Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
18 1 Crystal Structures
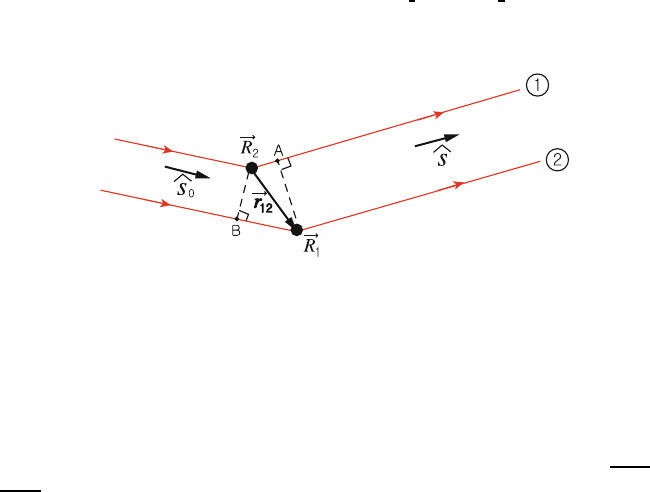
Fig. 1.18. Scattering of X-rays by a pair of atoms in a crystal
1. Let ˆs
0
be a unit vector in the direction of the incident wave, and ˆs be a 319
unit vector in the direction of the scattered wave. 320
2. Let R
1
and R
2
be the position vectors of a pair of atoms in a Bravais 321
lattice, and let r
12
= R
1
− R
2
. 322
Let us consider the waves scattered by R
1
and by R
2
and traveling different 323
path lengths as shown in Fig. 1.18. The difference in path length is | R
2
A − 324
BR
1
|. But this is clearly equal to |r
12
· ˆs − r
12
· ˆs
0
|. We define S as S =ˆs−ˆs
0
; 325
then the difference in path length for the two rays is given by 326
Δ=|r
12
· S|. (1.11)
For constructive interference, this must be equal to an integral number of
327
wave length. Thus, we obtain 328
r
12
· S = mλ, (1.12)
where m is an integer and λ is the wave length. To obtain constructive inter-
329
ference from every atom in the Bravais lattice, this must be true for every 330
lattice vector R
n
. Constructive interference will occur only if 331
R
n
·S = integer × λ (1.13)
for every lattice vector R
n
in the crystal. Of course there will be different 332
integers for different R
n
in general. Recall that 333
R
n
= n
1
a
1
+ n
2
a
2
+ n
3
a
3
. (1.14)
The condition (1.13) is obviously satisfied if
334
a
i
· S = ph
i
λ, (1.15)
where h
i
is the smallest set of integers and p is a common multiplier. We can 335
obviously express S as 336
S =(S · a
1
) b
1
+(S · a
2
) b
2
+(S · a
3
) b
3
. (1.16)

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.4 Diffraction of X-Rays 19
θ
θ
Fig. 1.19. Relation between the scattering vector S =ˆs −ˆs
0
and the Bragg angle θ
Therefore, condition (1.13) is satisfied and constructive interference from every 337
lattice site occurs if 338
S = p (h
1
b
1
+ h
2
b
2
+ h
3
b
3
) λ, (1.17)
or
339
S
λ
= pG
h
, (1.18)
340
where G
h
is a vector of the reciprocal lattice. Equation (1.18) is called the 341
Laue equation. 342
Connection of Laue Equations and Bragg’s Law 343
From (1.18) S must be perpendicular to the planes with Miller indices 344
(h
1
h
2
h
3
). The distance between two planes of this set is 345
d(h
1
h
2
h
3
)=
2π
|G
h
|
= p
λ
|S|
. (1.19)
We know that S is normal to the reflection plane PP
with Miller indices 346
(h
1
h
2
h
3
). From Fig. 1.19, it is apparent that |S| =2sinθ. Therefore, (1.19) 347
