Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.


aquatic vertebrates and invertebrates, LC
50
ranged from 2.0 to 3.8 mg/L. For phenan-
threne, LC
50
was 0.3 mg/L to the grass shrimp Palaemonetes pugio and 0.6 mg/L to
the marine copepod Neanthes arenaceodentata. The concentration of PAH that causes
a 50% reduction in the photosynthetic activity of the freshwater algae Chlamydom onas
angulosa was 9.6 mg/L for naphthalene and 0.9 mg/L for phenanthrene.
Of greater importance are the mutagenic and carcino genic properties of this class of
compounds. Benzo[a]pyrene (BaP) is the first known human carcinogen. In London,
Pott noticed the association betwee n scrotal cancer in men working as chimney sweeps
in 1775. They are exposed to high dosages of BaP and other PAHs in creosote. MFO acti-
vation is required for genotoxicity. Intermediates in biotransformation have been shown to
bind to cellular DNA.
Crude and fuel oils are mixtures of aliphatic, olefinic, aromatic, and polyaromatic
hydrocarbons. Because of their complex composition, fate and transport in the environ-
ment result in large changes in toxic potency. The processes of volatilization, dissolution,
and degradation produce changes in composition known as weathering. Despite their
hydrophobicity, petroleum hydrcarbons do not significantly bioaccumul ate. Apparently,
this is because of their ability to be metabolized to soluble forms for excretion. The
96-hour LC
50
of unweathered crude oil ranges from 1.34 to more than 19.8 mg/L in
adult fish and from 0.81 to above 19.8 mg/L in aquatic invertebrates. No. 2 fuel oil ranged
from 0.7 to 11.6 times as toxic, with a median of about 3.
Acute exposures, such as those due to oil spills, can cause considerable mortality, espe-
cially of intertidal organisms and seabirds. This leads to successional changes in popula-
tions. Substituted aromatics represent the relatively soluble fraction of these oils. These
can affect organisms throughout the water column. Chronic exposure to oil pollution, such
as in highly indus trialized estuaries, have been found to reduce the number of species and
individuals, to increase the proportion of opportunistic species, and to decrease ecological
energy flow. Benthic amphipods, such as the genus Ampelisca, have been found to be a
sensitive indicator organism for petroleum pollution.
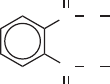
Phthalate esters are added to plastics to control their mechanical properties, especially
to reduce brittleness. In the structural formula shown below, the R groups may be a variety
of long-chain hydrocarbons, such as isooctyl phthalate, 2-ethylhexyl phthalate, or isode-
cyl phthalate. Although they have a very low vapor pressure, they can diffuse from plastic
materials into the environment, especially at elevated temperatures. Bioconcentrati on
TABLE 21.8. Transport Properties of Some PAHs and Bioaccumulation Factors for
Daphnia pulex
No. of Bioaccumulation
Compound Aromatic Rings Molar Mass Solubility (mg/kg) Factor SE
Naphthalene 2 128.2 22,000–31,700 13110
Fluorene 2 166.2 1685–1980
Anthracene 3 178.2 30–73 91748
Phenanthrene 3 178.2 1002–1290 32556
Fluoranthene 3 202.3 206–260 NA
Pyrene 4 202.3 132–171 2702245
Benzo[a]pyrene 5 228.3 9.4–14 NA
a
NA, not available.
Source: Rand and Petrucelli (1985).
818 TOXICITY OF SPECIFIC SUBSTANCES
factors as high as 720 and 3600 have been measured. Their acute toxicity is fairly low.
The oral LD
50
in lab animal s range from 8 to 64 g/kg. Short-chain phthalates tend to be
more toxic. The 96-hour LC
50
value for dibutyl phthalate ranged from 0.7 to 6.5 mg/L,
whereas di-2-ethylhexyl phthala te was usually greater than 10 mg/L. Terrestrial organisms
are not as likely to suffer exposures, whereas aquatic organisms receive exposure from
water pollution, and humans through various routes, such as food packaging. Phthalates
have also been implicated as possible xenoestrogens.
