Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
8 1 Crystal Structures
a
a
cos =
a
2
a
b
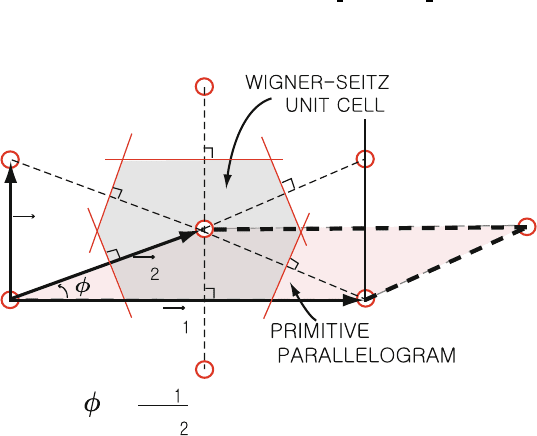
Fig. 1.7. Construction of the Wigner–Seitz cell of a two-dimensional centered
rectangular lattice. Note that cos φ = a
1
/2a
2
Primitive Unit Cell 92
From the three primitive translation vectors a
1
, a
2
, a
3
, one can form a paral- 93
lelepiped that can be used as a primitive unit cell. By stacking primitive unit 94
cells together (like building blocks) one can fill all of space. 95
Wigner–Seitz Unit Cell 96
From the lattice point (0, 0, 0) draw translation vectors to neighboring lat- 97
tice points (to nearest, next nearest, etc., neighbors). Then, draw the planes 98
which are perpendicular bisectors of these translation vectors (see, for exam- 99
ple, Fig. 1.7). The interior of these intersecting planes (i.e., the space closer 100
to (0, 0, 0) than to any other lattice point) is called the Wigner–Seitz unit 101
cell. 102
Space Group 103
For a Bravais lattice, the space group is simply the product of the operations 104
of the translation group and of the point group. For a lattice with a basis, 105
there can be other symmetry operations. Examples are glide planes and screw 106
axes; illustration of each is shown in Figs. 1.8 and 1.9, respectively. 107
Glide Plane 108
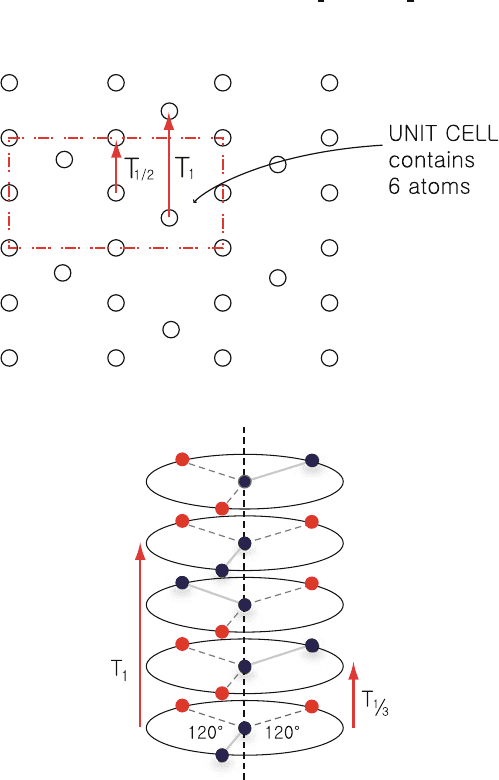
In Fig. 1.8, each unit cell contains six atoms and T
1/2
m
x
is a symmertry 109
operation even though neither T
1/2
nor m
x
is an operation of the symmetry 110
group by them. 111
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.1 Crystal Structure and Symmetry Groups 9
Fig. 1.8. Glide plane of a two-dimensional lattice. Each unit cell contains six atoms
