Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

on control coeffi cients for the various enzymes as opposed to the simpler view
of rate limiting enzymes. How is the activity of the cycle adjusted to various
conditions in a given tissue such as muscle, and how is the cycle ’ s capacity
regulated in different tissues?
Before some answers to these questions can be proposed, a presentation of
the cycle and a limited discussion of the individual steps are in order. It is
logical to start with one reaction outside of the cycle that produces the impor-
tant intermediate acetyl - CoA. The overall reaction is
pyruvate CoA NAD NADH acetyl-CoA CO
+
2
++ → + +
The enzyme involved is a multienzyme complex named pyruvate dehydroge-
nase, consisting of 132 subunits (30 E
1
dimers, 60 E
2
dimers, and 6 E
3
dimers in
mammals) with a total molecular mass of several hundred thousand kilodaltons.
Five coenzymes are necessary: thiamine pyrophosphate, lipoamide, CoA, FAD,
and NAD. Substrates and intermediates are shuttled between the subunits,
facilitated by the highly organized, three - dimensional structure of this complex.
For a detailed discussion of the structure and mechanism the reader is referred
to standard biochemistry textbooks, or to the original literature.
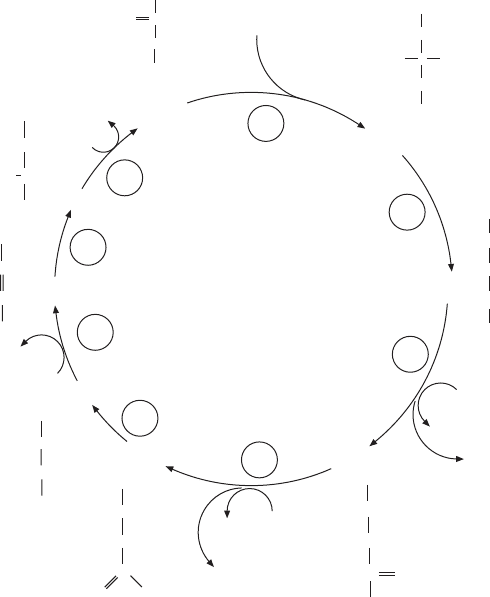
Figure 6.1 presents an overview of the intermediates and their sequence in
the citric acid cycle. In this scheme the individual reactions have been num-
bered, and some brief comments about salient features of each reaction and
the corresponding enzyme will have to suffi ce for the present treatment.
1. Citrate synthase catalyzes the condensation of acetyl - CoA with oxalo-
acetate to form citrate, a tricarboxylic acid from which a common name of the
cycle has been derived. The X - ray structure of the enzyme has been deter-
mined, and considerable detail about the mechanism is known. The reaction
can be described as a mixed aldol – Claisen condensation involving the stabili-
zation of the enol form of acetyl - CoA as an intermediate. Our current under-
standing of enzymes and their active sites also makes it clear why a specifi c
chiral carbon is formed with the carboxymethyl group from acetyl - CoA con-
stituting the pro - S arm. A consequence of this stereochemistry is that the two
carbons entering the cycle from acetyl - CoA will not be liberated as CO
2
during
the fi rst turn of the cycle.
2. Aconitase catalyzes the reversible interconversion between two struc-
tural isomers, citrate and isocitrate. Since citrate is prochiral, the two carboxy-
methyl groups of the symmetrical cityrate are distinguishable, and the hydroxyl
group is moved to the methylene carbon from oxaloacetate rather than to the
methylene group of the original acetyl - CoA. The enzyme is of special interest
because a nonheme iron – sulfur cluster ([4Fe – 4S]) participates in the active
site of the enzyme, although no electron transfer takes place. A specifi c Fe(II)
in the cluster serves to bind and stabilize the substrates and intermediates.
