Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

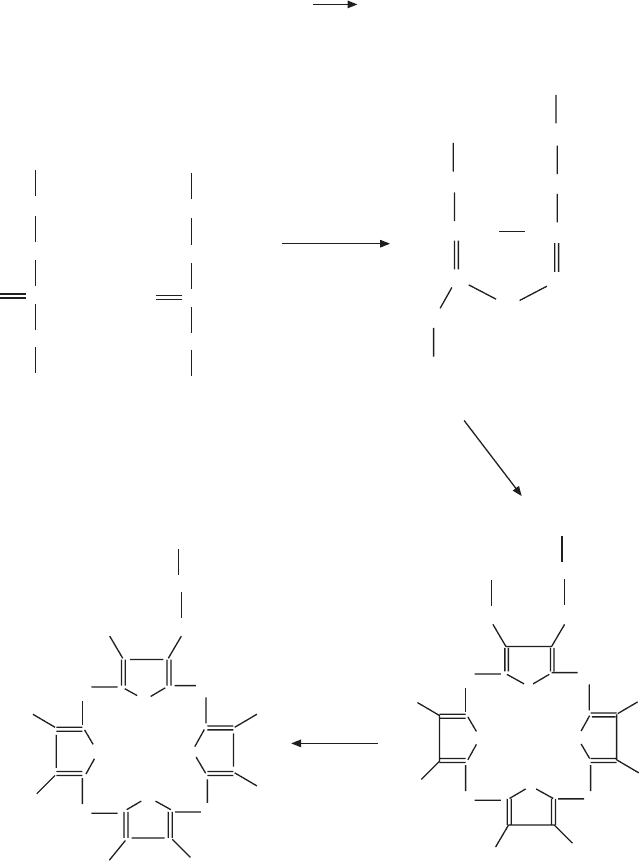
Figure 6.5 (A,B) Biosynthesis of heme. (C) Heme substituents in type a, b, and c
cytochromes.
COO
-
CH
2
CH
2
0C
CH
2
NH
3
+
COO
-
CH
2
CH
2
0
C
CH
2
NH
3
+
Porphobilinogen
COO
-
CH
2
CH
2
C
N
H
+
CH
2
COO
-
CH
2
CH
C
CH
2
NH
3
+
P
M
P
M
P
M
CH
2
COO
-
CH
2
N
H
HNNH
H
2
C
H
2
C
CH
2
CH
2
CH
3
H
N
A
P
CH
2
CH
2
H
2
C
H
2
C
NH HN
CH
2
COO
-
CH
2
COO
-
CH
2
A
P
A
P
N
H
H
N
Coproporphyrinogen III
Uroporphyrinogen III
δ-Aminolevulinic acid
Glycine + Succinyl-CoA δ-Aminolevulinic acid
A
BIOSYNTHESIS OF HEME 311
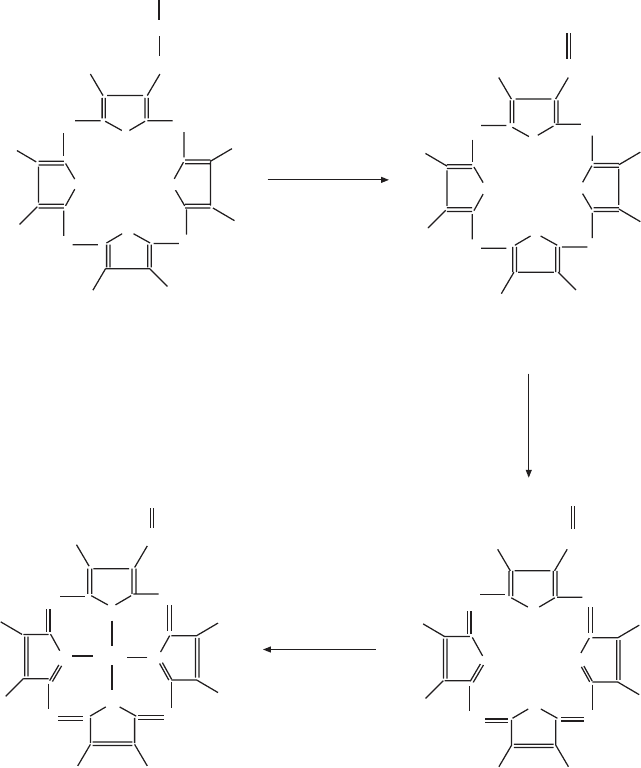
312 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
Coproporphyrinogen III
Protoporphyrinogen
Protoporphyrin IX
P
M
P
M
P
M
CH
2
COO
-
CH
2
HN
NH
H
2
C
2
HC
CH
2
CH
2
CH
3
N
H
H
N
CH
3
CH
2
CH
2
H
2
C
H
2
C
NH HN
M
V
M
P
M
P
CH
2
CH
N
H
H
N
CH
3
CH
CH
HC
HC
NN
M
V
M
P
M
P
CH
2
CH
H
N
N
H
CH
CH
2
P
M
PM
V
M
N
N
NN
HC
HC
CH
CH
CH
3
Fe
Heme
B
Figure 6.5 (Continued)
among the group of proteins that are associated with iron transport, storage,
and metabolism (see page 324; Section 6.8 .) and that are encoded by mRNAs
containing iron - responsive elements (IRE) in their untranslated regions. When
iron is defi cient, the cytosolic aconitase is in an open conformation (the iron –
sulfur center is dissociated) and capable of blocking the translation of ferritin
and ALA mRNAs by binding tightly to the IRE. This mechanism allows the
biosynthesis of heme to be adjusted to the availability of iron (Fe(II)) in the
cell and mitochondria.
As described in an earlier chapter, the expression of genes encoding mito-
chondrial proteins in yeast is sensitive to oxygen. More specifi cally, the need
for molecular oxygen in the fi nal oxidations in the pathway makes heme syn-
thesis sensitive to oxygen. In turn, heme can induce a number of genes required
for respiration, control of oxidative damage, and sterol synthesis, and it can
repress others (for review see reference 7 ).
As shown in Figures 6.5A and 6.5B , the porphyrin rings have various groups
attached, making the whole structure asymmetrical. In their fi nal form in a - ,
b - , and c - type cytochromes the Fe (II/III) ions are found in distinct microen-
vironments controlling their redox potential, not only because the peptides