Fig. 1.9. Screw axis. Unit cell contains three layers and T
1
is the smallest
translation. Occupied sites are shown by solid dots
Screw Axis 112
In Fig. 1.9, T
1/3
R
120
◦
is a symmetry operation even though T
1/3
and R
120
◦
113
themselves are not. 114
Two-Dimensional Bravais Lattices 115
There are only five different types of two-dimensional Bravais lattices. 116
1. Square lattice: primitive (P) one only 117
It has a = b and α = β =90
◦
. 118
2. Rectangular: primitive (P) and centered (C) ones 119
They have a = b but α = β =90
◦
. 120

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
10 1 Crystal Structures
3. Hexagonal: primitive (P) one only 121
It has a = b and α = 120
◦
(β =60
◦
). 122
4. Oblique: primitive (P) one only 123
It has a = b and α =90
◦
. 124
Three-Dimensional Bravais Lattices 125
There are 14 different types of three-dimensional Bravais lattices. 126
1. Cubic: primitive (P), body centered (I), and face centered (F) ones. 127
For all of these a = b = c and α = β = γ =90
◦
. 128
2. Tetragonal: primitive and body centered (I) ones. 129
For these a = b = c and α = β = γ =90
◦
. One can think of them as cubic 130
lattices that have been stretched (or compressed) along one of the cube 131
axes. 132
3. Orthorombic: primitive (P), body centered (I), face centered (F), and base 133
centered (C) ones. 134
For all of these a = b = c but α = β = γ =90
◦
. These can be thought 135
of as cubic lattices that have been stretched (or compressed) by different 136
amounts along two of the cube axes. 137
4. Monoclinic: primitive (P) and base centered (C) ones. 138
For these a = b = c and α = β =90
◦
= γ. These can be thought of as 139
orthorhombic lattices which have suffered shear distortion in one direction. 140
