Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.


Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4 1
Elements of Band Theory 2
4.1 Energy Band Formation 3
So far, we have completely neglected the effects of the ion cores on the motion 4
of the valence electrons. We consider “valence electrons” to be those outside 5
of a closed shell configuration, so that 6
1. Na has a single 3s valence electron outside a [Ne] core. 7
2. Mg has two 3s electrons outside a [Ne] core. 8
3. Ga has ten 3d electrons, two 4s electrons, and one 3p electrons outside an 9
[Ar] core. 10
The s and p electrons are usually considered as the “valence” electrons, since 11
they are responsible for “bonding”. Sometimes the mixing of d-electron atomic 12
states with “valence” electron states is important. 13
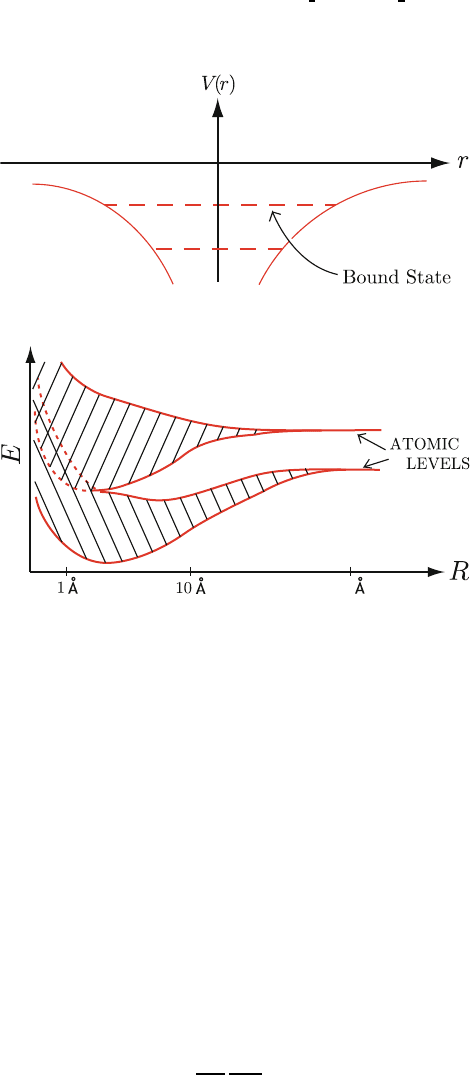
To get some idea about the potential due to the ion cores let’s consider 14
thesimplecaseofanisolatedNa
+
ion. This ion has charge +e and attracts 15
an electron via the Coulomb potential. (See Fig. 4.1.) 16
V (r)=−
e
2
r
if r>ion radius. (4.1)
For a pair of Na atoms separated by a large distance, each “conduction elec-
17
tron” (3s-electron in Na) has a well-defined atomic energy level. As the two 18
atoms are brought closer together, the atomic potentials V (r) begin to over- 19
lap. Then, each electron can feel the potential of both ions. This gives rise a 20
splitting of the degeneracy of atomic levels. 21
For a large number of atoms, the same effect occur. Think of a crystal 22
structure with a nearest neighbor separation of 1 cm. The energy levels of 23
the system will be atomic in character. However, as we decrease the nearest 24
neighbor separation the atomic energy levels will begin to broaden into bands. 25
(See Fig. 4.2.) The equilibrium separation of the crystal is the position at 26
which the total energy of the system is a minimum. In all crystalline solids the 27
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
110 4 Elements of Band Theory
Fig. 4.1. Coulomb potential
2
10
2
10
Fig. 4.2. Schematic illustration of energy band formation
electronic energies form bands of allowed energy values separated by energy 28
gaps (bands of forbidden energy values). These energy bands determine the 29
electrical properties of the solid. 30
4.2 Translation Operator 31
Because the crystalline potential seen by a single electron in a solid is a peri- 32