differ, but also because the substituents on the rings vary considerably (Figure
N
N
N
N
CH
3
CH
3
CH
3
Fe
3+
CH
2
CH
2
COO
-
CH
2
CH
2
COO
-
R
1
R
2
R
3
a-type
b-type
c-type
CH
3
CH
OH
CH
3
CH
3
CH
3
CH
2
CH CH
2
CH CH
2
CH CH
2
CH
3
CH
3
CHO
CH
S Cys
CH
3
CH
S Cys
CH
3
R
3
R
2
R
1
C
Figure 6.5 (Continued)
BIOSYNTHESIS OF HEME 313
314 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
6.5C ). In a - type cytochromes the heme is characteristically derivatized with an
isoprenyl side chain. In c - type cytochromes the heme is covalently linked to
cysteine side chains of the peptide.
6.6 CARDIOLIPIN AND LIPID BIOSYNTHESIS/METABOLISM
A comprehensive review of the lipid composition of mitochondria has been
published by Daum (8) . It is generally agreed that the inner membrane has
one of the highest protein : lipid ratios of all biological membranes studied, but
in spite of the dense packing of protein complexes and the various carriers
and import machinery, the membrane nevertheless behaves as a fl uid mosaic
(9, 10) . One of the most noteworthy aspects of the inner mitochondrial mem-
brane is that it is the exclusive location for the lipid diphosphatidylglycerol,
also named cardiolipin. This lipid has long been believed to be absolutely
essential for the functioning of various complexes and integral membrane
proteins. Some doubts were raised from the initial studies of a mutant yeast
strain about the “ absolute ” requirement. In the fi rst report of a yeast mutant
with the cardiolipin synthase gene, CLS1 , knocked out, and no cardiolipin
found in any of the membranes, it may have been a surprise to fi nd the cells
viable in both fermentable and nonfermentable carbon sources. Purifi ed, func-
tional preparations of such complexes have inevitably contained cardiolipin,
suggesting that this lipid has a special affi nity for such proteins. More discus-
sion on the need of this lipid for proper function of these complexes is found
in Chapter 5 . A comprehensive and expert review of the biosynthesis and
biological signifi cance of cardiolipin has been written by Schlame et al. (11)
The proposed functions of this unique lipid have also been broadened to
include a participation in mitochondrial protein import, protein folding, and
assembly — that is, in the assembly and maintenance of the inner membrane.