5. Triclinic: primitive (P) one. 141
This has the lowest symmetry with a = b = c and α = β = γ. 142
6. Trigonal: 143
It has a = b = c and α = β = γ =90
◦
< 120
◦
. The primitive cell is a 144
rhombohedron. The trigonal lattice can be thought of as a cubic lattice 145
which has suffered shear distortion. 146
7. Hexagonal: primitive (P) one only. 147
It has a = b = c and α = β =90
◦
, γ = 120
◦
. 148
1.2 Common Crystal Structures 149
1. Cubic 150
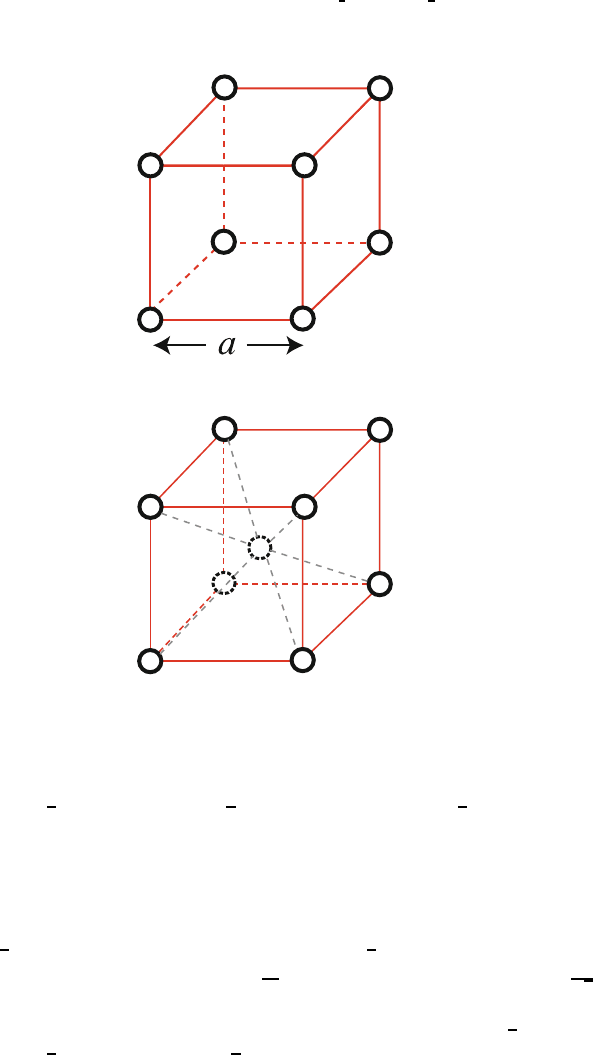
(a) Simple cubic (sc): Fig. 1.10 151
For simple cubic crystal the lattice constant is a and the volume per 152
atom is a
3
. The nearest neighbor distance is also a, and each atom has 153
six nearest neighbors. The primitive translation vectors are a
1
= aˆx, 154
a
2
= aˆy, a
3
= aˆz. 155
(b) Body centered cubic (bcc): Fig. 1.11 156
If we take a unit cell as a cube of edge a, there are two atoms 157
per cell (one at (0, 0, 0) and one at
1
2
,
1
2
,
1
2
). The atomic volume
158
is
1
2
a
3
, and the nearest neighbor distance is
√
3
2
a. Each atom has 159
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.2 Common Crystal Structures 11
Fig. 1.10. Crystallographic unit cell of a simple cubic crystal of lattice constant a
Fig. 1.11. Crystallographic unit cell of a body centered cubic crystal of lattice
constant a
eight nearest neighbors. The primitive translations can be taken as 160
a
1
=
1
2
a (ˆx +ˆy +ˆz), a
2
=
1
2
a (−ˆx +ˆy +ˆz), and a
3
=
1
2
a (−ˆx − ˆy +ˆz). 161
The parallelepiped formed by a
1
, a
2
, a
3
is the primitive unit cell (con- 162
taining a single atom), and there is only one atom per primitive unit 163
cell. 164
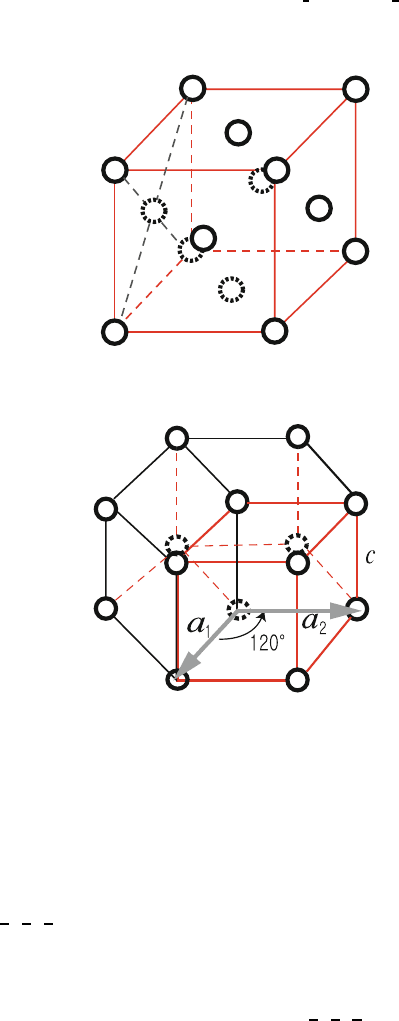