C
C
OR
1
OR
2
O
O
phthalate ester
Surfactants, surface-active agents, are substances that lower the surface tension of
liquids, especially water. They include soaps and synthetic detergents, of which the latter
are of greatest environmental interest. Structurally, their molecules are designed to have a
lipophilic (often hydrocarbon) portion and a hydrophilic portion. They may be anionic,
cationic, zwitterionic (capable of forming cations or anions), or nonionic. Above a charac-
teristic concentration, the critical micelle concentration (CMC) surfactants form
enclosed particles called micelles (see Figure 3.9). These can concentrate lipophilic sub-
stances from the water, decreasing their aqueous concentration but increasing the capacity
of the overall solution (water plus micelles) to dissolve those substances. They may increase
the uptake of lipophi lic substances, increasing their toxicity. Biodegradability of surfac-
tants decreases with branchin g of the hydrocarbon portion and increases with its length.
Toxicity occurs by various mechanisms, including the formation of complexes with
membranes and enzymes. Toxicity tends to increase with chain length. Some results
have shown greater correlation with surface tens ion than with surfactant concentr ation.
For example, the lethal concentration for hydra occurred at a surface tension of
494 dyn/cm. This corresponded with the level that would disrupt the cell membrane.
Some specific a cute toxicity results include a 24-hour EC
50
for Daphnia ranging from
12 to 13 mg/L, and a 96-hour LC
50
for fathead minnow ranging from 3.5 to 23 mg/L. A
number of investigators have noted damage to gills and external sensory organs at sub-
lethal concentrations.
21.4 HALOGENATED ORGANICS
Halogenated organic solvents displaced industrial uses for many hydrocarbon solvents
because their vapors have a flash point at much higher temperatures, making them non-
explosive. Others were developed for their special properties, such as the high dielectric
constant of polychlorinated biphenyls. Yet another group, such as chlorinated dioxins and
trihalomethanes in drinking water, are unintended by-products of other processes. Com-
pounding their toxic properties are their lipophilicity and their low rate of biodegradation,
both of which contribute to tendency to persist and bioaccumulate in the environment.
The halogenated alkanes refer mostly to chlorinated one- and two-carbon compounds,
various brominated compounds included among the trihalomethanes (THMs) formed
during chlorination of drinking water, and fluorinated alkanes such as a variety of Freons.
HALOGENATED ORGANICS 819

The properties of the halogenated alkanes are similar to their nonhalogenated counter-
parts. CNS depressant action and liver and the potential for kidney damage both increase
with degree of chlorination. Unsaturated compounds are more toxic than saturated. Repla-
cing chlorine with bromine increases toxicity, whereas replacing it with fluorine decreases
it. Many halogenated compounds induce liver cancer in laboratory tests with rodents.
Methylene chloride may be the least toxic of the chlorinated methanes. It has the low-
est irritancy and potential for CNS depression. It is commonly used as a laboratory sol-
vent. Chloroform and carbon tetrachloride are potent CNS depressants and liver and
kidney toxins. A summary of the inhalati on effects of chloroform in humans is:
200–300 ppmv Odor threshold
1000 ppmv Fatigue and headache
1500 ppmv Dizziness and salivation within minutes
4000 ppmv Fainting and nausea
14,000–16,000 ppmv Rapid narcosis
Vinyl chloride, a class A carcinogen, is one of the most dangerous compounds in this
group. CNS depression and death have been reported upon acute exposure. Chronic expo-
sure results in arthroosteolysis (destruction of bone at the joints), Raynaud’s phenomenon
(a vascular disease that can lead to gangrene), and sclerodermatous skin changes (hard-
ening and thickening of the skin). It is mutagenic and is a well-established cause of liver
cancer at levels formerly thought to be acceptable for occupational exposure.