odic function of position, with the period of the lattice, it is useful to introduce 33
a translation operator T defined by 34
Tf(x)=f(x + a), (4.2)
where f (x) is an arbitrary function of position and a is the period of the
35
lattice. It is clear that T commutes with the single particle Hamiltonian H 36
H = −
¯h
2
2m
∂
2
∂x
2
+ V (x), (4.3)
because if we let x
= x + a,wecanseethat∂/∂x
= ∂/∂x and V (x
)=V (x). 37

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.3 Bloch’s Theorem 111
One of the most useful theorems of linear algebra for the study of quantum 38
systems states that if two operatorscommute,onecanfind common eigen- 39
functions for them (i.e, they can both be diagonal in the same representation). 40
Let Ψ be an eigenfunction of H and of T 41
HΨ=EΨandT Ψ=λΨ. (4.4)
Here, E and λ are eigenvalues. Clearly applying T to Ψ N times gives
42
T
N
Ψ(x)=λ
N
Ψ(x)=Ψ(x + Na). (4.5)
If we apply periodic boundary conditions with period N ,thenΨ(x + Na)=
43
Ψ(x). This implies that 44
T
N
Ψ(x)=Ψ(x), (4.6)
or that λ
N
=1.Thus,λ itself must be the N th root of unity 45
λ =e
i
2π
N
n
, (4.7)
where n =0, ± 1,....Wecanwriteλ as
46
λ =e
ika
, (4.8)
where k =
2π
Na
× n. Then, it is apparent that two values of k which differ by 47
2π/a times an integer give identical values of λ. As usual, we choose the N 48
independent values of k to lie in the range −π/a < k ≤ π/a, the first Brillouin 49
zone of a one dimensional crystal. 50
For more than one dimension, T
R
translates through a lattice vector R 51
T
R
Ψ(r)=e
ik·R
Ψ(r), (4.9)
52
where k =
2π
N
(n
1
b
1
+n
2
b
2
+n
3
b
3
). Here n
1
,n
2
,n
3
are integers and b
1
, b
2
, b
3
53
are primitive translations of the reciprocal lattice. We have assumed a period 54
N for periodic boundary condition with L
1
= Na
1
, L
2
= N a
2
, L
3
= N a
3
, 55
and values of n
1
, n
2
, n
3
are chosen to restrict k to the first Brillouin zone. 56
4.3 Bloch’s Theorem 57
We have just demonstrated that for a one-dimensional crystal with N-atoms 58
and periodic boundary conditions 59
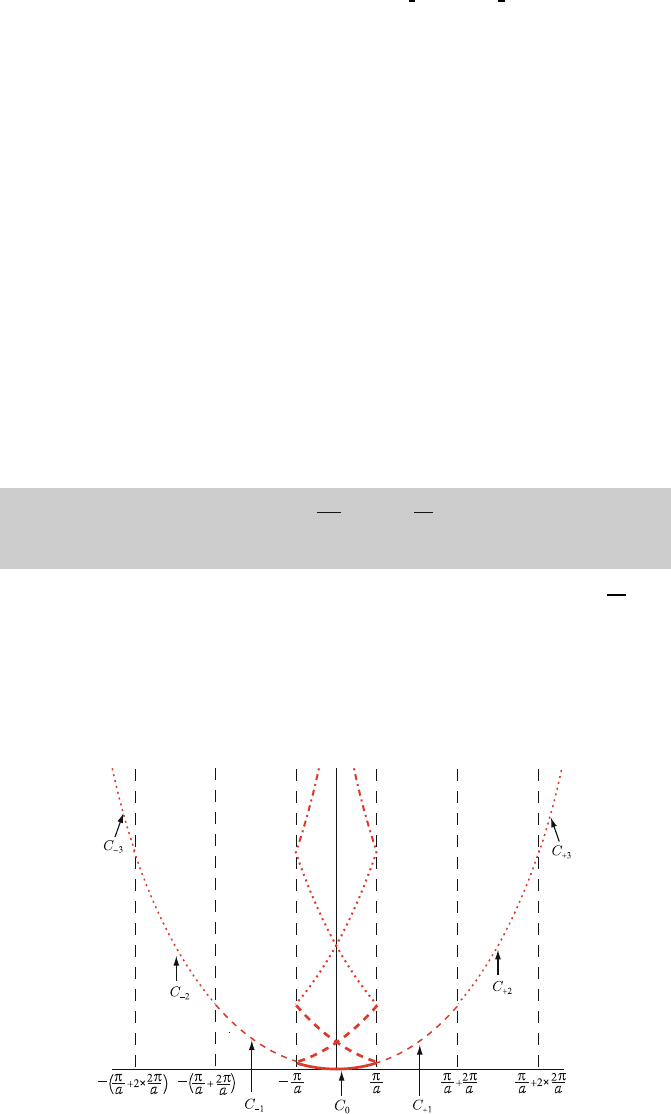
T Ψ(x) ≡ Ψ(x + a)=e
ika
Ψ(x), (4.10)
where the N independent values of k are restricted to the first Brillouin zone.
60
We can define a function 61
u
k
(x)=e
−ikx
Ψ(x). (4.11)

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
112 4 Elements of Band Theory
It is apparent that 62
Tu
k
(x)=T {e
−ikx
Ψ(x)} =e
−ik(x+a)
Ψ(x + a)=e
−ikx
Ψ(x)=u
k
(x).