More recently it was discovered that a cytosolic aconitase also serves as a
sensor of iron in the cytosol. Before its identity was established, this
THE KREBS CYCLE 301
302 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
iron - responsive protein (IRP) had been shown to interact with iron - responsive
elements (IREs) on certain mRNAs. These IREs can be recognized as specifi c
hairpin structures. When bound to IREs in the 5 ′ untranslated region of the
mRNAs for ferritin or 5 - aminolevulinate synthase, it prevents translation of
these mRNAs. Cytosolic aconitase binds to IREs when the iron concentration
is low; at high iron concentrations, aconitase is more tightly folded around the
[4Fe – 4S] cluster and incapable of binding the mRNA. An IRE is also found
in the 3 ′ untranslated region of the transferrin receptor mRNA. Binding of
aconitase (low iron) prevents turnover of the mRNA. At high iron concentra-
tions, degradation of the transferrin receptor mRNA is promoted. Thus, aconi-
tase as the iron sensor controls the transport of iron into the cell, the storage
of iron, a step in heme biosynthesis, and probably other pathways related to
iron (see references 1 – 3 for recent reviews).
3. Isocitrate dehydrogenase occurs in two forms in mammalian tissues. An
NAD - dependent form is found exclusively in mitochondria; a second form
Figure 6.1 The TCA (tri carboxylic acid) cycle or Krebs cycle.
acetyl - CoA
citrate
isocitrate
α-ketoglutarate
succinyl - CoA
succinate
fumarate
malate
oxaloacetate
3
4
2
8
1
5
7
6
NAD
NADH
NAD
NADH
FAD
FADH
NAD
NADH
COO
CH
COO
CH
KREBS
CYCLE
COO
COO
O
C
CH
CH
COO
CH
COO
H
O
2
CH
COO
CH
C
COO
2
O
COO
CH
2
CH
2
C
O
SCoA
CH
2
COO
CH
2
COO
COO
COO
2
CH
CH
2
H
H-C-COO
COO
C
2
COO
H-C-OH
CO
2
CO
2
2
2
HO
C COO
utilizes NADP and is localized both in mitochondria and the cytosol. The reac-
tion produces the fi rst of two CO
2
molecules produced per turn of the cycle,
and it links a decarboxylation to the oxidation of an alcohol to a keto group.
The fi rst of three NADH molecules is also produced here.
4. The enzyme α - ketoglutarate dehydrogenase is a multifunctional enzyme
complex like the pyruvate dehydrogenase; and the third enzyme, dihydrolipoyl
dehydrogenase, is identical to that in PDH. The fi rst two subunits are similar,
with specifi city for the succinyl instead of acetyl moiety, and the fi nal product
is another high - energy thioester, succinyl - CoA. NADH and CO
2
are the other
products of the reaction. At this point, two CO
2
molecules have been pro-
duced, accounting for the total input of two carbons from acetyl - CoA, but the
stereochemical considerations related to the formation of citrate and the sub-
sequent reactions can account for the observations that no radioactive CO
2
is
produced from [
14
C] acetyl - CoA during the fi rst turn of the cycle.
5. Succinyl - CoA synthetase carries out a substrate - level phosphorylation
with the slight twist that GDP is the phosphate acceptor:
succinyl-CoA P GDP succinate GTP CoA
i
++ → + +
There is persuasive evidence from isotope tracer experiments that the
inorganic phosphate displaces the CoA to form succinyl - phosphate. The phos-
phate is transferred to GDP via a 3 - phospho - histidine intermediate on the
enzyme.
6. Succinate is oxidized to fumarate by the membrane - linked SDH complex.
A large (70 - kDa) fl avoprotein forms the active site, and a covalently linked
fl avin becomes the hydrogen acceptor to form FADH
2
. The fl avin is linked to
a histidine side chain in a highly conserved portion of the protein. It is imme-
diately reoxidized with the release of protons and the transfer of two electrons
to the iron – sulfur centers of the iron – protein subunit of SDH. The Ip subunit
has three nonheme iron – sulfur centers ([2Fe – 2S], [3Fe – 4S], and [4Fe – 4S]) and
constitutes a prime example of a protein conducting electrons in the absence
of a heme group. The two integral membrane proteins of complex II are associ-
ated with a cytochrome, but it is still controversial whether this cytochrome
participates in the conduit of the electrons to the ubiquinone (CoQ). Succinate
dehydrogenase is the only Krebs cycle enzyme directly linked to the electron
transfer chain. A more detailed discussion of this complex can be found in
Chapter 5 .