can be written by 348
2d(h
1
h
2
h
3
)sinθ = pλ,
where p is an integer. According to Laue’s equation, associated with any
349
reciprocal lattice vector G
h
= h
1
b
1
+h
2
b
2
+h
3
b
3
, there is an X-ray reflection 350
satisfying the equation λ
−1
S = pG
h
,wherep is an integer. 351
1.4.3 Ewald Construction 352
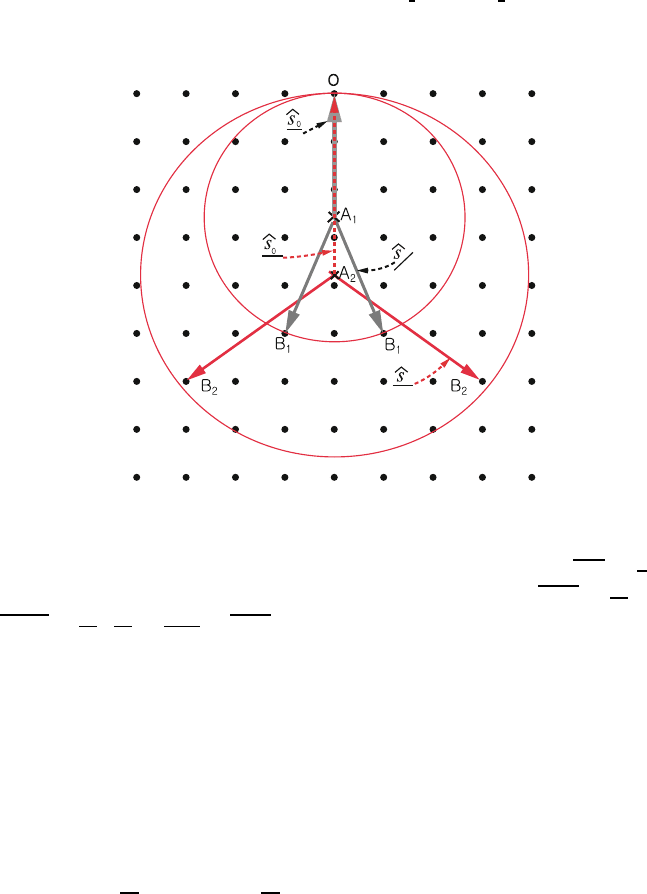
This is a geometric construction that illustrates how the Laue equation works. 353
The construction goes as follows: See Fig. 1.20. 354
1. From the origin O of the reciprocal lattice draw the vector AO of length 355
λ
−1
parallel to ˆs
0
and terminating on O. 356
2. Construct a sphere of radius λ
−1
centered at A. 357
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
20 1 Crystal Structures
λ
2
λ
1
λ
1
λ
2
Fig. 1.20. Ewald construction for diffraction peaks
If this sphere intersects a point B of the reciprocal lattice, then AB =
ˆs
λ
is 358
in a direction in which a diffraction maximum occurs. Since A
1
O =
ˆs
0
λ
1
and 359
A
1
B
1
=
ˆs
λ
1
,
S
λ
1
=
ˆs−ˆs
0
λ
1
= OB
1
is a reciprocal lattice vector and satisfies the 360
Laue equation. If a higher frequency X-ray is used, λ
2
, A
2
,andB
2
replace λ
1
, 361
A
1
,andB
1
. For a continuous spectrum with λ
1
≥ λ ≥ λ
2
, all reciprocal lattice 362
points between the two sphere (of radii λ
−1
1
and λ
−1
2
) satisfy Laue equation 363
for some frequency in the incident beam. 364
Wave Vector 365
It is often convenient to use the set of vectors K
h
=2πG
h
. Then, the Ewald 366
construction gives 367
q
0
+ K
h
= q, (1.20)
where q
0
=
2π
λ
ˆs
0
and q =
2π
λ
ˆs are the wave vectors of the incident and 368
scattered waves. Equation (1.20) says that wave vector is conserved up to 2π 369
times a vector of the reciprocal lattice. 370
1.4.4 Atomic Scattering Factor 371
It is the electrons of an atom that scatter the X-rays since the nucleus is so 372
heavy that it hardly moves in response to the rapidly varying electric field of 373
the X-ray. So far, we have treated all of the electrons as if they were localized 374
at the lattice point. In fact, the electrons are distributed about the nucleus of 375
the atom (at position r = 0, the lattice point) with a density ρ(r). If you know 376
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
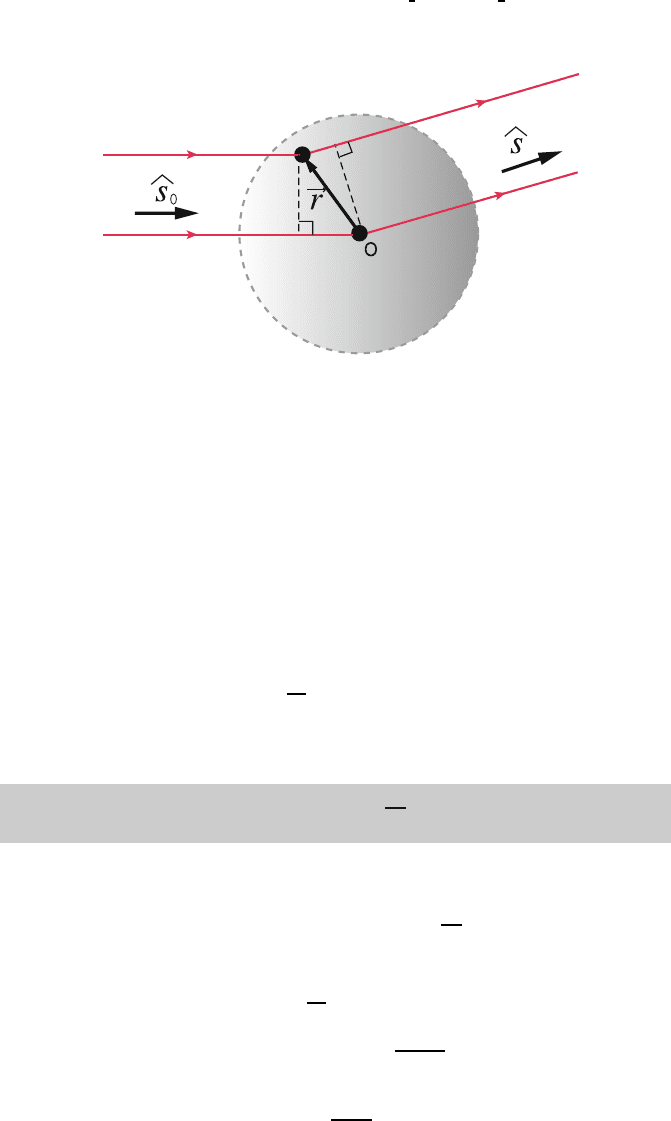
1.4 Diffraction of X-Rays 21
Fig. 1.21. Path difference between waves scattered at O and those at r
the wave function Ψ (r
1
, r
2
,...,r
z