However, most of these roles of cardiolipin must remain highly speculative. In
this discussion the focus will be on the synthesis of this lipid and some other
characteristics of its composition.
Cardiolipin is found in the membranes of eubacteria such as Escherichia
coli , as well as in the mitochondrial membranes of all eukaryotes studied so
far, but not in archaebacteria (12 – 14) . It is an unusual phospholipid with four
acyl chains. The synthesis in bacteria involves the combination of two mole-
cules of phosphatidyl glycerol with the elimination of one glycerol molecule.
In contrast, in eukaryotes (liver, Saccharomyces cerevisiae, Neurospora crassa ,
bean sprouts, and mussels) the two starting substrates have been shown to be
phosphatidylglycerol (PG) and CDP - diacylglycerol (see references 11 , 13 , and
15 for reviews and listing of additional references). The phosphatidyl group
from CDP - diacylglycerol is transferred to PG by the enzyme cardiolipin syn-
thase (Figure 6.6 ). The evolution of the reaction in eukaryotes from the bio-
synthetic pathway in bacteria has been postulated to be a consequence of the
different levels of the precursor present in bacteria and mitochondria (15) . In
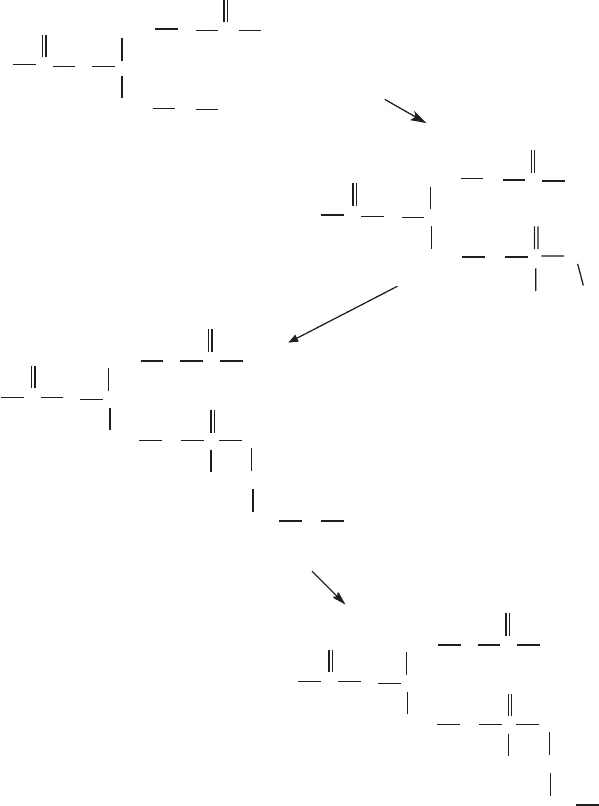
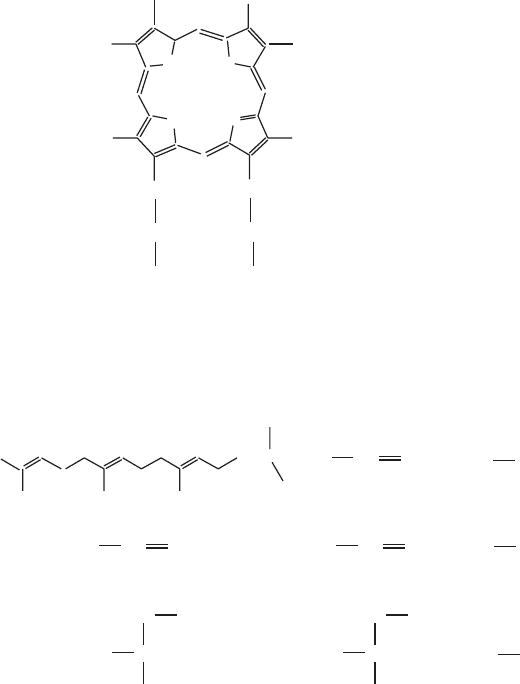
Figure 6.6 (A,B) Cardiolipin biosynthesis.