7. Fumarate is hydrated by the addition of H
2
O to the double bond, cata-
lyzed by fumarase. Controversy about the mechanism has centered on the
question of whether the addition of OH
–
is the fi rst step, leading to the forma-
tion of a carbanion that appears now to have been confi rmed by
18
O exchange
experiments.
8. The fi nal reaction catalyzed by malate dehydrogenase constitutes the last
oxidation step and hence production of another NADH.
THE KREBS CYCLE 303
304 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
A fi nal accounting of all the reactions with the inclusion of the pyruvate
dehydrogenase reactions leads to the following summation:
pyruvate P GDP NAD FAD 3 CO NADH FADH GTP
i
+
22
++ + + ⇒ + + +44
It should be reemphasized that pyruvate (C3) has become converted to three
CO
2
molecules without any consumption of oxygen. If a steady supply of
NAD
+
was available, glucose could thus be broken down to carbon dioxide in
the absence of oxygen. The cost of NAD
+
makes this impossible, and in reality
it has to be obtained by recycling. Hence, oxidation of NADH by complex I
of the electron transfer chain is essential to keep the citric acid cycle going.
Accumulation of NADH causes a strong inhibition of pyruvate dehydroge-
nase and α - ketoglutarate dehydrogenase, and the author ’ s laboratory has iso-
lated mammalian cell mutants with defects in the electron transfer chain based
on the almost total inhibition of the Krebs cycle (4) . Substrate and product
concentrations are the immediate controlling factors determining the fl ux
through the cycle, and ATP, ADP, and Ca
+2
are allosteric effectors of citric acid
cycle enzymes. Overall, oxygen consumption, NADH oxidation, and ATP syn-
thesis are tightly coupled not only to each other, but also to the citric acid
cycle by the above mechanisms.
In the end, Lavoisier was correct in viewing respiration as combustion, but
little did he suspect that many steps are necessary to make this “ burning ” of
glucose in a living cell such a controlled process. The many steps involved not
only ensure control over a “ slow burn, ” but also permit the effi cient capture
of the free energy released in the process, along with its interconversion and
utilization in many biological processes. It all started with attempts to under-
stand respiration, followed by a curiosity about the biochemistry and ener-
getics of muscular contraction. As a fi nal thought, it may be worthwhile to be
made aware that an average person turns over more than his/her own body-
weight of ATP per day, depending on the level of activity.
6.3 FATTY ACID METABOLISM
Fatty acid oxidation was one of the fi rst metabolic pathways fi rmly localized
in mitochondria by the pioneering studies of Lehninger and Kennedy in the
late 1940s. Since fats and fatty acids derived from them are obvious energy
sources, their degradation in mitochondria must have been suggestive. Soon
thereafter the localization of the Krebs cycle enzymes in the same organelle
allowed the integration of fatty acid oxidation with the TCA cycle via the
important intermediate acetyl - CoA. Peroxisomes had not been recognized at
that time, and therefore the discovery of β - oxidation of fatty acids in peroxi-
somes did not confuse the issue. It is now known that peroxisomes in animal
cells “ specialize ” in the degradation of very long fatty acids ( > 22 carbon
atoms), and they may even just shorten them to be accepted by the mitochon-
drial system. In plant cells, however, fatty acid degradation is restricted to
peroxisomes and glyoxysomes. The latter are specialized peroxisomes with
special signifi cance in metabolism during seed germination.