Therefore, we can write
63
Ψ(x)=e
ikx
u
k
(x), (4.12)
64
where u
k
(x) is a periodic function i.e. u
k
(x + a)=u
k
(x). This is known to 65
physicists as Bloch’s theorem, although it had been proven sometime earlier 66
than Bloch
1
and is known to mathematicians as Floquet’s theorem. 67
4.4 Calculation of Energy Bands 68
There are two very different starting points from which one can approach 69
energy bands in solids. The first approach is to start with atomic orbitals and 70
to form linear combinations which satisfy Bloch’s theorem. The second is to 71
start with a Sommerfeld free electron gas picture (for the electrons outside 72
a closed shell core) and to see how the periodic potential of ions changes 73
the ε
k
= k
2
¯h
2
/2m free electron dispersion. The first approach works well for 74
systems of rather tightly bound electrons, while the second works well for 75
weakly bound electrons. We will spend a good deal of time on the “nearly free 76
electron” model and how group theory helps to make the calculations easier. 77
Before doing that, we begin with the first approach called the “tight-binding 78
method” or the LCAO (linear combination of atomic orbitals). 79
4.4.1 Tight-Binding Method 80
Suppose that a free atom has a potential V
a
(r), so that a “conduction 81
electron,” like the 3s electron in sodium, satisfies the Schr¨odinger equation 82
−
¯h
2
2m
∇
2
+ V
a
(r) −E
a
φ(r)=0. (4.13)
Here, E
a
is the atomic energy level of this conduction electron. When atoms 83
form a crystal, the potentials of the individual atoms overlap, as indicated 84
schematically in Fig. 4.3. 85
solid
Fig. 4.3. Tight-Binding Potential
1
Felix Bloch, Z. Physik 52, 555 (1928).

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.4 Calculation of Energy Bands 113
In the tight-binding approximation, one assumes that the electron in the 86
unit cell about R
j
is only slightly influenced by atoms other than the one 87
located at R
j
. Its wave function in that cell will be close to φ(r − R
j
), the 88
atomic wave function, and its energy close to E
a
. One can make a linear 89
combination of atomic orbitals φ(r −R
j
) as a trial function for the electronic 90
wave function in the solid. 91
To satisfy Bloch’s theorem, we can write 92
Ψ
k
(r)=
1
√
N
j
e
ik·R
j
φ(r −R
j
). (4.14)
93
Clearly, the translation operator operating on Ψ
k
(r)gives 94
T
R
n
Ψ
k
(r)=Ψ
k
(r + R
n
)
=e
ik·R
n
1
√
N
j
e
ik·(R
j
−R
n
)
φ(r −R
j
+ R
n
)=e
ik·R
n
Ψ
k
(r).
The energy of a state Ψ
k
(r)isgivenby 95
E
k
=
Ψ
k
|H|Ψ
k
Ψ
k
|Ψ
k
, (4.15)
where H is the Hamiltonian for an electron in the crystal, and
96
Ψ
k
|Ψ
k
=
1
N
j,m
e
ik·(R
j
−R
m
)
d
3
rφ
∗
(r − R
m
)φ(r − R
j
). (4.16)
If we neglect overlap between φ(r − R
j
)andφ(r − R
m
), the d
3
r integration 97
gives δ
j,m
and the sum over j simply gives a factor N, the number of atoms 98
in the crystal. 99
The Hamiltonian for an electron in the solid contains the potential V (r). 100
Let’s write 101
V (r)=
˜
V (r − R
j
)+V
a
(r − R
j
). (4.17)
In other words,
˜
V (r −R
j
) is the full potential of the solid minus the potential 102
of an atom located at R
j
. It’s clear from Fig. 4.3 that V
a
is larger than V (r) 103
in the cell containing R
j
so that
˜
V (r − R
j
) is negative. Since 104
−
¯h
2
2m
∇
2
+ V
a
(r − R
j
) − E
a
φ(r −R
j
)=0, (4.18)
105
E
k
=
1
N
j
m
e
ik·(R
j
−R
m
)
d
3
rφ
∗
(r −R
m
)
E
a
+
˜
V (r −R
j
)
φ(r −R
j
).
