Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.


Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.7 Nearly Free Electron Model 119
4.7 Nearly Free Electron Model 187
If we take V
K
for | K |= 0 to be very small but non-zero, we can use “pertur- 188
bation theory” to solve the infinite set of coupled equations approximately. 189
For the lowest band (the one with C
(0)
0
= 1) we know that in zeroth order 190
(i.e. with V
K
=0for| K |=0) 191
E
(0)
= V
0
+
¯h
2
k
2
2m
,
C
(0)
0
=1 and C
(0)
K
=0for | K |=0.
Let us look at other values of C
K
(i.e. not | K |=0value)forV
K
= 0 (but 192
very small) when | K |= 0. The first order correction to C
(0)
K
is given by 193
[E − V
0
−
¯h
2
2m
(k + K)
2
]C
(1)
K
=
|H| =0
C
K−H
V
|H|
. (4.45)
On the right-hand side all the V
|H|
appearing are small; therefore to first order 194
we can use for C
K−H
the value C
(0)
K−H
which equals unity for K −H =0and 195
zero otherwise. Solving for C
(1)
K
gives 196
C
(1)
K
=
V
K
¯h
2
2m
[k
2
− (k + K)
2
]
(4.46)
Here,wehaveusedE
¯h
2
k
2
2m
+ V
0
for the zeroth order approximation to the 197
energy of the lowest band (the one we are investigating). We substitute this 198
result back into the equation for C
0
, which is approximately equal to unity. 199
E − V
0
−
¯h
2
k
2
2m
C
0
|H| =0
C
0−H
V
H
=
V
−H
V
H
¯h
2
2m
[k
2
− (k −H)
2
]
(4.47)
C
0
=1+C
(1)
0
, but C
(1)
0
can be neglected since the right-hand side is already 200
small. Setting C
0
1 and solving for E gives 201
E = V
000
+ ε
k
−
|K|=0
|V
K
|
2
ε
k+K
− ε
k
(4.48)
202
In this equation, we have used V
−H
= V
∗
H
and let −H = K.Aslongas 203
| ε
k+K
− ε
k
|| V
K
|, this perturbation expansion is rather good. It clearly 204
breaks down when 205
ε
k+K
= ε
k
or 206
|k + K| = |k|.
This is exactly the condition for a Bragg reflection; when k
− k = K we get 207
Bragg reflection. 208

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
120 4 Elements of Band Theory
4.7.1 Degenerate Perturbation Theory 209
Suppose that for some particular reciprocal lattice vector K 210
|k + K||k| (4.49)
Our simple perturbation theory result gave
211
C
(1)
K
=
V
K
ε
k
− ε
k+K
.
It is clear that this result is inconsistent with our starting assumption that
212
C
K
was very small except for K = 0. To remedy this shortcoming we assume 213
that both C
0
and C
K
(for the particular value satisfying |k| = |k + K|)are 214
important. This assumption gives us a pair of equations 215
(E − V
0
− ε
k
) C
0
= C
K
V
−K
(E − V
0
− ε
k+K
) C
K
= C
0
V
K
.
(4.50)
The solutions are obtained by setting the determinant of the matrix mul-
216
tiplying the column vector
C
0
C
K
equal to zero. The two roots are given
217
by 218
E
±
(k)=V
0
+
1
2
[ε
k
+ ε
k+K
] ±
(
|V
K
|
2
+
ε
k
− ε
k+K
2
2
'
1/2
(4.51)
For |k| = |k + K|, ε
k
− ε
k+K
= 0 and the solutions become 219
E
±
(k)=V
0
+ ε
k
±|V
K
| (4.52)
220
This behavior is shown in Fig. 4.6. If we introduce q =
K
2
+ k,whereq
K
2
, 221
we can expand the roots for small q and obtain 222
K
K
Fig. 4.6. Bandgap formation at the zone boundary k =
K
2

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.8 Metals–Semimetals–Semiconductors–Insulators 121
E
±
= V
0
+ ε K
2
+ ε
q
±|V
K
|
(
1+
1
2
ε
K
|V
K
|
2
ε
q
'
.