Polychlorinated biphen yls (PCBs) have the general structure shown below, with chlor-
ines substitut ed at various positions, forming a great variety of possible congeners. Their
empirical formula is C
12
H
12n
Cl
n
, where n is the number of chlorine substitutions. Thus, a
congener with five chlorines will have a molar mass of 328.5 and consists of 54% chlorine
by weight. PCBs were produced as mixtures of various congen ers plus impurities and sold
by the Monsanto Company under the trade name Aroclor 12xx, where xx indicates the
average percent chlorine. Aroclor 1254, one of the common mixtures sold, thus has an
average of five chlorines per molecu le. Their vapor pressure and aqueous solubility is
very low, and they adsorb strongly to soil and sediment. The solubility of Aroclor 1248
ranges from 0.034 to 0.175 mg/L; that of Aroclor 1268 is less than 0.007 mg/L. They both
bioconcentrate and biomagnify in aquatic systems. They have the chemical property of
being highly stable, even at high temperatures, thus are very persistent in the environment.
They are resistant to biochemical degradation. However, some breakdown has been
observed with PCBs having fewer than four chlorine atoms.
Cl
Cl Cl Cl
biphenyl
2,2',3,4'- tetrachlorobiphenyl,
a polychlorinated biphenyl (PCB)
11'
2'3'
4'
5'
6'
23
4
56
Their use has been discontinued because of the environmental problems they cause.
However, because of their persistance and of previous releases, they remain a problem
in a number of aquatic and marine environments. For example, an advisory has been
issued recommending that consumption of striped bass taken from the Hudson River estu-
ary be limited to not more than one per month because of PCB contamination of that river.
820 TOXICITY OF SPECIFIC SUBSTANCES

PCBs are stored in fatty tissues and excreted in milk. They are strong inducers of
microsomal enzymes. This results in enhancement of metabolism of some of the sex hor-
mones and produces interaction with other toxins. They can also act as a carcinogenic
promoter. In the liver they cause tumor formation and fibrosis. Their reproductive effects
include reduced fertility in mink and rhesus monkey. They also produce immunosuppres-
sion. Other effects included immunosuppression, numbness, coughing and expectoration,
fatigue, and eye discharge.
Chloracne is one of the major symptoms of exposure to halogenated PAHs, including
chlorinated phenols, PCBs, and chlorinated dioxins. Chloracne is an acute rash of acne
that may resemble a severe form of ordinary acne, but characterized by hundreds and hun-
dreds of com edomes, very persistent blackheads that develop into inflamed papules.
In 1968, 1000 people in Japan were poisoned by PCB-contaminated rice oil. Chloracne
was the major symptom. Polybrominated biphenyls (PBBs) have toxic properties similar
to those of PCBs. They are less common in the environment, although a major exposure
occurred in Michigan in 1973 when cattle feed was contaminated.
PCBs have low acute toxicity to most mammals tested (Table 21.9). Mink is an excep-
tion, as are rhesus monkeys. The latter show toxic effects after one year of eating food
with 2.5 to 5.0 mg/day of PCB 1254; 100 to 300 mg/kg in food per day results in mortality
in two to three months. Toxicity to birds is lower than that of DDT and seems to increase
with degree of chlo rination. In contrast to birds, toxicity of PCBs to fish decreases with
degree of chlorination, and the overall toxicity is much higher.
Because of the typically low exposures, sublethal effects are more important in
mammals than are lethal effects. Effects include liver enlargement and damage, reduced
growth, immunosuppresion, and liver enzyme induction that increases with degree of
TABLE 21.9 Acute Toxicity of PCBs
a
Organism PCB Grade Conditions Test Toxicity
Mammals
Rats 1254 NA LD
50
1.3–2.5 g/kg
Rats, mice, rabbits 1242 NA LD
50
4.2–29 g/kg
Rats 1254 100% m. in 53 days PCB in food 1000 ppm
Rats 1254 50% m., 8 months PCB in food 1000 ppm
Mink 1254 100% m., 105 days PCB in food 3.6 ppm
Birds
Mallard, pheasant, Six Aroclors 50% in 5 days LC
50
0.745–5.0 g/kg
bobwhite, quail (1232–1264) of toxic diet
Fish
Gammarus 1254 PCB colloidal and LC
50
0.001–0.1 mg/L
oceanicus solubilized in
emulsion
Goldfish Clophen A50 5, 21 days LC
50
4, 0.5 mg/L
Pinfish, spotfish 1254 12–18 days LC
50
0.005 mg/L
Cutthrout trout 1221–1260 96 h LC
50
1.2–61.0 mg/L
Fathead minnows 1242–1254 Newly hatched, 96 h LC
50
0.008, 0.015 mg/L
Crustaceans
Pink shrimp PCBs 48 h LC
50
0.1 mg/L
a
NA, not available; m., mortality.
