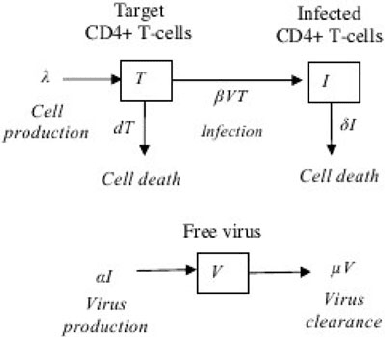
A simple model that has been proposed to explain some of the observed HIV viral
dynamics is shown in Fig. 18.3 [32]. The model consists of three variables: the
Fig. 18.3 Asimple
within-host model of viral
infection. T, I and V represent
the number of target cells,
infected cells and free virions,
respectively. Free virions are
produced at a rate, ˛,per
infected cell and infect target
cells at a rate proportional to
the abundance of free virions,
ˇV. In addition, target cells
are produced at rate and die
at a rate d per cell; free
virions are cleared at rate
per virion; and infected cells
die at rate ı per cell
18 CTL in HIV Infection 369
population size of target cells (i.e. uninfected CD4
C
T cells), T, infected cells, I,
and free virions, V. Free virions are produced at a rate, ˛, per infected cell and in-
fect target cells at a rate proportional to the abundance of free virus, ˇV. In addition,
target cells are produced at rate and die at a rate d per cell; free virions are cleared
at rate per virion and infected cells die at rate ı per cell. The model is prescribed
by the following set of coupled ordinary differential equations.
P
T D dT ˇV T
P
I D ˇV T ıI
P
V D ˛I V: (18.1)
Fitting the Simple Target-Cell-Limited Model to Viral Load Data
from Hosts with Natural HIV Infection
In the simplest form of this model, the death rate of infected cells, ı, can be as-
sumed to be constant through time. As such, the model includes no explicit CTL
response. Despite this, the model is nevertheless able to reproduce many of the viral
dynamics observed in HIV-infected individuals; namely it predicts that virus will
grow rapidly up to a peak before declining and settling at a stable equilibrium at
which there is continuous production and loss of virus. It is perhaps surprising that
a model that includes no explicit introduction of a CTL response during acute infec-
tion is able to reproduce the early containment of virus so often attributed to CTLs.
In fact, in this model it is saturation of the target cell population that is the cause of
viral suppression. The simple model of infection of target cells therefore puts into
question the hypothesis that acute viral containment is due to CTLs and suggests
that the observed temporal correlation may be coincidental. In support of this asser-
tion, Phillips [33] has cited three studies in which infected individuals suppressed
their primary viremia without mounting any detectable specific anti-viral immune
responses.
Although saturation can explain the drop in viral load qualitatively, how well it
can explain the drop quantitatively has been questioned. Stafford et al. [34] fitted
viral load data from ten patients in acute infection. They showed that, by assuming
only small variations in different model parameters across patients, the model could
accurately explain both the increase in viremia up to the peak and the set point that
was eventually reached, but could not explain the seemingly overly-rapid rates of
viral decay seen in some individuals. These fast decay rates could be explained by
the appearance of CTLs during acute infection. The fact that viral set points can
be explained without inclusion of a CTL response, however, implies that even if
the CTL response is important during acute infection, it is not explicitly important
during chronic infection.

370 H. Fryer and A. McLean
Does Progression to AIDS Happen Because of a Failing
CTL Response?
It has often been suggested that it is the failure of the CTL response that eventu-
ally disrupts the equilibrium observed during chronic phase and leads to increasing
viremia and eventual progression to AIDS. If this were true, it would favour the the-
ory that CTLs are crucial in controlling HIV during chronic infection, as has been
shown to be true in SIV/SHIV infected in macaques. In these monkeys, depletion of
CTLs during chronic infection leads to an increase in viremia.
It has been proposed that, if progression to AIDS results from a failing CTL
response, then individuals with more advanced disease would kill infected cells
more slowly. Conversely if AIDS results from the virus evolving to infect cells more
quickly, infected cells would be cleared more quickly in individuals with more ad-
vanced disease. To investigate this issue, curves showing the decay of viremia
following treatment with a protease inhibitor (PI) – a type of antiretroviral – in
chronically infected humans have been compared to decay curves predicted by a
version of the simple target-cell limited model that is adapted to include PI ther-
apy during chronic infection. Protease inhibitors do not affect the survival or rate of
virion production of infected cells, but do prevent the production of infectious viri-
ons. To model the impact of PI therapy, infectious virions V
I
and non-infectious
virions V
NI
are regarded separately and the production rate of infectious virions
is set to zero. Furthermore, for the short period over which viral decay has been
measured (approximately 1 week) the number of target cells is assumed to remain
constant at the steady state value attained prior to therapy, T
0
. Equations represent-
ing the adapted model are given by:
P
I D ˇV
I
T
0
ıI
P
V
I
DV
I
P
V
NI
D ˛I V
NI
: (18.2)
During the steady state achieved prior to therapy, the abundance of target cells, pro-
ductively infected cells and free virus can be regarded as constant. Thus by setting
T D I D V D 0 in the original model (18.1), expressions can be found for ˛ and
I