(c) Face centered cubic (fcc): Fig. 1.12 165
If we take a unit cell as a cube of edge a, there are four atoms per cell; 166
1
8
of one at each of the eight corners and
1
2
of one on each of the six 167
faces. The volume per atom is
a
3
4
; the nearest neighbor distance is
a
√
2
, 168
and each atom has 12 nearest neighbors. The primitive unit cell is the 169
parallelepiped formed from the primitive translations a
1
=
1
2
a (ˆx +ˆy), 170
a
2
=
1
2
a (ˆy +ˆz), and a
3
=
1
2
a (ˆz +ˆx). 171
All three cubic lattices have the cubic group as their point group. 172
Because the primitive translations are different, the simple cubic, bcc, 173
and fcc lattices have different translation groups. 174
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
12 1 Crystal Structures
Fig. 1.12. Crystallographic unit cell of a face centered cubic crystal of lattice
constant a
Fig. 1.13. Crystallographic unit cell of a simple hexagonal crystal of lattice
constants a
1
, a
2
,andc
2. Hexagonal 175
(a) Simple hexagonal: See Fig. 1.13. 176
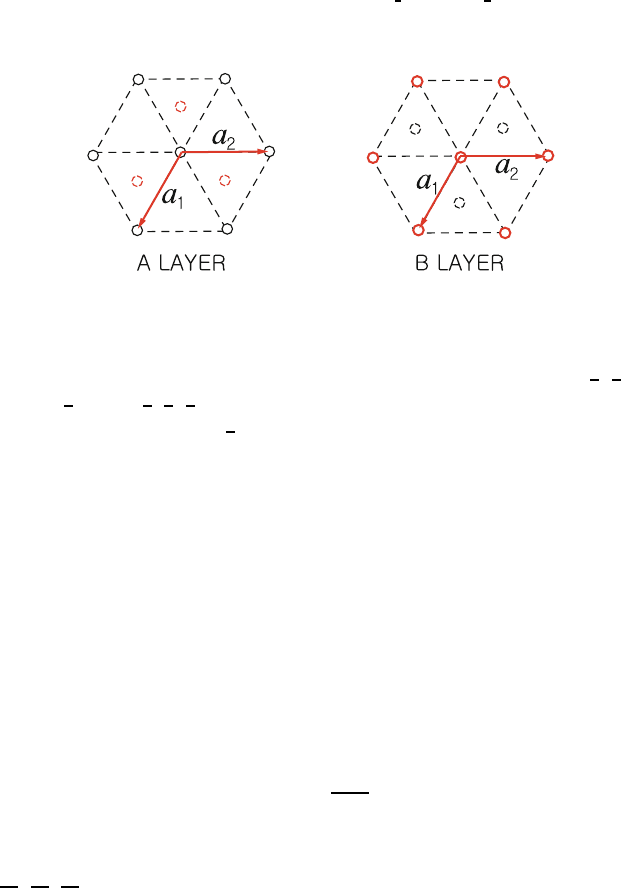
(b) Hexagonal close packed (hcp): 177
This is a non-Bravais lattice. It contains two atoms per primitive unit 178
cell of the simple hexagonal lattice, one at (0,0,0) and the second 179
at
1
3
,
2
3
,
1
2
. The hexagonal close packed crystal can be formed by
180
stacking the first layer (A) in a hexagonal array as is shown in Fig. 1.14. 181
Then, the second layer (B) is formed by stacking atoms in the alternate 182
triangular holes on top of the first layer. This gives another hexagonal 183
layer displaced from the first layer by
1
3
,
2
3
,
1
2
. Then the third layer
184
is placed above the first layer (i.e., at (0,0,1)). The stacking is then 185