) describing the z electrons of the atom, ρ(r) 377
is given by 378
ρ(r)=
z
i=1
δ (r − r
i
)
=
Ψ(r
1
,...,r
z
)
z
i=1
δ(r − r
i
)
Ψ(r
1
,...,r
z
)
.
(1.21)
Now, consider the difference in path length Δ between waves scattered at O
379
and those scattered at r (Fig. 1.21). 380
Δ=r · (ˆs − ˆs
0
)=r · S. (1.22)
The phase difference is simply
2π
λ
times Δ, the difference in path length. 381
Therefore, the scattering amplitude will be reduced from the value obtained 382
by assuming all the electrons were localized at the origin O by a factor z
−1
f, 383
where f is given by 384
f =
d
3
rρ(r)e
2πi
λ
r·S
. (1.23)
385
This factor is called the atomic scattering factor.Ifρ(r) is spherically sym- 386
metric we have 387
f =
∞
0
1
−1
2πr
2
dr d(cos φ)ρ(r)e
2πi
λ
Sr cos φ
. (1.24)
Recall that S =2sinθ,whereθ is the angle between ˆs
0
and the reflecting 388
plane PP
of Fig. 1.19. Define μ as
4π
λ
sin θ;thenf can be expressed as 389
f =
∞
0
dr4πr
2
ρ(r)
sin μr
μr
. (1.25)
If λ is much larger than the atomic radius, μr is much smaller than unity
390
wherever ρ(r) is finite. In that case
sin μr
μr
1andf → z,thenumberof 391
electrons. 392

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
22 1 Crystal Structures
1.4.5 Geometric Structure Factor 393
So far we have considered only a Bravais lattice. For a non-Bravais lattice the 394
scattered amplitude depends on the locations and atomic scattering factors 395
of all the atoms in the unit cell. Suppose a crystal structure contains atoms 396
at positions r
j
with atomic scattering factors f
j
. It is not difficult to see that 397
this changes the scattered amplitude by a factor 398
F (h
1
,h
2
,h
3
)=
j
f
j
e
2πi
λ
r
j
·S(h
1
h
2
h
3
)
(1.26)
for the scattering from a plane with Miller indices (h
1
h
2
h
3
). In (1.26) the 399
position vector r
j
of the jth atom can be expressed in terms of the primitive 400
translation vectors a
i
401
r
j
=
i
μ
ji
a
i
. (1.27)
Forexample,inahcplatticer
1
=(0, 0, 0) and r
2
=(
1
3
,
2
3
,
1
2
) when expressed 402
in terms of the primitive translation vectors. Of course, S(h
1
h
2
h
3
)equalto 403
λ
i
h
i
b
i
,whereb
i
are primitive translation vectors in the reciprocal lattice. 404
Therefore,
2πi
λ
r
j
·S(h
1
h
2
h
3
)isequalto2πi (μ
j1
h
1
+ μ
j2
h
2
+ μ
j3
h
3
), and the 405
structure amplitude F (h
1
,h
2
,h
3
) can be expressed as 406
F (h
1
,h
2
,h
3
)=
j
f
j
e
2πi
i
μ
ji
h
i
. (1.28)
If all of the atoms in the unit cell are identical (as in diamond, Si, Ge, etc.)