Source: Connell and Miller (1984).
HALOGENATED ORGANICS
821

chlorination. PCBs cause liver tumors in rats but are not mutagenic or teratogenic in mam-
mals. Birds, on the other hand, have exhibited teratogenesis from PCBs as well as eggshell
thinning and embryotoxicity. The carcinogenic potency of PCBs also varies with number
of chlorines. Molecules with five chlorines have the highest potency; those with six are
about half as potent; those with four are one-tenth as carcinogenic; molecules with fewer
than four or more than six are less than one-tenth as potent.
A number of reproductive effects have been noted in mammals. A single injection of
20 mg PCB per kilogram of body weight lengthens the estrus cycle of mice; 100 mg/kg in
the diet of Aroclor 1242 or 1254 decreased survival of rat offspring, and 20 mg/kg of Aro-
clor 1254 reduced the number of pups per litter. However, 100 mg of Aroclor 1260 per
kilogram showed no effect in rats. Monkeys showed lower fertility and reduced birth-
weights at a dosage of 2.5 ppm of Aroclor 1248 in the diet.
PCBs are of concern in ecosystems becau se of their persistance and tendency to bio-
concentrate and biomagnify. In addition, they show chron ic toxicity at exceedingly low
concentrations to certain species, which can cause significant population effects. As
low as 4.4 ppb decreases hatching, survival, and growth of coho salmon; 19 ppb produces
a 50% reduction in cell population growth of Daphnia magna. In addition, in an effect that
strikes at the heart of a natural aquatic ecosystem’s productivity, Aroclor 1242 inhibited
net phytoplankton production by 50% at exposures of 10 to 25 ppb. Changes also occur in
phytoplankton species distribution, with smaller species favored. This causes changes in
zooplankton species distribution, which may in turn affect community structure at higher
trophic levels.
Dioxins refer to chlo rinated dibenzo-p-dioxins. They are produced as a by-product of
various manufacturing processes, such as the production of the herbi cide 2,4,5-T or the
wood preservative pentachlorophenol, and are impurities in those products. They are
also produced in all combustion processes in which the feed material contains chlorine
in any form, such as chlorides. Thus, they may be found in fumes from incineration of
refuse, and even of plant material. However, they are not produced significantly in
exhausts from fossil-fuel combustion. The different congeners have different toxic and
physicochemical properties. Of greatest concern is 2,3,7,8-tetrachlorodibenzo-p-dioxin
(2,3,7,8-TCDD). This substance has the reputation of being the most toxic human-
made substance known.
O
O
O
OCl
Cl
Cl
Cl
1
2
3
4
5
10
9
8
7
6
dioxin 2,3,7,8-tetrachlorodibenzodioxin (TCDD)
Like PCBs, chlorinated dioxins tend to be very stable thermally and environmentally.
They photodegrade in the presence of organic material. However, they adsorb strongly to
particulate matter such as soil and sediment, protecting them from exposure to the neces-
sary ultraviolet radiation. They have very low solubility in both polar and nonpolar
solvents. The solubility in o-dichl orobenzene is 1.4 g/L, in n-octanol it is 0.048 g/L,
and in water it is 2 10
7
g=L. Thus, the K
OW
is 2:4 10
5
.
Table 21.10 shows the ranges of TCDD bioconcentration factors that have been
observed in model ecosystems. Data from the accident in Seveso, Italy (see below) and
elsewhere have shown that terrestrial organisms can bioconcentrate dioxin from the soil.
822 TOXICITY OF SPECIFIC SUBSTANCES

TCDD is absorbed in the liver by first-order kinetics and stored. It is slowly converted to
polar metabolites and excreted. Elimination in rats is also first order and can be described
by a one-compartment model. Half-lives of 17 and 31 days have been measured. How-
ever, biomagnification has not been proven for dioxin.
