Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

TRANSPORT OF SMALL SOLUTES INTO AND OUT OF MITOCHONDRIA 331
voltage gating parameters. For a detailed discussion the reader is referred to
the work by Colombini and co - workers (51) . How are these properties related
to function? It is suggested that the channel can exist in various functional
states differing in selectivity between anions and cations. In the open state the
selectivity for anions is not very large, while cations are favored by the closed
state. It is argued that pore size is one major determinant with regard to non -
electrolytes (e.g., PEG 3400), but the electrostatic profi le within the channel
can have an infl uence on the passage of charged molecules such as ATP or
NAD
+
. Thus, in the closed state the channel may prevent ATP/ADP exchange
with the cytosol. The question becomes, What controls the open and closed
states of the VDAC channel? Several suggestions are summarized in the
review by Rostovtseva et al. (51) , among them phosphorylation. Another
group (52) has elevated VDAC to the “ governator of global mitochondrial
function both in health and in disease. ” As has been pointed out, the lack of
a specifi c and potent pharmacological inhibitor of this channel has made physi-
ological studies diffi cult.
VDAC has also assumed a prominent place in discussions of early events
in apoptosis, where it has been implicated in the formation of an even larger
pore allowing the escape of cytochrome c from the IMS (51) . Either it could
become part of the pore, or its behavior could cause outer membrane permea-
bilization by an unknown mechanism. Alternatively, VDAC has been hypoth-
esized to stack on top of the adenine nucleotide transporter (ANT) to form a
contact site and a continuous pore through the outer and inner membrane.
Such a structure could be responsible for the permeability transition and
breakdown of ΔΨ at some stage during apoptosis. This topic will be expanded
on in the section on apoptosis.
6.9.3 The ADP/ATP Translocator
The ADP/ATP translocator (adenine nucleotide translocase, ANT) is an inter-
esting integral membrane protein with a rather unique function in mitochon-
dria. Its properties, shared with the uncoupling protein, UCP - 1 and with what
is believed to be a large family of mitochondrial carriers, have already been
emphasized (53) . To repeat: The protein is an integral membrane protein with
a molecular mass of ∼ 32 kDA (297 residues in the bovine protein). There are
three repeats of about 100 amino acids, each of which contains two shorter
motifs representing transmembrane helices. The protein therefore is localized
in the membrane with six transmembrane helices, and both the N terminal and
the C terminal are on the same side, facing the intermembrane space. The
loops on the matrix side contain an exceptionally polar combination of about
40 side chains. There are also signature motifs with short sequences conserved
in all members of the family. These have been exploited very successfully in
blast searches of whole genomes to identify novel family members (54) . Bio-
chemical and biophysical experiments had suggested that a dimeric structure
makes up the functional carrier, with a twofold axis of rotational symmetry
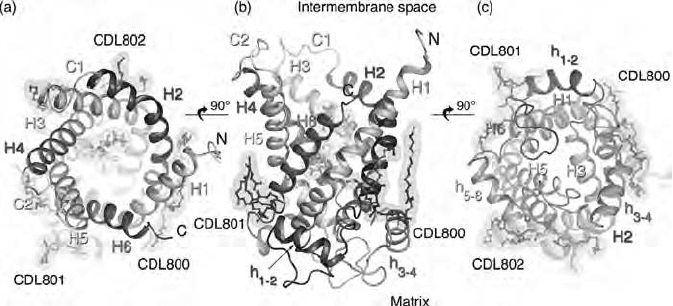
332 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
that has also been proposed to form the channel through which ADP/ATP
translocation occurs. A structure at a resolution of 2.2 Å by X - ray crystallo-
graphy in complex with the inhibitor carboxyatractyloside (see below) was
published in 2003, representing the fi rst high - resolution structure of a member
of this large carrier family (Figure 6.9 ) (55 – 57) . It has since become a model
structure for predicting and understanding other members of this mitochon-
drial carrier family (see page 334) . Six transmembrane α - helices are clearly
seen, but they are severely tilted relative to each other and relative to the
orthogonal axis through the membrane. In the crystal the unit cell contained
only the monomer. Furthermore, the six helices form a barrel - shaped structure
with the postulated nucleotide binding site. These results clearly contradict the
idea of a dimeric structure with 12 helices required for the formation of the
pore through the membrane. It is therefore possible that the proteins are
aggregated as dimers (or even higher - order oligomers) while each monomer
still functions independently (55, 57) . The crystal structure suggests a cavity
on one side lined with side chains that are presumably responsible for the
specifi city of the carrier. It is thought to represent one conformation of the
carrier, as stabilized by the inhibitor.