407
all of the atomic scattering factors f
j
are equal, and we can write 408
F (h
1
,h
2
,h
3
)=fS(h
1
h
2
h
3
). (1.29)
409
The S(h
1
h
2
h
3
) is called the geometric structure amplitude. It depends only 410
on crystal structure, not on the atomic constituents, so it is the same for all 411
hcp lattices or for all diamond lattices, etc. 412
Example 413
A useful demonstration of the geometric structure factor can be obtained by 414
considering a bcc lattice as a simple cubic lattice with two atoms in the simple 415
cubic unit cell located at (0,0,0) and (
1
2
,
1
2
,
1
2
). Then 416
S(h
1
h
2
h
3
)=1+e
2πi
(
1
2
h
1
+
1
2
h
2
+
1
2
h
3
)
. (1.30)
If h
1
+h
2
+h
3
is odd, e
iπ(h
1
+h
2
+h
3
)
= −1andS(h
1
h
2
h
3
)vanishes.Ifh
1
+h
2
+ 417
h
3
is even, S(h
1
h
2
h
3
) = 2. The reason for this effect is that the additional 418
planes (associated with the body centered atoms) exactly cancel the scattering 419
amplitude from the planes made up of corner atoms when h
1
+ h
2
+ h
3
is odd, 420
but they add constructively when h
1
+ h
2
+ h
3
is even. 421
The scattering amplitude depends on other factors (e.g. thermal motion 422
and zero point vibrations of the atoms), which we have neglected by assuming 423
a perfect and stationary lattice. 424

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.4 Diffraction of X-Rays 23
1.4.6 Experimental Techniques 425
We know that constructive interference from a set of lattice planes separated 426
by a distance d will occur when 427
2d sin θ = nλ, (1.31)
where θ is the angle between the incident beam and the planes that are scat-
428
tering, λ is the X-ray wave length, and n is an integer. For a given crystal 429
thepossiblevaluesofd are fixed by the atomic spacing, and to satisfy (1.31), 430
one must vary either θ or λ over a range of values. Different experimental 431
methods satisfy (1.31) in different ways. The common techniques are (1) the 432
Laue method,(2)therotating crystal method, and (3) the powder method. 433
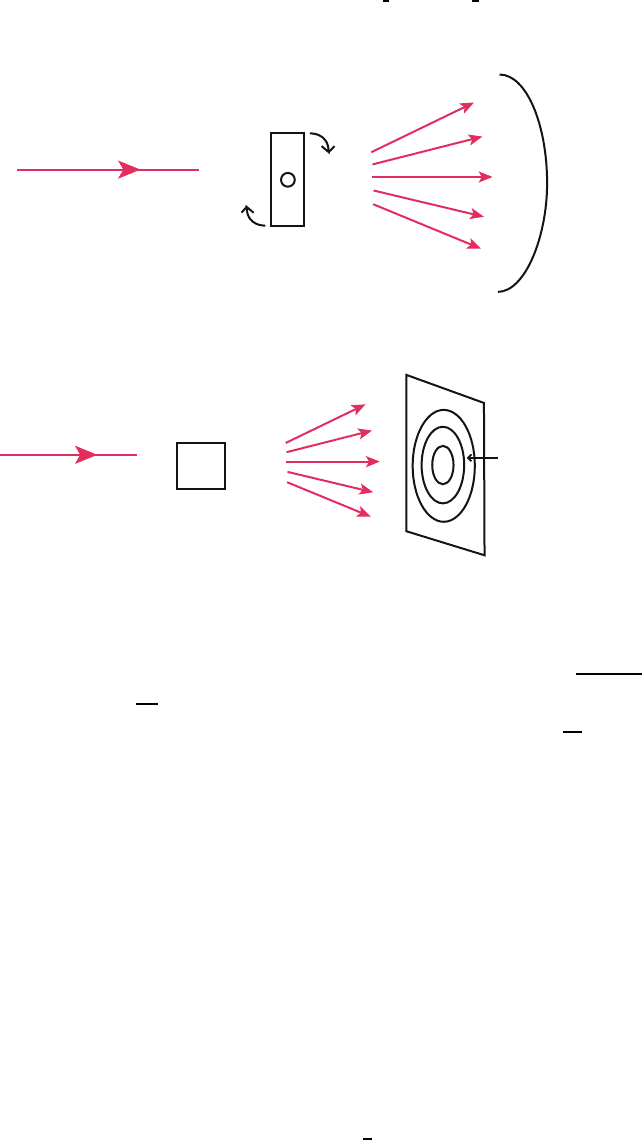
Laue Method 434
In this method a single crystal is held stationary in a beam of continuous wave 435
length X-ray radiation (Fig. 1.22). Various crystal planes select the appropri- 436
ate wave length for constructive interference, and a geometric arrangement of 437
bright spots is obtained on a film. 438
Rotating Crystal Method 439
In this method a monochromatic beam of X-ray is incident on a rotating 440
single crystal sample. Diffraction maxima occur when the sample orientation 441
relative to the incident beam satisfies Bragg’s law (Fig. 1.23). 442
Powder Method 443
Here, a monochromatic beam is incident on a finely powdered specimen. The 444
small crystallites are randomly oriented with respect to the incident beam, 445
so that the reciprocal lattice structure used in the Ewald construction must 446
be rotated about the origin of reciprocal space through all possible angles. 447
This gives a series of spheres in reciprocal space of radii K
1