The toxic effects of chlorinated dioxins are similar to PCBs. They are strong inducers
of microsomal enzymes. They are toxic to the liver, immune system, and to fetuses. Ter-
atogenic effects have been observed at dosages as low as 1/400 of the LD
50
for the mater-
nal rat. Dioxins cause cancer in laboratory animals, including rats at 2 ppb. Tumors appear
in the liver, respiratory tract, and oral cavity, among othe r places. In humans, chlorinated
dioxins are known to produce chloracne, but no evidence of carcinogenicity has been
found. The extrapolation to humans is complicated by the extreme variability in toxicity
between different animals. The oral LD
50
for guinea pigs is 0.6 to 2 mg per kilogram of
body weight in a single oral dose, for mouse it is 114 to 284 mg/kg, and in hamsters it is
5 mg/kg.
Dioxin has been responsible for a number of notorious pollution episodes. It is an
ingredient of Agent Orange, the military defoliant used in Vietnam to deprive the
enemy of cover. In that application it was blamed for numerous toxic effects to exposed
American soldiers. Catfish and carp taken from the Dong Nai River in South Vietnam
were found to have from 0.320 to 1.02 mg/kg body burdens. Agent Orange manufacturing
plants in the United States, including in Cleveland, Ohio and Newark, New Jersey, were
found to be heavily contaminated as well.
In 1971, a company that manufactured 2,4,5-trichlorophenol hired a used oil dealer to
dispose of manufacturing wastes. The oil dealer mixed the material with waste oil and was
subsequently hired to spray oil for dust control at several riding stables in Times Beach,
Missouri. Shortly after the spraying, it was noticed that birds and insects nearby had
begun to die. The horses also became ill, and many died, including 65 of 125 at one stable.
The incident came to the attention of authorities when a 6-year-old girl became ill with
hemorrhagic cystitis. Others affected included a 10-year-old girl with nosebleeds and
headaches and two 3-year-old boys with chloracne. It took three years before the problem
was traced to dioxin and the oil dealer. Up to 37 sites had been sprayed. Many had soil
contamination levels higher than 100 mg/kg, and some more than 1.0 mg/kg. A flood sub-
sequently spread the contamination over a large area, which was evacuated by the U.S.
EPA and subsequently remediated.
TABLE 21.10 TCDD Bioconcentration Factors
Organism BCF
Plants
Algae 6–2083
Duck weed 4000
Invertebrates
Daphnia 2198–48,000
Snails 735–24,000
Fish
Silverside 54
Mosquito fish 676–24,000
Catfish 2000
Source: Connell and Miller (1984).
HALOGENATED ORGANICS
823
Near Seveso, Italy (near Milan), in 1976, a trichlorophenol reaction vessel at a 2,4,5-T
manufacturing plant released an unknown amount of dioxin over an area of 2000 ha
(about 5000 acres). Soil TCDD dosages ranged from <0.75 to 5000 mg/m
2
, resulting in
concentrations averaging 3.5 mg/kg in the top 7 cm of the soil. Extensive plant and animal
mortality occurred, including hundreds of small pets. Body burden measured in small wild
animals include 4.5 mg/kg in field mice and 12.0 mg/kg in earthworms. In humans 135
cases of chloracne were confirmed, mostly in children, but no deaths; 1 4 required hospi-
talization within 8 days and some required treatment for some years. An area of 108 ha
(267 acres) was evacuated of all residents and remai ns so today.
21.5 AIR POLLUTANTS
Air pollutants include species discharged directly to the atmosphere (such as carbon mon-
oxide, sulfur dioxide, and nitrogen oxide) and those formed from other pollutants by reac-
tions in the atmosphere (ozone and nitrogen dioxide). Some of the sulfur and nitrogen
species contribute to acid deposition (acid rain and dry acid deposition).