A distinguishing feature of the translocator/carrier and UCP family is the
interaction with purine nucleotides. In the UCP - 1 the nucleotide is a regula-
tory ligand controlling unidirectional transport of other small ions (H
+
/OH
–
,
or Cl
–
), while in the ADP/ATP translocator the purine nucleotide is in fact
the substrate and activator of translocation. UCP - 1 binds the regulatory ligand
only on the outside (cytoplasmic side) of the inner membrane. The translocator
must bind nucleotides on either side, and the net reaction (ADP imported, or
ATP exported) is driven by the membrane potential, ΔΨ , since ATP has one
more negative charge than ADP. The function of this protein is therefore inti-
mately linked to the overall process of oxidative phosphorylation, although its
control coeffi cient may be modest (58) . Nevertheless, a more severe dysfunc-
Figure 6.9 X - ray structure of ANT (from reference 57 with permission).
TRANSPORT OF SMALL SOLUTES INTO AND OUT OF MITOCHONDRIA 333
tion may contribute to pathological effects that may be implicated in neuro-
muscular diseases (59) .
Two highly specifi c inhibitors have been employed in the study and in the
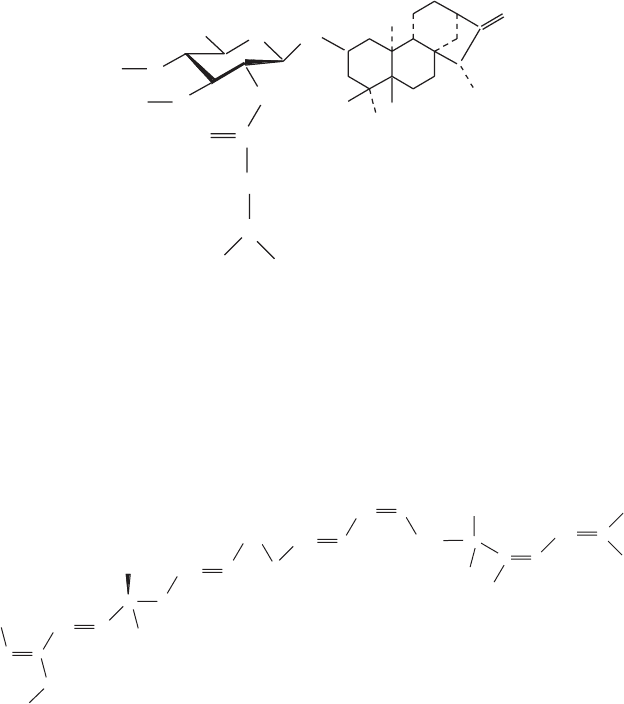
purifi cation of the ADP/ATP translocator (Figure 6.10 ). These inhibitors do
not bear much resemblance to each other or to ATP, except for the presence
of three or four negative charges. Binding sites for the inhibitor and ATP are
similar or overlapping, since binding of one precludes the binding of the other.
Carboxyatractyloside is derived from plants, and bongkrekic acid has been
isolated from certain Pseudomonas strains. Their structures are shown in
Figure 6.10 ). They differ in their ability to penetrate the inner membrane.