If we define
223
E
0
= V
0
+ ε K
2
−|V
K
|,
and choose zero of energy at E
0
, the two roots can be written (for small q) 224
E
−
=
¯h
2
q
2
2m
∗
−
E
+
= E
G
+
¯h
2
q
2
2m
∗
+
.
(4.53)
Here, the energy gap E
G
is equal to 2 |V
K
| and the effective masses m
∗
±
are 225
given by 226
m
∗
±
=
m
1 ∓
ε
K
2|V
K
|
. (4.54)
227
It is common for ε
K
to be larger than 2 | V
K
| so that m
∗
−
is negative. Then 228
the two roots are commonly expressed as 229
E
v
(k)=−
¯h
2
k
2
2m
v
,
E
c
(k)=E
G
+
¯h
2
k
2
2m
c
,
where m
v
= −m
∗
−
. These results are frequently used to describe the valence 230
band and conduction band in semiconductors. The results are only valid near 231
q = 0 since we expanded the original equations for small deviations q from 232
the extrema. This result is called the effective mass approximation. 233
4.8 Metals–Semimetals–Semiconductors–Insulators 234
The very simple Bloch picture of energy bands and energy gaps allows us to 235
understand in a qualitative way why some crystals are metallic, some insulat- 236
ing, and some in between. For a one-dimensional crystal there will be a gap 237
separating every band (assuming that V
K
is non-zero for all values of K). We 238
know that the gaps occur when |k| = |k + K|. This defines the first Brillouin 239
zone’s boundaries. 240
In more than one dimension, the highest energy levels in a lower band can 241
exceed the energy at the bottom of a higher band. This is referred to as band 242
overlap. It is illustrated for a two-dimensional square lattice in Fig. 4.7. The 243
square represents the first Brillouin zone bounded by |k
x
| = π/a and |k
y
| = 244
π/a. The point Γ indicates the zone center of k =0.ThepointsX=
π
a
, 0
and 245
M=
π
a
,
π
a
are particular k-values lying on the zone boundary. The Δ and Σ
246
are arbitrary points on the lines connecting Γ → XandΓ→ M, respectively. 247
If we plot the energy along these lines, we obtain the result illustrated in 248
Fig. 4.8. It is clear that if the gaps are not too large, the maximum energy in 249
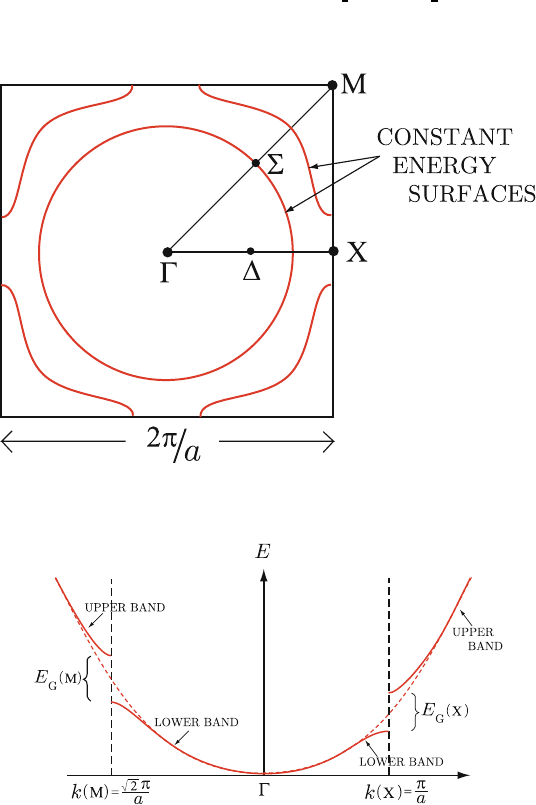
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
122 4 Elements of Band Theory
Fig. 4.7. Constant energy surface in two-dimensional square lattice
Fig. 4.8. Energy dispersion along the line M − Γ − X
the lower band E
LB
(M) is higher than the minimum energy in the upper band 250
E
UB
(X). If we fill all the lowest energy states with electrons, being mindful of 251
the exclusion principle, then it is clear that there will be some empty states 252
in the lower band as we begin to fill the low-energy states of the upper band. 253
The band overlap can be large (when the energy gaps are very small) or 254
non-existent (when the energy gaps are very large). 255
The existence of 256
1. Band gaps in the energy spectrum at Brillouin zone boundaries 257
2. Band overlap in more than one dimension when energy gaps are small and 258
3. The Pauli exclusion principle allows us to classify solids as follows: 259

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.8 Metals–Semimetals–Semiconductors–Insulators 123
• Metal 260
1. Monovalent metal. A material which contains one electron (outside a 261
closed core) on each atom and one atom per unit cell. Na, K, Rb, Cs 262
are good examples of monovalent metals. Because the total number of 263
allowed k values in the first Brillouin zone is equal to N,thenumber 264
of unit cells in the crystal, and because each k-state can accommodate 265
one spin up and one spin down electron, each band can accommodate 266