, K
2
, ... (we 448
include the factor 2π in these reciprocal lattice vectors) equal to the smallest, 449
COLLIMATOR
SAMPLE
FILM
X - RAY
BEAM
SPOT
PATTERN
Fig. 1.22. Experimental arrangement of the Laue method
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
24 1 Crystal Structures
ROTATABLE
SAMPLE
MONOCHROMATIC
X-RAY BEAM
FILM
Fig. 1.23. Experimental arrangement of the rotating crystal method
POWDER
SAMPLE
FILM
X - RAY BEAM
DIFFRACTION
RINGS
Fig. 1.24. Experimental arrangement of the powder method
next smallest, etc. reciprocal lattice vectors. The sequence of values
sin(φ
i
/2)
sin(φ
1
/2)
450
give the ratios of
K
i
K
1
for the crystal structure. This sequence is determined 451
by the crystal structure. Knowledge of the X-ray wave length λ =
2π
k
allows 452
determination of the lattice spacing (Fig. 1.24). 453
1.5 Classification of Solids 454
1.5.1 Crystal Binding 455
Before considering even in a qualitative way how atoms bind together to 456
form crystals, it is worthwhile to review briefly the periodic table and the 457
ground state configurations of atoms. The single particle states of electrons 458
moving in an effective central potential (which includes the attraction of the 459
nucleus and some average repulsion associated with all other electrons) can 460
be characterized by four quantum numbers: n, the principal quantum number 461
takes on the values 1, 2, 3,..., l, the angular momentum quantum number 462
takes on values 0, 1,...,n−1; m, the azimuthal quantum number (projection 463
of l onto a given direction) is an integer satisfying −l ≤ m ≤ l;andσ,the 464
spin quantum number takes on the values ±
1
2
. 465

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.5 Classification of Solids 25
The energy of the single particle orbital is very insensitive to m and σ (in 466
the absence of an applied magnetic field), but it depends strongly on n and l. 467
Of course, due to the Pauli principle only one electron can occupy an orbital 468
with given n, l, m, and σ. The periodic table is constructed by making an 469
array of slots, with l value increasing from l = 0 as one moves to the left, and 470
the value of n + l increasing as one moves down. (Table 1.4) Of course, the 471
correct number of slots must be allowed to account for the spin and azimuthal 472
degeneracy 2(2l +1)of a givenl value. One then begins filling the slots from 473
the top left, moving to the right, and then down when all slots of a given 474
(n + l) value have been used up. See Table 1.4, which lists the atoms (H, 475
He, ...) and their atomic numbers in the appropriate slots. As the reader can 476
readily observe, H has one electron, and it will occupy the n =1,l =0(1s) 477
state. Boron has five electrons and they will fill the (1s)and2s states with the 478
fifth electron in the 2p state. Everything is very regular until Cr and Cu. These 479
two elements have ground states in which one 4s electron falls into the 3d shell, 480
giving for Cr the atomic configuration (1s)