Figure 6.10 (A,B) Two highly specifi c inhibitors employed in the purifi cation of the
ADP/ATP translocator.
atractyloside R = H
carboxyatractyloside R = COOH
CH
2
OH
O
O
O
O
CH
2
CH
CH
3
CH
3
CH
2
OH
CH
3
C
O
COOH
H
R
O
O
3
S
-
O
3
S
-
bongkrekic acid
COOH
CH
2
C
HC
COOH
CH CH
C
CH
2
CH
3
CH
CH
CH
2
CH
2
CH CH
CH
CH
CH
2
C
OCH
3
H
C
CH
3
CH
CH C
CH
3
COOH
H
A
B
334 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
Atractyloside is restricted to the outside and hence binds to the cytoplasmic
side of the carrier, whereas the bongkrekic acid can enter the mitochondria,
and it binds only to the matrix side. The use of radiolabeled or fl uorescent
inhibitors can help in estimating the number of carrier sites per mitochondrion
(it is generally a very abundant protein — up to 15% of total protein). With the
atractyloside bound, the carrier can be stabilized during purifi cations and
protected against enzymatic degradation (see reference 53 for review and
earlier references). This form has also been successfully used for the crystalli-
zation and X - ray diffraction experiments (56, 57) (Figure 6.9 ).
Most interesting and revealing, however, has been the observation that no
ternary complexes have been found; that is, the inhibitors not only prevent the
binding of ATP, or ADP, but both inhibitors cannot bind at the same time, even
though they bind to opposite sides of the carrier. The interpretation by Klin-
genberg was that the carrier is alternately open for the substrate on one side
or the other. It is postulated to exist in two conformational states; and during
the transition from one state to the other, the substrate is translocated
across the membrane. In fact, it was further hypothesized that the transition
between the two states (c state and m state) is possible only with the substrate
(either ATP or ADP) present ( “ induced transition fi t theory of carrier trans-
port catalysis ” ). According to this model, the inhibitors can bind to the open
faces of two opposing “ ground states ” of the carrier, stabilizing a conformation
that is incapable of binding the adenine nucleotide. The crystal structure is
consistent with this model, but it would clearly be strengthened considerably,
if a crystal structure of the alternate conformation binding a nucleotide or
bongkrecic acid on the matrix side could be solved.
6.9.4 The Mitochondrial Carrier Protein Family
The previous sections have emphasized a variety of reactions and pathways
localized in the mitochondrial matrix. It is clear that many intermediates have
to transported into mitochondria, and others have to be exported. Because
metabolism is thus compartmentalized and because the inner membrane must
maintain a membrane potential, the transport of metabolites must be tightly
controlled. Mechanisms for transporting various metal ions have already been
described. Most/all metabolites are transported by a large group of mitochon-
drial transport proteins that can be grouped into 37 subfamilies with a total of
75 members in humans (54) . Several prominent family members have already
been introduced and discussed in a different context: The adenine nucleotide
transporter (ANT) and its isoforms are responsible for the import of ADP and
the export of ATP. A phosphate transporter brings phosphate from the cytosol
into the matrix for ATP synthesis. Multiple isoforms of uncoupling proteins
(UCP1, UCP2, . . . ) were identifi ed after the fi rst member of the group was
found to be a key factor in thermogenesis in brown adipose tissue. The role of
the others appears to be more subtle (see page 266 or see section 5.6.2 ). It is
now clear that most of these transporters have a structure very similar to that
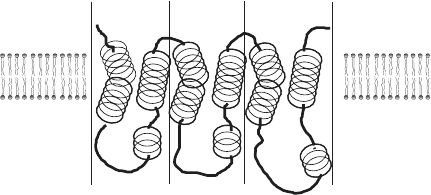
TRANSPORT OF SMALL SOLUTES INTO AND OUT OF MITOCHONDRIA 335
of the ANT: a molecular mass of 30 kDa, three degenerate repeats of ∼ 100
amino acids, six transmembrane helices, N and C terminals in the intermem-
brane space, two short loops in the IMS, and three larger loops in the matrix
(Figure 6.11 ). This topology, originally predicted from the sequence and hydrop-
athy profi les, has been confi rmed by a high - resolution structure (2.2 Å ) deter-
mined from X - ray diffraction (see page 332; also see references 55 and 56 ) A
blast of the human genome with 23 residue long sequences from 12 different
regions of 20 known human mitochondrial transporters has predicted 75 such
transporters (54) . Some of these are signifi cantly larger due to N - terminal
extensions forming a calcium binding site. Others are much smaller, with partial
sequences and truncations at either the N terminal or the C terminal. Their
functions, if any, remain to be established, although at least one has been found
to be expressed in mitochondria. Highly conserved sequence and structure
elements are constrained by the need to form homodimers for their activity.