repeated ABABAB ....IfonestacksABCABC..., where C is the 186
hexagonal array obtained by stacking the third layer in the other set 187
of triangular holes above the set B (instead of the set A), one gets 188
an fcc lattice. The closest possible packing of the hcp atoms occurs 189
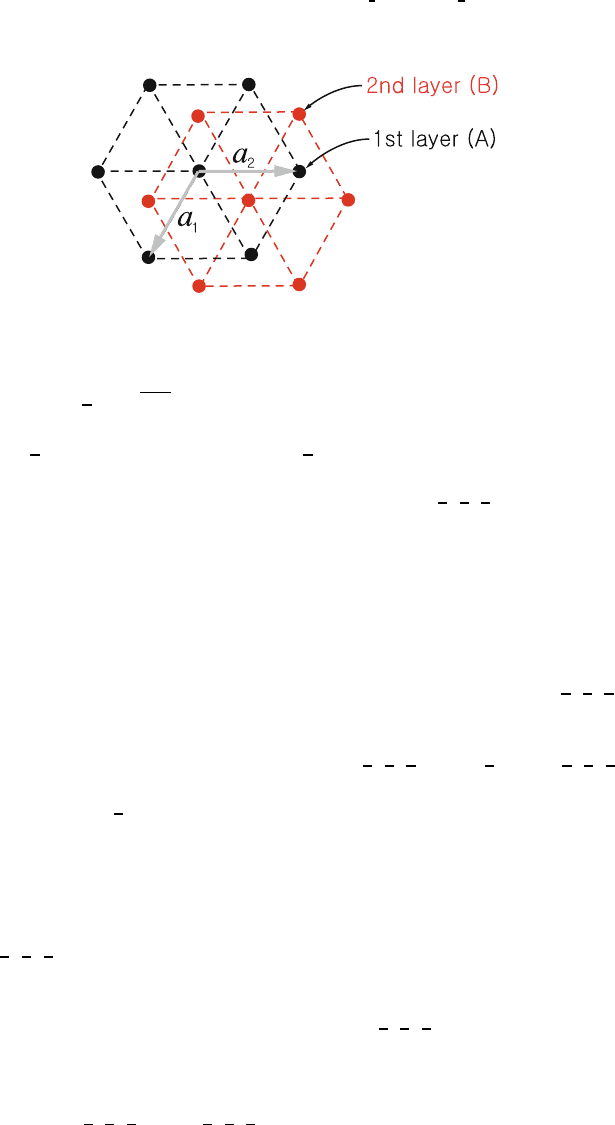
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.2 Common Crystal Structures 13
Fig. 1.14. Stacking of layers A and B in a hexagonal close packed crystal of lattice
constants a
1
, a
2
,andc
when
c
a
=
8/3 ≈ 1.633. We leave this as an exercise for the reader. 190
Zn crystalizes in a hcp lattice with a =2.66
˚
Aand c =4.96
˚
Agiving 191
c
a
≈ 1.85, larger than the ideal
c
a
value. 192
3. Zincblende Structure This is a non-Bravais lattice. It is an FCC with two 193
atoms per primitive unit cell located at (0,0,0) and
1
4
,
1
4
,
1
4
. The structure
194
can be viewed as two interpenetrating fcc lattices displaced by one fourth 195
of the body diagonal. Examples of the zincblende structure are ZnS (cubic 196
phase), ZnO (cubic phase), CuF, CuCl, ZnSe, CdS, GaN (cubic phase), 197
InAs, and InSb. The metallic ions are on one sublattice, the other ions on 198
the second sublattice. 199
4. Diamond Structure This structure is identical to the zincblende structure, 200
except that there are two identical atoms in the unit cell. This struc- 201
ture (unlike zincblende) has inversion symmetry about the point
1
8
,
1
8
,
1
8
.
202
Diamond, Si, Ge, and gray tin are examples of the diamond structure. 203
5. Wurtzite Structure This structure is a simple hexagonal lattice with 204
four atoms per unit cell, located at (0,0,0),
1
3
,
2
3
,
1
2
,
0, 0,
3
8
,and
1
3
,
2
3
,
7
8
.