CTP
Glycerol-3-
phosphatidylglycerol
CH
2
CHOH
CH
2
O
O
P
R
2
O
CO
OCH
2
CH
R
1
O
C
OCH
2
OH
O
Cytidine
R
O
2
O
C
O
P
O
CH
2
CH
R
1
O
C
O
CH
2
O
CDP-diacylglycerol
phosphatidylglycerol phosphate
R
=
CH
2
O
C
O
R
1
CH
CH
2
O
OC
O
2
P
O
O
CH
2
CHOH
CH
2
O
PO
3
=
R
2
O
C
O
PO
3
OCH
2
CH
R
1
O
C
O
CH
2
phosphatidic acid
A
the former, phosphatidyl glycerol is abundant, and a simple, reversible trans-
esterifi cation can be driven toward cardiolipin by substrate availability. In
mitochondria the precursor may be limited, and the forward reaction is driven
by the irreversible cleavage of a high - energy anhydride bond in phosphatidyl -
CMP (CDP - diacylglycerol). Hence, the mitochondrial reaction is highly exer-
gonic. Properties of this enzyme from various mitochondrial preparations have
been described (13, 15, 16) . Although it had long been suspected to be present
CARDIOLIPIN AND LIPID BIOSYNTHESIS/METABOLISM 315
316 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
exclusively in mitochondria, a detailed study has localized it to the matrix side
of the inner membrane in rat liver mitochondria (17) ; that is, it is an integral
membrane protein in this membrane with a large hydrophilic domain exposed
to the matrix. The enzyme has been solubilized with the help of detergents
and purifi ed to a signifi cant degree from yeast and mammalian sources. Several
of the properties of this enzyme appear to be similar in all eukaryotes exam-
ined, in support of the monophyletic origin of mitochondria (13) , but there are
also some distinctions between the yeast enzyme and the mammalian enzyme
of as yet uncertain signifi cance. Most notably, the molecular mass of the yeast
enzyme has been reported to be 20 – 30 kDa, in contrast to a molecular mass
of ∼ 50 kDa for the mammalian enzyme.
Schlame and co - workers (11, 13, 15, 16) also addressed the problem of the
molecular acyl species found in cardiolipin of various species. In all organisms
a high percentage of symmetrical acyl species was found — that is, species with
identical diacylglycerol moieties. Acyl chains either were identical or differed
only by two carbons or one double bond, respectively. Minimum energy confor-
mations were calculated by these authors, but they point out that the conforma-
tion in a real membrane and in association with protein complexes may be quite
different. Not surprisingly, the models suggest all acyl chains to be arranged
more or less in parallel, and in a highly cartoon - like fashion one could represent
cardiolipin as a single polar head group with four aliphatic chains (Figure 6.6B ).
Another notable property of cardiolipin is that it is an anionic phospholipid.
R
1
CH
2
CH
O
C
OCH
2
O
C
O
R
2
O
O
P
O
O
O
CO
CH
2
CH
O
C
O
CH
2
R
2
R
1
O
O
P
O
O
CH
2
CH
2
C
H
HO
Cardiolipin
B
Figure 6.6 (Continued)
The Barth syndrome is an X - linked cardiomyopathy due to a defect in a
protein called tafazzin (gene: TAZ). This enzyme is involved in a remodeling
reaction of cardiolipin following the de novo biosynthesis of this lipid. Remod-
eling leads to cardiolipin highly enriched in linoleic acid. In the absence of
tafazzin, cardiolipin levels are signifi cantly reduced, and the remaining cardio-
lipin has a more diverse set of acyl chains (18, 19) . Model systems for the study
of tafazzin have been established in yeast (20) and in Drosophila (21) , where
the tafazzin mutation causes reduced motor activity and morphologically
abnormal mitochondria in fl ight muscles.