Only 21 of the human mitochondrial transport proteins have been identifi ed
with regard to their specifi city and function. There is likely to be a certain
degree of redundancy, leading to an estimate of a possible total of 39 functions
(60) . Known and expected functions include the import/export of amino acids
including citrulline and ornithine for the urea cycle, keto acids (pyruvate),
di - and tricarboxylic acids (malate, citrate, α - ketoglutarate), carnitine and acyl
carnitine (fatty acid oxidation), nucleotides for DNA and RNA metabolism,
coenzyme A, NAD
+
, FAD, S - adenosyl methionine, and more (see Table 6.1 ).
The specifi city of the transporter for a metabolite is not predictable from the
amino acid sequence, and it has to be established from genetic experiments
(gene knockouts) or biochemical experiments. The latter typically involve
overexpression of a carrier in bacteria or yeast, followed by purifi cation and
reconstitution in liposomes.
6.9.5 Cation Transport
The most signifi cant cations other than protons are Na
+
, K
+
, Ca
2+
, Mg
2+
, and
the trace metals Fe
2+
, Cu
2+
, Zn
2+
, and others. Because of its special importance
Figure 6.11 Structure of a phosphate transporter. (Adapted from reference 54 .)
I M
N-ter
C-ter
M1 M2 M3
Matrix
IMS

336 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
in specifi c regulatory mechanisms, the uptake and release of Ca
2+
will be
considered under its own heading below. One primary mechanism of cation
uptake/exchange involves antiporters, and several such proteins with distinct
specifi cities have been described (61, 62) . Na
+
ions can traverse the inner
membrane via an apparently unregulated Na
+
/H
+
antiport, or a slightly more
complex Na
+
/Ca
2+
antiport (see below). The Na
+
ion concentration is generally
low in the cytosol, and it is also kept low in the mitochondrial matrix. The
presence of the Na
+
/H
+
antiport keeps the electrochemical Na
+
gradient equal
to the H
+
gradient. K
+
ions have a major role in maintaining mitochondrial
volume, and their uptake is achieved by three mechanisms: an electroneutral
K
+
/H
+
antiporter that may be regulated by free Mg
2+
in the matrix, a K
+
channel activated by ATP (63) , and a leakage driven by the high electrochemi-
cal gradient across the inner membrane (64) . The purifi ed mitK
ATP
channel has
been studied in synthetic lipid bilayer to prove that regulatory nucleotides
(and also acyl - CoA esters) bind to the cytosolic side of the membrane (65, 66) .
Thus, a balance must be maintained between (a) an outward fl ux via the anti-
porter driven by the proton gradient and (b) an inward fl ux through the leak
and channel driven by the electrochemical gradient. The precise physiological
signifi cance of the ATP - sensitive channel remains to be fully explained, but
it has been suggested that it serves in regulated energy dissipation at times
of low ATP demand by the cell, when the fl ow and metabolism of reducing
equivalents between matrix and cytosol has to be maintained. Mitochondrial
TABLE 6.1 Carrier Proteins in the Inner Mitochondrial Membrane
Substrate 1 Substrate 2
Antiporters
Malate Phosphate
ATP ADP
ATP dNTP
S - Adenosyl - methionine S - Adenosyl - homocysteine
Carnitine Acyl - carnitine
Ornithine Citrulline
Glutamate Aspartate
Malate
α - Ketoglutarate
Malate Citrate
Uniporters (or antiporters with one proton)
Pyruvate
Glutamate
Phosphate
Glutamine
NAD+
FAD
Coenzyme A
TRANSPORT OF SMALL SOLUTES INTO AND OUT OF MITOCHONDRIA 337
volume has been shown to infl uence the rate of electron transport and respira-
tion (61, 67) . The mitK
ATP
channel has received particular attention in relation
to cardiac function (64, 68, 69) . Based on co - purifi cation and re - constitution
experiments Garlid ’ s group has proposed a possible signaling mechanism from
the observed association of the mitK
ATP
channel with the protein kinase C ε .