Before considering the details, it will be instructive to take a more global
view. The fatty acids are fi rst made available in the cytosol — for example,
from the hydrolysis of cholesterol esters of low - density lipoprotein (LDL) in
lysosomes or from the degradation of phospholipids by lipases. Their trans-
port into mitochondria is not a trivial issue (see below). The fi nal product of
their degradation is acetyl - CoA that can feed into the Krebs cycle. Under
different conditions, fatty acids can also be synthesized, also starting from
acetyl - CoA. The biosynthesis of fatty acids does not occur simply by the
reversal of the reactions of the β - oxidation, an observation that generally
applies to other pathways such as glycolysis and gluconeogenesis. Different
enzymes are utilized; and most signifi cantly, the biosynthesis of fatty acids
takes place in the cytosol. During elongation in the cytosol the acyl group is
attached to a large multifunctional protein (in animal cells) taking the place
of CoA.
Fatty acid degradation generates acetyl - CoA as the fi nal product, but inter-
mediates are also attached to CoA during the entire process. Thus, the fi rst
step in fatty acid degradation requires an activation:
Fatty acid CoA ATP acyl-CoA AMP PP
i
++→ + +
Several acyl - CoA - sythetases (thiokinases) serve in this activation in the
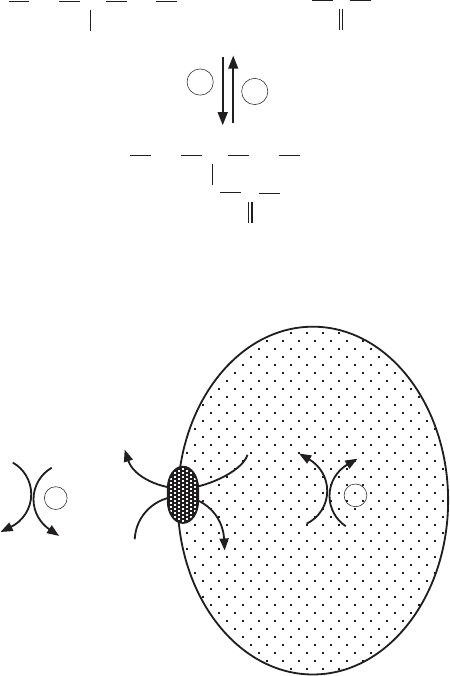
cytosol. To reach the mitochondrial matrix, a shuttle system has evolved, which
is shown in Figure 6.2 . The acyl group is transferred to carnitine, liberating
CoA. Acyl - carnitine is transported into the mitochondria by a specifi c carrier
protein; and once it is inside, the transesterifi cation is reversed to form carni-
tine and acyl - CoA. The carrier protein is operating like an antiporter, trans-
porting free carnitine out of the mitochondria.
The β - oxidation reactions (Figure 6.3 ) are well known and found in all
standard biochemistry textbooks. A brief listing of all reactions and some
select comments will suffi ce here.
The fi rst reaction is an an elimination of hydrogen to form a double bond
between the α and β carbons of acyl - CoA.
acyl-CoA FAD - 2-enoyl-CoA FADH
2
+⇒ +trans Δ
(1)
The FAD is a cofactor of acyl - CoA dehydrogenase, and the FADH
2
is reoxi-
dized via two proteins, the electron transfer fl avoprotein (ETF) and the ETF:
ubiquinone oxidoreductase transferring electrons to ubiquinone (to yield
QH
2
), which is reoxidized via the electron transfer chain. Superfi cially, the
acyl - CoA dehydrogenase therefore resembles succinate dehydrogenase in the
use of fl avin as the immediate acceptor of hydrogen, followed by electron
transfers via iron – sulfur centers to ubiquinone.
trans- 2-enoyl-CoA H O 3- hydroxyacyl-CoA
2
L-Δ+⇒
(2)
FATTY ACID METABOLISM 305
306 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
This hydration reaction resembles to formation of malate from fumarate and
is catalyzed by enoyl - CoA hydratase. It is followed by an oxidation to
β - ketoacyl - CoA:
3- hydroxyacyl-CoA NAD -ketoacyl-CoA NADHL-
+
+⇒ +β (3)
In the fi nal step, another CoA is a second substrate, and the reaction splits off
a two - carbon unit to form acetyl - CoA, leaving acyl - CoA, but now two carbon
atoms shorter than at the start:
() ( )C acyl-CoA CoA C acyl-CoA acetyl-CoA
nn
+⇒ +
−2
(4)
Figure 6.2 Acyl - CoA/carnitine shuttle in mitochondria.