Since cardiolipin is synthesized on the inside of the inner membrane and is
therefore most likely found in the inner leafl et of the bilayer, one has to ask
whether it can be distributed beyond this particular location. A “ fl ippase ”
would be required to translocate it to the outer leafl et. On the other hand, in
the immediate vicinity of the multisubunit integral membrane protein com-
plexes where cardiolipin is found, the distinction between outer and inner
leafl ets may be meaningless. Movement to the outer membrane is even less
likely but still not defi nitely excluded by experiments. Since mitochondria may
fuse with each other, but not (normally) with other membranous organelles
in a cell, the exclusive presence of cardiolipin in mitochondria can be
explained.
The problem is less settled for the precursor phosphatidylglycerol (PG). It
is derived from the precursor phopatidylglycerophosphate (PGP) (catalyzed
by a phosphatase). PGP is made from CDP - diacylglycerol, and PGP synthase
has also been localized in mitochondria. Such a fi nding would explain the
presence of cardiolipin and PG in mitochondria, but PG appears to be a minor
constituent of other intracellular membranes of mammalian cells, and it may
be necessary to postulate a second, cytoplasmic isozyme for PGP synthase
(22) .
The biosynthesis of the other common phospholipids found in mitochon-
dria is not exclusive, and detailed reviews on phospholipid metabolism are
beyond the scope of this treatise (8, 12, 14) . If the synthesis of these lipids takes
place in the smooth ER, a mechanism must be found for distributing these
lipids to other cellular locations. Vesicular traffi c from the ER via the Golgi
to the plasma membrane, as well as vesicular traffi c in both directions between
these membranes and various intermediate compartments, can be used to
rationalize the distribution of lipids within a cell. Nevertheless, mechanisms
must be postulated to explain the nonuniform distribution of lipids such as
cholesterol in these membranes. Less attention appears to have been devoted
to the delivery of lipids to mitochondria: Are specialized shuttle vesicles
involved? Are lipids delivered individually by means of specialized carrier
proteins? “ Confl uencies ” between the outer mitochondrial membrane and
the endoplasmic reticulum have been observed (see reference 23 for a review).
It is not clear whether it is a transient state, and whether it serves to
import components into mitochondria, or whether it represents an early step
in degradation. When mitochondria are vitally stained with the cyanine dye
DiOC
6
, the dye is fi rst accumulated by mitochondria, but subsequently it can
CARDIOLIPIN AND LIPID BIOSYNTHESIS/METABOLISM 317
318 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
be observed in the ER after the mitochondria have lost their elongated
shape (23) .
A few generalizations and basic principles are expertly reviewed by Daum
(8) , with special reference to mitochondria, and more recently by Kent (24)
and Dowhan (14) from a more general perspective. While the lipid content of
mitochondria is somewhat variable as expected, differing between tissues as
well as between organisms, it is clear that the major phospholipids of mito-
chondria in addition to cardiolipin (10 – 20%) are phosphatidylethanolamine,
PE (20 – 40%), and phosphatidylcholine, PC (35 – 50%). The other phospholip-
ids are present at a level generally less than 5%, and cholesterol is present
only in traces. These generalizations apply to vertebrate tissues, as well as to
a variety of microorganisms and plant mitochondria. The latter also contain a
variety of free and derivatized sterols (see reference 8 for review and original
references). A further distinction can be made between the outer and inner
membranes of mitochondria, and a few attempts have been made to character-
ize the differences. The presence of cardiolipin in the outer membrane is still
controversial, since contamination is a possibility. Other differences in the
ratio of PE : PC, or the relative amounts of phosphatidylinositol, PI, have been
observed, but the functional signifi cance is still obscure.