2
(2s)
2
(2p)
6
(3s)
2
(3p)
6
(4s)
1
(3d)
5
, 481
and for Cu the atomic configuration (1s)
2
(2s)
2
(2p)
6
(3s)
2
(3p)
6
(4s)
1
(3d)
10
. 482
Other exceptions occur in the second transition series (the filling of the 4d 483
levels) and in the third transition series (filling the 5d levels), and in the rare 484
earth series (filling the 4f and 5f levels). Knowing this table allows one to 485
write down the ground state electronic configuration of any atom. Note that 486
the inert gases He, Ne, Kr, Rn, complete the shells n =1,n =2,n =3,and 487
n = 4, respectively. Ar and Xe are inert also; they complete the n =3shell 488
(except for 3d electrons), and n = 4 shell (except for 4f electrons), respec- 489
tively. Na, K, Rb, Cs, and Fr have one weakly bound s electron outside these 490
closed shell configurations; Fl, Cl, Br, I and At are missing one p electron from 491
the closed shell configurations. The alkali metals easily give up their loosely 492
bound s electrons, and the halogens readily attract one p electron to give a 493
closed shell configuration. The resulting Na
+
− Cl
−
ions form an ionic bond 494
which is quite strong. Atoms like C, Si, Ge, and Sn have an (np)
2
(n +1s)
2
495
configuration. These four valence electrons can be readily shared with other 496
atoms in covalent bonds, which are also quite strong.
1
Compounds like GaAs, 497
1
In Table 1.4, we note exceptions (i)–(vii):
i Dropping a 4s electron into the 3d shell while filling 3d shell(Cr,Cu)
ii Dropping a 5s electron into the 4d shell while filling 4d shell(Nb,Mo,Ru,Rh,
Ag)
iii Dropping both 5s electrons into the 4d shell while filling 4d shell (Pd)
iv Dropping both 6s electrons into the 5d shell while filling 5d shell (Pt)
v Dropping one 6s electron into the 5d shell while filling 5d shell (Au)
vi Adding one 5d electron before filling the entire 4f shell(La,Gd)
vii Adding one 6d electron before filling the entire 5f shell(Ac,Pa,U,Cm,Cf)
viii (h) Adding two 5d electrons before filling the entire 5f shell (Th, Bk)

Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
26 1 Crystal Structures
t4.1 Tabl e 1. 4. Ground state electron configurations in a periodic table: Note that n =1, 2, 3,...; l<n; m = −l, −l +1,...,l; σ = ±
1
2
t4.2 ← ln + l ↓
t4.3
l =0
t4.4
1
H
2
He
1
(1s)
t4.5 l =1
3
Li
4
Be
2
(2s)
t4.6
5
B
6
C
7
N
8
O
9
F
10
Ne
11
Na
12
Mg
3
(2p, 3s)
t4.7 l =2
13
Al
14
Si
15
P
16
S
17
Cl
18
Ar
19
K
20
Ca
4
(3p, 4s)
t4.8
21
Sc
22
Ti
23
V
24
(i)
Cr
25
Mn
26
Fe
27
Co
28
Ni
29
(i)
Cu
30
Zn
31
Ga
32
Ge
33
As
34
Se
35
Br
36
Kr
37
Rb
38
Sr
5
(4p, 5s)
t4.9 l =3
39
Y
40
Zr
41
(ii)
Nb
42
(ii)
Mo
43
Tc
44
(ii)
Ru
45
(ii)
Rh
46
(iii)
Pd
47
(ii)
Ag
48
Cd
49
In
50
Sn
51
Sb
52
Te
53
I
54
Xe
55
Cs
56
Ba
6
(4d, 5p, 6s)
t4.10
57
(vi)
La
58
Ce
59
Pr
60
Nd
61
Pm
62
Sm
63
Eu
64
(vi)
Gd
65
Tb
66
Dy
67
Ho
68
Er
69
Tm
70
Yb
71
Lu
72
Hf
73
Ta
74
W
75
Re
76
Os
77
Ir
78
(iv)
Pt
79
(v)
Au
80
Hg
81
Tl
82
Pb
83
Bi
84
Po
85
At
86
Rn
87
Fr
88
Ra
7
(4f, 5d, 6p, 7s)
t4.11
89
(vii)
Ac
90