2N electrons. A monovalent metal has N electrons, so the conduction 267
band will be half filled. The Fermi energy is far (in comparison to 268
k
B
T ) from the band edges and band gaps. Therefore, the crystal acts 269
very much like a Sommerfeld free electron model. The same picture 270
holds for any odd valency solid containing 1, 3, 5, ... electrons per 271
unit cell. 272
2. Even-valency metals When the band gaps are very small, there can be 273
a very large overlap between neighboring bands. The resultant solid 274
will have a large number n
h
of empty states in the lower band and 275
an equal number n
e
(n
e
= n
h
of electrons in the higher band. If n
e
276
and n
h
are of the order of N, the number of unit cells, the material is 277
metallic. 278
• Insulator. For a material with an even number of electrons per unit cell 279
and a large gap (≥ 4eV) between the highest filled state and the lowest 280
empty state, an insulating crystal results. The application of a modest 281
electric field cannot alter the electron distribution function because to do 282
so would require a large energy E
G
. 283
• Semiconductor. A material which is insulating at low temperature, but 284
whose band gap E
G
is small (0.1eV≤ E
G
≤ 2 eV) is called a semiconduc- 285
tor. At finite temperature a few electrons will be excited from the filled 286
valence band to the empty conduction band. These electrons and holes 287
(empty states in the valence band) can carry current. Because the con- 288
centration of electrons in the conduction band varies like e
−E
G
/2k
B
T
,the 289
conductivity increases with increasing temperature. 290
• Semimetal. These materials are even-valency materials with small band 291
overlap. The number of electrons n
e
equals the number of holes n
h
but 292
both are small compared to N, the number of unit cells in the crystal. 293
Typically n
e
and n
h
might be 10
−3
or 10
−4
times the number of unit 294
cells. 295
Examples 296
Monovalent metals Li,Na,K,Rb,Cs,Cu,Ag,Au 297
Divalent metals Zn, Cd, Ca, Mg, Ba 298
Polyvalent metals Al,Ga,In,Tl 299
Semimetals As, Sb, Bi, Sn, graphite 300
Insulators Al
2
O
3
,diamond 301
Semiconductors Ge, Si, InSb, GaAs, AlSb, InAs, InP 302

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
124 4 Elements of Band Theory
Problems 303
4.1. In an infinite linear chain of A and B atoms (...ABABAB ...)with304
equal spacings R between each atom, the energies of electrons in the system 305
are given by E
k
= ±(ε
2
+4β
2
cos
2
kR)
1/2
,wherek is the wavevector of the 306
electron state. What is the band gap in the electronic band structure for this 307
system? How would you expect the electrical and optical properties of this 308
structure to depend on ε and β? 309
4.2. Consider a crystal of sodium with a volume 0.10 cm
3
, estimate the average 310
spacing between the energy levels in the 3s band given that the 3s electron 311
eneregies span a range of 3.20 eV. The electron concentration of a sodium 312
crystal is approximately 2.65 × 10
28
m
−3
. (You can estimate this value by 313
yourself.) 314
4.3. A BCC lattice has eight nearest neighbors at r =
a
2
(±ˆx ± ˆy ± ˆz). Use the 315
s-band tight-binding formula, (4.22), to evaluate E(k). 316
4.4. A single graphite sheet (called graphene) has a honeycomb structure. 317
Let us assume that there is one p
z
orbital, which is oriented perpendicular to 318
the sheet, on each carbon atom and forms the active valence and conduction 319
bands of this two-dimensional crystal. 320
(a) Using the tight-binding method and only nearest-neighbor interactions, 321