Various activators of this kinase could also activate K
+
fl ux in liposomes with
these two components (70) .
Uniports for Na
+
and K
+
have also been identifi ed, but their characteristics
and functional importance are still under investigation. Similarly, the passage
of Mg
2+
ions through the inner mitochondrial membrane is still poorly under-
stood (71) . It appears that mitochondria take up Mg
2+
through membrane
leakage channels driven by the membrane potential, rather than by a specifi c
transporter (72) . Interesting speculations have centered on the possibility of
hormonal control of [Mg
2+
] in both the cytosol and matrix.
6.9.5.1 Transport of Calcium and Its Physiological Role Intramito-
chondrial free calcium [Ca
2+
]
m
, and the mechanisms associated with its control
have been examined extensively, since its recognition as a potential mediator
of a variety of metabolic activities. Additional importance is derived from the
recognition that certain pathological conditions may be associated with abnor-
mal Ca
2+
levels in mitochondria (49, 73) . The nature of the transporters, the
bioenergetics of transport, and the regulation of Ca
2+
transport all have been
investigated since the discovery of mitochondrial Ca transport in the early
1960s (74) . The existing membrane potential favors infl ux, and effl ux is ener-
getically uphill. An expert discussion of the energetics of Ca
+
transport is pre-
sented in a review by Gunter and Gunter (48) . From typical values for ΔΨ and
internal and external [Ca
2+
], it is estimated that ∼ 29 kJ/mole Ca
2+
is required
to get Ca
2+
out of mitochondria. From another point of view, this considerable
free energy difference explains why mitochondria have been thought of as
intracellular “ sinks ” or reservoirs for Ca
2+
, leading to the challenge to under-
stand how they contribute to the control to the overall cytosolic level of this
ion. This idea has been challenged in the past decade from the discovery that
intracellular Ca
2+
is largely found in the ER, from where it is mobilized by
inositol triphosphate (IP
3
). Mitochondria became the target for Ca
2+
signaling
rather than the origin of regulatory Ca
2+
(75) .
A major player in Ca
2+
transport in vertebrate mitochondria is a uniporter
that can also transport Mn
2+
, Ba
2+
, Fe
2+
, Pb
2+
in decreasing order of selectivity,
although, curiously, Mg
2+
uptake may not involve this route. The conclusion
that it is a uniporter rests on a variety of experiments summarized in reference
48 . The membrane potential (negative inside) is essential, favoring the net
charge transfer of 2; that is, Ca
2+
diffuses down the Ca
2+
electrochemical gra-
dient, thereby diminishing the membrane potential. Detailed kinetic studies
have been interpreted to show the existence of two sites for Ca
2+
on the uni-
porter: one for activation and the second for the actual transport. In other
words, Ca
2+
itself modulates the activity of this uniporter and can inhibit its
338 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
own uptake; experiments show that it is a calmodulin - mediated process (see
reference 75 for a further discussion and references). Another interesting
observation is that this uniporter does not appear to allow Ca
2+
ions to pass
down a chemical gradient alone; that is, a membrane potential is required to
induce a conformation necessary for transport.
Two distinct Ca
2+
effl ux mechanisms in vertebrate mitochondria are respon-
sible for protection against the buildup of toxic levels of Ca
2+
in the matrix.
It was a challenge to prove that the Na
+
- independent mechanism was not
an artifact or simply a reversed uniport (reviewed by in reference 48 ). It now
appears established that Ca
2+
effl ux is accompanied by proton infl ux, and a
ratio 1Ca
2+
/2H
+
appears to be favored. However, bioenergetic considerations
conclude that it cannot be a simple passive exchanger, and some additional
input of energy from the electron transport chain must be postulated. The
energy coupling mechanism is still obscure. An Na
+
- dependent mechanism can
be distinguished on the basis of certain ion selectivities (Sr
2+
but not Mn
2+
),
stimulation by Na
+
, spermidine, and spermine, and inhibition by a variety of
inhibitors. Current thinking favors an electrogenic Ca
2+
/3 Na
+
exchanger with
the evidence pointing to a ΔΨ - dependent electrophoretic antiport (76) .