(4.19)
In the first term, E
a
is the constant value of the atomic energy level and it can 106
be taken out of the integration. All that remains in the integral is Ψ
k
|Ψ
k
107
which is 1, so the first term is just E
a
. We can define 108

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
114 4 Elements of Band Theory
α = −
d
3
rφ
∗
(r −R
j
)
˜
V (r −R
j
)φ(r − R
j
), (4.20)
and
109
γ = −
d
3
rφ
∗
(r −R
m
)
˜
V (r −R
j
)φ(r − R
j
). (4.21)
In the definition of γ we assume that the only terms that are not negligible
110
are terms in which R
m
is a nearest neighbor of R
j
. Then, we have 111
E
k
= E
a
− α −γ
m
e
ik·(R
j
−R
m
)
(4.22)
112
where the sum is over all nearest neighbors of R
j
. We chose minus signs in the 113
definition of α and γ to make α and γ positive (since
˜
V (r − R
j
) is negative). 114
Now look at what happens for a simple cubic lattice. There are six nearest 115
neighbors of R
j
located at R
j
±aˆx, R
j
±aˆy,andR
j
±aˆz. Substituting into 116
(4.22) gives 117
E
k
= E
a
− α −2γ (cos k
x
a +cosk
y
a +cosk
z
a) . (4.23)
Because γ is positive
118
E
Min
k
= E
a
− α −6γ, (4.24)
and
119
E
Max
k
= E
a
− α +6γ. (4.25)
The result is sketched in Fig. 4.4.
120
For k π/a 121
E
k
E
a
− α −6γ + γa
2
k
2
; k
2
= k
2
x
+ k
2
y
+ k
2
z
= E
Min
k
+
¯h
2
k
2
2m
∗
.
The effective mass m
∗
=
¯h
2
2γa
2
.Asγ decreases, the band width ΔE gets smaller 122
and the effective mass near E = E
Min
k
increases. 123
[1,1,1]
Fig. 4.4. Tight-binding dispersion for simple cubic lattice
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.4 Calculation of Energy Bands 115
4.4.2 Tight Binding in Second Quantization Representation 124
Suppose a system of free electrons is described by the Hamiltonian 125
H
0
=
k
ε
k
c
†
k
c
k
, (4.26)
where ε
k
=
¯h
2
k
2
2m
is the kinetic energy. In the presence of a periodic potential 126
V (r)=
K
V
K
e
iK·r
, we can write the potential energy of the electrons as 127
H
=
k,K
V
K
c
†
k+K
c
k
. (4.27)
Now, introduce the operators c
n
and c
†
n
which annihilate or create electrons 128
at site R
0
n
. 129
c
n
=
1
√
N
k
c
k
e
ik·R
0
n
. (4.28)
The inverse transformation is
130
c
k
=
1
√
N
n
c
n
e
−ik·R
0
n
. (4.29)
Substitute the latter equation into H
0
to obtain 131
H
0
=
k
ε
k
nm
1
N
c
†
n
c
m
e
ik·(R
0
n
−R
0
m
)
. (4.30)
Define
132
T
nm
=
1
N
k
ε
k
e
ik·(R
0
n
−R
0
m
)
. (4.31)
Then
133
H
0
=
nm
T
nm
c
†
n
c
m
. (4.32)
Now, look at H
134
H
=
k,K
V
K
1
N
nm
c
†
n
e
i(k+K)·R
0
n
c
m
e
−ik·R
0
m
=
Knm
k
1
N
e
ik·(R
0
n
−R
0
m
)
V
K
e
iK·R
0
n
c
†
n
c
m
.
Since
1
N
k
e
ik·(R
0
n
−R
0
m
)
= δ
nm
,wehave 135
H
=
K,n
V
K
e
iK·R
0
n
c
†
n
c
n
. (4.33)
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
116 4 Elements of Band Theory
But we note that
K
V
K
e
iK·R
0
n
= V (R
0
n
), and hence H
becomes 136
H
=
n
V (R
0
n
)c
†
n
c
n
. (4.34)
Adding H
0
and H
gives 137
H =
n
T
nn
+ V (R
0
n
)
c
†
n
c
n
+
n=m
T
nm
c
†
n
c
m
=
n
ε
n
c
†
n
c
n
+
n=m
T
nm
c
†
n
c
m
, (4.35)
138
where ε
n
= T
nn
+ V (R
0
n
) represents an energy on site n and T
nm
denotes the 139
amplitude of hopping from site m to site n. Starting with atomic levels ε
n
and 140
allowing hopping to neighboring sites results in energy bands, and the band 141
width depends on the hopping amplitude T
nm
. Later we will see that starting 142
with free electrons and adding a periodic potential V (r)=
K
V
K
e
iK·r
also 143
results in energy bands. The band gaps between bands depend on the Fourier 144
components V
K
of the periodic potential. 145
4.5 Periodic Potential 146
Because the potential experienced by an electron is periodic with the period 147
of the lattice, it can be expanded in a Fourier series 148
V (r)=
K
V
K
e
iK·r
, (4.36)
where the sum is over all vectors K of the reciprocal lattice, and
149
V
K
=
1
Ω
d
3
rV(r)e
−iK·r
.