CYTOSOL
MITOCHONDRIA
1
2
B
carnitine
acyl-CoA
acyl-carnitine
+
SCoAR
C
O
COO
-
CH
2
OH
CHCH
2
+
N(CH )
33
COO
-
CH
2
O
CHCH
2
RC
O
+
N(CH )
33
2
1
A
H-SCoA
+
acyl-CoA
H-SCoA
carnitine
acyl-carnitine
carnitine
acyl-carnitine
acyl-CoA
H-SCoA
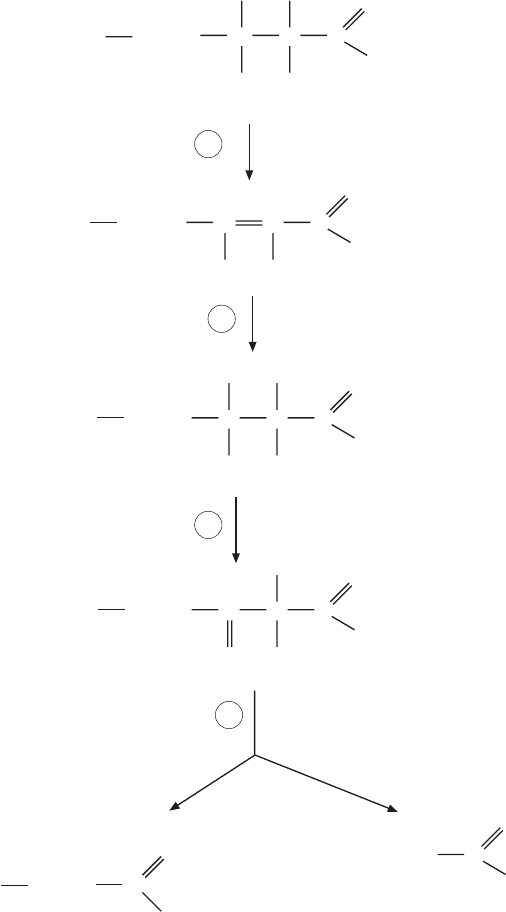
Figure 6.3 β - oxidation reactions.
acyl-CoA dehydrogenase
enoyl-CoA-hydratase
3-L-hydroxyacyl-CoA-dehydrogenase
β-ketoacyl-CoA-thiolase
1
3
2
4
SCoA
O
C
H
C
H
OH
C
H
α
β
CH (CH )
3
2
n
SCoA
O
C
H
C
H
O
C
α
β
CH (CH )
3
2
n
C
O
SCoA
CH
3
O
C
SCoA
CH (CH )
3
2
n
SCoA
O
C
H
C
H
C
CH (CH )
3
2
n
α
β
H
C
H
H
C
H
C
O
SCoA
CH (CH )
3
2
n
α
β
FATTY ACID METABOLISM 307
308 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
The series of reactions 1 – 4 is repeated, and a saturated fatty acid with an
even number of carbons can be converted to acetyl - CoA with no further
complications, except that different acyl - CoA dehydrogenases are used as
the carbon chain of the acyl group gets shorter. Unsaturated fatty acids
can be dealt with by a combination of isomerases and reductases (requiring
NADPH), depending on the position of the double bonds and their con-
fi guration. The β - oxidation of odd - chain fatty acids leads to the formation
of propionyl - CoA, which must be dealt with by a distinct series of
reactions:
(1)
propionyl-CoA ATP CO -methylmalonyl-CoA ADP P
2i
++→ + +()S
(2)
( -methylmalonyl-CoA -methylmalonyl-CoASR)()→
(3)
()R -methylmalonyl-CoA succinyl-CoA→
Reactions 1 – 3 are catalyzed by a carboxylase, a racemase and a mutase,
respectively. The methyl - malonyl - CoA mutase is an unusual enzyme requiring
5 - deoxyadenosylcobalamin (coenzyme B
12
) as a cofactor; this cofactor con-
tains Co(III) in the center of a ring resembling heme and has the only carbon –
metal bond known in biology. Vitamin B
12
defi ciency was fi rst shown in 1926
to cause pernicious anemia, and humans must acquire it from their food
(meat). Uptake and transport in the bloodstream requires specifi c proteins,
cobalamins. The solution of the crystal structure of cobalamin by D. Hodgkin
was a notable achievement in crystallography in 1956, which was later
dwarfed only by the determination of the crystal structure of whole proteins.