Our understanding of the physiological role of mitochondrial Ca
2+
transport
is still expanding (49, 75, 77) . As stated above, mitochondria can constitute a
buffering compartment with a role in modulating cytosolic calcium, [Ca
+
]
cytosol
.
A relevant question was whether transport in and out of mitochondria was
suffi ciently rapid to keep up with rapid, pulsed changes in cytosolic [Ca
2+
],
found, for example, in beating cardiomyocytes or in nerve cells engaged in
active signaling (exocytosis triggered by elevated [Ca
+
]
cytosol
. A second major
insight was that two prominent mitochondrial dehydrogenases (pyruvate
dehydrogenase and α - ketoglutarate dehydrogenase) are activated by [Ca
2+
] m .
It should be noted that the critical concentrations are those of the free Ca
2+
.
Estimates are that only one part of 10
3
of total mitochondrial Ca
2+
is free, and
this fact must also be considered in the context of the buffering capacity of
mitochondria. The stimulation of the dehydrogenases increases the NADH/
NAD
+
ratio and hence oxidative phosphorylation. One can consider a plausi-
ble scenario in which increased [Ca
+
]cytosol in stimulated skeletal or cardiac
muscle is rapidly coupled to increased [Ca
2+
]
m
and hence increased rates of
oxidative phosporylation. Such a mechanism could serve to signal increased
energy demand in the cytosol to the OXPHOS machinery.
The use of fl uorescent Ca
+
indicators (e.g., fura - 2) developed by Tsien and
colleagues (77a) has revolutionized the measurement of [Ca
+
]
cytosol
, and has
even been applied to isolated mitochondria. Subsequently, recombinant aequo-
rin has been targeted to mitochondria to serve as a sensitive indicator of
[Ca
2+
]
m
(78) . The combination of fura - 2 and mitochondrial aequorin in single
cells permitted simultaneous measurements of [Ca
+
]
cytosol
and [Ca
2+
]
m
, with
intriguing results (reviewed in reference 79 and 80 ). Raising [Ca
+
]
cytosol
by
infl ux from extracellular Ca
2+
affected [Ca
2+
]
m
less and at a slower rate than
an increase in [Ca
+
]
cytosol
by a release (IP
3
- induced) from intracellular stores
TRANSPORT OF SMALL SOLUTES INTO AND OUT OF MITOCHONDRIA 339
(ER). These observations can now be explained by observations made in the
light/fl uorescent microscope showing that the ER and mitochondria can form
intracellular “ synaptic ” regions. At these junctions the IP
3
- stimulated Ca
2+
release channel in the ER is physically close to the mitochondrial Ca
2+
uni-
porter, and the ion can be taken up directly into the mitochondria without
signifi cant spreading into the cytosol (81, 82) . An interesting paper by Rapizzi
et al. (83) demonstrates that overexpression of VDAC can enhance the trans-
fer of Ca
2+
from the ER to mitochondria. As a consequence, cytoplasmic Ca
2+
microdomains are generated within the cellular architecture that fl uctuate
both in space and in time. Subcellular Ca
2+
concentrations are dynamically
heterogeneous from one location to another. They can thus regulate a diverse
group of cellular events stimulated by various extracellular factors (77) .
6.9.6 The Mitochondrial Permeability Transition
The mitochondrial permeability transition (MPT) was originally defi ned as
sudden increase in the inner membrane permeability to solutes of molecular
mass less than ∼ 1500 Da. It is now believed to be due to the opening of a mega
channel, which is referred to in some publications as the mitochondrial perme-
ability transition pore (MTP). It has received particular attention in recent
years because pore opening is sensitive to cyclosporin A (see below) and
because it has been implicated in the pathway of apoptosis (Chapter 8 ). The
transition is detectable and measured with isolated mitochondria in protein -
free buffers by an uptake of radioactive molecules such as sucrose, by a change
in optical density due to swelling, and even by patch - clamp techniques (84,
85) . The original observations were made with animal mitochondria (beef
heart, rat liver), but recent publications suggest that yeast mitochondria are
also capable of forming a transition pore. However, the regulation of this pore
in yeast may be different (86) .