(viii)
Th
91
(vii)
Pa
92
(vii)
U
93
Np
94
Pu
95
Am
96
(vii)
Cm
97
(viii)
Bk
98
(vii)
Cf
99
Es
100
F
101
Md
102
No
8
(5f, 6d, 7p, 8s)
We note exceptions (i)–(vii):
i Dropping a 4s electron into the 3d shell while filling 3d shell(Cr,Cu)
ii Dropping a 5s electron into the 4d shell while filling 4d shell(Nb,Mo,Ru,Rh,Ag)
iii Dropping both 5s electrons into the 4d shell while filling 4d shell (Pd)
iv Dropping both 6s electrons into the 5d shell while filling 5d shell (Pt)
v Dropping one 6s electron into the 5d shell while filling 5d shell (Au)
vi Adding one 5d electron before filling the entire 4f shell(La,Gd)
vii Adding one 6d electron before filling the entire 5f shell(Ac,Pa,U,Cm,Cf)
viii (h) Adding two 5d electrons before filling the entire 5f shell (Th, Bk)
Uncorrected Proof
BookID 160928 ChapID 01 Proof# 1 - 29/07/09
1.6 Binding Energy of Ionic Crystals 27
GaP, GaSb, or InP, InAs, InSb, etc., are formed from column III and column 498
V constituents. With the partial transfer of an electron from As to Ga, one 499
obtains the covalent bonding structure of Si or Ge as well as some degree of 500
ionicity as in NaCl. Metallic elements like Na and K are relatively weakly 501
bound. Their outermost s electrons become almost free in the solid and act as 502
a glue holding the positively charged ions together. The weakest bonding in 503
solids is associated with weak Van der Waals coupling between the constituent 504
atoms or molecules. To give some idea of the binding energy of solids, we will 505
consider the binding of ionic crystals like NaCl or CsCl. 506
1.6 Binding Energy of Ionic Crystals 507
The binding energy of ionic crystals results primarily from the electrostatic 508
interaction between the constituent ions. A rough order of magnitude estimate 509
of the binding energy per molecule can be obtained by simply evaluating 510
V =
e
2
R
0
=
4.8 × 10
−10
esu
2
2.8 × 10
−8
cm
8 × 10
−12
ergs ∼ 5eV.
Here, R
0
is the observed interatomic spacing (which we take as 2.8
˚
A,the 511
spacing in NaCl). The experimentally measured value of the binding energy 512
of NaCl is almost 8 eV per molecule, so our rough estimate is not too bad. 513
Interatomic Potential 514
For an ionic crystal, the potential energy of a pair of atoms i, j can be taken 515
to be 516
φ
ij
= ±
e
2
r
ij
+
λ
r
n
ij
. (1.32)
Here, r
ij
is the distance between atoms i and j.The± sign depends on 517
whether the atoms are like (+) or unlike (−). The first term is simply the 518
Coulomb potential for a pair of point charges separated by r
ij
. The second 519
term accounts for core repulsion. The atoms or ions are not point charges, and 520
when a pair of them gets close enough together their core electrons can repel 521
one another. This core repulsion is expected to decrease rapidly with increas- 522
ing r
ij
. The parameters λ and n are phenomenological; they are determined 523
from experiment. 524
Total Energy 525
The total potential energy is given by 526
U =
1
2
i=j
φ
ij
. (1.33)