calculate and sketch the p-electron band structure E
n
(k) for graphene. 322
You may assume the overlap matrix is the unity matrix. 323
(b) Show that this is a zero-gap semiconductor or a zero-overlap semimetal. 324
Note that there is one p electron per carbon atom. 325
(c) Locate the position where the zero gap occurs in the momentum space. 326
4.5. A one-dimensional attractive potential is given by V (z)=−λδ(z). 327
(a) Show that the lowest energy state occurs at ε
0
= −
¯h
2
K
2
2m
,where328
K =
mλ
¯h
2
. 329
(b) Determine the normalized wave function φ
0
(z). 330
4.6. Consider a superlattice with V (z)=−λ
∞
l=−∞
δ(z − la). 331
(a) Obtain ε
0
(k
z
), the energy of the lowest band as a function of k
z
by using 332
the tight-binding approximation (i.e. overlap between only neighboring 333
sites is kept). 334
(b) Use the tight binding form of the wave function Ψ
0
(k
z
,z)andshow 335
that it can be expressed as 336
Ψ
0
(k
z
,z)=e
ik
z
z
u(k
z
,z),
337
where u(k
z
,z) is periodic with period a. Determine an expression for 338
u(k
z
,z). 339

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.8 Metals–Semimetals–Semiconductors–Insulators 125
4.7. Let us consider electrons in a one-dimensional Bravais lattice described 340
by the wave function and potential written as Ψ(x)=αe
ikx
+ βe
i(kG)x
and 341
V (x)=2V
1
cos(Gx). The zone boundary is at k = G/2=π/a,wherea is the 342
lattice constant. 343
(a) Obtain the band structure by solving the Schr¨odinger equation, and 344
sketch the band for 0 ≤ k ≤ G for V
1
=0andV
1
=0.2¯h
2
G
2
/2m. 345
(b) What kind of material do we have when V
1
=0? 346
(c) If V
1
=0.2¯h
2
G
2
/2m and each atom has three electrons, what kind of 347
material do we have? Explain the reason. 348

Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
126 4 Elements of Band Theory
Summary 349
In this chapter, we studied the electronic states from the consideration of the 350
periodicity of the crystal structure. In the presence of periodic potential the 351
electronic energies form bands of allowed energy values separated by bands of 352
forbidden energy values. Bloch functions were introduced as a consequence of 353
the translational symmetry of the lattice. Energy bands obtained by a simplest 354
tight-binding method and nearly free electron model are discussed. 355
The eigenfunction of the Hamiltonian H can be written as 356
Ψ(r)=e
ik·R
u
k
(r),
where u
k
(r) is a lattice periodic function i.e. u
k
(r + R)=u
k
(r). This is the 357
Bloch’s theorem. For an electron in a crystalline potential, we have 358
T
R
Ψ(r)=e
ik·R
Ψ(r),
where T
R
is a lattice translation operator and k =
2π
N
(n
1
b
1
+ n
2
b
2
+ n
3
b
3
). 359
Here n
1
,n
2
,n
3
are integers and b
1
, b
2
, b
3
are primitive translations of the 360
reciprocal lattice. 361
In the tight-binding approximation one assumes that the electron in the 362
unit cell about R
j
is only slightly influenced by atoms other than the one 363
located at R
j
. Its wave function in that cell will be close to φ(r − R
j
), the 364
atomic wave function, and its energy close to E
a
. One can make a linear 365
combination of atomic orbitals φ(r −R
j
) as a trial function for the electronic 366
wave function in the solid: 367
Ψ
k
(r)=
1
√
N
j
e
ik·R
j
φ(r −R
j
).
The energy of a state Ψ
k
(r)isgivenby 368
E
k
= E
a
− α −γ
m
e
ik·(R
j
−R
m
)
where the sum is over all nearest neighbors of R
j
and 369
α = −
d
3
rφ
∗
(r − R
j
)
˜
V (r − R
j
)φ(r − R
j
)
and
370
γ = −
d
3
rφ
∗
(r −R
m
)
˜
V (r −R
j
)φ(r − R
j
).