For any reciprocal lattice vector K
150
K · R =2π ×integer,
if R is any translation vector of the lattice. Thus
151
V (r + R)=
K
V
K
e
iK·(r+R)
,
=
K
V
K
e
iK·r
= V (r).
The periodic part of the Bloch function can also be expanded in Fourier series.
152
We can write 153
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.5 Periodic Potential 117
u
n
(k, r)=
K
C
K
(n, k)e
iK·r
, (4.37)
For the moment, let us omit the band index n and wave number k and simply
154
write 155
Ψ
k
(r)=e
ik·r
u(r)=
K
C
K
e
i(k+K)·r
. (4.38)
Use the Fourier expansion of V (r)andu(r) in the Schr¨odinger equation; this
156
gives 157
K
¯h
2
2m
(k + K
)
2
+
K
V
K
e
iK
·r
C
K
e
i(k+K
)·r
= E
K
C
K
e
i(k+K
)·r
. (4.39)
We multiply by e
−i(k+K)·r
and integrate recalling that
"
d
3
re
iK·r
=Ωδ(K) 158
where Ω is the volume. This gives 159
E − V
0
−
¯h
2
2m
(k + K)
2
C
K
=
H=0
V
H
C
K−H
(4.40)
160
Here, we have set K
= H and have separated the H = 0 term from the other 161
terms in the potential. This is an infinite set of linear equations for the coeffi- 162
cients C
K
. The non-trivial solutions are obtained by setting the determinant 163
of the matrix multiplying the column vector 164
⎛
⎜
⎜
⎝
.
.
.
C
K
.
.
.
⎞
⎟
⎟
⎠
equal to zero. The roots give the energy levels (an infinite number – one for
165
each value of K) in the periodic potential of a crystal. We can express the 166
infinite set of linear equations in the following matrix notation: 167
⎛
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎝
ε
K
1
+ K
1
|V |K
1
−E
K
1
|V |K
2
··· K
1
|V |K
n
···
K
2
|V |K
1
ε
K
2
+ K
2
|V |K
2
−E ···
K
2
|V |K
n
···
.
.
.
.
.
.
.
.
.
K
n
|V |K
1
K
n
|V |K
2
···
ε
K
n
+ K
n
|V |K
n
−E ···
.
.
.
.
.
.
.
.
.
⎞
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎠
⎛
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎝
C
K
1
C
K
2
.
.
.
C
K
n
.
.
.
⎞
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎠
=0
(4.41)
Here, ε
K
=
¯h
2
2m
(k + K)
2
and K |V |K
= V
K−K
,where|K =
1
√
Ω
e
iK·r
.The 168
object of energy band theory is to obtain a good approximation to V (r)and 169
to solve this infinite set of equations in an approximate way. 170
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
118 4 Elements of Band Theory
4.6 Free Electron Model 171
If V
K
=0forallK = 0, then in the notation used earlier 172
(E − V
0
− ε
k+K
) C
K
=0. (4.42)
This is exactly the Sommerfeld model of free electrons in a constant potential
173
V
0
.Wecanwrite 174
E
(0)
K
= V
0
+ ε
k+K
C
(0)
K
=
#
1 For the band K
0 For all other bands
(4.43)
Let us discuss this in detail by considering a simple one-dimensional case, for
175
example, as shown in Fig. 4.5. The allowed values of the Bloch wave vector k 176
are restricted to the first Brillouin zone. Values of k outside the first Brillouin 177
zone are obtained by adding a reciprocal lattice vector K to k. The labels C
n
178
refer to the non-zero coefficients for that particular band; e.g. for C
+2
we have 179
E
(0)
2
= V
0
+
¯h
2
2m
k +2
2π
a
2
Ψ
(0)
nk
(r)=e
ik·r
u
(0)
nk
(r).
'
(4.44)
180
But u
nk
(r)=
K
C
K
(k)e
iK·r
;withC
(0)
K
(k)=1for| K |= K
n
=
2π
a
· n,
181
we have Ψ
(0)
nk
(r)=e
ik·r
·e
iK
n
·r
. All of this is simply a restatement of the free 182
electron model in the “reduced” zone scheme (i.e. all Bloch k−vectors are in 183
the first Brillouin zone, but energies of higher bands are obtained by adding 184
reciprocal lattice vectors K to k; the periodic part of the Bloch function is 185
u
K
=e
iK·r
). 186
Fig. 4.5. One dimensional free electron band