Cobinamide, a precursor in cobalamin biosynthesis, has an extremely high
affi nity for cyanide, and it has been proposed to be used in cyanide detoxifi ca-
tion (5) .
While the acetyl - CoA formed during β - oxidation could enter the Krebs
cycle as expected, its actual fate in liver may be different. A process referred
to as ketogenesis converts acetyl - CoA into compounds called ketone bodies,
which include acetone, acetoacetate, and β - hydroxybutyrate. The solubility of
ketone bodies makes them readily transportable by the blood to other organs
and tissues where they serve as alternate metabolic fuels. For example, when
fats are mobilized during starvation, ketone bodies can substitute for glucose
in the brain.
Although the biosynthesis of fatty acids takes place in the cytosol, and its
details get short shrift in this book on mitochondria, it is appropriate and rel-
evant to discuss one of the fi rst steps required for this cyctosolic sequence to
take place: Acetyl - CoA has to get out of the mitochondria. This is achieved
by making citrate in the mitochondria, which can be exported by a transport
system for tribarboxylic acids. In the cytosol, acetyl - CoA is regenerated by
ATP - citrate lyase:
citrate ATP CoA acetyl-CoA ADP P oxaloacetate
i
++⇒ + ++
6.4 THE UREA CYCLE
The Krebs cycle ketoacids α - ketoglutarate and oxaloacetate can be withdrawn
to form amino acids by transamination or added to the cycle by the easily
reversible reaction, and it may be worth mentioning that mammalian cells
in tissue culture thrive in media with added glutamine even though it could
be considered a nonessential amino acid. It enters the Krebs cycle as a -
ketoglutarate, thus providing nitrogen in a transamination reaction. This
nitrogen is recaptured when oxaloacetate is converted to aspartate (and aspar-
agine). In fi broblasts these two nonessential amino acids are derived almost
exclusively from glutamine via the operation of the Krebs cycle (4, 6) .
A most signifi cant role of mitochondria in amino acid metabolism is the par-
ticipation in the urea cycle. The urea cycle was the fi rst metabolic cycle discov-
ered and described by Krebs fi ve years before his more famous citric acid cycle.
The cycle is crucial for the breakdown of excess amino acids and the conversion
of excess nitrogen into urea, the excretable form of nitrogen in many terrestial
animals. The major activity takes place in the liver, from where urea is deliverd
via the bloodstream to the kidneys for excretion in urine.
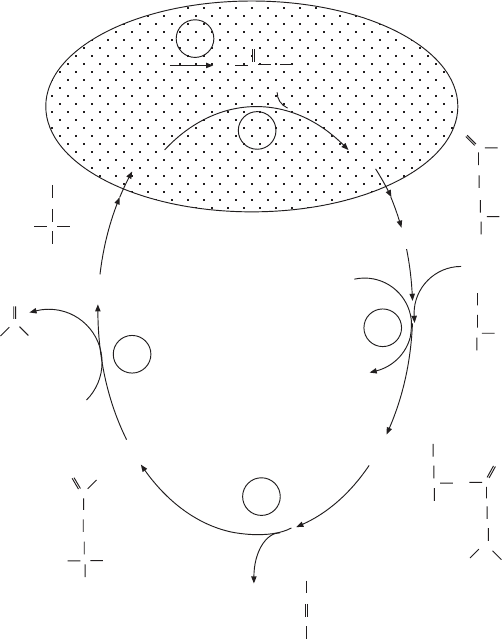
As expected, there is one reaction creating a precursor, carbamoyl -
phosphate, which can enter the cycle by condensation with a cycle interme-
diate, and products including urea are generated in various steps in the cycle.