In second quantization representation, the tight-binding Hamiltonian is given
371
by 372
H =
n
ε
n
c
†
n
c
n
+
n=m
T
nm
c
†
n
c
m
Uncorrected Proof
BookID 160928 ChapID 04 Proof# 1 - 29/07/09
4.8 Metals–Semimetals–Semiconductors–Insulators 127
where ε
n
= T
nn
+ V (R
0
n
) represents an energy on site n and T
nm
denotes the 373
amplitude of hopping from site m to site n. 374
The periodic part of the Bloch function is expanded in Fourier series as 375
u
n
(k, r)=
K
C
K
(n, k)e
iK·r
and the energy eigenfunction is simply written as 376
Ψ
k
(r)=e
ik·r
u(r)=
K
C
K
e
i(k+K)·r
.
The Schr¨odinger equation of an electron in a lattice periodic potential is
377
written as an infinite set of linear equations for the coefficients C
K
: 378
E − V
0
−
¯h
2
2m
(k + K)
2
C
K
=
H=0
V
H
C
K−H
.
We can express the infinite set of linear equations in a matrix notation:
379
⎛
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎝
ε
K
1
+ K
1
|V | K
1
−E
K
1
|V |K
2
··· K
1
|V |K
n
···
K
2
|V |K
1
ε
K
2
+ K
2
|V |K
2
−E ···
K
2
|V |K
n
···
.
.
.
.
.
.
.
.
.
K
n
|V |K
1
K
n
|V |K
2
···
ε
K
n
+ K
n
|V |K
n
−E ···
.
.
.
.
.
.
.
.
.
⎞
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎠
⎛
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎜
⎝
C
K
1
C
K
2
.
.
.
C
K
n
.
.
.
⎞
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎟
⎠
=0.
Here, ε
K
=
¯h
2
2m
(k + K)
2
and K |V |K
= V
K−K
,where|K =
1
√
Ω
e
iK·r
. 380
In the nearly free electron method, the energies near the zone boundary 381
become 382
E
±
(k)=V
0
+ ε
k
±|V
K
|.
The two roots can be written, for small q, as
383
E
−
=
¯h
2
q
2
2m
∗
−
; E
+
= E
G
+
¯h
2
q
2
2m
∗
+
,
where q =
K
2
+ k. Here, the energy gap E
G
is equal to 2 |V
K
| and the effective 384
masses m
∗
±
are given by 385
m
∗
±
=
m
1 ∓
ε
K
2|V
K
|
.
The results are only valid near q = 0 since we expanded the original equations
386
for small deviations q from the extrema. This result is called the effective 387
mass approximation. Crystalline solids are classified as metals, semimetals, 388
semiconductors, and insulators according to the magnitudes and shapes of 389
the energy gap of the material. 390
Uncorrected Proof
BookID 160928 ChapID 05 Proof# 1 - 29/07/09
5 1
Use of Elementary Group Theory 2
in Calculating Band Structure 3
5.1 Band Representation of Empty Lattice States 4
For a three-dimensional crystal, the free electron energies and wave functions 5
can be expressed in the Bloch function form in the following way: 6
1. Write the plane-wave wave vector as a sum of a Bloch wave vector and a 7
reciprocal lattice vector. The Bloch wave vector k is restricted to the first 8
Brillouin zone; the reciprocal lattice vectors are given by 9
K
=2π[l
1
b
1
+ l
2
b
2
+ l
3
b
3
] (5.1)
10
where (l
1
,l
2
,l
3
)= are integers and b
i
are primitive translations of the 11
reciprocal lattice. Then 12
Ψ
(k, r)=e
ik·r
e
iK
·r
. (5.2)
The second factor has the periodicity of the lattice since e
iK
l
·R
=1forany 13
translation vector R. 14
2. The energy is given by 15
E
(k)=
¯h
2
2m
(k + K
l
)
2
. (5.3)
16
3. Each band is labeled by =(l
1
,l
2
,l
3
) and has Ψ
(k, r) given by (5.2) and 17
E
(k) given by (5.3). 18
5.2 Review of Elementary Group Theory 19
In our brief discussion of translational and rotational symmetries of a lattice, 20
we introduced a few elementary concepts of group theory. The object of this 21
chapter is to show how group theory can be used in evaluating the band 22
structure of a solid. We begin with a few definitions. 23