Several broad questions can be raised, and answers to these questions are
beginning to accumulate. The discussion will focus on the following issues: (1)
What components of the inner membrane constitue the pore? (2) What mech-
anisms regulate the opening and closing of the pore? (3) What is the physio-
logical signifi cance of the operation of this megachannel? To some extent the
answers are interrelated.
The exact composition of the pore is still not completely clear, except for
a general agreement that the adenine nucleotide translocase (ANT) is a par-
ticipant in channel formation, perhaps by forming complexes with outer mem-
brane proteins to create contact sites and hence to stabilize and/or enlarge the
channel. The peripheral benzodiazepine receptor and VDAC in the outer
membrane have been implicated, and there is speculation about the inclusion
of Bcl2, cardiolipin synthase, 3 - b - hydroxysteroid isomerase, and phospholipid
hydroperoxide glutathione peroxidase (87) . On the cytosolic side, hexokinase
may also be associated, and on the matrix side the participation of cyclophilin
D is suggested by the sensitivity to cyclosporin.
340 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
The channel can be opened when Ca
2+
is accumulated in the matrix by
exposure of mitochondria to high concentrations of Ca
2+
(generally above
physiological levels), and the sensitivity to Ca
2+
is heightened under various
conditions including oxidative stress, adenine nucleotide depletion, elevated
phosphate concetrations, and low membrane potenial, ΔΨ ? The role of
nucleotides points to an involvement of ANT, but a more direct indication is
the effect of inhibitors of ANT on the MPT. Inhibitors that stabilize the “ c ”
conformation of ANT (e.g., atractyloside) favor the MPT, while inhibitors
stabilizing the “ m ” conformation (e.g., bongkrekic acid) also inhibit pore
opening. A high ΔΨ or a low pH prevents the transition (88) . Of special inter-
est was the observation that submicromolar concentrations of cyclosporin A
are very effective in keeping the pore from opening. The target for cyclosporin
is the matrix peptidyl - proline cis – trans isomerase, a member of the cyclophilin
family. One interpretation was that the (soluble) matrix isomerase may inter-
act with an integral membrane protein, and when triggered by Ca
2+
, the pore
opening is induced. Cyclosporin A would interfere with this required interac-
tion. A further speculation was that the relevant IMP was the ANT (89) . The
idea was supported by the observed attachment of cyclophilin to the inner
membrane under certain conditions (oxidative stress, thiol reagents, increased
matrix volume), which favor or enhance pore opening (90, 91) . Even though
the MPT makes an appearance in a large fraction of publications related
to apoptosis, necrosis, oxidative stress, and other pathologies, there is still no
clear distinction between the “ MPT ” and the “ MTP. ” The fi rst describes a
phenomenon, while the second implies the existence of a structure that has
never been isolated and whose composition is hypothesized on the basis of
indirect studies with various inhibitors. The suggestion has even been made
that the “ pore ” is constituted of unfolded integral membrane proteins damaged
by ROS (e.g., oxidation of sulfhydryls, etc.) (92) .
The combined observations have served to support a variety of potential
functions for the MTP, which may not be exclusive (87) : (1) a voltage sensor,
(2) a matrix pH sensor, (3) a divalent cation sensor (role in calcium homeo-
stasis (e.g., 93 )), (4) a sensor of adenine nucleotide concentrations, (5) a sensor
of the redox state of the pyridine nucleotide pool, and (6) a thiol sensor (redox
status of glutathione). The context in which the MPT is most commonly
encountered is the triggering and progression of apoptosis, a subject that will
be expanded in a following chapter.
It is clear that a prolonged opening of a number of pores, or the simultane-
ous opening of many pores, would deenergize and inactivate mitochondria
rapidly, and it may be that such an event is associated with the irreversible
breakdown of ΔΨ observed in the course of apoptosis (see Chapter 8 ).
However, at this time it is still a matter of much speculation as to whether the
MTP makes a signifi cant contribution to mitochondrial activities under phy-
siological conditions. It could be imagined as a safety valve under stressfull
conditions, but the lack of specifi city for any small - molecular - weight solute
precludes any fi ne - tuning for specifi c ions or solutes.