205
It can be pictured as consisting of two interpenetrating hcp lattices sepa- 206
rated by
0, 0,
3
8
. In the wurtzite phase of ZnS, the Zn atoms sit on one hcp
207
lattice and the S atoms on the other. ZnS, BeO, ZnO (hexagonal phase), 208
CdS, GaN (hexagonal phase), and AlN are materials that can occur in the 209
wurtzite structure. 210
6. Sodium Chloride Structure It consists of a face centered cubic lattice with 211
a basis of two unlike atoms per primitive unit cell, located at (0,0,0) and 212
1
2
,
1
2
,
1
2
. In addition to NaCl, other alkali halide salts like LiH, KBr, RbI
213
form crystals with this structure. 214
7. Cesium Chloride Structure It consists of a simple cubic lattice with two 215
atoms per unit cell, located at (0,0,0) and
1
2
,
1
2
,
1
2
. Besides CsCl, CuZn
216
(β-brass), AgMg, and LiHg occur with this structure. 217
8. Calcium Fluoride Structure It consists of a face centered cubic lattice with 218
three atoms per primitive unit cell. The Ca ion is located at (0,0,0), the F 219
atoms at
1
4
,
1
4
,
1
4
and
3
4
,
3
4
,
3
4
.
220
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
14 1 Crystal Structures
Fig. 1.15. Stacking of layers A and B in a graphite structure
9. Graphite Structure This structure consists of a simple hexagonal lat- 221
tice with four atoms per primitive unit cell, located at (0,0,0),
2
3
,
1
3
, 0
, 222
0, 0,
1
2
,and
1
3
,
2
3
,
1
2
. It can be pictured as two interpenetrating HCP lat-
223
tices separated by
0, 0,
1
2
. It, therefore, consists of tightly bonded planes
224
(as shown in Fig. 1.15) stacked in the sequence ABABAB .... The individ- 225
ual planes are very tightly bound, but the interplanar binding is rather 226
weak. This gives graphite its well-known properties, like easily cleaving 227
perpendicular to the c-axis. 228
Miller Indices 229
Miller indices are a set of three integers that specify the orientation of a crystal 230
plane. The procedure for obtaining Miller indices of a plane is as follows: 231
1. Find the intercepts of the plane with the crystal axes. 232
2. Take the reciprocals of the three numbers. 233
3. Reduce (by multiplying by the same number) this set of numbers to the 234
smallest possible set of integers. 235
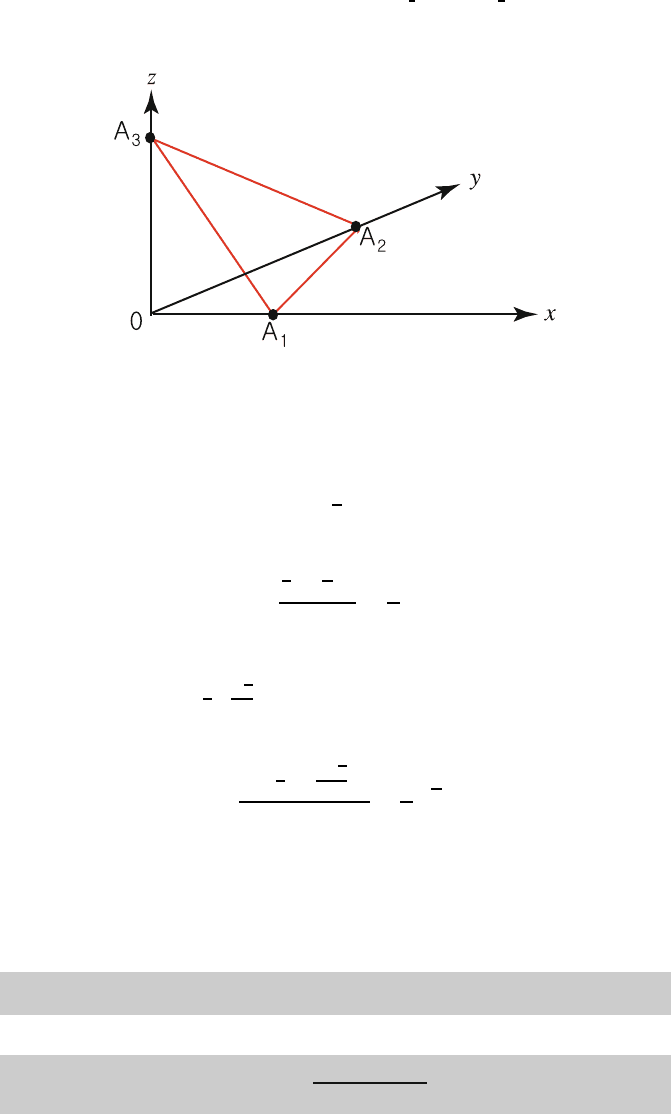
As an example, consider the plane that intersects the cubic axes at A
1
,A
2
,A
3
236
as shown in Fig. 1.16. Then x
i
a
i
= OA
i
. The reciprocals of (x
1
,x
2
,x
3
) 237
are
x
−1
1
,x
−1
2
,x
−1
3
, and the Miller indices of the plane are (h
1
h
2
h
3
)=238
p
x
−1
1
,x
−1
2
,x
−1
3
,where(h
1
h
2
h
3
) are the smallest possible set of integers 239
p
x
1
,
p
x
2
,
p
x
3
.