Nitrogen oxides are formed whenever oxygen and nitroge n are heated together to flame
temperatures. Thus, all combustion processes using air produce nitric oxide (NO). The
product formed remains when the mixture is cooled. The NO then reacts with O
2
in
the atmosphere to form nitrogen dioxide (NO
2
). Other species include nitrous oxide
(N
2
O), nitrogen trioxide (N
2
O
3
), nitrogen tetroxide (N
2
O
4
), and nitrogen pentoxide
(N
2
O
5
). These are often collectively called NO
x
. Only NO and NO
2
are environmentally
significant. NO
2
is the most abundant and most toxic form. It is also involved in reactions
that generate ozone and photochemical smog (see below). NO
2
is absorbed in water, but
NO is very insoluble. A seventh species, nitric acid (HNO
3
), is significant both toxicolo-
gically and as a component of acid deposition. Nitric acid is formed primarily by reaction
of NO
2
and hydroxide radicals, but also by a series of reactions involving NO
2
, ozone, and
water.
Plants take up and respond to NO
2
similarly to SO
2
. Plants can use atmospheric NO
2
nutritionally, assimilating them through the pathway
NO
x
! NO
3
! NO
2
! NH
3
! aminoacids ! proteins
This pathway can compete with carbon assimilation for NADPH
2
. Toxic effects can be
caused by accumulation of ammonia as well as other mechanisms, such as oxidation of
unsaturated fatty acids. However, it is believed that none of these effects are significant at
ambient NO
x
concentrations.
In humans and animals, most studies have been done at concentrations much higher
than ambient. Various cellular and structural changes have been observed in lungs
exposed to 10 to 25 ppmv of NO
2
for 24 hours. Brief inhalation of 200 to 700 ppmv
can be fatal. Systemically, NO
2
is converted to nitrite and nitrate in the lungs and can
be detected in the blood and urine. Some blood changes have been observed, although
methemoglobin is only formed at high concentrations. Lipids from the lungs of rats
exposed to NO
2
are oxidized, and more so if the rats are deficient in vitamin E. Asthmatics
have been shown to experience significant increase in hyperreactivity of the airway at
0.1 ppmv of NO
2
.NO
x
exposure from ambient air is less likely to be a problem than
the indoor air pollution produced by unvented use of combustion for cooking or heating
in homes.
824 TOXICITY OF SPECIFIC SUBSTANCES
Ozone (O
3
) is formed in various ways, including direct photolysis of oxygen (O
2
)by
ultraviolet light from sunlight or from equipment, or by spark discharges from motors, sta-
tic discharges, electrostatic air purifiers, and by lightning. UV from sunlight produces the
stratospheric ozone layer that protects the surface from the same ultraviolet radiation.
However, in the troposphere (the lower atmosphere) an environmentally more significant
mechanism is the complex sequence of reactions that form photochemical smog. In these
reactions, NO
2
and oxygen react to form ozone and NO. Normally, the equilibrium would
favor a low ozone concentration. However, atmospheric hydrocarbons, such as from
incomplete products of combustion, react to form free radicals. The organic radicals sca-
venge the NO, shifting the equilibrium to the right. The entire sequence is a chain reaction
regenerating the NO
2
, resulting in many ozone molecules produced for each NO
2
initially
available. Another important product of photochemical smog reactions is peroxyacyl
nitrate (PAN), formed by the reaction of the free radical ROO,NO
2
, and oxygen:
ROO + NO
2
+O
2
RC
O
OONO
2
peroxyacyl nitrate
⋅
In plants, ozone produces numerous growth and metabolic change s. Both ozone and
PAN can oxidize protein sulfhydryl groups, affecting enzyme function. For example,
exposure to 0.05 ppmv ozone inhibited the hydrolysis of starch for 2 to 6 hours in cucum-
ber, bean, and monkey flower.
Ozone and other oxidants, such as chlorine gas, can produce eye and respiratory tract
irritation. The effect has been likened to a ‘‘sunburn inside the lungs.’’ The ambient air
quality standard for ozone is 0.1 ppmv. Levels of 0.6 to 0.8 ppmv O
3
for 1 hour caused
headaches, nausea, anorexia, and increased airway resistanc e. Exposure has been shown
to produce hyperreactivity (asthma) in dogs and humans. Pulmonary edema has been
observed at concentrations only slightly higher than levels that occur ambiently in Los
Angeles, California. Humans and other animals are known to develop tolerance to
ozone exposure. Exposing rodents to 0.3 ppmv ozone makes them tolerant of later expo-
sures of several ppmv, which if applied to the same animals at the outset would cause
massive pulmonary edema. Inhalation of 10 to 20 ppmv of Cl
2
causes immediate irrita-
tion, and 1000 ppmv can be fatal after only brief exposure.