Only some reactions of the cycle take place in mitochondria; it is completed
in the cytosol. The complete cycle is shown in Figure 6.4 . The two reactions
taking place in mitochondria are the formation of carbamoyl phosphate from
ammonia and bicarbonate, driven by the hydrolysis of ATP, and the condensa-
tion of carbamoyl phosphate with ornithine to form citrulline. Carbamoyl
phosphate synthetase I, the enzyme required for step 1, is allosterically regu-
lated by N - acetylglutamate, made from glutamate and acetyl - CoA. Increased
glutamate concentrations signal excess amino acids (nitrogen), and an increase
in N - acetylglutamate activates carbamoyl phosphate synthesis.
6.5 BIOSYNTHESIS OF HEME
Studies on respiration, cytochromes, and heme leading to the elucidation of
the role of mitochondria have been intricately linked for over a century. In
hemoglobin in the blood, the heme group is used to bind and transport oxygen.
In the electron transfer chain, heme groups associated with peptides form the
cytochromes. Another important cytochrome is the cytochrome P
450
, an enzyme
system used primarily in the liver for metabolizing various toxic compounds
such as carcinogens. An intermediate in heme biosynthesis is a tetrapyrrole
derivative (see below), a conjugated, planar ring structure without the metal
ion, and in plants this intermediate is the precursor of chlorophyll.
The use of isotope tracers made it possible in the late 1940s to investigate
the precursors for heme biosynthesis, and the whole pathway was established
BIOSYNTHESIS OF HEME 309
310 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
in the following decade (Figure 6.5 ). It starts in the mitochondria with a Krebs
cycle intermediate, succinyl - CoA, and the formation of δ - aminolevulinic acid
(ALA), involving the rate - limiting enzyme ALA synthase. It was described
earlier how ALA protein levels can be controlled by regulating the effi ciency
with which ALA mRNA is translated (see also below). ALA leaves the mito-
chondria to be converted to coproporphyrinogen III in a complex series of
reactions. An oxidative modifi cation of the ring structure is coupled to its
transport back into mitochondria, where the synthesis of heme is completed
with another oxidation and the fi nal addition of the iron to the center of the
porphyrin ring. It it noteworthy that the latter reactions require molecular
oxygen and thus are sensitive to oxygen concentrations.
ALA synthase is the key regulatory enzyme in the liver, but may not play
this role in immature erythrocytes, another tissue in which heme biosynthesis
is prominent. Under these conditions, ALA synthase may be feedback inhib-
ited by its product. Its import into mitochondria may be controlled. It is also
Figure 6.4 Urea cycle.
COO
CH
2
HC
COO
NH
C
NH
2
NH
(CH
2
)
3
HC
NH
3
COO
H
2
O
carbamoyl phosphate
citrulline
arginino-
succinate
fumarate
arginine
ornithine
urea
ornithine
citrulline
aspartate
ATP
AMP + PP
MITOCHONDRION
UREA
CYCLE
CYTOSOL
COO
HC
CH
COO
2
NH
2
C
NH
(CH
2
)
3
H
C
NH
3
COO
HN
H
2
N
C
NH
2
O
COO
C
H
NH
3
NH
3
(CH
2
)
3
H
2
N
C
O
O
P
3
O
O
C
NH
2
NH
(CH
2
)
3
HC NH
3
COO
COO
CH
2
HC
NH
3
COO
1
2
3
4
5
2ATP + HCO + NH
3
3
2ADP + P
i
+