240
Indices of a Direction 241
A direction in the lattice can be specified by a vector V = u
1
a
1
+u
2
a
2
+u
3
a
3
, 242
or by the set of integers [u
1
u
2
u
3
]chosentohavenocommonintegralfactor.For 243
cubic lattices the plane (h
1
h
2
h
3
) is perpendicular to the direction [h
1
h
2
h
3
], 244
but this is not true for general lattices. 245
Packing Fraction 246
The packing fraction of a crystal structure is defined as the ratio of the volume 247
of atomic spheres in the unit cell to the volume of the unit cell. 248
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.3 Reciprocal Lattice 15
Fig. 1.16. Intercepts of a plane with the crystal axes
Examples 249
1. Simple cubic lattice: 250
We take the atomic radius as R =
a
2
(then neighboring atoms just touch). 251
The packing fraction p will be given by 252
p =
4
3
π
a
2
3
a
3
=
π
6
≈ 0.52
253
2. Body centered cubic lattice: 254
Here, we take R =
1
2
√
3
2
a
, i.e., half the nearest neighbor distance. For 255
the non-primitive cubic cell of edge a, we have two atoms per cell giving 256
p =
2 ×
4
3
π
a
√
3
4
3
a
3
=
π
8
√
3 ≈ 0.68
1.3 Reciprocal Lattice 257
If a
1
, a
2
, a
3
are the primitive translations of some lattice, we can define the 258
vectors b
1
, b
2
, b
3
by the condition 259
a
i
·b
j
=2πδ
ij
, (1.5)
260
where δ
ij
=0ifi is not equal to j and δ
ii
= 1. It is easy to see that 261
b
i
=2π
a
j
× a
k
a
i
·(a
j
× a
k
)
, (1.6)
262

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
16 1 Crystal Structures
where i, j,andk are different. The denominator a
i
· (a
j
× a
k
) is simply the 263
volume v
0
of the primitive unit cell. The lattice formed by the primitive trans- 264
lation vectors b
1
, b
2
, b
3
is called the reciprocal lattice (reciprocal to the lattice 265
formed by a
1
, a
2
, a
3
), and a reciprocal lattice vector is given by 266
G
h
1
h
2
h
3
= h
1
b
1
+ h
2
b
2
+ h
3
b
3
. (1.7)
267
Useful Properties of the Reciprocal Lattice 268
1. If r = n
1
a
1
+ n
2
a
2
+ n
3
a
3
is a lattice vector, then we can write r as 269
r =
i
(r · b
i
) a
i
. (1.8)
270
2. The lattice reciprocal to b
1
, b
2
, b
3
is a
1
, a
2
, a
3
. 271
3. A vector G
h
from the origin to a point (h
1
,h
2
,h
3
) of the reciprocal lattice 272
is perpendicular to the plane with Miller indices (h
1
h
2
h
3
). 273
4. The distance from the origin to the first lattice plane (h
1
h
2
h
3
)is274
d (h
1
h
2
h
3
)=2π |G
h
|
−1
. This is also the distance between neighboring 275
{h
1
h
2
h
3
} planes. 276
The proof of 3 is established by demonstrating that G
h
is perpendicular to 277
the plane A
1
A
2
A
3
shown in Fig. 1.16. This must be true if G
h
is perpendic- 278
ular to both A
1
A
2
and to A
2
A
3
.ButA
1
A
2
= OA
2
− OA
1
= p
a
2
h
2
−
a
1
h
1
.