One incident of the release of an extremely hazardous air pollutant has had great influ-
ence on the regulation of these materials. On December 2, 1984, at an Indian subsidiary of
the Union Carbide Corporation in Bhopal, India, water was added to a storage tank con-
taining the pesticide precursor methyl isocyanate (MIC). The addition may have been
deliberate sabotage. In any case, the water reacted with the MIC to create a large amount
of heat, vaporizing the MIC. Several tons of vapors escaped into a residential area sur-
rounding the plant, waking the residents to choking, blinding fumes. Some 3800 people
died as an immediate resu lt, and 11,000 were left permanently disabled. (Unofficial esti-
mates put the total number of deaths as high as 16,000.) Chronic effects may be affecting
tens or hundreds of thousands more. Regardless of the exact numb er, there is little doubt
that this was the greatest single industrial disaster in history. The incident has led to strict
control over the storage of extremely hazardous air pollutants, including the chlorine gas
used at many water and wastewater treatment plants for disinfection.
H
3
CN
C
O
methyl isocyanate
AIR POLLUTANTS 825

Carbon monoxide (CO) is produced by automobile engines and other combustion pro-
cesses. It accumulates to toxicologically significant levels in high-traffic areas and areas
where vehicles must accelerate, such as at intersections and toll booths. Typical levels in
air range from 5 to 100 ppmv. The toxic effect of CO is related to the percent carboxy-
hemoglobin in the blood. Equation (17.2) and Table 21.11 show the equilibrium relation
between this and the CO concentration in the air. It takes 8 to 10 hours for the blood to
reach equilibrium. Thus an exposure to 50 ppmv for 90 minutes has been found to be
equivalent to 10 to 15 ppmv for 8 hours or more.
Sulfur dioxide in the atmosphere originates primarily from combustion of fossil fuels,
especially coal and some petroleum oils, and from metals smelting. Emissions are in the
form of SO
2
. The solubility of sulfur dioxide is 11.3 g per 100 mL, facilitating its absorp-
tion to aqueous aerosols. In solution it forms sulfite (SO
2
3
) and bisulfite (HSO
3
). It reacts
both in the gas phase and in the aerosols to form H
2
SO
4
. The sulfuric acid form is
removed from the atmosphere by dry and wet deposition.
Sulfur dioxide enters plant leaves by molecular diffusion through the stomata. Photo-
synthesis and transpira tion are increased by short-term low-concentration exposures, but
decreased by longer-term or higher-concentration levels. The resulting sulfite and bisulfite
are toxic to the plant. They can be oxidized with or without enzymes to the less toxic
sulfate, but damaging free radicals can be formed. Effects include chlorosis (loss of chlor-
ophyll) and necrosis as well as changes in phosphorus metabolism and photosynthesis.
The effects are highly species dependent. Atmospheric sulf ate is also a contributor to
acid deposition.
In animals, sulfur dioxide is irritating to the eyes and upper respiratory tract. However,
these effects appear only at levels 50 or 100 times ambient levels. Necrosis of nasal
epithelium occurred in mice exposed to 10 ppmv for 72 hours. However, at high humidity
or atmospheric particulate levels, increased irritation occurs. It is likely that it is the
conversion to sulfuric acid that is responsible. At 5 ppmv, humans exhibit increased
respiratory frequency and decreased tidal volume (faster and shallower breathing).
Asthmatics are more sensitive than the rest of the population, but less so when breathing
through the nose than through the mouth. SO
2
interacts positively with ozone and smoke
particles. Concentrations of SO
2
and smoke both exceeding 500 mg/m
3
are thought to
produce excess mortality among the elderly and the chronically ill; 130 mg/m
3
of SO
2
(0.046 ppmv) together with 130 mg/m
3
of particulates resulted in increased number and
severity of respiratory disease in schoolchildren.