0
(˛ D ı=ˇT
0
and I
0
D ˇV
0
T
0
=ı) which enable (18.2)tobesolvedtogivean
expression for the decay of virus (V D V
NI
C V
I
) upon commencement of therapy:
V.t/ D V
0
e
t
C
V
0
ı
Â
ı
.e
ıt
e
t
/ ıte
t
Ã
(18.3)
This function fits patient data in which plasma viral load after PI treatment is seen
to decay in an exponential fashion following an initial lag [35](Fig.18.4(a)). Im-
portantly, this equation shows that the exponential viral decay rate will be faster
if clearance of infected cells (rate ı) is faster. It is noteworthy that this correlation
would hold true irrespective of whether clearance is CTL mediated. The hypothesis
18 CTL in HIV Infection 371
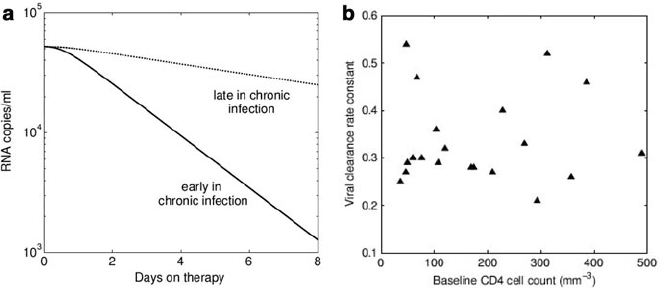
Fig. 18.4 The decline of viremia upon commencement of treatment with a protease inhibitor in
individuals with chronic HIV infection. (a) An example of the predicted decline in viral load fol-
lowing therapy in patients late in chronic infection and in patients early in chronic infection under
the assumption that patients with further progressed disease clear infected cells more slowly (see
(18.3)). The rate of viral decline would be faster in patients early on during chronic infection. (b)A
comparison of pre-therapy CD4 counts and viral clearance rates in 20 individuals. No relationship
exists between these two variables, implying that progression to AIDS does not result from a failing
CTL response [36]
that individuals with more advanced disease clear infected cells more slowly can
therefore be tested by comparing therapy-induced viral decay rates to pre-therapy
CD4 T cell counts. Interestingly, a comparison of such data shows that not only is
there no relationship between pre-therapy CD4 T cell counts and infected cell clear-
ance rates, but that infected cell clearance rates are remarkably consistent across
different individuals [36]. These results imply that people do not progress to AIDS
because clearance of infected cells diminishes. They therefore also provide no evi-
dence that CTLs are important in controlling HIV during chronic infection.
In summary, a simple within-host mathematical model of viral dynamics can
be used to understand what observed viral dynamics reveal about the importance
of the CTL response in HIV infection. This analysis shows that much of the ob-
served viral dynamics seen in natural infection can be explained qualitatively by a
model that includes no explicit CTL response. This includes containment of virus
during acute infection through saturation of the target cells. When the model and
the data are compared quantitatively, there remains no explicit evidence that CTLs
are important during chronic infection; however, in some individuals, rates of viral
containment during acute infection are faster than predicted by the simple model,
suggesting that CTLs may indeed be functionally important during acute infection,
as has been shown to be to be the case for macaques using CTL depletion experi-
ments. To investigate further whether CTLs are important during chronic infection,
viral clearance rates have been compared to pre-therapy CD4 T cell counts. No rela-
tionship has been found between these two variables, suggesting that progression to
AIDS does not occur because of a failing CTL response. This comparison therefore
372 H. Fryer and A. McLean
also provides no evidence in support of CTLs being functionally important dur-
ing chronic HIV infection. This is in disagreement with results from macaques in
which depletion of CTLs during chronic SIV/SHIV infection leads to significantly
increased levels of viremia.
Modelling Within-Host Dynamics of CTL Escape and Reversion
to Estimate the Proportion of Cell Lysis Attributable to CTLs
There is good evidence to show that the average life expectancy of productively
infected CD4
C
T cells is considerably less than that of CD4
C
T cells present in
uninfected individuals [35–40]. What is not known is what proportion of this cell
death is attributable to CTLs and what proportion is attributable to other factors–
notably viral cytotopathicity and by-stander activation.
Longitudinal data relating to the outgrowth of CTL escape mutants within indi-
viduals is able to provide us with insight into this issue (Fig. 18.5(a)). For a start,
it is clear from the fact that selection of CTL escape mutants occurs, that CTLs are
responsible for some proportion of infected cell death. Secondly, it is intuitive that
the rate of outgrowth of a particular CTL escape mutant should reflect three main
quantities: the strength of the CTL response that is being evaded, the fitness con-
straints associated with the mutant and the rate of cell turnover. Fitness costs cannot
be measured directly. However, fitness costs are believed to drive the reversion of
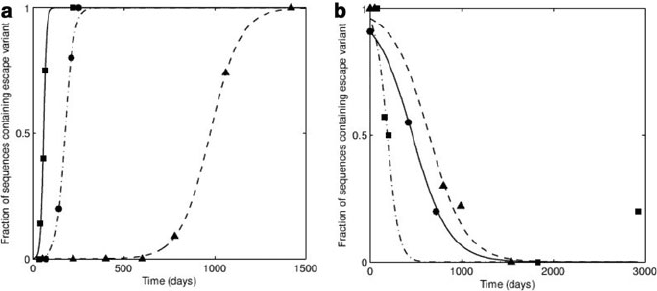
Fig. 18.5 Escape and reversion data and theoretical fits. (a) Three examples of the selection of
an escape mutant in an individual who makes an immune response to the epitope of interest. The
function 1=.ge
kt
C1/ is fitted to each of the escape curves to estimate the value of the escape rate,
k. The estimated escape rates for these three events are, 0.012, 0.119 and 0.41 day
1
.(b)Three
examples of the reversion of an escape mutant in an individual who does not make an immune
response to the epitope of interest. The function 1=.ge
t
C1/ is fitted to each the reversion curves
to estimate the value of the fitness cost (or reversion rate), . The estimated fitness costs for these
three events are, 0.016, 0.005 and 0.005 day
1