The origin and site of synthesis of mitochondrial lipids are areas still under
investigation and in a state of evolution (25) . Conclusions are somewhat
subject to the success of fractionation schemes — most notably, fractionation
of mitochondria from microsomes and other vesicular structures, identifi ed by
marker enzymes. Phosphatidylethanolamine is made in mitochondria from the
decarboxylation of phosphatidylserine, PS. A search for the cellular location
(in yeast) of phosphatidylserine synthase has identifi ed a microsomal subfrac-
tion, but the absence of typical microsomal marker enzymes has led the authors
to propose a novel particle population. One is forced to conclude that PS is
imported into mitochondria, but some of the PE made must also be exported
to other cellular membranes where it is found. Alternatively, a second PS
decarboxylase may exist in a cytosolic membrane, and indications for its exis-
tence are found from the study of a yeast mutant defective in the mitochon-
drial PS decarboxylase (see reference 24 for discussion).
While cell fractionation studies have proved their worth over the decades,
the fractionation of subcellular membranous structures has been a technical
challenge because of microheterogeneity, and results are subject to misinter-
pretation when contamination cannot be ruled out. In the future, one can
expect that model organisms like yeast will allow systematic gene disruptions
to explore how the absence of a particular enzyme (and hence the product
from this pathway) affects growth and other cellular activities. An example of
the unexpected type of result derived from a genetic approach is the fi nding
that unsaturated fatty acids are essential for the distribution of mitochondria
to the daughter cells in the yeast Saccharomyces cerevisiae , presumably because
the necessary shape changes (elongation) are dependent on the fl uidity of the
mitochondrial membranes (26) .
6.7 BIOSYNTHESIS OF UBIQUINOL (COENZYME Q)
The biosynthesis of ubiquinol was elucidated some time ago from studies in
Escherichia coli and yeast Saccharomyces cerevisiae . Genetic studies were
critical, since CoQ - defi cient strains of each organism could be isolated, and
intermediates accumulating at different stages in the biosynthetic pathway
gave clues about the sequence of reactions. Although the pathways differ
slightly in some intermediate steps in prokaryotes and yeast, 10 genes have
been defi ned in each organism by the isolation of eight complementation
groups ( coq1 – coq10 in yeast). The CoQ - defi cient yeast strains are respiration -
defi cient and fail to grow on nonfermentable carbon sources, but they do grow
on glucose (27) . The laboratory of Clarke has made a number of signifi cant
contributions to the detailed functional analysis of a number of different coq
mutants in yeast (28 – 32) . The defi ciency can in many cases be overcome by
the addition of exogenous Q
2
.
The reactions defi ned by mutations in CoQ/ubiquinone - defi cient bacteria
or yeast are the fi nal reactions unique for the biosynthesis of ubiquinone. Key
starting materials in this pathway are p - hydroxybenzoic acid and polyisoprene
diphosphate. The fi nal polyprenyl tail contains n isoprene units, where n varies
between organisms. In mammalian mitochondria n = 10, and hence the com-
pound is also referred to as Q
10
. The Q
10
derivative is extremely insoluble in
water, and in in vitro experiments as well as in therapeutic applications deriva-
tives with shorter tails are often used.
The synthesis of the polyprene tail begins in the well - known pathway
from acetyl - CoA via hydroxymethylglutaryl - CoA , mevalonate, geranyl -
pyrophosphate, and farnesyl - pyrophosphate, the initial reactions established
fi rst by K. Bloch in the biosynthesis of cholesterol. It has now become clear
that farnesyl - pyrophosphate is a key intermediate and branch - point in several
very important pathways leading to cholesterol synthesis, dolichol synthesis,
ubiquinone synthesis and protein isoprenylation (see references 33 and 34 for
recent reviews). The reactions up to the synthesis of farnesyl - pyrophosphate
are also referred to as the mevalonate pathway, and they are illustrated in
Figures 6.7A and 6.7B . Three acetyl - CoA molecules are fi rst combined succes-
sively to form hydroxymethylglutaryl - CoA, the precursor for mevalonate.