279
Therefore, 280
G
h
· A
1
A
2
=(h
1
b
1
+ h
2
b
2
+ h
3
b
3
) · p
a
2
h
2
−
a
1
h
1
, (1.9)
which vanishes. The same can be done for
A
2
A
3
. The proof of 4 is established 281
by noting that 282
d(h
1
h
2
h
3
)=
a
1
h
1
·
G
h
|G
h
|
.
The first factor is just the vector
OA
1
for the situation where p =1,and283
the second factor is a unit vector perpendicular to the plane (h
1
h
2
h
3
). Since 284
a
1
· G
h
=2πh
1
, it is apparent that d(h
1
h
2
h
3
)=2π |G
h
|
−1
. 285
1.4 Diffraction of X-Rays 286
Crystal structures are usually determined experimentally by studying how 287
the crystal diffracts waves. Because the interatomic spacings in most crystals 288
are of the order of a few
˚
As(1
˚
A =10
−8
cm), the maximum information 289
can most readily be obtained by using waves whose wave lengths are of that 290

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.4 Diffraction of X-Rays 17
order of magnitude. Electromagnetic, electron, or neutron waves can be used 291
to study diffraction by a crystal. For electromagnetic waves, E = hν,where 292
E is the energy of the photon, ν =
c
λ
is its frequency and λ its wave length, 293
and h is Planck’s constant. For λ =10
−8
cm, c =3× 10
10
cm/sandh = 294
6.6 × 10
−27
erg · s, the photon energy is equal to roughly 2 × 10
−8
ergs or 295
1.24 × 10
4
eV. Photons of energies of tens of kilovolts are in the X-ray range. 296
For electron waves, p =
h
λ
6.6 × 10
−19
g · cm/swhenλ =10
−8
cm. This 297
gives E =
p
2
2m
e
,wherem
e
0.9 × 10
−27
g, of 2.4 × 10
−10
ergs or roughly 298
150 eV. For neutron waves, we need simply replace m
e
by m
n
=1.67×10
−24
g 299
to obtain E =1.3 × 10
−13
ergs 0.08 eV. Thus neutron energies are of the 300
order of a tenth of an eV. Neutron scattering has the advantages that the low 301
energy makes inelastic scattering studies more accurate and that the magnetic 302
moment of the neutron allows the researcher to obtain information about 303
the magnetic structure. It has the disadvantage that high intensity neutron 304
sources are not as easily obtained as X-ray sources. 305
1.4.1 Bragg Reflection 306
We have already seen that we can discuss crystal planes in a lattice structure. 307
Assume that an incident X-ray is specularly reflected by a set of crystal planes 308
as shown in Fig. 1.17. Constructive interference occurs when the difference in 309
path length is an integral number of wave length λ. It is clear that this occurs 310
when 311
2d sin θ = nλ, (1.10)
312
where d is the interplane spacing, θ is the angle between the incident beam 313
and the crystal planes, as is shown on the figure, and n is an integer. Equation 314
(1.10) is known as Bragg’s law. 315
1.4.2 Laue Equations 316
A slightly more elegant discussion of diffraction from a crystal can be obtained 317
as follows: 318
θ
θ
INCIDENT
WAVE
REFLECTED
WAVE
Fig. 1.17. Specular reflection of X-rays by a set of crystal planes separated by a
distance d