Particul ates include dust and smoke. Smoke is made up of carbon and products of
incomplete combustion, including polynuclear aromatic compounds. The toxic effects
TABLE 21.11 Relation Between Percent Carboxyhemoglobin and Equilibrium Exposure
Concentration with Health Effects in Humans
Health Effect Percent Carboxyhemoglobin Exposure Concentration (ppmv)
No symptom to reduced acuity 0–10 0–200
Headache and reduced acuity 10–20 200–400
Throbbing headache 20–30 400–600
Vomit and collapse 30–40 600–800
Coma 40–60 800–1200
Death Over 60 Over 1200
Source: Based on Seinfeld (1975).
826 TOXICITY OF SPECIFIC SUBSTANCES
of smoke are thought to be primarily due to compounds that are adsorbed onto its surface.
The solids serve to carry the compounds into the lungs.
Ambient particulate concentrations as low as 80 to 100 mg/m
3
(annual geometric mean)
have been reported to result in increased death rates for people over 50 years old. A
24-hour level of 300 mg/m
3
together with 630 mg/m
3
of SO
2
(0.22 ppmv) produced
acute worsening of symptoms of patients suffering from chronic bronchitis.
Cigarette smoke contains more than 2000 identified compounds. It is a significant
source of radionuclides and carbon monoxide. Local toxic effects include chronic bron-
chitis, emphysema, and cancer of the lung and other parts of the respiratory tract. Sys-
temic effects include arteriosclerosis of coronary arteries and cancer of the bladder,
pancreas, and other organs. Nicotine in tobacco smoke causes addiction. Smoking para-
lyzes the mucociliary escalator that clears particles from the respiratory tract. It also inhi-
bits macrophages in the alveoli, making the lungs more susceptible to disease. Pregnant
women who smoke have more miscarriages and their babies have lower birthweights than
the infants of nonsmokers. Users of snuff or cigars who do not inhale smoke are suscep-
tible to cancer of the lips and oral cavity. Nonsmokers subjected to ‘‘secondhand smoke’’
also are at increased risk. Children of smokers have more respiratory illnesses than do
children of nonsmokers.
Epidemiological data have been collected on cigarette smoking and cancer only since
the 1940s. As mentioned above, the relative risk between smokers and nonsmokers for all
cancers is about 5.0, and for lung cancer is 10 to 15. The data on lung cancer can be sum-
marized in more detail by the following models. The rate of death by lung cancer
increases with the first to second power of the number of cigarettes smoked per day,
and with the fourth to fifth power of the number of years the person smoked. This
means that doubling the number of cigarettes smoked per day triples the risk of lung can-
cer, but doubling the number of years that one smokes increases the risk by a factor of
20. Another model that takes into account the smoker’s age in years (a) and years of
exposure (y)is
probability of death by lung cancer per year ¼ 10
11
a
4
þ 10
9
y
4
ð21:2Þ
According to this model, the annual lung cancer death rate for 70-year-old nonsmokers is
24 per 100,000. For 70-year-olds that have been smoking since the age of 20, the rate is
649 per 100,000. This is a relative risk of 27.
Smoking is als o known to increase the cancer rates of the oral cavity, the bladder, the
kidney, the pancreas, and the esophagus, as well noncancer diseases such as cerebrovas-
cular disease (e.g., strokes), coronary heart disease, and pulmonary disease (e.g., emphy-
sema).
21.6 WATER POLLUTANTS
The nitrogen species of intere st in water are ammonia, nitrite, and nitrate. The transfor-
mations and other relationships among these species are described in detail elsewhere.
Here we focus only on their toxicological properties. Ammonia distributes between the
nonionized form and the ammonium ion. The distribution, and thus the toxicity, depend
on the pH (Secti on 3.4). The nonionized form seems to be much more toxic, possibly
because of a greater ability to pass through membranes. Ammonia also increases in
toxicity at lower dissolved oxygen levels.
WATER POLLUTANTS 827