Phosphorylations and a decarboxylation yield isopentenyl - pyrophosphate
(IPP), which is reversibly isomerized to dimethylallyl - pyrophosphate (DMA -
PP). A condensation of IPP and DMA - PP yields geranyl - pyrophosphate
(GPP), and the addition of another isopentene unit leads to farnesyl - pyro-
phosphate (FPP). The enzymes are soluble cytosolic enzymes, with one excep-
tion: The enzyme HMG - CoA reductase is an integral membrane protein in
the endoplasmic reticulum. It has received a considerable amount of attention
as a key regulatory enzyme in cholesterol biosynthesis. It may therefore also
infl uence the rate of synthesis of the other products at the end of the various
branches, but while its regulatory role in cholesterol biosynthesis is undis-
puted, modulating its activity by dietary conditions or inhibitors has been
BIOSYNTHESIS OF UBIQUINOL (COENZYME Q) 319
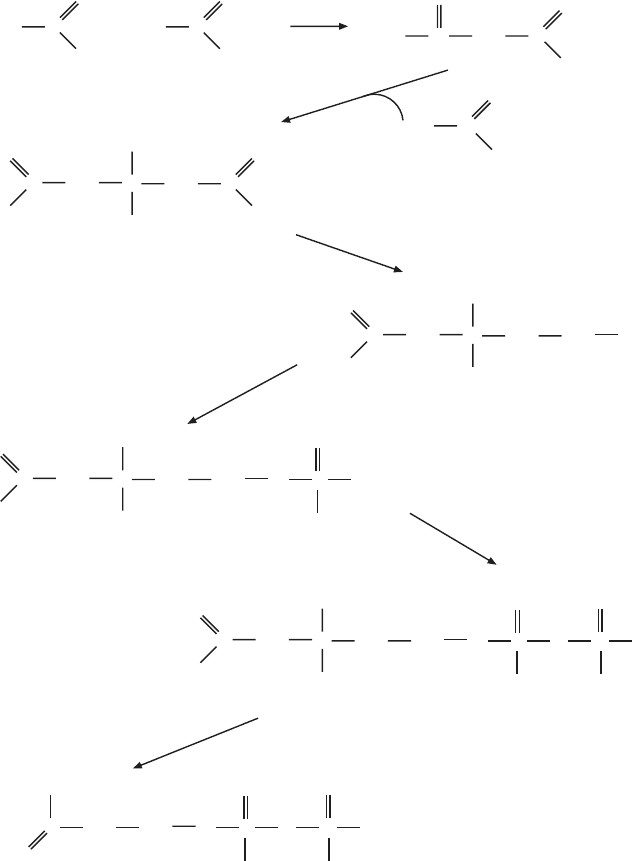
320 METABOLIC PATHWAYS INSIDE MITOCHONDRIA
Figure 6.7 (A – D) Ubiquinone, cholesterol, and Dolichol synthesis.
C
O
SCoA
C
O
SCoA
CH
3
C
O
CH
2
C
O
SCoA
C
O
SCoA
-
-
3
SCoA
HMG-CoA reductase
mevalonate-5-phosphotransferase
phospho-mevalonate kinase
pyrophospho-mevalonate
decarboxylase
HMG-CoA
mevalonate
isopentenyl pyrophosphate
CH
3
CH
3
CH
3
-
O
O
O
PO
O
O
P
OCH
2
CH
2
CH
3
C
CH
2
-
O
O
CCH
2
C
OH
CH
3
CH
2
CH
2
O
P
O
O
O P
O
O
O
-
-
-
-
O
O
CCH
2
C
OH
CH
3
CH
2
CH
2
O
P
O
O
O
-
-
OHCH
2
CH
2
CH
3
OH
CCH
2
C
O
O
-
O
O
CCH
2
C
OH
CH
CH
2
C
O
A
found to have no signifi cant infl uence on the biosynthesis of dolichol, ubiqui-
none, or the isoprenylation of proteins. This apparent dilemma has been
addressed by several authors, and a plausible model referred to as the “ fl ow
diversion hypothesis ” has been postulated. According to this hypothesis, most
